A Standardized Quality Control Framework for Cerebral Cortical Organoids: Enhancing Reproducibility in Neuroscience Research and Drug Development
Cerebral cortical organoids represent a transformative in vitro model for studying human brain development, neurological disorders, and neurotoxicity.
A Standardized Quality Control Framework for Cerebral Cortical Organoids: Enhancing Reproducibility in Neuroscience Research and Drug Development
Abstract
Cerebral cortical organoids represent a transformative in vitro model for studying human brain development, neurological disorders, and neurotoxicity. However, significant challenges in reproducibility and quality standardization have limited their broader adoption in research and industrial applications. This article explores a newly developed hierarchical quality control (QC) framework that systematically evaluates 60-day cortical organoids across five critical criteria: morphology, size and growth profile, cellular composition, cytoarchitectural organization, and cytotoxicity. We examine the framework's methodological implementation, validation through controlled stress tests, and practical application for troubleshooting organoid variability. Furthermore, we discuss emerging complementary technologies for functional assessment and maturity benchmarking, providing researchers and drug development professionals with a comprehensive toolkit for enhancing experimental reliability and accelerating the translation of organoid technology into preclinical and pharmaceutical contexts.
The Critical Need for Standardization in Cerebral Organoid Research
The Promise and Pitfalls of Cerebral Organoids in Neuroscience
Human brain organoids have emerged as groundbreaking three-dimensional (3D) in vitro models that fundamentally transform how we study brain development, disease mechanisms, and therapy discovery [1]. Derived from human pluripotent stem cells (hPSCs), including induced pluripotent stem cells (iPSCs), these self-organizing structures closely mimic the cellular diversity, spatial organization, and functional connectivity of the early human brain [2] [1]. This biological fidelity offers significant advantages over traditional two-dimensional (2D) cell cultures and animal models, which often fail to replicate the human brain's 3D architecture and species-specific features [1].
Despite their transformative potential, cerebral organoids face significant challenges regarding quality, reproducibility, and standardization [2]. Morphological inconsistencies, variations in size, differences in cellular composition, and discrepancies in functional activities often arise from the stochastic nature of stem cell differentiation and spontaneous self-organization within organoids [2]. These limitations compromise their reliability in crucial applications like disease modeling, neurotoxicity testing, and preclinical drug screening [2]. This article examines both the promise and pitfalls of cerebral organoid technology, with particular focus on how emerging quality control frameworks address these critical challenges.
The Promise: Unprecedented Opportunities in Neuroscience
Enhanced Physiological Relevance
Cerebral organoids provide a physiologically relevant microenvironment that surpasses conventional models. Unlike 2D cultures, they enable realistic cell-cell interactions, molecular transport, and spatial organization that mirrors native brain tissue [1]. This 3D architecture supports autocrine and paracrine signaling, enabling more accurate modeling of cellular proliferation, migration, and differentiation [1]. Through guided differentiation protocols, researchers can generate region-specific organoids (cortical, midbrain, striatal) that recapitulate developmental trajectories observed in the human fetal brain [2] [3].
Diverse Research and Clinical Applications
The applications of brain organoids span multiple neuroscience domains:
Disease Modeling: When derived from patient-specific iPSCs, organoids reproduce disease-specific phenotypes for conditions including Alzheimer's disease [2] [1], Parkinson's disease [2] [1], microcephaly [2] [4], autism spectrum disorders [1], and Zika virus infection [4]. They enable studies of pathological mechanisms in a human-specific context.
Drug Screening and Neurotoxicity Testing: Organoids provide a human-relevant platform for evaluating drug efficacy and safety, as well as assessing the developmental neurotoxicity of chemicals, pollutants, and pharmaceuticals [2] [5]. Studies have successfully investigated effects of valproic acid, nicotine, cannabis, bisphenol S, cadmium, and nanoplastics [2].
Personalized Medicine: The combination of patient-derived iPSCs with CRISPR/Cas9 gene editing enables highly precise mechanistic studies and scalable drug screening for personalized therapeutic development [1].
Innovative Computing Applications
Emerging research explores brain organoids for highly efficient biocomputing systems. These organic processors demonstrate remarkable energy efficiency, potentially operating with 10³ to 10⁶ times greater efficiency than traditional silicon-based computers [6]. Their inherent parallelism, adaptivity through plasticity, and event-driven spiking behavior offer advantages for complex AI tasks and pattern recognition [6]. Early implementations already enable simple computations, image processing, and speech recognition [6].
The Pitfalls: Critical Challenges Limiting Adoption
Variability and Reproducibility Issues
The most significant challenge facing cerebral organoid technology is inter-organoid heterogeneity [2] [1] [4]. This variability manifests across multiple dimensions:
- Morphological variability: Within a single batch, organoids may display optimal morphology with dense structure and well-defined borders, while others appear poorly compacted with degraded structures [2].
- Cellular composition differences: Proportions of neural cell types (progenitors, neurons, glia) vary significantly, with some organoids containing unintended cell populations like mesenchymal cells [4].
- Structural inconsistencies: While some organoids develop proper ventricular-like structures, others form disorganized architectures or develop suboptimal cystic cavities [2] [4].
This heterogeneity stems from multiple sources, including the stochastic nature of stem cell differentiation, variations in self-organization, differences in stem cell sources, reagent quality variations, and manual handling differences [2] [1].
Technical and Biological Limitations
Current organoid models face several biological constraints:
- Limited maturation: Organoids typically model early developmental stages rather than mature brain circuitry [1].
- Absence of vascularization: The lack of blood vessels limits nutrient diffusion, potentially leading to hypoxic core regions and necrotic centers [2] [1].
- Incomplete cell type representation: While containing multiple neural cell types, organoids may lack all relevant cellular components found in native brain tissue [1].
- Simplified connectivity: Although exhibiting some functional activity, organoids lack the complex inter-regional connectivity of the human brain [1].
Standardization and Characterization Gaps
The field suffers from a notable absence of standardized quality metrics and characterization methods [2] [7]. Current assessment approaches often rely on qualitative, subjective evaluations that introduce inconsistencies and bias between research groups [2]. Furthermore, analysis methods commonly used in 2D cultures are difficult to transpose to 3D organoid systems, complicating the standardization of their characterization [2]. This methodological gap hinders the comparability of results across different laboratories and limits the broader adoption of organoid technology in industrial and clinical applications [2] [5].
Quality Control Frameworks: Addressing the Standardization Challenge
A Comprehensive QC Scoring System
Recent research has introduced standardized quality control (QC) frameworks specifically designed for cerebral organoids [2] [8]. One comprehensive approach proposes a hierarchical scoring system for 60-day cortical organoids based on five critical criteria [2] [8]:
- Morphology: Assessing compactness, border integrity, and absence of cysts
- Size and Growth Profile: Ensuring proper developmental dynamics
- Cellular Composition: Verifying presence of expected neural populations
- Cytoarchitectural Organization: Evaluating structural integrity and organization of neural layers
- Cytotoxicity: Measuring DNA damage and cell viability
This framework operates hierarchically, beginning with non-invasive assessments (morphology and size) to exclude low-quality organoids early, while reserving in-depth analyses for those passing initial evaluation thresholds [2]. The system supports both Initial QC for pre-study selection and Final QC for comprehensive post-study analysis [2].
Table 1: Quality Control Scoring Criteria for 60-Day Cortical Organoids
| QC Criterion | Assessment Method | Key Parameters | Quality Threshold |
|---|---|---|---|
| Morphology | Brightfield imaging | Compactness, border integrity, cyst formation | Minimum score-based threshold |
| Size & Growth | Diameter measurement, time-series analysis | Growth dynamics, Feret diameter | ~3050 μm Feret diameter [4] |
| Cellular Composition | Immunostaining, flow cytometry | Neural progenitors (SOX2, PAX6), neurons (MAP2) | Presence of expected cell types |
| Cytoarchitectural Organization | Immunohistochemistry | Ventricular-like structures, cortical layers | Organized neural rosettes |
| Cytotoxicity Viability assays | DNA damage markers, cell death assays | Low cytotoxicity levels |
Simplified Morphological Metrics
Complementing comprehensive frameworks, researchers have identified simplified morphological parameters that strongly correlate with organoid quality. The Feret diameter (maximal caliper diameter) has emerged as a particularly reliable, single-parameter predictor of organoid quality [4]. Studies demonstrate that classifying organoids using a Feret diameter threshold of approximately 3050 μm accurately reflects expert quality evaluations with 94.4% positive predictive value [4]. This metric correlates with underlying biological factors; low-quality organoids with larger Feret diameters typically show higher proportions of unintended mesenchymal cells, indicating aberrant differentiation [4].
Experimental Validation of QC Frameworks
To validate quality control systems, researchers have exposed 60-day cortical organoids to graded doses of hydrogen peroxide (H₂O₂), inducing controlled oxidative stress and generating a spectrum of quality outcomes [2] [8]. The QC framework successfully discriminated organoid qualities across this range, demonstrating its sensitivity and reliability [2]. This experimental approach provides a robust validation method for assessing QC system performance.
Table 2: Experimental Approaches for QC Framework Validation
| Validation Method | Implementation | Outcome Measures | Significance |
|---|---|---|---|
| Controlled Stress Induction | H₂O₂ exposure at graded doses | QC system's ability to discriminate quality levels | Establishes system sensitivity and reliability [2] |
| Multi-Protocol Comparison | scRNA-seq across 4 protocols, multiple cell lines | Cell-type recapitulation, differentiation propensity (NEST-Score) | Identifies protocol-specific strengths [3] |
| Multi-Center Analysis | 72 organoids from 12 hPSC lines | Morphological parameters, transcriptomic signatures | Identifies reproducible quality markers [4] |
| Longitudinal Analysis | Time-resolved RNA-seq up to day 120 | Developmental trajectories, maturation markers | Tracks quality across maturation timeline [3] |
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for Cerebral Organoid QC
| Reagent/Material | Function | Application Examples |
|---|---|---|
| Human Pluripotent Stem Cells (hPSCs) | Starting material for organoid generation | Embryonic stem cells (H9, H1), induced pluripotent stem cells [1] [4] |
| Artificial ECMs (Matrigel, Geltrex) | Provide structural support, promote tissue organization | Embedding organoids, inclusion in culture media [1] |
| Patterning Molecules | Direct regional specification | Generating region-specific organoids (cortical, midbrain) [1] |
| Differentiation Media | Support neural differentiation, maintenance | Various protocol-specific formulations [3] [4] |
| Immunostaining Reagents | Cellular composition and organization analysis | Antibodies against SOX2, MAP2, PAX6 for neural markers [2] [4] |
| Viability Assays | Cytotoxicity assessment | Measuring DNA damage, cell death [2] |
| Multielectrode Arrays (MEAs) | Functional characterization | Recording electrical activity, stimulation [6] |
Cerebral organoids represent a transformative technology with immense potential for advancing neuroscience research, disease modeling, and therapeutic development. However, realizing this potential requires addressing significant challenges in quality control, standardization, and reproducibility. The emerging QC frameworks detailed herein provide essential tools for objective, reproducible quality assessment that enhances result consistency and comparability across laboratories [2] [8]. By implementing these standardized approaches—ranging from comprehensive scoring systems to simplified morphological metrics—researchers can minimize observer bias, improve experimental reliability, and accelerate the adoption of cerebral organoids in both academic and industrial settings [2] [5]. As the field continues to evolve, these quality control foundations will be crucial for harnessing the full promise of cerebral organoids while effectively navigating their current pitfalls.
A significant challenge in cerebral cortical organoid research is the inherent variability in morphology, size, and cellular composition. This variability poses a substantial barrier to the reproducibility and reliability of experiments, particularly in disease modeling and drug screening [2]. This guide objectively compares the key sources of this variability and details the experimental methodologies used to quantify them, framing the discussion within the essential context of developing robust quality control frameworks.
Comparative Analysis of Variability in Cerebral Organoids
The table below summarizes the primary sources of variability in cerebral organoids, their impact on research, and the corresponding quality control metrics proposed to address them.
| Variability Parameter | Manifestation in Low-Quality Organoids | Manifestation in High-Quality Organoids | Impact on Research Reproducibility | Proposed QC Metrics [2] |
|---|---|---|---|---|
| Morphology | Irregular shape; presence of large, fluid-filled cysts; poorly defined borders; migrating cells; degraded or non-compact structure [2] [4]. | Spherical shape with well-defined, dense borders; presence of neuroepithelial buds [2] [4]. | Introduces bias in structural analyses; affects consistency in toxicity and drug response assays [2]. | Scoring system (0-5) for morphology, cyst amount, and cyst area. |
| Size | High degree of size dispersion; Feret diameter >3050 μm at day 30, correlating with lower quality [4]. | Consistent size within batches; Feret diameter below ~3050 μm at day 30 [4]. | Influences diffusion gradients, leading to necrotic cores; creates disparities in cell number for assays [2] [9]. | Scoring system (0-5) for size and growth profile; Feret diameter threshold. |
| Cellular Composition | Presence of unintended cell types, particularly mesenchymal cells (can range up to 74% of composition) [4]. Non-cerebral structures like neural crest or choroid plexus cells [10]. | Predominantly neural lineage cells (can exceed 99%); presence of expected cells like SOX2+ neural progenitors and MAP2+ neurons [4]. | Confounds disease-specific phenotypes; leads to incorrect interpretation of transcriptomic or functional data [2] [4]. | Scoring system (0-5) for cellular composition; single-cell RNA-seq validation. |
| Cytoarchitectural Organization | Failure to form ventricular-like structures (VLS); disorganized cellular architecture [4]. | Formation of multiple, well-defined VLS populated with SOX2+ neural stem cells [4]. | Limits modeling of neurodevelopmental processes; reduces physiological relevance for studying neurogenesis [2]. | Scoring system (0-5) for cytoarchitectural organization. |
Experimental Protocols for Quantification
Standardized experimental protocols are critical for objectively assessing the variability outlined above.
Hierarchical Quality Control Framework
A proposed QC methodology for 60-day cortical organoids uses a hierarchical scoring system to efficiently classify organoid quality [2].
- Objective: To provide a standardized, user-friendly framework for quality assessment of cerebral organoids, enabling reliable selection for downstream applications.
- Procedure:
- Initial QC (Pre-Study): Organoids are first evaluated using non-invasive criteria:
- Threshold Check: Organoids failing minimum thresholds for Criteria A and B are excluded from further study.
- Final QC (Post-Study): Organoids passing the initial QC undergo in-depth, often invasive, analysis:
- Criterion C - Cellular Composition: Analyzed via immunohistochemistry for markers like SOX2 (neural progenitors), MAP2 (neurons), and PAX6 (CNS progenitors), or by flow cytometry. scRNA-seq can identify off-target cells [4] [10].
- Criterion D - Cytoarchitectural Organization: Assessed via immunohistochemistry on cryosections for the presence and organization of ventricular-like structures [4].
- Criterion E - Cytotoxicity: Measured using assays like lactate dehydrogenase release [2].
- Validation: This framework was validated by exposing organoids to hydrogen peroxide (H₂O₂), which induced a range of quality outcomes. The QC system successfully discriminated between these quality levels [2].
Morphometry and Transcriptomic Correlation
This protocol links morphological measurements directly to underlying cellular composition.
- Objective: To identify straightforward morphological parameters that predict organoid quality and cellular purity [4].
- Procedure:
- Bright-field Imaging: Organoids are imaged at a standard stage (e.g., day 30).
- Morphometric Analysis: Images are analyzed with software (e.g., ImageJ, MOrgAna) to extract parameters like Feret diameter (maximal caliper distance), area, perimeter, and cyst area [4] [11].
- Unsupervised Classification: Parameters are subjected to k-means clustering (k=2) to objectively classify organoids into high- and low-quality groups without expert bias [4].
- Transcriptomic Deconvolution: Bulk RNA sequencing is performed on the same organoids. Computational tools like BayesPrism are used with reference single-cell atlases (e.g., Human Neural Organoid Cell Atlas) to estimate cellular composition, particularly the proportion of mesenchymal cells [4].
- Key Finding: The Feret diameter is a highly reliable single parameter for quality assessment. Organoids with a larger Feret diameter consistently showed a higher proportion of unintended mesenchymal cells, confirming that simple morphological analysis can predict cellular composition [4].
Experimental Workflow and Signaling Relationships
The following diagram illustrates the logical workflow for the morphometry and transcriptomic correlation protocol.
The Scientist's Toolkit: Key Research Reagents and Materials
Successful execution of the described protocols relies on a suite of specific reagents and tools. The table below details these essential items.
| Tool/Reagent | Function in Experimental Protocol |
|---|---|
| hPSCs (H9, H1, iPSCs) [4] | The starting biological material for generating cerebral organoids; source of donor-specific genetic information. |
| Matrigel / Extracellular Matrix [9] [4] | Provides a 3D scaffold that supports organoid formation, growth, and self-organization. |
| Defined Culture Media & Supplements [9] | Contains specific growth factors, hormones, and small molecules to direct neuroectodermal differentiation and maintain organoid culture. |
| MOrgAna / ImageJ Software [11] [4] | Machine learning-based or standard image analysis tools for segmenting organoids and quantifying morphological parameters (Feret diameter, area, etc.). |
| Antibodies for IHC (SOX2, MAP2, PAX6) [4] | Enable visualization and quantification of specific cell types (neural progenitors, neurons) and cytoarchitectural features (ventricular-like structures). |
| scRNA-seq / Bulk RNA-seq Platforms [4] [10] | Used for comprehensive analysis of cellular composition, identification of off-target cell populations, and validation of neural identity. |
Cerebral cortical organoids have emerged as groundbreaking tools in neuroscience, providing unprecedented in vitro models that recapitulate aspects of the human brain's complexity. These three-dimensional structures derived from pluripotent stem cells self-organize into architectures containing neural progenitors, neurons, and astrocytes, offering unique insights into neurodevelopmental processes. However, challenges related to reproducibility and quality have historically limited their translational potential. The establishment of standardized quality control frameworks represents a critical advancement for harnessing organoids in key applications including disease modeling, drug screening, and neurotoxicity testing. This guide objectively compares organoid performance across these applications, examining how quality control methodologies enhance data reliability and experimental outcomes.
Quality Control Framework for Cerebral Cortical Organoids
Standardized QC Criteria and Scoring System
A comprehensive Quality Control framework specifically designed for 60-day cortical organoids employs a hierarchical scoring system across five critical criteria [2] [8]:
- Morphology: Assessing compactness, border integrity, and absence of cystic structures
- Size and Growth Profile: Ensuring proper developmental dynamics and dimensional consistency
- Cellular Composition: Verifying presence of expected neural populations (neural progenitors, neurons, astrocytes)
- Cytoarchitectural Organization: Evaluating structural integrity and organization of neural layers
- Cytotoxicity: Detecting DNA damage and cell viability markers
The framework operates through two distinct assessment phases [2]:
- Initial QC: Utilizes non-invasive criteria (morphology and size) for pre-study organoid selection
- Final QC: Implements comprehensive analysis of all five criteria for post-study evaluation
Table 1: QC Scoring Thresholds for 60-Day Cortical Organoids
| QC Phase | Criteria Assessed | Minimum Threshold | Assessment Method |
|---|---|---|---|
| Initial QC | Morphology | Score >3 | Brightfield microscopy |
| Initial QC | Size and Growth Profile | Score >3 | Diameter measurement |
| Final QC | Cellular Composition | Score >3 | Immunohistochemistry |
| Final QC | Cytoarchitectural Organization | Score >3 | Histological analysis |
| Final QC | Cytotoxicity | Score >3 | Viability assays |
Experimental Validation of QC Framework
The QC system was rigorously validated by exposing 60-day cortical organoids to graded doses of hydrogen peroxide (H₂O₂) to induce controlled quality variations [2]. Organoids were first selected using Initial QC criteria before H₂O₂ exposure, followed by a one-week recovery period and comprehensive Final QC assessment. The framework successfully discriminated organoid quality levels across the induced variability spectrum, demonstrating robust sensitivity to detect quality variations [2].
Disease Modeling Applications
Recapitulation of Neurodevelopmental Processes
Quality-controlled cerebral organoids effectively model key neurodevelopmental aspects including neurogenesis, neuronal migration, neuromorphogenesis, and synaptogenesis [2]. Transcriptomic and epigenetic analyses confirm that these models closely mimic developmental trajectories observed in the human fetal brain, providing unprecedented opportunities to study neurodevelopmental disorders [2].
Specific Disease Models
When derived from patient-specific cells or combined with genetic engineering techniques, quality-controlled organoids have successfully modeled [2]:
- Microcephaly through disruption of neural progenitor proliferation
- Trisomy 21 (Down syndrome) with characteristic neurodevelopmental alterations
- Neurological cancers utilizing tumor-specific mutations
- Neurodegenerative diseases including Alzheimer's disease, Parkinson's disease, and Creutzfeldt-Jakob disease
Enhanced Protocol for Outer Radial Glia Enrichment
Advanced protocols generating cortical organoids enriched in outer radial glia (oRG) through guided differentiation with leukemia inhibitory factor (LIF) treatment have demonstrated improved reproducibility and cellular complexity [12]. These organoids develop an expanded outer subventricular zone (oSVZ), recapitulating a critical human-specific developmental feature essential for studying cortical expansion disorders [12].
Table 2: Disease Modeling Performance Comparison
| Disease Model | Organoid Type | Key Features Recapitulated | QC Requirements |
|---|---|---|---|
| Microcephaly | Unguided whole-brain | Reduced neuronal output, progenitor defects | Size, Cellular composition |
| Trisomy 21 | Cortical forebrain | Altered neurogenesis, delayed maturation | Growth profile, Cytoarchitecture |
| Alzheimer's Disease | Mature cortical | Aβ aggregation, tau pathology | Cellular composition, Cytotoxicity |
| Parkinson's Disease | Midbrain organoids | Dopaminergic neuron vulnerability | Cellular composition, Morphology |
| Cortical Malformations | oRG-enriched cortical | OSVZ expansion, neuronal migration | Cytoarchitecture, Cellular composition |
Drug Screening Applications
High-Content Screening Methodologies
Advanced drug screening approaches for organoids have evolved beyond bulk viability measurements to high-content imaging techniques that capture heterogeneous responses at the individual organoid level [13] [14]. These methodologies employ fluorescent indicators including:
- Hoechst 33342: Nuclear staining for cell counting and segmentation
- Caspase 3/7 Green: Apoptosis detection
- Propidium Iodide: Cell viability assessment through membrane integrity
Experimental Protocol for Drug Screening
Optimized drug testing procedures for prostate cancer patient-derived xenograft organoids (PDXOs) demonstrate key methodological considerations applicable to cerebral organoids [13]:
- Organoid Preparation: Passage organoids through 100μm strainers to reduce size heterogeneity
- Hydrogel Embedding: Use synthetic hydrogels (Noviogel-P5K) with medium-gel ratio of 64.5:35.5
- Drug Exposure: Initiate treatment 4 days after plating, maintain for 10 days with logarithmic dosing
- Viability Assessment: Utilize CellTiter-Glo 3D with preliminary ice incubation for hydrogel dissolution
- Image Acquisition: Employ confocal high-content systems (Opera Phenix) with 40× water immersion objectives
Performance Comparison with Traditional Models
Quality-controlled organoids demonstrate superior performance in drug screening compared to traditional models [13] [15]:
Table 3: Drug Screening Model Comparison
| Screening Model | Throughput | Clinical Predictive Value | Heterogeneity Capture | Cost Efficiency |
|---|---|---|---|---|
| 2D Cell Culture | High | Limited | No | High |
| Animal Models | Low | Moderate (species differences) | Limited | Very Low |
| Uncontrolled Organoids | Medium | Variable | Partial | Medium |
| QC-Controlled Organoids | Medium-High | Improved | Yes | Medium |
Neurotoxicity Testing Applications
Addressing Limitations of Conventional Models
Traditional neurotoxicity testing relies on animal behavioral studies and 2D cell culture cytotoxicity assays with endpoints that poorly correlate to human functional network disruptions [16]. Approximately one in four safety-related drug failures stem from undetected central nervous system toxicity, with nearly 80% of issues remaining undetected until clinical trials [16]. Quality-controlled cerebral organoids address this translational gap by providing human-relevant functional endpoints for neurotoxicity assessment.
Organoid Intelligence for Developmental Neurotoxicity
Organoid intelligence (OI) leverages brain organoids to study neuroplasticity in vitro, bringing a new approach to measure impacts of xenobiotics on plasticity mechanisms—critical processes not adequately covered in current developmental neurotoxicity (DNT) in vitro assays [17]. OI assays include:
- Open-loop experiments: Organoids receive electrical, chemical, or optogenetic stimuli with measured network dynamic responses
- Closed-loop experiments: Feedback provided based on organoid responses, testing capability to "learn" and modify output
Regulatory Context and Standardized Testing
U.S. regulatory policy is accelerating the shift toward organoid-based neurotoxicity testing [16]:
- FDA Roadmap (April 2025): Outline of phased strategy to integrate New Approach Methodologies (NAMs) including organoids
- NIH Funding (July 2025): New opportunities requiring NAMs alongside animal models
- SOM Center: Standardized Organoid Modeling Center with $87 million funding for model validation
Commercial Neurotoxicity Testing Platforms
The 28bio CNS-3D platform exemplifies industrial application of quality-controlled organoids for neurotoxicity testing [16]:
- Functional Assessment: Detects neuromodulatory and seizurogenic effects through high-throughput calcium imaging
- Consistency: Maintains uniform organoid size, cellular ratios, and pharmacological response across batches
- Endpoint Measurement: Quantifies functional signatures including synchrony, frequency, and amplitude shifts in network activity
Table 4: Neurotoxicity Testing Model Comparison
| Testing Model | Human Relevance | Functional Assessment | Regulatory Acceptance | Standardization Level |
|---|---|---|---|---|
| Animal Behavioral Tests | Low | Indirect behavioral correlates | High (declining) | High |
| 2D Cytotoxicity Assays | Medium | Limited to cell death | Medium | Medium |
| Traditional Organoids | High | Variable functional response | Emerging | Low |
| QC-Framework Organoids | High | Consistent functional metrics | Growing (NAM adoption) | High |
The Scientist's Toolkit: Essential Research Reagents
Table 5: Key Reagents for Cerebral Organoid Research
| Reagent/Category | Specific Examples | Function | Application Context |
|---|---|---|---|
| Extracellular Matrices | Matrigel, Synthetic PEG hydrogels | Structural support for 3D growth | Organoid formation, Self-assembly protocols [18] |
| Neural Induction Media | APCOM, PGM basic | Guided differentiation toward neural lineages | Cortical specification, Regional patterning [13] [12] |
| Small Molecule Inhibitors | Y-27632 (ROCK inhibitor) | Enhance cell survival after passage | Routine organoid maintenance [13] |
| Viability Assay Kits | CellTiter-Glo 3D | ATP-based viability quantification | Drug screening, Cytotoxicity assessment [13] |
| Cell Death Detection | Hoechst 33342, Propidium Iodide, Caspase 3/7 Green | Nuclear, membrane integrity, and apoptosis staining | High-content imaging, Mechanism of action studies [13] |
| Functional Probes | Calcium indicators (e.g., Cal-520) | Network activity monitoring | Neurotoxicity screening, Functional assessment [16] |
The implementation of standardized quality control frameworks represents a transformative advancement for cerebral cortical organoid applications across disease modeling, drug screening, and neurotoxicity testing. The hierarchical QC system evaluating morphology, size, cellular composition, cytoarchitecture, and cytotoxicity enables reliable discrimination of organoid quality, directly addressing previous challenges with reproducibility. Quality-controlled organoids demonstrate superior performance in recapitulating disease phenotypes, predicting drug responses, and detecting functional neurotoxicity compared to traditional models. As regulatory momentum builds for New Approach Methodologies, with the FDA and NIH actively promoting alternatives to animal testing, quality-controlled cerebral organoids are poised to become indispensable tools in neuroscience research and drug development pipelines.
Current Limitations in Organoid Characterization Methods
Organoid technology has revolutionized biomedical research by providing complex three-dimensional (3D) in vitro models that mimic the structure and function of human organs, notably the brain [19] [20]. These models offer unprecedented opportunities for studying human brain development, neurological diseases, and drug interactions, effectively addressing ethical and practical limitations inherent in traditional animal models [20] [21]. However, the transformative potential of cerebral organoids is hampered by significant challenges in characterization methods. Variability in morphology, cellular composition, and cytoarchitectural organization across batches creates substantial obstacles for reproducibility, hindering both fundamental research and preclinical applications [5] [8]. This review objectively compares current characterization approaches within the emerging context of quality control (QC) frameworks, synthesizing experimental data to highlight methodological limitations and propose standardized pathways forward for researchers and drug development professionals.
Fundamental Limitations in Current Characterization Practices
The absence of standardized, universally accepted characterization protocols leads to inconsistent assessment of organoid quality, compromising data comparability across studies and laboratories.
Variability in Morphological and Cellular Composition
A primary limitation lies in the significant heterogeneity observed even within organoids generated from the same stem cell line under identical conditions [4]. This variability manifests in several key areas:
- Morphological Diversity: Organoids exhibit considerable differences in size, shape, and structural integrity. Undesired morphological features such as fluid-filled cysts, irregular shapes, and migrating cells are common [4].
- Inconsistent Cellular Makeup: The presence and proportion of intended neural lineages versus unintended cell populations vary substantially. Transcriptomic analyses consistently reveal significant variation in the proportion of mesenchymal cells (ranging from 0.5% to 74% across samples), which correlates strongly with reduced organoid quality [4].
- Structural Formation Disparities: The development of critical ventricular-like structures (VLS) within organoids is inconsistent; some organoids form multiple VLS populated with SOX2+ neural stem cells, while others fail to form these structures entirely [4].
Table 1: Key Sources of Variability in Cerebral Organoid Generation
| Variability Factor | Impact on Characterization | Experimental Evidence |
|---|---|---|
| Stem Cell Line Differences | Donor-specific epigenetic signatures influence differentiation potential and final cellular composition [19]. | Coefficient of variation of mean mesenchymal cell composition across different cell lines: 80.98% [4]. |
| Protocol Selection | Guided vs. unguided protocols yield different regional identities and cellular diversity [22]. | Unguided protocols (36/114 studies) produce multiple brain regions; guided protocols (78/114 studies) generate region-specific identities [22]. |
| Extracellular Matrix (ECM) | ECM source and batch effects significantly impact neuroepithelial morphogenesis and reproducibility [22]. | Matrigel, used in 67/114 protocols, contains >1800 unique proteins with batch-to-batch variability [22]. |
| Culture Duration | Extended cultures (≥6 months) for maturation exacerbate metabolic stress and hypoxia-induced necrosis [23]. | Necrotic cores in prolonged cultures diminish cellular diversity and structural integrity, complicating accurate characterization [23]. |
Challenges in Functional Maturation Assessment
Current organoid models consistently fail to achieve full functional maturity, particularly those characteristics essential for modeling adult-onset neurological disorders and performing clinically predictive drug screening.
- Developmental Arrest: Brain organoids predominantly remain at fetal-to-early postnatal stages of development, even after extended culture periods exceeding 100 days [23]. This precludes their effective use in modeling late-onset disorders like Alzheimer's disease, which require mature amyloid-β processing pathways [23].
- Immature Network Activity: While organoids demonstrate electrical activity and synaptic connections, they often lack the sophisticated network plasticity and oscillatory patterns characteristic of mature human brain circuits [23].
- Deficient Supportive Cell Functions: The maturation of key supportive cells, particularly astrocytes, remains a major bottleneck. Current organoid paradigms frequently fail to robustly form essential structures like the glia limitans and a fully functional blood-brain barrier (BBB) [23].
Quantitative Frameworks for Standardizing Organoid Assessment
To address these characterization challenges, researchers have begun developing quantitative QC frameworks that establish objective metrics for evaluating organoid quality.
Hierarchical Quality Control Scoring System
A comprehensive QC framework specifically designed for 60-day cortical organoids introduces a scoring-based system evaluating five essential criteria [8]:
- Morphology - assessing compactness, border integrity, and absence of cysts
- Size and Growth Profile - ensuring proper developmental dynamics
- Cellular Composition - presence of expected neural populations
- Cytoarchitectural Organization - structural integrity and organization of neural layers
- Cytotoxicity - assessment of DNA damage and cell viability
This framework operates hierarchically, with an initial non-invasive QC (morphology and growth) for pre-selection, and a final comprehensive QC integrating all criteria for post-study evaluation [8]. When validated with hydrogen peroxide exposure experiments, this system successfully distinguished organoids of different quality levels, demonstrating sensitivity, reliability, and scalability for industrial applications [8].
Morphological Parameters as Quality Predictors
Research has identified specific, easily measurable morphological parameters that strongly correlate with organoid quality, potentially simplifying initial quality assessment:
Table 2: Morphological Parameters Correlated with Organoid Quality
| Parameter | Correlation with Quality | Optimal Threshold (Youden Index) | Predictive Value |
|---|---|---|---|
| Feret Diameter (maximal caliper) | Strong inverse correlation | 3050 μm (Youden index: 0.68) [4] | PPV: 94.4%; NPV: 69.4% [4] |
| Area | Significant inverse correlation | Not specified | Included in top 5 parameters [4] |
| Cysts Amount | Significant direct correlation | Not specified | Included in top 5 parameters [4] |
| Perimeter | Significant inverse correlation | Not specified | Included in top 5 parameters [4] |
| Cysts Area | Significant direct correlation | Not specified | Included in top 5 parameters [4] |
The Feret diameter has emerged as a particularly reliable single parameter for characterizing brain organoid quality, with high-quality organoids consistently displaying a lower diameter (<3050 μm) accompanied by a reduced proportion of unintended mesenchymal cells [4]. K-means clustering using the top five morphological parameters achieved a 93.3% positive predictive value for quality classification, confirming that morphological measurements can effectively objectify visual expert evaluation [4].
Methodological Limitations in Experimental Characterization Protocols
Structural and Cellular Characterization Methods
Current methodologies for assessing structural architecture and cellular diversity face significant limitations in standardization and interpretation:
Table 3: Experimental Methods for Brain Organoid Characterization
| Method Category | Specific Techniques | Key Limitations | Typical Output Metrics |
|---|---|---|---|
| Structural Architecture | Immunofluorescence/Immunohistochemistry (IF/IHC) with confocal microscopy; Electron Microscopy (EM) [23] | Manual quantification; Sample destruction; Limited 3D reconstruction; Expertise-intensive [23] | Cortical lamination (SATB2, TBR1, CTIP2); VLS formation; Synaptic density (SYB2, PSD-95) [23] |
| Cellular Diversity | Fluorescence-Activated Cell Sorting (FACS); scRNA-seq; IF/IHC [23] | Marker specificity issues; Cell loss during processing; High cost for sequencing; Technical variability [4] | Neural populations (NEUN, TUBB3); Maturity stage (DCX vs. MAP2); Astrocytes (GFAP); Oligodendrocytes (MBP) [23] |
| Regional Identity | IF/IHC; scRNA-seq [23] [22] | Regional marker overlap; Protocol-dependent biases; Limited spatial resolution in sequencing [22] | Forebrain (FOXG1); Dorsal telencephalic (PAX6); Ventral (NKX2.1) [23] |
Functional Maturation Assessment Techniques
Evaluating the functional maturity of brain organoids requires multiple complementary approaches, each with inherent methodological constraints:
Each functional assessment method provides unique insights but captures only partial aspects of organoid maturity. The lack of standardized multimodal evaluation frameworks combining these approaches complicates comprehensive functional characterization and cross-study comparisons [23].
Research Reagent Solutions for Organoid Characterization
A standardized toolkit of research reagents is essential for consistent organoid characterization. The table below details essential materials used in comprehensive organoid assessment.
Table 4: Essential Research Reagents for Organoid Characterization
| Reagent Category | Specific Examples | Function in Characterization | Protocol Considerations |
|---|---|---|---|
| Extracellular Matrices | Matrigel; Synthetic hydrogel scaffolds; Decellularized tissue-derived scaffolds [22] | Provides 3D scaffolding supporting neuroepithelial morphogenesis and polarization [22] | Batch-to-batch variability requires validation; Engineered scaffolds offer more defined alternatives [22] |
| Neural Lineage Markers | SOX2 (neural stem cells); PAX6 (radial glia); MAP2 (mature neurons); DCX (immature neurons) [23] [4] | Identifies neural progenitor populations and neuronal maturation stages [4] | Quantification by flow cytometry (FACS) or immunofluorescence; Critical for QC cellular composition [4] |
| Regional Identity Markers | FOXG1 (forebrain); PAX6 (dorsal telencephalon); NKX2.1 (ventral/GE) [23] | Confirms region-specific patterning success in guided protocols [23] [22] | Combinatorial marker signatures more reliable than single markers [23] |
| Functional Assessment Tools | GCaMP calcium indicators; Tetrodotoxin (TTX) channel blocker [23] | Measures neural/glial activity and network functionality [23] | Calcium imaging spatial patterns; MEA recordings of network oscillations [23] |
| Synaptic Markers | Synaptobrevin-2 (SYB2; presynaptic); PSD-95 (postsynaptic) [23] | Evaluates synaptic density and maturation at ultrastructural level [23] | Electron microscopy required for nanoscale validation of synaptic structures [23] |
The limitations in current organoid characterization methods represent a significant bottleneck in the field, impeding reproducibility and translational applications. The development of integrated quality control frameworks that combine morphological metrics like Feret diameter with standardized molecular and functional assessments offers a promising path forward [8] [4]. Future directions must focus on establishing universally accepted maturity benchmarks, optimizing non-destructive real-time monitoring technologies, and creating comprehensive atlases for cross-protocol comparison [24] [23]. By addressing these characterization challenges, the research community can enhance the reliability and predictive validity of cerebral organoid models, ultimately accelerating their utility in drug discovery and personalized medicine approaches for neurological disorders.
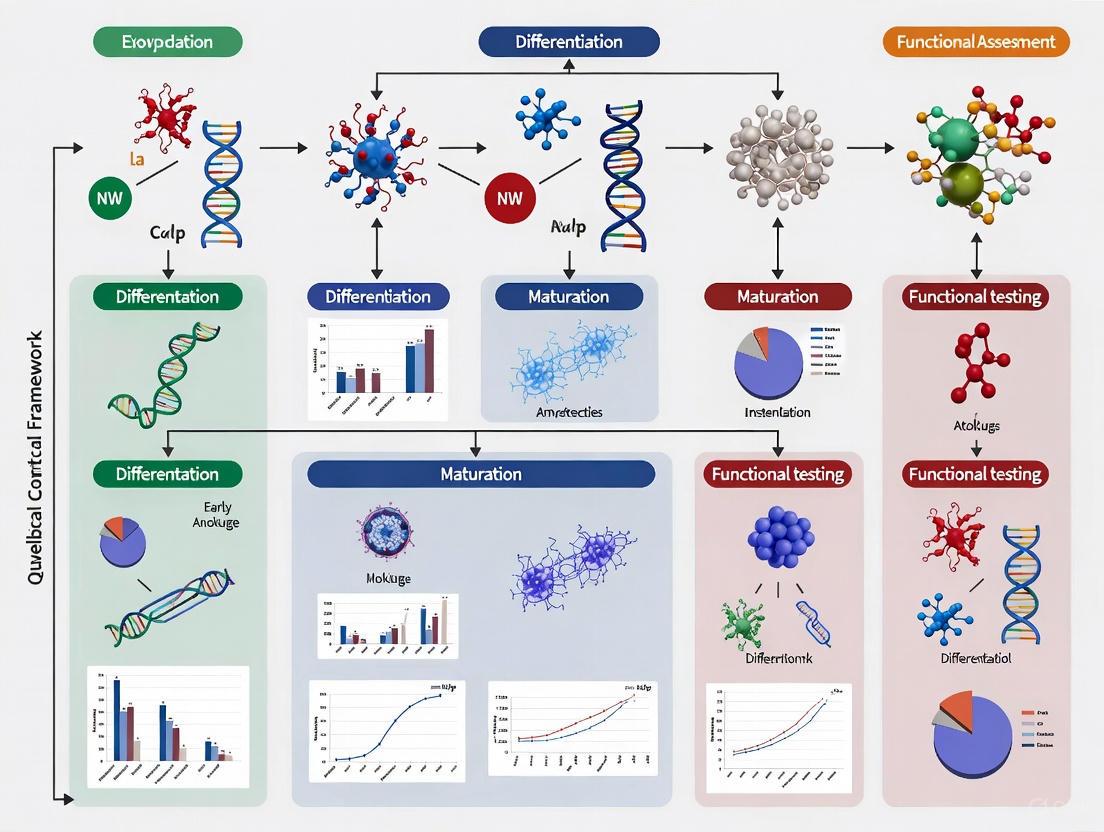
Implementing a Hierarchical QC Framework: From Non-Invasive Screening to Comprehensive Analysis
Cerebral organoids have emerged as transformative tools in neuroscience, providing unprecedented in vitro models for studying human brain development, neurological diseases, and neurotoxicology. However, their potential has been constrained by significant challenges in reproducibility and quality control, with variations in morphology, size, cellular composition, and structural organization leading to inconsistent experimental outcomes [25] [8]. To address this critical bottleneck, researchers have developed a standardized, five-pillar quality control (QC) framework specifically for assessing 60-day cortical organoids [25]. This systematic scoring methodology enables robust, objective classification of organoid quality, enhancing reliability for both academic research and industrial applications like drug screening and toxicology testing [25] [8].
The Essential Five-Pillar Scoring Framework
The QC framework evaluates cerebral organoids against five critical criteria, each subdivided into specific indices scored from 0 (low quality) to 5 (high quality) [25]. The system employs a hierarchical approach, prioritizing non-invasive assessments initially and reserving more in-depth analyses for organoids that pass preliminary thresholds [25] [8].
Table 1: The Five-Pillar Scoring System for Organoid Assessment
| Pillar | Key Assessment Parameters | Evaluation Methods | Quality Thresholds |
|---|---|---|---|
| Morphology | Compactness, border integrity, absence of cysts or necrotic cores [25] | Brightfield imaging, visual inspection [25] | Minimum score required; excludes organoids with large fluid-filled cysts or irregular shapes [25] [4] |
| Size & Growth Profile | Diameter (e.g., Feret diameter), growth dynamics, developmental appropriateness [25] [4] | Brightfield imaging, diameter measurement (e.g., ≥3050 µm potentially indicates lower quality) [4] | Specific size range for 60-day cortical organoids; tracks growth profile over time [25] |
| Cellular Composition | Presence/ratio of expected neural populations: neural progenitors (SOX2, PAX6), neurons (MAP2, βIII-tubulin), astrocytes (GFAP, S100β) [25] [23] [26] | Immunofluorescence, immunohistochemistry, flow cytometry [25] [23] | Expected proportions of cell types for developmental stage; minimal unintended differentiation (e.g., mesenchymal cells) [25] [4] |
| Cytoarchitectural Organization | Cortical lamination (SATB2, TBR1, CTIP2), ventricular-like structures, rosette formation, synaptic density (PSD-95, SYB2) [25] [23] | Immunofluorescence, confocal microscopy, electron microscopy [25] [23] | Well-defined organized structures; specific layered organization for cortical models [25] [23] |
| Cytotoxicity | Cell viability, DNA damage, apoptosis markers, necrotic core presence [25] | Viability assays, lactate dehydrogenase (LDH) release, caspase activation assays [25] | Low cytotoxicity levels; minimal cell death [25] |
The framework is designed for two primary applications [25]:
- Initial QC (Pre-study): Uses only non-invasive Criteria A (Morphology) and B (Size/Growth) to screen organoid eligibility before study inclusion.
- Final QC (Post-study): Employs all five criteria for comprehensive analysis after experiment completion.
Experimental Validation & Comparative Performance
Validation Using Hydrogen Peroxide Exposure
To validate the scoring system's discriminative power, researchers exposed 60-day cortical organoids to graded doses of hydrogen peroxide (H₂O₂), a chemical known to induce oxidative stress and cell death [25] [8]. Organoids were first selected using the Initial QC, then exposed to H₂O₂ followed by a one-week recovery period. Subsequent Final QC evaluation demonstrated the system's robustness in accurately discriminating organoid quality across the induced quality spectrum [25]. This controlled validation confirmed the framework's sensitivity to quality variations and its reliability for assessing experimental outcomes.
Comparative Analysis with Alternative Assessment Methods
The five-pillar system addresses significant limitations of other common organoid assessment approaches.
Table 2: Comparison of Organoid Assessment Methodologies
| Assessment Method | Key Advantages | Major Limitations | Role in QC Framework |
|---|---|---|---|
| Traditional Morphology-Only Assessment | Simple, non-invasive, low-cost [25] | Subjective, qualitative, insufficient for comprehensive quality determination [25] | Forms initial screening pillar (A); requires supplementation with other criteria [25] |
| Molecular Profiling (Transcriptomics/Proteomics) | Detailed cellular composition, pathway analysis, highly quantitative [27] | Destructive, expensive, complex data analysis, difficult for routine use [27] | Validates cellular composition; used for framework development rather than routine QC [25] [27] |
| Electrophysiological Functional Assessment | Measures neuronal activity, network functionality [23] [26] | Requires specialized equipment, technically challenging, may not reflect overall structural quality [23] [26] | Not a core pillar in basic QC; used for specialized functional studies beyond quality assessment [25] |
| Five-Pillar Integrated Framework | Standardized, hierarchical, combines multiple dimensions, objective scoring [25] [8] | More complex than single-method assessments, requires multiple assays | Comprehensive quality assessment for reproducibility and industrial application [25] [8] |
Detailed Methodologies for Key Assessment Protocols
Morphological and Size Assessment Protocol
Workflow:
- Image Acquisition: Capture high-resolution brightfield images of individual organoids daily or at standardized time points [25].
- Parameter Measurement: Use image analysis software (e.g., ImageJ) to quantify:
- Scoring: Assign scores (0-5) for each parameter based on predefined thresholds specific to the organoid type and developmental stage [25].
Organoid Morphology Assessment Workflow
Immunohistochemical Analysis for Cellular Composition
Protocol Details:
- Fixation: Use 4% paraformaldehyde for 15-60 minutes depending on organoid size [25] [23].
- Sectioning: Embed in OCT compound, cryosection at 10-20µm thickness [4].
- Staining: Standard immunofluorescence protocols with antigen retrieval if needed [23].
- Imaging: Confocal microscopy for three-dimensional structural analysis [23].
- Key Markers:
- Quantification: Use image analysis software to determine cell type proportions and spatial distribution [25].
Signaling Pathways in Cortical Patterning
The successful generation of high-quality cortical organoids requires precise manipulation of key developmental signaling pathways during differentiation [26].
Signaling Pathways for Brain Region Specification
Protocol Application:
- Dorsal Forebrain Organoids: Combine dual SMAD inhibition with Wnt and SHH pathway inhibition to achieve anterior dorsal identity [26].
- Ventral Forebrain Organoids: Apply dual SMAD inhibition, Wnt inhibition, and SHH pathway activation using agonists like SAG or purmorphamine [26].
- Unguided Whole-Brain Organoids: Rely on self-organization with minimal patterning factors, often resulting in more heterogeneous regional identities [26].
Essential Research Reagent Solutions
Successful implementation of the five-pillar QC framework requires specific research reagents and materials for proper organoid generation, maintenance, and assessment.
Table 3: Essential Research Reagents for Organoid QC
| Reagent Category | Specific Examples | Function in QC Process | Application Notes |
|---|---|---|---|
| Patterning Factors | SMAD inhibitors (LDN-193189, SB431542), Wnt inhibitors (IWR-1, IWP-2), SHH modulators (SAG, purmorphamine, cyclopamine) [26] | Direct regional specification during organoid differentiation [26] | Critical for generating region-specific organoids; concentrations and timing vary by protocol [26] |
| Extracellular Matrix | Matrigel, Basement Membrane Extract, synthetic hydrogels [25] [21] | Provides 3D structural support, enhances cell-cell interactions, improves viability [25] [21] | Matrigel embedding common for unguided protocols; concentration affects organoid structure and nutrient diffusion [25] |
| Cell Line Sources | H9 (WA09), H1 (WA01) hESC lines; IMR90, Kucg2 iPSC lines [4] | Starting material for organoid generation; influences inherent variability [4] | Multiple cell lines recommended for robust findings; pluripotency validation (e.g., TRA-1-60 staining >90%) essential [4] |
| Immunostaining Markers | SOX2, PAX6 (progenitors); MAP2, TBR1, SATB2 (neurons); GFAP, S100β (astrocytes) [23] [26] | Cellular composition and cytoarchitectural assessment in Pillars C and D [25] [23] | Antibody validation critical; 3D staining requires longer incubation times and potential antigen retrieval [23] |
| Viability/Cytotoxicity Assays | Lactate dehydrogenase (LDH) assay, Live/Dead staining (calcein-AM/ethidium homodimer), caspase assays [25] | Cytotoxicity evaluation for Pillar E [25] | Normalize to organoid size/protein content; multiple time points recommended for dynamic assessment [25] |
The implementation of a standardized, five-pillar scoring system represents a significant advancement in cerebral organoid quality assessment, directly addressing the critical reproducibility challenges that have limited their broader adoption [25] [8]. By integrating hierarchical evaluation of morphology, size, cellular composition, cytoarchitectural organization, and cytotoxicity, this framework provides researchers with a comprehensive, objective methodology for organoid qualification [25].
The validation of this system through controlled hydrogen peroxide exposure demonstrates its robust discriminative capacity across quality gradients [25]. Furthermore, the structured approach—separating initial non-invasive screening from comprehensive final assessment—offers practical efficiency while maintaining analytical rigor [25]. This balanced methodology makes the framework particularly valuable for industrial applications requiring scalability, such as high-throughput drug screening and toxicology testing, where consistency and reliability are paramount [25] [8].
As the field progresses toward more complex organoid models, including those with integrated vascularization and multiple brain region identities, the adaptation and expansion of such QC frameworks will be essential [23] [21]. The five-pillar system provides a foundational approach that can be modified for specific organoid types, developmental stages, and research applications, promising to enhance data reliability and cross-study comparability in this rapidly evolving field [25] [28].
In the evolving field of 3D cell culture, cerebral organoids have emerged as groundbreaking models for neuroscience research, offering unprecedented insights into human brain development, disease modeling, and drug screening [2] [8]. However, the significant potential of these complex in vitro models is often hampered by challenges related to their quality and reproducibility. Variability in morphology, size, cellular composition, and cytoarchitectural organization across batches compromises experimental reliability and consistency, particularly in disease modeling and neurotoxicity testing [2]. To address these challenges, researchers have developed standardized quality control (QC) frameworks that implement hierarchical evaluation systems, beginning with non-invasive morphological and size-based assessments as an initial screening step [2] [8].
This Initial QC approach enables researchers to efficiently identify and exclude low-quality organoids before committing valuable resources to more extensive, invasive analyses [2]. By leveraging simple, reproducible, and cost-effective morphological evaluations, scientists can significantly enhance the reliability of their experimental results while minimizing unnecessary expenditure of time and reagents. The implementation of such standardized QC methodologies is particularly valuable for drug development professionals seeking to incorporate organoid-based models into preclinical screening pipelines, where consistency and predictability are paramount [5] [29].
The QC Framework: A Hierarchical Approach to Organoid Assessment
The quality control framework for cerebral organoids employs a strategic hierarchical approach that prioritizes efficiency and resource allocation [2]. This system is structured to maximize information gain while minimizing unnecessary invasive procedures, organized into two distinct phases:
Initial QC (Pre-study): This first-line assessment relies exclusively on non-invasive criteria—morphology and size/growth profile—to determine organoid eligibility before initiating studies [2]. This phase serves as a gatekeeping step, allowing researchers to quickly identify and exclude organoids with obvious quality issues without consuming limited resources.
Final QC (Post-study): After initial screening, organoids that pass the Initial QC undergo comprehensive analysis using all five QC criteria, including invasive assessments of cellular composition, cytoarchitectural organization, and cytotoxicity [2]. This phased approach ensures that more valuable resources are reserved only for organoids that meet basic quality thresholds.
The scoring methodology employs a standardized system where organoids are evaluated on a scale of 0-5 for each parameter, with minimum thresholds established for progression to subsequent evaluation stages [2]. Organoids failing to meet these thresholds at any stage are excluded from further analysis, streamlining the QC process and enhancing overall experimental efficiency.
Key Advantages of the Hierarchical QC Approach
- Resource Optimization: By reserving invasive, costly analyses for only high-quality organoids, researchers significantly reduce reagent costs and labor requirements [2].
- Early Decision-Making: The non-invasive nature of Initial QC enables early identification of problematic batches, allowing researchers to make timely decisions about continuing or aborting experiments [2].
- Standardization: The structured scoring system minimizes observer bias and introduces much-needed standardization to organoid research [2] [8].
- Scalability: The efficient Initial QC process makes large-scale organoid studies more feasible, supporting industrial applications in drug discovery and toxicology testing [2] [5].
The following diagram illustrates the decision-making workflow within this hierarchical QC framework:
Hierarchical QC Workflow: The sequential evaluation process for cerebral organoids, beginning with non-invasive assessments.
Core Principles of Non-Invasive Initial QC
Morphological Assessment Criteria
Morphological evaluation serves as the primary non-invasive quality indicator in the Initial QC framework [2]. This assessment focuses on visual characteristics that correlate with overall organoid health and developmental potential:
- Compactness and Structural Integrity: High-quality organoids display dense overall structure with well-defined borders, indicating proper cell-cell interactions and structural organization [2]. Poorly compact organoids that appear loose or disorganized typically indicate suboptimal differentiation or health issues.
- Border Integrity and Surface Characteristics: The presence of irregular borders, protruding cystic structures, or excessive surface irregularities suggests quality concerns [2]. These features often correlate with improper development or degenerative processes.
- Absence of Degeneration Signs: Quality organoids should not show signs of cellular debris, excessive cell shedding, or necrotic areas, which would indicate health problems or culture stress [2].
Size and Growth Profile Evaluation
The second component of Initial QC involves monitoring size parameters and growth dynamics over time [2]. This quantitative assessment provides objective measures of organoid development:
- Size Consistency: Organoids should fall within expected diameter ranges for their developmental stage (e.g., 60-day cortical organoids) [2]. Significant deviations from expected size ranges may indicate aberrant development.
- Growth Trajectory: The rate of size increase over time should follow expected patterns for the specific organoid type and protocol [2]. Arrested growth or explosive enlargement both signal potential quality issues.
- Batch Consistency: Within a batch, organoids should demonstrate relatively uniform size distribution, with outliers potentially indicating quality concerns [2].
Experimental Validation: Establishing QC Reliability
Validation Methodology
To validate the Initial QC framework, researchers conducted controlled experiments using 60-day cortical organoids exposed to graded doses of hydrogen peroxide (H₂O₂) to induce controlled variations in quality [2]. This approach generated organoids with predictable quality differences that could be used to test the discrimination power of the QC system.
The experimental protocol followed these key steps:
- Organoid Culture: Cerebral cortical organoids were generated from pluripotent stem cells and maintained for 60 days, a critical intermediate stage characterized by the presence of neural progenitors, neurons, and astrocytes [2].
- Quality Perturbation: Organoids were exposed to increasing concentrations of H₂O₂, a chemical known to cause oxidative stress-induced cellular death, to create a spectrum of quality levels [2].
- QC Application: Both Initial QC (morphology and size) and Final QC (comprehensive analysis) were applied to all organoids following treatment and a recovery period [2].
- System Validation: The accuracy of Initial QC predictions was verified by comparing them with Final QC results, confirming that non-invasive assessments could reliably identify quality issues [2].
Key Experimental Findings
The validation study demonstrated that the Initial QC framework based on morphological and size criteria successfully discriminated between organoids of different quality levels [2]. The non-invasive assessments showed strong correlation with more comprehensive invasive analyses, supporting their use as reliable screening tools. Specifically, organoids identified as low-quality through Initial QC consistently showed poor performance in subsequent cellular composition, cytoarchitectural organization, and cytotoxicity assessments [2].
Table 1: QC Scoring Criteria for Cerebral Organoids
| QC Criterion | Assessment Method | High-Quality Indicators (Score 4-5) | Low-Quality Indicators (Score 0-1) |
|---|---|---|---|
| Morphology | Bright-field microscopy | Dense structure, well-defined borders, no cysts | Poor compactness, irregular borders, cystic structures |
| Size & Growth | Diameter measurement, time-series tracking | Appropriate size for age, consistent growth | Size outliers, arrested growth, excessive variation |
| Cellular Composition | Immunohistochemistry, flow cytometry | Expected neural populations, proper ratios | Missing cell types, aberrant composition |
| Cytoarchitectural Organization | Histology, immunostaining | Organized layers, rosette structures | Disorganized structures, missing features |
| Cytotoxicity Cell viability assays, DNA damage markers | Low cell death, minimal DNA damage | High cytotoxicity, significant DNA damage |
Comparative Analysis: Initial QC Versus Alternative Methods
When evaluating quality assessment methods for cerebral organoids, researchers have multiple approaches available, each with distinct advantages and limitations. The following comparison highlights how Non-Invasive Initial QC compares to other common strategies:
Table 2: Method Comparison for Organoid Quality Assessment
| Assessment Method | Key Features | Advantages | Disadvantages |
|---|---|---|---|
| Non-Invasive Initial QC | Hierarchical approach starting with morphology/size | Preserves samples, cost-effective, rapid, scalable | Limited depth of information, may miss subtle defects |
| Comprehensive QC | Full analysis of all 5 criteria simultaneously | Maximum information gain, detailed quality profile | Resource-intensive, requires sample destruction |
| Molecular Analysis | qPCR, RNA-Seq, flow cytometry | Detailed molecular information, precise characterization | Invasive, destructive, requires specialized expertise |
| Functional Assessment | Electrophysiology, calcium imaging | Direct functional data, physiological relevance | Technically challenging, low throughput, specialized equipment |
| AI-Powered Morphology | Machine learning analysis of images | High predictability, automated, quantitative | Requires training data, computational resources |
Integration with Advanced Technologies
Recent advances in imaging and computational analysis have enhanced the capabilities of non-invasive QC approaches. Machine learning algorithms applied to bright-field images have demonstrated remarkable ability to predict molecular markers and functional properties based solely on morphological features [30] [31]. For example, studies have successfully predicted pluripotency markers like OCT4 and NANOG from bright-field images of stem cells using unsupervised and semi-supervised learning models [30]. Similarly, morphological heterogeneity descriptors have enabled early prediction of T-cell proliferation inhibitory potency and growth rates in mesenchymal stem cells [31]. These technological enhancements are progressively strengthening the predictive power of non-invasive Initial QC methods.
Implementation Protocols: Establishing Initial QC in the Laboratory
Standard Operating Procedure for Initial QC
Implementing a robust Initial QC process requires standardized protocols to ensure consistency and reproducibility:
Imaging Protocol:
- Acquire bright-field images of organoids using standardized magnification (recommended 20× high numerical aperture objectives) [30].
- Capture multiple focal planes (z-stacks) to ensure comprehensive morphological assessment [30].
- Maintain consistent lighting and exposure settings across all imaging sessions (e.g., 50-ms exposure for bright-field) [30].
- Establish regular imaging intervals (e.g., daily or every 48 hours) to track growth dynamics [2].
Morphological Scoring Protocol:
- Evaluate each organoid against standardized morphological criteria using a 0-5 scoring system [2].
- Assess compactness, border integrity, and absence of cysts or surface irregularities [2].
- Establish threshold scores for inclusion (typically minimum score of 3-4 per criterion) [2].
- Implement blind scoring or automated analysis to minimize observer bias [2].
Size Assessment Protocol:
Quality Thresholds and Decision Points
Establishing clear quality thresholds is essential for consistent application of Initial QC:
- Passing Threshold: Organoids must achieve minimum scores in both morphology and size criteria to proceed to further studies [2]. The specific thresholds may vary based on organoid type and research application.
- Borderline Cases: Organoids scoring near thresholds may require additional evaluation or be allocated to less critical experiments [2].
- Batch Assessment: If a high percentage of organoids within a batch fail Initial QC, the entire batch may need to be excluded, indicating potential protocol issues [2].
Essential Research Tools for Implementation
Successful implementation of Initial QC requires specific reagents and equipment designed to support standardized assessment:
Table 3: Research Reagent Solutions for Initial QC Implementation
| Item | Function | Application Notes |
|---|---|---|
| Bright-field Microscopy Systems | Non-invasive imaging for morphological assessment | Systems with high numerical aperture objectives (20×) and z-stack capability recommended [30] |
| Image Analysis Software | Quantitative assessment of size and morphology | Capable of batch processing, diameter measurement, and morphological characterization [2] |
| Standardized Culture Matrices | Consistent 3D support for organoid development | Matrigel or synthetic alternatives; batch-to-batch consistency is critical [32] |
| Quality Control Scoring Templates | Standardized evaluation against criteria | Visual guides with reference images for each score level (0-5) [2] |
| Time-Lapse Imaging Systems | Continuous monitoring of growth dynamics | Systems with environmental control to maintain culture conditions during imaging [30] |
The implementation of standardized Initial QC focusing on non-invasive morphological and size-based assessments represents a significant advancement in cerebral organoid research. By providing a structured, hierarchical approach to quality evaluation, this framework addresses one of the most pressing challenges in the field: reproducibility and reliability [2] [8] [5]. The validated methodology enables researchers to make informed decisions about organoid quality early in the experimental process, conserving valuable resources while enhancing the consistency of research outcomes.
As organoid technology continues to evolve and find broader applications in disease modeling, drug screening, and personalized medicine, standardized QC frameworks will play an increasingly critical role in ensuring data reliability and translational relevance [5] [29]. The integration of these QC approaches with emerging technologies such as artificial intelligence, automated image analysis, and machine learning will further enhance their predictive power and accessibility [30] [29]. By adopting these standardized methodologies, the research community can accelerate the translation of organoid technology from basic research to clinical and industrial applications, ultimately advancing our understanding of brain development and neurological disorders.
Cerebral cortical organoids have emerged as transformative tools in neuroscience, providing unprecedented in vitro models that recapitulate aspects of human brain development and dysfunction [2] [33]. However, their significant potential is hampered by challenges related to quality and reproducibility, which limit reliability in disease modeling, drug screening, and neurotoxicity testing [2]. While initial quality control (QC) can filter organoids using non-invasive morphological assessments, the final QC—encompassing detailed analysis of cellular composition and cytoarchitectural organization—is indispensable for validating model fidelity and experimental conclusions [2]. This in-depth analysis moves beyond superficial assessments to quantify the fundamental biological structures and cell populations that determine whether an organoid truly mimics the developing human cortex. This guide provides a structured framework for conducting this essential final QC evaluation, comparing methodological approaches and establishing benchmarks for researchers requiring rigorous organoid characterization.
Core Quality Control Criteria: A Comparative Framework
A comprehensive QC framework for cerebral organoids integrates multiple assessment dimensions. The table below compares the five core criteria, detailing their role in QC and key assessment parameters.
Table 1: Core QC Criteria for Cerebral Cortical Organoids
| QC Criterion | Role in Quality Assessment | Key Parameters & Markers |
|---|---|---|
| Morphology | Initial, non-invasive screening of overall health and structural integrity [2]. | Compactness, border integrity, absence of large cysts [2] [4]. |
| Size & Growth Profile | Ensures proper developmental dynamics and identifies growth abnormalities [2]. | Diameter (e.g., Feret diameter), volume, growth consistency [2] [4]. |
| Cellular Composition | Verifies presence and proportion of expected neural and non-neural cell types [2]. | Neural progenitors (SOX2, PAX6), neurons (MAP2, NeuN), astrocytes (GFAP, S100β), undesired cells [2] [23] [4]. |
| Cytoarchitectural Organization | Assesses structural integrity and layered organization mimicking cortical development [2] [23]. | Cortical lamination (TBR1, CTIP2, SATB2), ventricular zone-like structures, synaptic density (PSD-95, SYB2) [23]. |
| Cytotoxicity | Measures cell health and death, indicating culture stress or toxicity [2]. | DNA damage, cell viability assays, necrotic core presence [2]. |
The final QC process is hierarchically structured, beginning with simpler, non-destructive assays and progressing to complex, multi-parameter analyses that often require organoid fixation or dissociation [2]. This staged approach efficiently allocates resources while ensuring only organoids passing basic benchmarks undergo labor-intensive cellular and cytoarchitectural analysis.
Experimental Protocols for Cellular Composition Analysis
Determining the cellular makeup of a cortical organoid is crucial for confirming its relevance to the in vivo brain region and identifying contamination by off-target cell types. The following protocols outline standard methodologies for this analysis.
Immunohistochemistry (IHC) and Immunofluorescence (IF)
Detailed Protocol:
- Fixation: Immerse 60-day cortical organoids in 4% paraformaldehyde (PFA) for 24-48 hours at 4°C to ensure complete penetration [34].
- Cryoprotection: Transfer organoids to a sucrose gradient (15% followed by 30% sucrose solution) until they sink, then embed in Optimal Cutting Temperature (O.C.T.) compound [4].
- Sectioning: Cryosection organoids into 10-20 μm thick sections using a cryostat and mount on glass slides [4].
- Staining: Perform antigen retrieval if required. Block sections with a buffer containing a detergent (e.g., 0.1-0.3% Triton X-100) and 5-10% normal serum. Incubate with primary antibodies (see Table 2 for key antibodies) overnight at 4°C, followed by appropriate fluorescently conjugated secondary antibodies [4].
- Imaging and Analysis: Acquire high-resolution images using confocal microscopy. Quantify marker-positive cells manually or using image analysis software (e.g., ImageJ) across multiple organoids and sections to determine cellular proportions [2] [4].
Flow Cytometry
Detailed Protocol:
- Organoid Dissociation: Mechanically dissociate PFA-fixed organoids or gently dissociate live organoids using papain or accutase enzymes to create a single-cell suspension [4].
- Staining: For intracellular markers (e.g., transcription factors), permeabilize cells using a permeabilization buffer. Incubate with fluorescently conjugated antibodies against target proteins (e.g., anti-PAX6 for cortical progenitors) [4].
- Data Acquisition and Analysis: Analyze the cell suspension using a flow cytometer. Use unstained and isotype controls to set gating parameters and determine the percentage of positive cells for each marker within the population [4].
Key Reagent Solutions for Cellular Analysis
Table 2: Essential Research Reagents for Cellular Composition Analysis
| Reagent / Assay | Function in QC | Example Application |
|---|---|---|
| Anti-SOX2 / PAX6 Antibodies | Labels neural progenitor cells to quantify the progenitor pool and identify ventricular-like structures [4]. | IHC/IF staining on organoid sections [4]. |
| Anti-MAP2 / NeuN Antibodies | Identifies mature and immature neurons to assess neuronal differentiation and maturation stage [23]. | IHC/IF staining; flow cytometry [4]. |
| Anti-GFAP / S100β Antibodies | Marks astrocytes to evaluate the presence and distribution of key glial cells [23]. | IHC/IF staining to visualize astrocyte networks [23]. |
| Anti-TBR1 / CTIP2 / SATB2 Antibodies | Critical for assessing cortical layer identity and organization within the cytoarchitecture [23]. | IHC/IF staining to visualize distinct cortical layers [23]. |
| Papain Dissociation Kit | Gently dissociates live 3D organoids into single-cell suspensions for downstream flow cytometry analysis [35]. | Preparing cells for flow cytometry analysis of surface and intracellular markers [35]. |
| Single-Cell RNA Sequencing (scRNA-seq) | Provides a high-resolution, unbiased profile of all cell types present, identifying both desired neural lineages and undesired off-target populations [23] [4]. | Comprehensive cellular deconvolution to quantify heterogeneity and identify confounders like mesenchymal cells [4]. |
Experimental Protocols for Cytoarchitectural Organization Analysis
The presence of specific cell types alone is insufficient; their spatial organization into structures resembling the developing cortex is a hallmark of a high-quality organoid.
Multimarker Immunofluorescence and Confocal Analysis
Detailed Protocol:
- Sample Preparation: Follow the IHC/IF protocol in Section 3.1 to obtain stained organoid sections.
- Multiplexed Staining: Use a combination of antibodies against layer-specific neuronal markers simultaneously. A standard panel includes:
- High-Resolution Imaging: Use confocal microscopy to acquire z-stacks through the entire thickness of the organoid section. This allows for 3D reconstruction of the cytoarchitecture.
- Analysis: Assess the presence and relative organization of distinct layers. High-quality organoids will show a structured arrangement, often with TBR1+ and CTIP2+ cells located in deeper regions and SATB2+ cells in more superficial areas, mimicking the in vivo cortical plate [23].
Synaptic Density Analysis
Detailed Protocol:
- Staining: Co-stain organoid sections with antibodies for a presynaptic protein, such as Synaptobrevin-2 (SYB2), and a postsynaptic protein, such as PSD-95 [23].
- Imaging and Quantification: Acquire high-magnification confocal images. The density and co-localization of PSD-95 and SYB2 puncta can be quantified using image analysis software to provide an index of synaptic maturity and density [23].
The following workflow diagram illustrates the hierarchical steps for the final QC process, from initial sample selection to comprehensive analysis and final classification.
Final QC Workflow for Cortical Organoids
Quantitative Benchmarking and Data Integration
For the final QC to be objective, qualitative observations must be translated into quantitative scores. The framework proposed by Castiglione et al. uses a scoring system from 0 (low quality) to 5 (high quality) for each criterion [2]. The tables below summarize key quantitative benchmarks for cellular composition and cytoarchitecture.
Table 3: Benchmarking Cellular Composition in 60-Day Cortical Organoids
| Cell Population | Target Presence | Key Quality Indicators | Confounding Factors |
|---|---|---|---|
| Neural Progenitors (SOX2+, PAX6+) | High proportion in ventricular-like zones [4]. | Well-defined rosette structures; active neurogenesis [2] [4]. | Disorganized distribution; absence of clear structures [4]. |
| Neurons (MAP2+, NeuN+) | High density of neurons across the organoid [4]. | Presence of both deep (CTIP2+) and upper (SATB2+) layer neurons [23]. | Homogeneous, disorganized neuronal distribution. |
| Astrocytes (GFAP+, S100β+) | Emerging population at 60 days [2]. | Presence of astrocytic networks [23]. | General lack of glial cells indicates immaturity. |
| Mesenchymal Cells | Minimal to absent [4]. | Low Feret diameter (<~3050 μm) correlates with low mesenchymal content [4]. | High mesenchymal cell proportion indicates off-target differentiation and poor quality [4]. |
Table 4: Benchmarking Cytoarchitectural Organization in 60-Day Cortical Organoids
| Structural Feature | High-Quality Benchmark | Assessment Method |
|---|---|---|
| Cortical Lamination | Emergence of distinct neuronal layers (TBR1+ deep layers, SATB2+ upper layers) with spatial segregation [23]. | Multiplex IHC/IF with confocal microscopy and 3D analysis [23]. |
| Ventricular Zone-like Structures | Multiple, well-formed SOX2+ rosettes surrounding clear lumens [4]. | IHC/IF for SOX2/PAX6 and brightfield microscopy [4]. |
| Synaptic Density | Presence of co-localized pre- (SYB2) and post-synaptic (PSD-95) puncta [23]. | High-resolution IF and puncta quantification [23]. |
| Structural Integrity | Absence of large, fluid-filled cysts and significant necrotic cores [2] [4]. | Brightfield microscopy, H&E staining, and cytotoxicity assays [2]. |
The implementation of a rigorous final QC for cellular composition and cytoarchitecture is not merely a endpoint check but a critical feedback mechanism that elevates the entire field of organoid research. By adopting the standardized protocols and benchmarks outlined in this guide, researchers can move beyond subjective morphological assessments to generate quantifiable, reproducible quality data. This enables reliable discrimination between high- and low-quality organoids, as demonstrated in validation studies using controlled stressors like hydrogen peroxide [2]. Furthermore, correlating simple, non-invasive metrics like the Feret diameter with deeper molecular and cellular data creates powerful tools for pre-screening [4]. Ultimately, the consistent application of these in-depth analyses is foundational for building reproducible, high-fidelity models that can accelerate drug discovery, advance our understanding of neurodevelopmental diseases, and fulfill the transformative potential of cerebral organoid technology.
Cerebral cortical organoids have emerged as revolutionary tools in neuroscience, providing complex three-dimensional in vitro models that mimic human brain development [2]. These self-organizing structures recapitulate aspects of the cellular composition, architecture, and functional characteristics of the developing human brain, offering unprecedented opportunities for studying neurodevelopmental processes, disease mechanisms, and neurotoxicity [36]. However, significant challenges related to reproducibility, quality control, and standardization have hindered their broader adoption and reliability, particularly in pharmaceutical testing and disease modeling applications [2] [4].
The intrinsic variability of cerebral organoids presents a major obstacle for consistent research outcomes. Discrepancies in morphology, size, cellular composition, and cytoarchitectural organization limit their applications, especially in contexts requiring high reproducibility such as drug screening and neurotoxicity testing [2]. This variability stems from the stochastic nature of stem cell differentiation and spontaneous self-organization processes, leading to substantial heterogeneity even within organoids generated from the same cell line under identical conditions [4]. Consequently, researchers face significant challenges in objectively selecting organoids for experiments and comparing results across different laboratories [4].
To address these challenges, recent research has focused on developing standardized quality assessment frameworks. This article examines and compares two prominent approaches: a comprehensive hierarchical quality control system for 60-day cortical organoids and a simplified methodology based on a single morphological parameter. We present practical workflows, defined thresholds, and experimental protocols to guide researchers in implementing these quality control systems, ultimately enhancing the reliability and comparability of cerebral organoid research.
Comparative Analysis of Quality Control Frameworks
Table 1: Comparison of Cerebral Organoid Quality Control Frameworks
| Feature | Comprehensive QC Framework [2] | Feret Diameter Approach [4] |
|---|---|---|
| Target Organoid | 60-day cortical organoids | 30-day unguided brain organoids |
| Number of Parameters | 5 primary criteria with multiple indices | Single parameter (Feret diameter) |
| Assessment Level | Hierarchical: from non-invasive to in-depth | Single-step measurement |
| Key Thresholds | Minimum scores for each criterion; composite thresholds | 3050 μm Feret diameter |
| Implementation Complexity | High (requires multiple assays) | Low (brightfield imaging only) |
| Validation Method | Hydrogen peroxide exposure inducing quality gradient | Expert evaluation correlation |
| Diagnostic Performance | Robust classification across multiple parameters | Youden index: 0.68; PPV: 94.4%; NPV: 69.4% |
| Best Application Context | High-stakes research requiring comprehensive characterization | High-throughput screening and rapid assessment |
Table 2: Key Research Reagent Solutions for Cerebral Organoid Quality Control
| Reagent/Category | Specific Examples | Function in QC Framework |
|---|---|---|
| Extracellular Matrix | Corning Matrigel [37] | Supports neuroepithelial bud formation and polarization; critical for proper morphological development |
| Neural Induction Media Components | Dual SMAD inhibitors (BMP inhibition + TGF-β inhibition) [38] | Directs neuroectodermal differentiation; ensures correct lineage specification |
| Patterning Factors | WNT inhibitors, SHH activators, FGF, BMP7, retinoic acid [38] | Generates region-specific organoids; enables assembloid formation for circuit studies |
| Quality Stress Agent | Hydrogen peroxide (H₂O₂) [2] | Induces controlled oxidative stress for QC system validation |
| Immunostaining Markers | SOX2 (neural stem cells), MAP2 (mature neurons), PAX6 (CNS progenitors) [4] | Evaluates cellular composition and cytoarchitectural organization |
| Live Imaging Labels | Endogenous tags: ACTB (actin), TUBA1B (tubulin), HIST1H2BJ (nucleus) [37] | Enables tracking of tissue morphodynamics and cellular behaviors |
The Comprehensive Hierarchical QC Framework: Structure and Workflow
The comprehensive quality control framework for 60-day cortical organoids employs a hierarchical scoring methodology based on five critical criteria, each with specific indices and scoring thresholds [2]. This system is designed to streamline the assessment process by prioritizing non-invasive evaluations before proceeding to more complex, destructive analyses.
The Five Core Quality Criteria
The framework evaluates organoids across five essential domains [2]:
- Morphology: Assesses overall structure, including compactness, border definition, and presence of undesirable features such as suboptimal cystic cavities or necrotic cores.
- Size and Growth Profile: Tracks dimensional parameters and growth dynamics over time.
- Cellular Composition: Evaluates the presence and proportion of key cell types, including neural progenitors, neurons, and astrocytes.
- Cytoarchitectural Organization: Examines the formation and organization of characteristic structures such as neural rosettes and ventricular-like structures.
- Cytotoxicity Level: Measures cell death and stress markers.
Each criterion is subdivided into specific indices scored from 0 (low quality) to 5 (high quality), with defined minimum thresholds that must be met for an organoid to proceed to the next evaluation level [2].
Hierarchical Application Workflow
The quality control process follows a structured, hierarchical pathway that maximizes efficiency by reserving resource-intensive analyses only for organoids that pass initial non-invasive assessments [2].
Diagram 1: Hierarchical QC workflow for cerebral organoids
Experimental Protocol: Validation via Hydrogen Peroxide Exposure
To validate the hierarchical QC framework, researchers implemented a systematic protocol using hydrogen peroxide exposure to generate organoids with controlled quality variations [2]:
Materials:
- 60-day cortical organoids
- Hydrogen peroxide (H₂O₂) solutions at graded concentrations
- Control culture medium
- Recovery culture medium
- Immunostaining reagents (SOX2, MAP2, PAX6 antibodies)
- Viability/cytotoxicity assay kits
Procedure:
- Initial QC Screening: At day 60 of culture, subject all organoids to non-invasive assessment of morphology and size using brightfield imaging.
- Organoid Selection: Select organoids meeting Initial QC thresholds for the validation experiment.
- Quality Perturbation: Expose selected organoids to increasing doses of H₂O₂ (0 μM, 50 μM, 100 μM, 200 μM) for 24 hours to induce a gradient of quality outcomes.
- Recovery Period: Transfer all organoids to recovery medium for one week.
- Final QC Assessment: Perform comprehensive evaluation using all five criteria:
- Morphology and Size: Brightfield imaging and measurement
- Cellular Composition: Flow cytometry and immunostaining for neural markers
- Cytoarchitectural Organization: Immunofluorescence for structural markers
- Cytotoxicity: Cell viability assays and death marker staining
- Scoring and Classification: Apply the scoring system to classify organoids into quality categories based on achieved thresholds.
This systematic validation demonstrated the QC framework's robustness by accurately discriminating organoid qualities across the induced quality spectrum [2].
The Feret Diameter Approach: A Simplified Quality Metric
For researchers requiring a rapid assessment method, a simplified approach based on the Feret diameter (maximal caliper diameter) has been developed and validated for 30-day unguided brain organoids [4].
Experimental Protocol: Feret Diameter Measurement and Validation
Materials:
- 30-day brain organoids
- Brightfield microscope with camera
- Image analysis software (e.g., ImageJ)
- RNA sequencing reagents (for validation studies)
Procedure:
- Image Acquisition: Capture brightfield images of day 30 organoids under standardized magnification and lighting conditions.
- Morphological Parameter Measurement: Using ImageJ or similar software, measure nine morphological parameters:
- Feret diameter
- Area
- Perimeter
- Circularity
- Cysts amount
- Cysts area
- Solidity
- Aspect ratio
- Roundness
- Expert Quality Rating: Have domain experts classify organoids as high or low quality based on established morphological hallmarks:
- High-quality: Spherical shape with neuroepithelial buds growing into Matrigel embedding
- Low-quality: Large fluid-filled cysts, migrating cells, or irregular shape
- Statistical Correlation Analysis: Perform point-biserial correlation analysis between measured parameters and expert ratings.
- Threshold Determination: Apply Youden's J statistics to identify optimal Feret diameter threshold that best discriminates quality classifications.
- Validation: Verify threshold performance through transcriptomic analysis and cellular deconvolution.
This methodology identified 3050 μm as the optimal Feret diameter threshold with a Youden index of 0.68, positive predictive value of 94.4%, and negative predictive value of 69.4% [4].
Biological Basis: Correlation with Mesenchymal Cell Content
The Feret diameter threshold correlates with a critical biological factor affecting organoid quality: the proportion of mesenchymal cells (MC) [4]. Transcriptomic analysis revealed that organoids classified as low-quality by Feret diameter consistently showed higher proportions of MC (0.5% to 74%), representing unintended mesodermal differentiation [4]. High-quality organoids with appropriate Feret diameter consistently displayed lower MC presence, indicating more specific neural differentiation [4].
Signaling Pathways Governing Organoid Development and Quality
Understanding the signaling pathways that guide brain organoid development is essential for proper quality assessment, as disruptions in these pathways often manifest as quality defects.
Diagram 2: Key signaling pathways in brain organoid development
The proper functioning of these signaling pathways directly impacts organoid quality metrics assessed in both QC frameworks. For example, disruptions in WNT signaling can lead to incorrect regional patterning, while inadequate ECM support can impair neuroepithelial formation and lumen expansion [37].
Framework Selection and Application
Choosing between the comprehensive hierarchical framework and the simplified Feret diameter approach depends on research objectives, resources, and context of use:
Comprehensive Hierarchical Framework is preferable for:
- Studies requiring deep characterization of organoid properties
- Regulatory applications and safety assessment
- Disease modeling where specific cellular and architectural features are critical
- Protocol development and optimization
Simplified Feret Diameter Approach is suitable for:
- High-throughput screening applications
- Rapid quality assessment during routine culture
- Initial screening before committing to more extensive analyses
- Studies with limited resources for extensive characterization
Both quality control frameworks represent significant advances in addressing the reproducibility challenges in cerebral organoid research. The comprehensive hierarchical system offers robust, multi-parameter assessment for applications demanding high reliability, while the Feret diameter approach provides an efficient, validated method for rapid quality screening. Implementation of these standardized quality assessment methods will enhance the consistency, reliability, and comparability of cerebral organoid research, ultimately accelerating their application in disease modeling, drug discovery, and neurodevelopmental studies.
Researchers should select the appropriate framework based on their specific needs, considering the trade-offs between comprehensiveness and efficiency. As the field evolves, these quality control standards are expected to undergo further refinement and validation, potentially incorporating additional parameters such as functional electrophysiological measurements and advanced molecular profiling to more comprehensively assess organoid quality and maturity.
Addressing QC Challenges and Integrating Advanced Functional Readouts
Common Organoid Failures and Their Identification via QC Scoring
Cerebral organoids have emerged as transformative tools in neuroscience research, providing unprecedented opportunities to model human brain development, study neurological disorders, and screen therapeutic compounds in a biologically relevant three-dimensional context [2] [8]. However, the significant potential of these models is tempered by substantial challenges related to reproducibility, quality control, and standardization. Variability in morphology, size, cellular composition, and structural organization across organoid batches compromises experimental reliability and hinders broader adoption in both academic and industrial settings [2] [39]. This guide systematically examines common organoid failures and presents standardized quality control frameworks that enable researchers to identify, classify, and address these limitations through quantitative scoring methodologies.
Common Organoid Failures: Types and Causes
Organoid cultures face multiple technical challenges that can lead to experimental failure. Understanding these failure modes is essential for implementing effective quality control measures.
Table 1: Common Organoid Failure Types and Their Primary Characteristics
| Failure Category | Primary Characteristics | Underlying Causes |
|---|---|---|
| Structural Defects | Irregular shape, poor border integrity, cystic formations | Improper matrix formation, stochastic differentiation [2] [4] |
| Size Variability | Significant inter-organoid size differences, growth arrest | Inconsistent initial aggregate formation, nutrient diffusion limitations [40] [4] |
| Aberrant Cellular Composition | Presence of non-neural lineages, incorrect neural subtype ratios | Spontaneous differentiation, protocol-specific biases [3] [4] |
| Cytoarchitectural Disorganization | Lack of ventricular zone-like rosettes, disrupted radial organization | Defective self-organization, signaling pathway imbalances [2] [39] |
| Viability Issues | Necrotic cores, apoptosis, cytotoxicity | Hypoxia, metabolic stress, passaging damage [41] [39] |
Technical Failures in Culture Maintenance
Beyond intrinsic biological variability, technical aspects of organoid culture present significant failure risks, particularly during passaging procedures:
- Matrix Gel Handling: Temperature sensitivity during matrix gel solidification can damage organoids. Recommended approaches include 4°C for 30 minutes or 0°C for 30 minutes for temperature-sensitive organoids, or -20°C for 5 minutes for duration-sensitive cultures [41].
- Centrifugation Damage: Mechanical stress from centrifugation can cause cell membrane rupture and internal structural damage. Horizontal rotor centrifuges at 200g-300g, 4°C, for ≤5 minutes are recommended to minimize damage [41].
- Digestion Problems: Over-digestion with trypsin or other enzymes can increase apoptosis rates and damage cell surface receptors. Digestion at room temperature for 2-3 minutes with specialized organoid passaging digestion medium is advised over traditional trypsin [41].
- Pipetting-Induced Stress: Mechanical damage and shear stress from improper pipetting techniques can cause significant organoid injury. Using sterile Pasteur pipettes instead of standard pipette tips and limiting pipetting to under 30 actions reduces this risk [41].
QC Scoring Frameworks for Failure Identification
Comprehensive QC Framework for Cortical Organoids
A hierarchical quality control framework specifically designed for 60-day cortical organoids provides a standardized approach to identify and classify organoid failures [2] [8]. This system evaluates five critical criteria through a quantitative scoring methodology:
Table 2: QC Scoring Framework for 60-Day Cortical Organoids
| QC Criterion | Assessment Method | High-Quality Indicators | Failure Indicators |
|---|---|---|---|
| Morphology | Brightfield imaging, visual inspection | Spherical shape with neuroepithelial buds, well-defined borders | Cystic cavities, irregular shape, migrating cells [2] [4] |
| Size & Growth Profile | Feret diameter measurement, area calculation | Consistent size within batch (e.g., ~3050μm Feret diameter) | Significant deviation from expected size range [40] [4] |
| Cellular Composition | Immunostaining, flow cytometry, scRNA-seq | Expected neural populations (SOX2+, MAP2+, PAX6+) | Presence of mesenchymal cells, incorrect neural ratios [2] [4] |
| Cytoarchitectural Organization | Immunohistochemistry for layer markers | Ventricular-like structures, polarized rosettes | Disorganized structure, lack of defined zones [2] [39] |
| Cytotoxicity Cell viability assays, DNA damage markers | Low cytotoxicity, minimal DNA damage | Significant cell death, activation of stress pathways [2] [40] |
The scoring system operates on a 0-5 scale for each parameter, with minimum thresholds established for organoid qualification [2]. The framework is applied hierarchically, beginning with non-invasive assessments (morphology and size) to exclude low-quality organoids before proceeding to more resource-intensive cellular and molecular analyses [2] [8].
Feret Diameter as a Key Quality Indicator
Research indicates that the Feret diameter (the longest distance between any two points of the organoid) serves as a particularly reliable single parameter for assessing brain organoid quality [4]. Studies demonstrate that classification using a Feret diameter threshold of 3050μm accurately reflects expert quality evaluation with a positive predictive value of 94.4% and negative predictive value of 69.4% [4]. This measurement correlates with critical quality aspects, as organoids exceeding this diameter threshold typically show higher proportions of unintended mesenchymal cells and reduced neural differentiation capacity [4].
Hierarchical QC Assessment Workflow
Experimental Validation of QC Frameworks
Hydrogen Peroxide-Induced Stress Model
To validate the QC scoring system, researchers exposed 60-day cortical organoids to graded doses of hydrogen peroxide (H₂O₂) to induce controlled quality variations [2]. This experimental approach generated organoids with predictable quality deficits, allowing systematic testing of the scoring framework's ability to discriminate between quality levels. Organoids were first selected using the Initial QC based on non-invasive criteria before H₂O₂ exposure, followed by a one-week recovery period and comprehensive Final QC assessment [2]. The framework successfully distinguished organoid quality across the induced stress spectrum, demonstrating its sensitivity and reliability for quality discrimination [2] [8].
High-Quantity (Hi-Q) Organoid Protocol
Alternative approaches address quality concerns through protocol optimization rather than post-hoc assessment. The Hi-Q method generates thousands of uniform-sized organoids using custom-designed spherical plates with 185 equally-sized microwells (1×1mm opening, 180μm diameter base) [40]. This system eliminates the embryoid body stage and extracellular matrix embedding, instead directly differentiating hiPSC into neurospheres under identical diffusion conditions [40]. The resulting organoids show minimal disintegration (1-2 per batch of 300), consistent size progression across multiple hiPSC lines, and reduced activation of cellular stress pathways [40].
Table 3: Comparison of QC Approaches for Brain Organoids
| QC Method | Key Features | Advantages | Limitations |
|---|---|---|---|
| Comprehensive QC Framework | Hierarchical scoring of 5 criteria (0-5 scale), minimum thresholds | Identifies specific failure modes, applicable to existing protocols | Resource-intensive for full assessment [2] |
| Feret Diameter Screening | Single-parameter assessment (3050μm threshold) | Rapid, non-invasive, high positive predictive value | May miss some quality issues detectable only molecularly [4] |
| Hi-Q Protocol | Custom microwells, direct neural induction, spinner bioreactors | High quantity, minimal stress pathways, cryopreservable | Requires specialized equipment, protocol-specific [40] |
| NEST-Score Computational Tool | scRNA-seq based, compares to in vivo references | Comprehensive cell type evaluation, protocol optimization | Expensive, computational expertise required [3] [42] |
The Scientist's Toolkit: Essential Research Reagents
Successful organoid culture and quality assessment depend on specialized reagents and materials:
Table 4: Essential Research Reagents for Organoid Culture and QC
| Reagent Category | Specific Examples | Function & Importance | QC Considerations |
|---|---|---|---|
| Extracellular Matrix | Matrigel, EHS-derived matrices | 3D scaffold, provides structural support | Batch-to-batch variability affects results; store at -20°C, use within weeks at 4°C [41] [43] |
| Passaging Solutions | Specialized organoid passaging digestion medium (e.g., abs9520) | Gentle dissociation using multiple mild enzymes | Avoid trypsin; store aliquots at -20°C, use within 2 weeks at 4°C [41] |
| Culture Media Components | Noggin, FGF-10, EGF, B-27, N-Acetyl cysteine, ROCK inhibitor | Direct differentiation, support viability and growth | Factor activity declines; aliquot and freeze at -20°C for long-term storage [43] [40] |
| QC Assessment Reagents | Antibodies (SOX2, MAP2, PAX6), viability dyes, DNA damage markers | Evaluate cellular composition and viability | Validate antibodies for 3D cultures; optimize penetration [2] [4] |
The implementation of standardized quality control frameworks is essential for advancing cerebral organoid research from exploratory studies to reproducible experimental systems and preclinical applications. The comprehensive QC scoring system enables objective identification of common organoid failures through hierarchical assessment of morphological, structural, and cellular parameters. For researchers requiring high-throughput applications, the Hi-Q protocol provides an alternative approach through standardized production methods. Integrating these quality assessment and assurance methods throughout organoid culture and experimentation will enhance data reliability, improve cross-study comparability, and accelerate the translation of organoid technology into meaningful biomedical advances.
Limitations of the 60-Day Framework and Adapting to Other Timepoints
Cerebral cortical organoids have emerged as transformative tools in neuroscience, offering unprecedented insights into human brain development, disease modeling, and drug screening. However, the inherent variability in organoid quality poses significant challenges for reproducibility and reliable data interpretation. A standardized quality control (QC) framework specifically designed for 60-day cortical organoids represents a critical advancement toward addressing these challenges [25] [2]. This guide objectively examines the capabilities and constraints of this 60-day framework, explores its adaptability to other developmental timepoints, and provides supporting experimental data and protocols for researchers and drug development professionals.
The 60-day QC framework for cerebral cortical organoids employs a hierarchical scoring system to standardize quality assessment. This system evaluates organoids across five critical criteria, prioritizing non-invasive assessments to efficiently exclude low-quality specimens before proceeding to more in-depth analyses [25] [8].
Table 1: Core Quality Criteria in the 60-Day QC Framework
| Criterion | Assessment Focus | Key Indices | Scoring Scale |
|---|---|---|---|
| A. Morphology | Macroscopic structure | Compactness, border integrity, absence of cysts | 0 (low quality) to 5 (high quality) |
| B. Size & Growth Profile | Developmental dynamics | Diameter, growth progression | 0 to 5 |
| C. Cellular Composition | Presence of expected cell types | Neural progenitors, neurons, astrocytes | 0 to 5 |
| D. Cytoarchitectural Organization | Microstructural arrangement | Neural rosette formation, layered organization | 0 to 5 |
| E. Cytotoxicity | Cell health and death | DNA damage, cell viability markers | 0 to 5 |
The framework operates through two sequential applications: an Initial QC (Criteria A-B) for pre-study selection and a Final QC (Criteria A-E) for comprehensive post-study analysis [25] [2]. This structure ensures resource-efficient evaluation while maintaining rigorous quality standards.
Experimental Validation of the Framework
Researchers validated this QC system by exposing 60-day cortical organoids to graded doses of hydrogen peroxide (H₂O₂) to induce controlled oxidative stress and create a spectrum of quality outcomes [25] [2]. The scoring system successfully discriminated between organoids of differing quality levels, demonstrating its robustness and sensitivity in detecting quality variations induced by external stressors [25].
Diagram Title: Hierarchical QC Scoring Workflow
Key Limitations of the 60-Day Framework
While the 60-day QC framework provides crucial standardization, several important limitations restrict its direct application to other research contexts and developmental stages.
Temporal Specificity and Developmental Scope
The framework targets a specific maturation point—60 days—which represents a critical intermediate stage characterized by the presence of neural progenitors, neurons, and astrocytes, alongside frequent rosette structures modeling neural tube development [25] [2]. This narrow focus creates significant constraints:
- Inapplicability to Early Developmental Stages: The framework cannot adequately assess organoids during early morphogenetic events (before day 30-40), such as neuroepithelium formation, lumen expansion, and initial patterning phases documented in live imaging studies [37].
- Limited Relevance for Late Maturation: Organoids cultured for extended periods (≥90 days) develop more complex neural networks, diverse glial populations, and different maturation markers that the 60-day criteria do not capture [1].
Regional Patterning and Organoid Type Restrictions
The framework was specifically validated for cortical organoids, limiting its utility for other region-specific models:
- Lacks Ventral Forebrain Assessment: Ventral organoids requiring Sonic Hedgehog (SHH) activation exhibit distinct cellular compositions (e.g., GABAergic neurons) not evaluated by the standard cortical-centric criteria [22].
- Incompatible with Multi-Regional Models: Complex assembloid systems combining multiple brain regions possess unique quality parameters related to regional boundary formation and neuronal connectivity that the framework does not address [33] [22].
Technical and Scalability Challenges
Implementation barriers affect the framework's broad adoption:
- Throughput Limitations: The hierarchical approach, while efficient, may still present bottlenecks for industrial-scale applications requiring thousands of uniform organoids, such as high-throughput drug screening [40].
- Dependency on Protocol-Specific Features: The framework assumes specific differentiation protocols, particularly those using extracellular matrix (ECM) components like Matrigel, which significantly influence neuroepithelial morphogenesis and cytoarchitecture [37] [22].
Table 2: Key Limitations and Their Experimental Implications
| Limitation Category | Specific Constraint | Impact on Research Applications |
|---|---|---|
| Temporal Specificity | Optimized for 60-day maturation | Not validated for early (<30 days) or late-stage (>90 days) organoids |
| Regional Specificity | Designed for dorsal cortical identity | Limited utility for ventral, midbrain, or cerebellar organoids |
| Protocol Dependency | Assumes ECM embedding (e.g., Matrigel) | Performance uncertain for matrix-free or synthetic hydrogel protocols |
| Technical Implementation | Requires multiple assessment modalities | May be resource-intensive for labs without established histology pipelines |
| Scalability | Hierarchical but manual scoring | Potentially slow for generating thousands of organoids for drug screening |
Adapting the QC Framework to Other Timepoints
Successfully extending the QC framework to different developmental stages requires substantial modifications to assessment criteria and scoring thresholds based on stage-specific biological features.
Early-Stage Organoids (Day 15-30)
Early brain organoids undergo rapid morphogenetic changes requiring fundamentally different quality metrics [37]:
- Critical Quality Parameters: Neuroepithelium formation, lumen expansion dynamics, rosette number and organization, and apoptosis levels in core regions.
- Modified Assessment Techniques: Live imaging to track lumen volume and number over time becomes essential, replacing static endpoint analyses [37].
- Key Signaling Pathways: WNT and Hippo (YAP1) signaling pathways regulate early neuroepithelium formation and regional specification, serving as potential biomarkers for quality assessment [37].
Diagram Title: Early-Stage Matrix Signaling Pathways
Late-Stage Organoids (Day 90-180)
Mature organoids require assessment of advanced developmental features absent at 60 days:
- Enhanced Functional Criteria: Electrophysiological network activity, synaptic density and maturity, and advanced glial differentiation (including myelination potential) should be incorporated into quality scoring [1] [44].
- Vascularization and Metabolism: Assessment of necrotic core reduction, vascular structure formation, and metabolic activity becomes crucial for long-term culture health [1].
- Cell Type Diversity Metrics: Scoring should evaluate the emergence of upper-layer cortical neurons, oligodendrocytes, and astrocyte subtypes that mature after 60 days [44].
Alternative Organoid Generation Protocols with Enhanced Reproducibility
Recent technological advances address fundamental reproducibility challenges that the QC framework aims to manage:
Hi-Q Brain Organoid Platform
This innovative approach bypasses traditional embryoid body formation, instead using custom-designed microwells to generate highly uniform neurospheres directly from dissociated hiPSCs [40]:
- Enhanced Standardization: Produces thousands of organoids per batch with minimal size variability and consistent cytoarchitecture.
- Reduced Cellular Stress: Omits ROCK inhibitor after initial plating, minimizing ectopic stress pathway activation that can compromise cell-type specification [40].
- Experimental Validation: scRNA-seq confirmed highly similar cell diversities across independent batches and successful modeling of microcephaly and glioma invasion [40].
Matrix-Free and Engineered Substrate Approaches
Protocols eliminating or replacing ill-defined matrices like Matrigel address batch variability concerns:
- Defined Synthetic Hydrogels: Offer reproducible composition and mechanical properties to support neuroepithelial formation without introducing uncontrolled variables [22].
- Early ECM Independence: Studies demonstrate that cell types and tissue morphology over long-term development can be comparable without exogenous ECM supplementation [22].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for Cerebral Organoid Research
| Reagent/Category | Specific Examples | Function in Organoid Generation |
|---|---|---|
| Extracellular Matrices | Matrigel, Geltrex, synthetic hydrogels | Provides structural support, promotes neuroepithelial morphogenesis [37] [22] |
| SMAD Inhibitors | SB431542 (TGF-β inhibitor), Dorsomorphin (BMP inhibitor) | Induces neuroectodermal differentiation [22] [40] |
| Patterning Molecules | Wnt agonists/antagonists, SHH, RA, FGFs | Directs regional specificity (dorsal/ventral, anterior/posterior) [22] |
| Neural Induction Media | N2, B27 supplements | Supports neural differentiation and survival [37] [40] |
| Cell Dissociation Reagents | Accutase, Trypsin | Dissociates pluripotent stem cells for initial aggregation [40] |
The 60-day QC framework for cerebral cortical organoids represents a significant stride toward standardizing organoid quality assessment, yet its application remains constrained by temporal specificity, regional limitations, and protocol dependencies. Adapting this framework to other developmental timepoints requires substantial modifications addressing stage-specific biological features—from early morphogenesis to late maturation. Incorporating emerging technologies like the Hi-Q platform and defined culture systems will be crucial for developing more comprehensive, flexible quality standards that accommodate the diverse and evolving applications of brain organoid research. As the field advances, iterative refinement of such QC frameworks will be essential for ensuring the reproducibility and reliability necessary for both fundamental neuroscience and translational drug development.
Cerebral cortical organoids have emerged as revolutionary tools in neuroscience, providing unprecedented in vitro models that recapitulate aspects of human brain development. While significant progress has been made in standardizing structural assessments, a comprehensive quality control (QC) framework must extend beyond morphology to incorporate electrophysiological and functional maturity metrics. Current challenges in organoid research include substantial functional heterogeneity, where organoids within the same batch exhibit varying degrees of electrophysiological maturation and network complexity [2] [45]. This variability compromises the reproducibility of scientific results, particularly in disease modeling, neurotoxicity testing, and preclinical drug screening where consistent functional performance is essential [23].
The integration of functional metrics is not merely complementary but fundamental to establishing biologically relevant organoid models. Research indicates that electrophysiological maturation correlates strongly with molecular and cellular development, including synaptic formation, astrocyte integration, and neural circuit establishment [45]. Without standardized assessment of these functional parameters, organoids may possess appropriate structural characteristics while lacking the functional fidelity required for modeling complex neurological processes or screening pharmaceutical compounds [46] [23]. This review compares current methodologies for assessing electrophysiological and functional maturity, providing researchers with experimental data and protocols to enhance their QC frameworks.
Comparative Analysis of Electrophysiological Assessment Platforms
Multielectrode Arrays (MEAs) for Network-Level Analysis
Multielectrode Arrays (MEAs) have emerged as a powerful platform for non-invasive, long-term monitoring of neural network activity in cerebral organoids. This technology enables researchers to quantify spontaneous electrical activity across multiple sites simultaneously, providing insights into network formation and synchronization.
Table 1: MEA Parameters for Organoid Functional Assessment
| Parameter | Measurement | Developmental Significance | Reported Values in Maturing Organoids |
|---|---|---|---|
| Mean Firing Rate | Number of spikes per second | Indicates neuronal excitability and maturation | Increases from negligible to ~18 Hz by 40 weeks [46] |
| Burst Detection | Synchronized spike patterns | Reflects functional synaptic connectivity | Emerges at ~120 days, frequency reaches 0.25 Hz by 10 months [45] |
| Network Burst Duration | Temporal length of synchronized activity | Demonstrates network integration | 985 ± 152 ms in day 161 organoids [45] |
| Synchronized Burst Firing | Coordinated firing across electrodes | Indicates functional neural circuit formation | Emerges at day 120, increases through day 161 [45] |
| Spike Amplitude | Voltage magnitude of action potentials | Correlates with neuronal health and maturation | Increases significantly from day 64 to day 99 [45] |
The developmental trajectory of electrophysiological properties follows a predictable pattern, with weak spiking activity emerging as early as day 34-64 in culture, followed by increased mean spike rates and field spike amplitudes by day 99 [45]. The appearance of synchronized burst firings at approximately day 120 represents a critical milestone, indicating the establishment of functional cortical neural networks—a hallmark of synaptic signal propagation in the developing mammalian cortex [45].
Patch-Clamp Electrophysiology for Single-Cell Resolution
While MEAs provide excellent network-level analysis, patch-clamp electrophysiology offers unparalleled resolution for investigating intrinsic properties of individual neurons within organoids. This technique typically requires slicing organoids or using whole-mount preparations to achieve optimal recording conditions.
Table 2: Single-Cell Electrophysiological Properties in Cortical Organoids
| Property | Measurement Technique | Significance | Reported Values |
|---|---|---|---|
| Sodium Current (INa) | Voltage-clamp recording | Indicates expression of voltage-gated sodium channels | Peak amplitude: -1,466.86 pA [46] |
| Potassium Current (IK) | Voltage-clamp recording | Reflects potassium channel expression and maturation | Peak amplitude: 3,031.79 pA [46] |
| Spontaneous EPSC Frequency | Current-clamp at -60 mV | Measures presynaptic glutamate release | 0.25 Hz in forebrain organoids [46] |
| Spontaneous EPSC Amplitude | Current-clamp at -60 mV | Indicates postsynaptic receptor density | -19.92 pA in forebrain organoids [46] |
| Action Potential Firing | Depolarizing current steps | Demonstrates neuronal excitability | 80% of neurons fire APs upon depolarization [46] |
| Resting Membrane Potential | Current-clamp recording | Reflects ion channel homeostasis | RMP = -60 mV to -80 mV [46] |
Studies utilizing patch-clamp electrophysiology have confirmed that a high percentage (80-86%) of neurons in mature cortical organoids fire action potentials upon depolarization and exhibit spontaneous excitatory postsynaptic currents (EPSCs) [46]. These measurements provide critical validation of neuronal functionality at the cellular level, complementing network-level data obtained through MEA recordings.
Experimental Protocols for Functional Assessment
MEA Setup and Recording Protocol for Cerebral Organoids
The following protocol adapts traditional MEA methodology for 3D cerebral organoid cultures:
Materials Required:
- Multi-electrode array system (64-channel recommended)
- BrainPhys medium or similar neuronal maintenance medium
- Data acquisition software (e.g., Axion Biosystems, Multi Channel Systems)
- Environmental chamber maintained at 37°C and 5% CO₂
- Organoid transfer tools (wide-bore pipettes)
Procedure:
- Preparation: Condition BrainPhys medium to 37°C and pre-equilibrate the MEA system environment.
- Organoid Transfer: Using wide-bore pipettes, carefully transfer individual organoids onto the MEA probe, ensuring direct contact with multiple electrodes.
- Acclimation: Allow organoids to stabilize for 15-30 minutes before initiating recordings.
- Recording Session: Acquire data in 10-minute intervals across multiple channels simultaneously.
- Data Extraction: Extract spike times, burst events, and network parameters using manufacturer software.
- Analysis: Calculate key parameters including mean firing rate, interspike interval, burst frequency, and network burst duration [45].
This protocol enables non-destructive functional assessment, allowing for longitudinal studies of the same organoids over extended periods, which is crucial for tracking maturation trajectories.
Calcium Imaging for Network Activity Visualization
Calcium imaging provides an alternative approach for visualizing network activity, particularly valuable for mapping spatial patterns of activation across organoid structures:
Materials Required:
- Genetically encoded calcium indicators (GCaMP) or synthetic dyes (e.g., Cal-520)
- Confocal or light-sheet microscope system
- Perfusion system for maintained physiological conditions
- Analysis software (e.g., ImageJ, MATLAB)
Procedure:
- Loading: Incubate organoids with cell-permeable calcium indicator dyes (30-60 minutes).
- Imaging Setup: Mount organoids in appropriate imaging chambers with continuous perfusion.
- Acquisition: Capture time-lapse sequences at 2-10 Hz sampling rate depending on temporal resolution requirements.
- Processing: Motion correction and background subtraction.
- Analysis: Identify regions of interest (ROIs) corresponding to individual neurons and extract fluorescence transients (ΔF/F) [23].
While calcium imaging offers superior spatial resolution for mapping activity patterns, it provides indirect measurement of electrical activity with slower temporal resolution compared to direct electrophysiological techniques [23].
Signaling Pathways in Functional Maturation
Activity-Dependent Maturation Pathways
Electrical activity in developing organoids activates critical signaling cascades that promote further maturation and functional refinement. Research has identified several key pathways that translate neural activity into developmental programs:
Diagram 1: Activity-dependent maturation pathway. Electrical stimulation triggers calcium influx, activating CAMKII and PKA signaling that converges on pCREB to mediate gene expression changes supporting neuronal maturation and synapse formation [47].
Neurotrophin Signaling in Circuit Development
The neurotrophin/TRK signaling pathway emerges as a crucial mediator of functional maturation in cerebral organoids. Studies have documented the activation of this pathway in organoids by 5 months in culture, coinciding with advanced stages of electrophysiological development [45]. This pathway influences synaptic plasticity, neuronal survival, and circuit refinement, providing a molecular correlate to observed functional improvements in maturing organoids.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Electrophysiological Assessment of Cortical Organoids
| Reagent/Category | Specific Examples | Function in Assessment | Protocol Considerations |
|---|---|---|---|
| Culture Media | BrainPhys Medium | Supports neuronal synaptic function and electrophysiological maturation | Enhanced EP properties compared to standard media [45] |
| Electrical Recording Platforms | 64-channel MEA Systems | Network-level activity monitoring | Non-invasive, enables longitudinal studies [46] [45] |
| Patch-Clamp Equipment | Micromanipulators, Amplifiers | Single-cell resolution recordings | Requires organoid slicing or whole-mount preparation [46] |
| Calcium Indicators | GCaMP, Cal-520 | Optical monitoring of neural activity | Genetic encoding vs. dye loading approaches [23] |
| Signaling Agonists/Antagonists | Neurotrophins, Kynurenic acid | Pathway modulation and receptor blockade | Validate specific neurotransmission systems [46] |
| Extracellular Matrix | Matrigel | Supports neuroepithelium formation and maturation | Enhances lumen expansion and regionalization [37] |
Integration with Structural QC Frameworks
A comprehensive QC framework for cerebral cortical organoids must integrate both structural and functional assessment parameters. The recently proposed QC methodology establishes a hierarchical approach evaluating five key criteria: morphology, size and growth profile, cellular composition, cytoarchitectural organization, and cytotoxicity [2]. Incorporating electrophysiological metrics into this existing framework enhances its utility for ensuring organoid reproducibility and predictive validity.
Functional assessment should be implemented at two critical points:
- Initial QC: Following structural screening, organoids should undergo basic electrophysiological assessment (e.g., MEA screening for spontaneous activity) to exclude functionally compromised specimens before entry into studies.
- Final QC: Comprehensive functional characterization including network complexity analysis, synaptic function assessment, and pharmacological responsiveness should be conducted at study endpoint [2] [45].
This integrated approach addresses a critical limitation in current organoid research, where structural integrity does not necessarily predict functional competence. Organoids with appropriate morphological characteristics may lack mature electrophysiological properties essential for modeling neural network function or drug responses [23].
The integration of electrophysiological and functional maturity assessment into quality control frameworks represents an essential evolution in cerebral organoid research. As the field progresses toward applications in disease modeling, drug screening, and potentially regenerative medicine, standardized functional metrics will become increasingly critical for validating model reliability and reproducibility. The methodologies and data presented here provide researchers with practical tools for implementing these assessments, bridging the gap between structural organization and functional competence in cerebral cortical organoids.
Future developments should focus on establishing standardized functional benchmarks across laboratories, correlating in vitro electrophysiological metrics with specific developmental stages, and integrating functional assessment more seamlessly with high-throughput screening platforms. Through the adoption of comprehensive QC frameworks that value functional maturity alongside structural fidelity, the research community can fully leverage the transformative potential of cerebral organoid technology.
Cerebral cortical organoids have emerged as groundbreaking three-dimensional in vitro models that mimic the complexity of the human brain, offering unprecedented opportunities for neuroscience research, disease modeling, and drug screening [2]. Unlike traditional two-dimensional cultures or simpler 3D models, cerebral organoids recreate physiologically relevant cellular microenvironments that enhance cell-cell and cell-matrix interactions, fostering improved differentiation and maturation [2]. These complex structures self-organize into architectures that recapitulate aspects of human brain development, making them invaluable for studying neurodevelopmental disorders, neurological cancers, and neurodegenerative diseases [2] [8].
However, the transformative potential of cerebral organoids is hampered by significant challenges in quality control and reproducibility [2] [8]. The stochastic nature of stem cell differentiation and spontaneous self-organization within organoids leads to substantial variability in morphology, size, cellular composition, and cytoarchitectural organization [2]. This variability compromises the reliability of scientific results, particularly in disease modeling, neurotoxicity testing, and preclinical drug screening where high-quality and consistent models are essential [2]. Researchers commonly report discrepancies where some organoids within the same batch display optimal morphology with dense overall structure and well-defined borders, while others exhibit poor compactness, degrade over time, develop suboptimal cystic cavities, or present disorganized structures with lower proportions of expected cell types [2].
The integration of artificial intelligence and automated technologies presents a promising pathway to overcome these challenges by enabling standardized, quantitative, and high-throughput quality assessment of cerebral organoids [48] [49]. This comparison guide examines emerging AI-driven and automated platforms for cerebral cortical organoid analysis, providing researchers with experimental data, methodological details, and technical specifications to inform technology adoption decisions.
Comparative Analysis of AI and Automated Platforms
The table below provides a systematic comparison of currently available technologies for AI-driven analysis and automated quality screening of cerebral cortical organoids, synthesizing data from recent peer-reviewed studies and commercial implementations.
Table 1: Comparative Analysis of AI and Automated Platforms for Cerebral Organoid Quality Assessment
| Platform/Technology | Primary Function | Key Metrics | Performance Data | Technical Basis |
|---|---|---|---|---|
| Feedback-Driven IoT Platform [48] | Automated maintenance & neural activity monitoring | Median firing rates of neural units, recording frequency | Increased median firing rates; hourly electrophysiology recordings; feeding did not affect firing rates | IoT-enabled microfluidics, HD-MEA, computer vision fluid estimation |
| QC Scoring Framework [2] [8] | Quality classification of 60-day cortical organoids | 5 criteria scoring (0-5): Morphology, Size/Growth, Cellular Composition, Cytoarchitecture, Cytotoxicity | Successfully discriminated H₂O₂-induced quality variations; hierarchical exclusion of low-quality organoids | Multimodal assessment with defined thresholds and composite scoring |
| BioMARS Autonomous Lab System [50] | Fully automated biological experiments | Protocol execution accuracy, error detection capability | Requires human supervision for customized experiments; depends on existing procedures | Multi-agent AI (LLMs + VLMs) with robotic control integration |
| MO:BOT Automated Platform [51] | Standardized 3D cell culture automation | Throughput (6-well to 96-well formats), data yield | Up to 12x more data on same footprint; automated seeding, media exchange, and QC | Biology-first automation with pre-screening quality control |
| AI-Enabled Organoids Framework [49] | Construction, analysis, and preclinical evaluation | Multiscale image feature extraction, multi-omics integration | Rapid screening of construction strategies; precise preclinical evaluation | End-to-end AI integration across organoid lifecycle |
Experimental Protocols and Methodological Approaches
Validation of Automated IoT-Enabled Organoid Maintenance
A 7-day validation study comparing automated versus manual protocols for mouse cerebral cortex organoids employed a sophisticated experimental setup integrating multiple technologies [48]. The platform utilized custom 3D-printed chambers affixed to commercial microelectrode arrays with periodic feeding achieved through programmable microfluidic pumps. A critical innovation was the development of a computer vision fluid volume estimator that provided real-time feedback to rectify deviations in microfluidic perfusion during media feeding and aspiration cycles.
The experimental protocol involved continuous monitoring of neural activity through high-density microelectrode arrays (HD-MEA) with recording sessions conducted hourly throughout the experiment. Surprisingly, the results demonstrated that media exchange during recording did not show acute effects on firing rate, enabling the use of this automated platform for reagent screening studies [48]. The median firing rates of neural units increased for each sample throughout the experiment, and dynamic patterns of organoid firing rates were revealed by high-frequency recordings that would be impractical with manual methods.
Quality Control Framework Implementation and Validation
The quality control framework for 60-day cortical organoids was validated through a systematic experiment exposing organoids to graded doses of hydrogen peroxide (H₂O₂) to induce controlled variations in quality [2]. The experimental protocol followed a hierarchical assessment approach:
Initial QC Screening: Organoids were first evaluated using non-invasive criteria (morphology and size/growth profile) to establish baseline quality and exclude unsuitable samples prior to H₂O₂ exposure.
Controlled Stress Induction: Organoids that passed initial QC were exposed to increasing concentrations of H₂O₂, a chemical known to cause oxidative stress-induced cellular death at non-physiological doses.
Recovery Period: Following H₂O₂ exposure, organoids underwent a one-week recovery period before final assessment.
Comprehensive Final QC: Post-treatment evaluation applied all five QC criteria (morphology, size/growth profile, cellular composition, cytoarchitectural organization, and cytotoxicity) using the standardized scoring system.
The framework successfully discriminated organoid quality levels across the H₂O₂ gradient, demonstrating its robustness and sensitivity for quality assessment [2]. The hierarchical approach efficiently excluded low-quality organoids while reserving in-depth analyses for samples that met initial thresholds, optimizing resource utilization in research settings.
Cross-Site Reproducibility Assessment
A significant multi-site study investigated the reproducibility of human cortical organoids across three independent research groups using a harmonized miniaturized spinning bioreactor protocol [52]. The experimental design involved multiple differentiation replicates from one induced pluripotent stem cell line across a three-month differentiation period.
The protocol assessed consistency in cell type proportions, organization in cortical wall-like buds, organoid size, and expression of metabolism and cellular stress genes. Results demonstrated that organoids predominantly contained cortical progenitor and neuronal cell types in reproducible proportions consistently organized in cortical wall-like buds across sites [52]. However, cross-site differences were detected in organoid size and expression of metabolism and cellular stress genes, with variability correlating with stem cell gene expression prior to differentiation and technical factors associated with seeding.
Visualization of Workflows and System Architectures
Automated Feedback-Driven Organoid Culture Platform
Automated Organoid Maintenance System
Hierarchical Quality Control Framework
Hierarchical QC Assessment Workflow
Essential Research Reagents and Materials
The table below details key research reagents and materials essential for implementing AI-driven analysis and automated quality screening of cerebral cortical organoids, as identified from the experimental protocols analyzed.
Table 2: Essential Research Reagents and Materials for AI-Driven Organoid Analysis
| Reagent/Material | Function | Application Context | Technical Specifications |
|---|---|---|---|
| Programmable Microfluidic Pumps [48] | Precise media delivery and aspiration | Automated maintenance platforms | IoT-enabled; computer vision feedback integration |
| High-Density Microelectrode Arrays (HD-MEA) [48] | Neural activity monitoring | Electrophysiological recording | High-frequency recording capability (hourly intervals) |
| Custom 3D-Printed Chambers [48] | Organoid housing and positioning | Compatible with commercial MEAs | Tailored dimensions for specific organoid types |
| Hydrogen Peroxide (H₂O₂) [2] | Induced oxidative stress for QC validation | Quality control framework testing | Graded doses for quality variation induction |
| Miniaturized Spinning Bioreactors [52] | Scalable organoid culture | Cross-site reproducibility studies | Harmonized protocol compatibility |
| Multimodal Imaging Reagents [2] [53] | Cellular composition and viability assessment | QC criteria evaluation | Immunohistochemistry, cytotoxicity staining |
| AI-Assisted Analysis Software [50] [53] [49] | Image analysis and pattern recognition | Automated quality screening | Computer vision, neural networks, multimodal data integration |
The integration of AI-driven analysis and automated quality screening technologies for cerebral cortical organoids represents a significant advancement toward standardized, reproducible, and scalable organoid research [48] [2] [49]. The comparative data presented in this guide demonstrates that while various approaches show promising results, implementation decisions must consider specific research objectives, technical capabilities, and resource constraints.
For academic research settings, the hierarchical QC framework provides a cost-effective approach to standardize organoid quality without requiring substantial infrastructure investments [2] [8]. For industrial applications requiring high-throughput screening, fully automated platforms like the IoT-enabled maintenance system or MO:BOT platform offer scalability advantages despite higher initial implementation costs [48] [51]. The emerging trend of multi-agent AI systems like BioMARS and CRISPR-GPT indicates a future direction where autonomous experiment design and execution could further reduce human-dependent variability [50].
Critical implementation considerations include the trade-off between comprehensive assessment and throughput, the importance of metadata standardization for AI training, and the need for flexible systems that can adapt to diverse organoid types and research applications [51]. As the field progresses, the development of universally accepted standards for organoid quality assessment will be essential for maximizing the potential of these innovative technologies in both basic research and clinical translation.
Framework Validation and Comparative Analysis with Emerging Technologies
Within the advancing field of cerebral organoid research, the lack of standardized quality control (QC) frameworks presents a significant barrier to experimental reproducibility and reliability. This is particularly critical for applications in disease modeling and preclinical drug screening, where consistency is paramount. A promising validation strategy involves subjecting organoids to controlled oxidative stress to deliberately induce a spectrum of quality outcomes. This guide objectively compares the performance of a recently established QC framework against traditional, often subjective, assessment methods, using hydrogen peroxide (H₂O₂) stress testing as a benchmark for evaluation. The supporting data and protocols herein are designed to provide researchers with a clear, actionable comparison to enhance the rigor of their cerebral organoid studies.
The QC Framework: A Systematic Scoring Approach
A comprehensive QC framework has been developed specifically for 60-day cortical organoids, an intermediate stage characterized by the presence of neural progenitors, neurons, and astrocytes [2]. This framework transforms subjective qualitative assessments into a structured, quantitative scoring system.
The system is hierarchically organized around five critical criteria, each subdivided into specific indices scored from 0 (low quality) to 5 (high quality) [2]:
- A. Morphology: Assessing compactness, border integrity, and the absence of undesirable cystic structures.
- B. Size and Growth Profile: Ensuring proper developmental dynamics.
- C. Cellular Composition: Verifying the presence of expected neural populations.
- D. Cytoarchitectural Organization: Evaluating structural integrity and layered organization.
- E. Cytotoxicity: Detecting DNA damage and assessing cell viability.
This hierarchy allows for an efficient two-stage evaluation process. An Initial QC utilizes non-invasive criteria (Morphology and Size) to pre-select organoids for a study. The Final QC incorporates all criteria for a comprehensive post-study evaluation, ensuring robust and reproducible results [2] [8].
Workflow of the Quality Control Framework
The following diagram illustrates the hierarchical workflow for applying the QC scoring system to cerebral cortical organoids, from initial non-invasive checks to final comprehensive evaluation.
Experimental Validation: H₂O₂ Stress Testing
To validate the discriminative power of the QC framework, researchers exposed 60-day cortical organoids to graded doses of hydrogen peroxide (H₂O₂) [2]. H₂O₂ is a known inducer of oxidative stress that can lead to cell death at non-physiological doses, making it an ideal agent for generating organoids with a controlled range of quality outcomes [2]. After exposure and a subsequent recovery period, both exposed and non-exposed organoids were evaluated using the complete Final QC scoring system.
Experimental Workflow for H₂O₂ Validation
The diagram below outlines the key stages of the experimental protocol used to validate the QC framework through hydrogen peroxide stress testing.
The framework successfully distinguished organoids across the quality spectrum, confirming its sensitivity and reliability in identifying both high-quality and compromised tissues [2] [5]. Organoids with optimal morphology, dense structure, and well-defined borders received high scores, whereas those with poor compactness, disorganized structures, or suboptimal cavities were accurately flagged as low-quality [2].
Performance Comparison: Quantitative Data
The quantitative nature of the QC scoring system allows for direct comparison between organoids subjected to varying degrees of H₂O₂-induced stress. The data below summarizes how the framework discriminates between quality levels based on key metrics.
Table 1: QC Scoring Discrimination Following H₂O₂ Stress
| QC Criterion | High-Quality Organoid (Low/No H₂O₂) | Medium-Quality Organoid (Moderate H₂O₂) | Low-Quality Organoid (High H₂O₂) |
|---|---|---|---|
| Morphology Score | 4-5 (Dense structure, defined borders) | 2-3 (Moderate compactness, minor cysts) | 0-1 (Poor compactness, severe degradation) |
| Cellular Composition | 4-5 (Expected neural populations) | 2-3 (Proportion of non-target cells) | 0-1 (Disorganized, incorrect cell types) |
| Cytoarchitectural Organization | 4-5 (Structured layering, rosettes) | 2-3 (Disrupted organization) | 0-1 (No clear architecture) |
| Cytotoxicity Level | 4-5 (Low DNA damage, high viability) | 2-3 (Moderate damage) | 0-1 (High damage, low viability) |
| Overall QC Classification | PASS | Conditional PASS/FAIL | FAIL |
Table 2: Comparison of Organoid Assessment Methods
| Feature | Novel QC Framework | Traditional Morphology-Only | Non-Standarded In-Lab Methods |
|---|---|---|---|
| Basis of Assessment | Quantitative, multi-parameter scoring [2] | Qualitative and subjective [2] | Variable, often qualitative [2] |
| Scope of Evaluation | Comprehensive (5 key criteria) [2] | Narrow (primarily visual) | Inconsistent between labs |
| Inter-Rater Reliability | High (minimizes observer bias) [5] | Low (prone to individual bias) [2] | Low to very low |
| H₂O₂ Stress Test Performance | Accurately discriminates quality levels [2] | Limited sensitivity and reliability | Unreliable and non-comparable |
| Scalability for Industry | High (standardized and user-friendly) [8] | Low | Very Low |
| Best Use Case | Preclinical research, drug screening, reproducible studies [8] | Initial, non-invasive daily checks | General lab monitoring without need for cross-lab comparison |
Detailed Experimental Protocols
Hydrogen Peroxide Stress Test Protocol
This protocol is adapted from methods used to validate the QC framework [2].
- Step 1: Organoid Selection: At day 60 of culture, subject a batch of cortical organoids to the Initial QC (Criteria A & B). Select only those organoids that pass the minimum thresholds for morphology and size to ensure a uniform starting population for the stress test.
- Step 2: H₂O₂ Exposure Preparation: Prepare a concentration gradient of H₂O₂ in the organoid culture medium. For example, 0 μM (control), 50 μM, 100 μM, 200 μM, and 500 μM. Ensure all solutions are sterile and freshly prepared.
- Step 3: Application and Incubation: Expose the selected organoids to the different H₂O₂ concentrations for a specified duration (e.g., 24 hours). Include a control group in H₂O₂-free medium.
- Step 4: Recovery: After exposure, replace the medium with standard organoid culture medium and allow the organoids to recover for a predetermined period (e.g., 7 days) [2].
- Step 5: Final Analysis: After the recovery period, perform the Final QC assessment on all organoids, scoring each according to the full set of criteria (A-E). Correlate the final scores with the H₂O₂ exposure doses to validate the framework's discrimination capability.
Orthogonal Assay for H₂O₂ Detection
While the QC framework assesses phenotypic outcomes, understanding the underlying oxidative stress requires quantifying H₂O₂. The following plate reader-based method provides a high-throughput compatible approach [54].
- Principle: Use of coumarin-7-boronic acid (CBA), which reacts with H₂O₂ to form the fluorescent product 7-hydroxycoumarin (COH) [54].
- Procedure:
- Cell Culture Preparation: Plate cells (e.g., HEK293 cells overexpressing Nox isoforms as a ROS source) or organoid supernatants in a 96-well plate.
- Probe Addition: Add CBA stock solution to a final concentration optimized for detection (e.g., from a 0.1 M stock in DMSO) [54].
- Stimulation: Activate ROS-producing pathways if necessary (e.g., with PMA for Nox2).
- Fluorescence Measurement: Monitor fluorescence in a plate reader using excitation/emission wavelengths of 355/460 nm [54].
- Data Analysis: Calculate H₂O₂ production relative to a standard curve of authentic COH.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of the QC framework and associated stress tests relies on key reagents and tools. The following table details essential solutions for this field of research.
Table 3: Research Reagent Solutions for Organoid QC and Stress Testing
| Reagent / Solution | Function / Application | Key Details & Considerations |
|---|---|---|
| Hydropropidine (HPr+) | Cell-impermeable fluorescent probe for detecting extracellular superoxide (O₂⨪) [54] | Used with DNA to form a fluorescent product; excitation/emission: 485/574 nm [54]. |
| Coumarin-7-Boronic Acid (CBA) | Peroxidase-independent fluorescent probe for detecting hydrogen peroxide (H₂O₂) [54] | Reaction with H₂O₂ forms fluorescent 7-hydroxycoumarin; excitation/emission: 355/460 nm [54]. |
| Hydrogen Peroxide (H₂O₂) | Inducer of controlled oxidative stress for experimental validation of QC frameworks [2] | Use high-grade; prepare fresh solutions for dosing experiments to ensure accuracy and sterility. |
| Antibody Panels (for Immunostaining) | Characterization of cellular composition and cytoarchitectural organization [55] | Critical for Criteria C & D. Examples: EMX1 (cortical neurons), GAD2 (GABAergic neurons), COL1A1 (fibroblasts) [55]. |
| Viability/Cytotoxicity Assay Kits | Quantification of cell death and health status for Cytotoxicity criterion [2] | Enables scoring of Criterion E. Use assays validated for 3D cultures. |
| Single-Cell RNA Sequencing (scRNA-seq) | Gold-standard orthogonal method for validating cellular composition [55] | Provides high-resolution data to confirm correlations between organoid morphology and cell identity [55]. |
The experimental validation using hydrogen peroxide stress testing provides compelling evidence for the efficacy of the systematic QC framework. By replacing subjective assessment with a quantitative, multi-parameter scoring system, this framework significantly enhances the reproducibility and reliability of cerebral organoid research. The ability to accurately discriminate between organoids of varying quality, as induced by controlled oxidative stress, makes this framework a powerful tool for ensuring consistent and meaningful results in both academic studies and industrial applications like drug discovery and neurotoxicity testing. Its adoption is a critical step towards standardizing the field and fully realizing the potential of cerebral organoids in neuroscience.
Comparative Analysis with Morphology-Based Selection Methods
Cerebral cortical organoids have emerged as transformative tools in neuroscience, offering unprecedented in vitro models for studying human brain development, disease mechanisms, and neurotoxicity [2] [33]. However, the inherent variability in self-organizing 3D systems presents significant challenges for experimental reproducibility and reliability [2] [4]. This variability stems from the stochastic nature of stem cell differentiation and spontaneous self-organization, leading to inconsistencies in morphology, size, cellular composition, and cytoarchitectural organization [2].
To address these challenges, morphology-based selection methods have developed as crucial non-destructive approaches for quality assessment. These methods leverage visual characteristics as proxies for internal cellular composition and structural integrity, enabling researchers to select organoids with desired properties without invasive procedures that compromise the tissue [56] [57] [4]. This comparative analysis examines the current landscape of morphology-based selection methodologies, their validation approaches, and performance characteristics, providing researchers with a framework for implementing quality control in cerebral cortical organoid research.
Established Morphology-Based QC Frameworks
Comprehensive QC Scoring System
A hierarchical quality control framework specifically designed for 60-day cortical organoids represents one of the most comprehensive morphology-based approaches [2] [8]. This system employs a structured scoring methodology across five critical criteria, with morphology serving as the initial gatekeeping parameter:
- Morphology: Evaluates compactness, border integrity, and absence of cysts or necrotic cores
- Size and Growth Profile: Ensures proper developmental dynamics and excludes outliers
- Cellular Composition: Assesses presence of expected neural populations
- Cytoarchitectural Organization: Examines structural integrity and organization of neural layers
- Cytotoxicity: Measures DNA damage and cell viability markers
The framework operates hierarchically, beginning with non-invasive assessments (morphology and size) to exclude low-quality organoids initially, while reserving more in-depth analyses for those passing the initial evaluation [2]. This practical design efficiently allocates resources while maintaining rigorous quality standards. The system was validated using hydrogen peroxide exposure experiments, which induced controlled variations in quality, demonstrating the framework's robustness in discriminating organoid quality levels [2].
Morphology-Cellular Composition Correlation
Ikeda et al. established a direct correlation between external morphological features and internal cellular composition through paired morphological and single-cell RNA-seq analyses [56] [57]. This research demonstrated that non-destructive morphological analysis can accurately distinguish organoids composed of cerebral cortical tissues from those containing other cerebral tissues [56].
Table 1: Morphological Classification and Cellular Composition Correlation
| Morphological Type | Primary Cellular Composition | Key Identifying Features | Application Potential |
|---|---|---|---|
| Rosette-like concentric layered structures | Primarily cortical neurons | Distinct concentric layered organization | High suitability for cell replacement therapies |
| Low transparency with no clear internal structures | Predominantly GABAergic neurons | Opaque appearance, disorganized interior | Disease modeling of inhibitory neuron pathologies |
| Unique morphological features | Non-neuronal cells (neural crest, choroid plexus) | Distinct from neural tissue morphology | Specialized applications requiring non-neuronal cells |
This classification system enables researchers to select organoids with specific neuronal populations based solely on visual characteristics, significantly enhancing experimental accuracy and reliability [57]. The remarkable reproducibility of gene expression profiles among organoids sharing the same morphological characteristics underscores the validity of this approach [57].
Quantitative Morphological Parameters
Feret Diameter as a Key Metric
Deciphering brain organoid heterogeneity through morphological analysis identified Feret diameter (the longest distance between any two points of the organoid) as a reliable, single parameter characterizing brain organoid quality [4]. Through systematic analysis of 72 organoids from 12 different hPSC lines, researchers established a quantitative threshold of 3050μm, with organoids exceeding this diameter demonstrating poorer quality outcomes [4].
The Feret diameter exhibited the best performance among morphological parameters with a Youden index of 0.68, accurately reflecting expert quality evaluation with a positive predictive value of 94.4% and negative predictive value of 69.4% [4]. This straightforward measurement provides an accessible, quantitative metric for laboratories implementing basic quality control.
Automated 3D Morphological Characterization
Advanced image analysis workflows have been developed for comprehensive three-dimensional morphological characterization of cortical organoids [58]. This customized approach utilizes machine-learning-based cell nuclei segmentation implemented via Cellpose v2.2, with subsequent processing in MATLAB through a custom Graphical User Interface [58].
Table 2: Quantitative Morphological Parameters for Quality Assessment
| Parameter | Measurement Method | Quality Correlation | Practical Implementation |
|---|---|---|---|
| Feret Diameter | Brightfield imaging + ImageJ analysis | >3050μm correlates with poor quality [4] | Simple, requires standard lab equipment |
| Area/Perimeter | Brightfield imaging + computational analysis | Correlates with expert evaluation [4] | Easily quantifiable through image analysis |
| Cyst Formation Visual assessment scoring | Significant fluid-filled cysts indicate poor quality [2] [4] | Qualitative but can be standardized with training | |
| Necrotic Core Volume | 3D fluorescence imaging + automated segmentation | Larger cores indicate viability issues [58] | Requires specialized imaging and analysis |
This automated workflow can process large datasets of tiled and z-series images, analyzing over 1.5 million cells to quantify organoid health, development, morphology, and cellular composition over time [58]. Particularly valuable is its ability to identify and separate areas of cell death in non-viable regions while quantifying cell number and density in viable tissue areas [58].
Experimental Validation Protocols
Validation Through Transcriptomic Analysis
The correlation between morphological selection and cellular composition requires rigorous validation, typically achieved through single-cell RNA sequencing (scRNA-seq) [56] [4]. This approach involves:
- Morphological Classification: Organoids are first categorized based on morphological characteristics [56] [57]
- Single-Cell Dissociation: Selected organoids are processed into single-cell suspensions
- scRNA-seq Library Preparation: Using standard platforms (10X Genomics, etc.)
- Cell Type Annotation: Identifying cellular composition based on marker gene expression
- Correlation Analysis: Establishing relationships between morphological classes and cell type distributions
In the Feret diameter study, bulk RNA sequencing and BayesPrism deconvolution analysis revealed that high-quality organoids consistently displayed lower presence of mesenchymal cells, with the proportion of mesenchymal cells positively correlating with Feret diameter [4]. This provided a molecular basis for the observed morphological differences.
Experimental Workflow for QC Validation
The following diagram illustrates the typical experimental workflow for validating morphology-based selection methods:
Performance Comparison
Methodology Comparison
The various morphology-based selection methods offer different advantages depending on research objectives, technical capabilities, and required throughput:
Table 3: Performance Comparison of Morphology-Based Selection Methods
| Methodology | Key Metrics | Validation Approach | Throughput | Technical Requirements |
|---|---|---|---|---|
| Hierarchical QC Framework [2] | Multi-parameter scoring system | Hydrogen peroxide-induced quality variance | Moderate | Standard lab equipment + imaging |
| Morphology-RNA-seq Correlation [56] | Visual morphological classes | scRNA-seq cellular composition | Lower | Advanced sequencing capabilities |
| Feret Diameter Threshold [4] | Single parameter (3050μm) | Bulk RNA-seq + cellular deconvolution | High | Basic imaging + analysis software |
| Automated 3D Characterization [58] | Multiple volumetric parameters | Immunohistochemistry + cell counting | Moderate-High | Advanced imaging + ML expertise |
Signaling Pathways in Organoid Morphogenesis
Understanding the molecular mechanisms underlying organoid morphogenesis provides context for morphology-based quality assessment. Recent research on brain organoid morphodynamics has identified key signaling pathways active during these processes:
The diagram illustrates how extracellular matrix (ECM) influences organoid morphology through mechanosensing pathways, particularly via Hippo (YAP1) and WNT signaling, ultimately affecting brain regionalization and lumen formation – key morphological quality indicators [37].
Research Reagent Solutions
Implementing robust morphology-based selection methods requires specific research reagents and tools. The following table details essential solutions used across the cited studies:
Table 4: Essential Research Reagents for Morphology-Based QC
| Reagent/Category | Specific Examples | Research Function | Example Usage |
|---|---|---|---|
| Stem Cell Lines | H9 (WA09), H1 (WA01), WIBR1/2/3, IMR90, Kucg2 [4] | Organoid generation foundation | Multiple studies used established hPSC lines [4] |
| Differentiation Matrix | Matrigel [37] | Provides extracellular matrix environment | Support neuroepithelium formation in unguided organoids [37] |
| Morphological Validation | Hydrogen peroxide (H₂O₂) [2] | Induces controlled quality variance | QC framework validation [2] |
| Image Analysis Software | ImageJ, CellProfiler, Imaris, Cellpose v2.2 [4] [58] | Quantifies morphological parameters | Feret diameter measurement [4] |
| Transcriptomic Validation | Single-cell RNA sequencing [56] [4] | Correlates morphology with cellular composition | Validation of morphology-based selection [56] |
Morphology-based selection methods for cerebral cortical organoids have evolved from subjective visual assessment to quantitative, validated frameworks that reliably predict internal cellular composition and structural integrity [2] [56] [4]. The comparative analysis presented here demonstrates that these methods offer varying levels of sophistication, from simple Feret diameter measurements [4] to comprehensive multi-parameter scoring systems [2] and advanced computational approaches [58].
The choice of methodology depends on specific research needs, with simpler approaches suitable for routine quality control and more complex frameworks appropriate for rigorous preclinical applications. Crucially, the correlation between external morphological features and internal cellular composition has been consistently validated through transcriptomic analyses [56] [57] [4], establishing morphology-based selection as a scientifically sound approach for enhancing reproducibility in cerebral organoid research.
As the field advances, integrating these quality control frameworks with emerging technologies such as automated imaging systems and machine learning algorithms will further standardize cerebral organoid research, accelerating their application in disease modeling, drug screening, and therapeutic development [2] [8] [58].
Synergy with Advanced Maturation Benchmarking and Electrophysiological Profiling
Cerebral organoids have emerged as transformative tools in neuroscience, providing unprecedented in vitro models that recapitulate aspects of human brain development, disease pathology, and drug responses [59]. However, their full potential in both academic research and pharmaceutical development remains hampered by significant challenges in quality control and reproducibility [2]. The inherent variability in organoid morphology, cellular composition, cytoarchitectural organization, and functional maturation creates substantial barriers to reliable data interpretation and cross-study comparisons [2] [8]. This review examines how the synergistic integration of advanced maturation benchmarking with comprehensive electrophysiological profiling can establish a robust quality control framework, ultimately enhancing the translational relevance of cerebral cortical organoids for disease modeling and drug development.
The fundamental challenge stems from the stochastic nature of stem cell differentiation and the spontaneous self-organization processes that, while enabling remarkable complexity, also lead to substantial inter-organoid heterogeneity [2]. Without standardized assessment protocols, researchers face difficulties in distinguishing technical variability from biologically significant phenotypes, particularly in disease modeling contexts [8]. Recent initiatives have begun addressing these concerns through systematic quality control frameworks that evaluate organoids across multiple dimensions, including morphology, size, cellular composition, cytoarchitectural organization, and cytotoxicity [2]. When combined with functional assessments of electrophysiological maturation, these approaches provide a powerful foundation for validating organoid quality and physiological relevance.
Multidimensional Assessment of Brain Organoid Maturity
Structural and Cellular Maturation Benchmarks
Evaluating brain organoid maturity requires a multimodal approach that encompasses structural, cellular, functional, and molecular dimensions [23]. The structural architecture of mature organoids demonstrates progressive acquisition of layered cytoarchitecture, synaptic connectivity, and region-specific molecular identities [23]. Critical benchmarks include cortical lamination validated by layer-specific markers: SATB2 for upper-layer (II-IV) populations, TBR1 for deep-layer (VI) neurons, and CTIP2 expression in layer V [23]. The development of essential barrier structures, including the glia limitans externa (visualized via aquaporin-4 expressing astrocyte endfeet) and rudimentary blood-brain barrier units (with CD31+ endothelial tubes ensheathed by PDGFRβ+ pericytes), further indicates advanced maturation [23].
The diversity of cell types represents another critical dimension for assessment. Precise characterization of neural populations relies on cell-type-specific molecular markers, including general neuronal markers (NEUN, βIII-tubulin), maturity-stage markers (DCX and NeuroD1 for immature neurons; MAP2 for mature neurons), and neurotransmitter identity markers (VGLUT1 for glutamatergic neurons; GAD65/67 for GABAergic neurons) [23]. Beyond neuronal populations, the presence and maturation of glial cells—astrocytes (GFAP, S100β) and oligodendrocytes (MBP, O4)—provide essential indicators of progressing maturation [23]. These structural and cellular benchmarks are typically visualized through immunofluorescence and immunohistochemistry, with ultrastructural validation achieved via electron microscopy to resolve synaptic vesicles, postsynaptic densities, and intercellular junctions at nanoscale resolution [23].
Table 1: Key Structural and Cellular Maturation Markers for Brain Organoid Assessment
| Assessment Category | Specific Marker | Cellular/Structural Significance | Detection Methods |
|---|---|---|---|
| Cortical Layer Identity | SATB2 | Upper-layer (II-IV) neurons | IF/IHC |
| TBR1 | Deep-layer (VI) neurons | IF/IHC | |
| CTIP2 | Layer V neurons | IF/IHC | |
| Neuronal Maturity | DCX, NeuroD1 | Immature neurons | IF/IHC, scRNA-seq |
| MAP2 | Mature neurons | IF/IHC, scRNA-seq | |
| NEUN (RBFOX3) | Mature neuronal nuclei | IF/IHC | |
| Neurotransmitter Identity | VGLUT1 | Glutamatergic neurons | IF/IHC |
| GAD65/67 | GABAergic neurons | IF/IHC | |
| Glial Cells | GFAP, S100β | Astrocytes | IF/IHC, scRNA-seq |
| MBP, O4 | Oligodendrocytes | IF/IHC, scRNA-seq | |
| Synaptic Maturation | SYB2 | Presynaptic vesicles | IF/IHC, EM |
| PSD-95 | Postsynaptic densities | IF/IHC, EM | |
| Barrier Structures | Aquaporin-4 | Glia limitans formation | IF/IHC |
| CD31/PDGFRβ | BBB-like units | IF/IHC |
Electrophysiological Functional Maturation
The functional maturation of brain organoids represents perhaps the most critical dimension for assessing their physiological relevance, particularly for modeling neurological disorders and screening neuroactive compounds [45] [60]. Electrophysiological maturation follows a developmental trajectory that can be quantitatively monitored using multiple complementary technologies [45]. Early weak spiking activity typically emerges around day 30-40 in culture, with progressive increases in mean spike rate and amplitude over subsequent months [45]. The appearance of synchronized burst firings after approximately 120 days represents a critical milestone, indicating the formation of functional cortical neural networks—a hallmark of synaptic signal propagation in the developing mammalian cortex [45].
Multiple electrode arrays (MEAs) have emerged as particularly valuable tools for functional assessment, enabling non-invasive, multi-site recording of extracellular field potentials across the organoid surface [45] [60]. This approach allows researchers to quantify key network parameters including spike rates, burst patterns, and synchronized oscillations that resemble early brain activity [61]. By day 161 in culture, organoids typically demonstrate highly interconnected neural networks with complex burst patterns characterized by network burst durations of approximately 985 ± 152 ms, spike numbers of 1700 ± 300 per network burst, and inter-burst intervals of 25.3 ± 4.2 seconds [45]. These functional benchmarks provide critical validation of network-level integration that complements structural and molecular assessments.
Table 2: Electrophysiological Maturation Timeline and Key Benchmarks in Cerebral Organoids
| Culture Period | Observed Electrophysiological Features | Network Characteristics | Associated Cellular Developments |
|---|---|---|---|
| Day 30-40 | Weak spontaneous spiking activity | Limited synchronization | Early neurogenesis, neural progenitor expansion |
| Day 60-99 | Increased mean spike rate and amplitude | Emerging local connectivity | Neuronal maturation, early synaptic formation |
| Day 120 | Synchronized burst firings (SBFs) | Functional cortical network formation | Astrocyte expansion, synaptic refinement |
| Day 160+ | Complex oscillatory dynamics | Highly interconnected networks | Diverse neuronal populations, glial maturation |
Quality Control Frameworks: Standardizing Organoid Assessment
Hierarchical Scoring Systems
The development of systematic quality control frameworks represents a critical advancement in addressing reproducibility challenges in brain organoid research [2] [8]. Recent initiatives have proposed comprehensive QC methodologies that evaluate organoids across five essential criteria: morphology, size and growth profile, cellular composition, cytoarchitectural organization, and cytotoxicity [2]. These frameworks employ hierarchical scoring systems that prioritize non-invasive assessments to efficiently exclude low-quality organoids before proceeding to more detailed analyses [2]. This approach streamlines the evaluation process while ensuring that only organoids meeting stringent quality thresholds advance to experimental applications.
The QC scoring system operates through a structured evaluation of each criterion on a scale from 0 (low quality) to 5 (high quality), with defined minimum thresholds that must be met for organoids to advance in the assessment pipeline [2]. The initial QC phase focuses on non-destructive parameters—morphology (assessing compactness, border integrity, and absence of cysts) and size/growth profile—enabling rapid pre-selection of suitable organoids before committing valuable resources to more intensive studies [2]. Organoids passing this initial screening then undergo comprehensive final QC incorporating the full spectrum of assessment criteria, including cellular composition (verifying expected neural populations), cytoarchitectural organization (evaluating structural integrity and layer formation), and cytotoxicity (measuring DNA damage and cell viability) [2]. This systematic approach minimizes observer bias and establishes quantitative standards that enhance cross-study comparability.
Experimental Validation of QC Frameworks
The robustness of these QC frameworks has been experimentally validated through controlled perturbation experiments [2]. Exposure of 60-day cortical organoids to graded doses of hydrogen peroxide successfully induced a range of quality outcomes, allowing researchers to demonstrate the system's sensitivity in discriminating organoid quality levels across the proposed criteria [2]. This validation approach confirms that the framework can reliably identify subtle quality variations, making it particularly valuable for disease modeling and toxicological studies where distinguishing pathological phenotypes from technical artifacts is essential [2].
The implementation of such standardized QC frameworks addresses a critical need in the field, where current assessment methods often lack standardization and face limitations in transposing conventional 2D culture analyses to complex 3D systems [2]. By providing clearly defined, quantitative benchmarks, these systems enable both academic and industrial researchers to establish consistent quality standards essential for reliable disease modeling, drug screening, and toxicological assessment [2] [5]. Furthermore, the adaptability of these frameworks allows for application across diverse organoid types and developmental timepoints, enhancing their utility as the field continues to evolve toward more complex model systems [2].
Methodological Approaches for Electrophysiological Profiling
Multi-Electrode Array (MEA) Platforms
Multi-electrode array technology has emerged as a cornerstone methodology for functional analysis of brain organoids, enabling non-invasive, long-term monitoring of network activity with high temporal resolution [45] [60]. MEA systems typically consist of 64-electrode arrays that detect extracellular field potential changes, providing spatial information about activity patterns across the organoid surface [45]. The application of MEAs to 3D organoids requires specialized approaches, as traditional planar MEA systems have limited recording distances (typically up to 60μm), primarily capturing activity from superficial neuronal layers [45]. Recent methodological adaptations have enabled reliable detection of spontaneous electrical activity in organoids as early as 34 days in culture, with progressive increases in spike rates and synchronization over subsequent months [45].
Critical methodological considerations for MEA analysis of organoids include the development of specialized recording protocols that accommodate the three-dimensional structure and suspension culture requirements of organoids [45]. The integration of BrainPhys medium—specifically formulated to enhance neuronal synaptic function and promote electrophysiological maturation—has proven particularly valuable for long-term culture and functional analysis [45]. This optimized culture condition supports the development of complex network properties including synchronized bursting and oscillatory dynamics that more closely resemble in vivo neural activity [45]. The analytical output from MEA recordings provides quantifiable metrics of network maturation, including mean spike rates, burst patterns, inter-spike intervals, and network synchronization indices that serve as functional benchmarks for quality assessment [45].
Complementary Electrophysiological Techniques
While MEA platforms provide excellent temporal resolution and network-scale analysis, a comprehensive electrophysiological assessment incorporates complementary techniques to capture different aspects of functional maturation [60]. Patch clamp electrophysiology offers unparalleled resolution for examining intrinsic membrane properties and synaptic transmission at the single-cell level, allowing researchers to characterize action potential kinetics, receptor properties, and synaptic currents in individual neurons within organoids [60]. However, this approach is technically challenging in 3D cultures and provides limited spatial sampling of network activity [60].
Calcium imaging represents another essential methodology, utilizing fluorescent indicators (such as GCaMP) to visualize dynamic calcium transients associated with neuronal and increasingly glial activity [23] [60]. This technique excels at mapping spatial patterns of activity across cell populations with single-cell resolution, though it sacrifices temporal resolution due to the inherently slow kinetics of calcium signaling and practical limitations of optical imaging systems [23] [60]. The emergence of microfluidic platforms compatible with live imaging has further enhanced calcium imaging applications by improving oxygenation and nutrient diffusion during recording sessions [60]. Together, these complementary methodologies provide a comprehensive assessment of functional maturation across cellular and network levels, each contributing unique insights to the overall quality control framework.
Integrated Workflow: From QC Framework to Functional Analysis
The synergistic relationship between quality control assessment and electrophysiological profiling establishes a robust integrated workflow for validating cerebral organoid models. This workflow begins with systematic quality control screening using non-invasive morphological and growth assessments, followed by more detailed histological and molecular characterization of cellular composition and cytoarchitectural organization. Organoids meeting established quality thresholds then advance to functional validation through comprehensive electrophysiological profiling, ultimately generating thoroughly characterized models suitable for disease modeling, mechanistic studies, or compound screening.
Essential Research Reagent Solutions for Organoid Assessment
The implementation of robust quality control and functional assessment protocols requires specific research reagents and specialized materials. The following table details key solutions essential for cerebral organoid generation, maturation, and comprehensive characterization.
Table 3: Essential Research Reagent Solutions for Organoid Assessment
| Reagent Category | Specific Examples | Function in Organoid Assessment | Application Context |
|---|---|---|---|
| Cell Culture Media | BrainPhys Medium | Enhances neuronal synaptic function and promotes electrophysiological maturation | Long-term functional cultures [45] |
| Extracellular Matrices | Matrigel, Synthetic ECM | Provides structural support and biomechanical cues for 3D organization | Organoid formation and patterning [60] [62] |
| Neural Induction Agents | SMAD inhibitors, Morphogens (FGF, EGF) | Directs neural differentiation and regional specification | Region-specific organoid generation [62] |
| Immunohistochemistry Reagents | Antibodies against SATB2, TBR1, CTIP2, GFAP, MAP2 | Enables visualization of cellular diversity and architectural organization | Structural and cellular QC assessment [23] [2] |
| Functional Imaging Reagents | Calcium indicators (GCaMP), Voltage-sensitive dyes | Reports neural and glial activity dynamics | Functional maturation assessment [23] [60] |
| Molecular Analysis Kits | scRNA-seq reagents, Bulk RNA-seq kits | Enables transcriptomic profiling and cellular heterogeneity assessment | Molecular validation of maturity [23] [27] |
| Viability Assays | Live/Dead staining, LDH cytotoxicity kits | Assesses cell viability and detects cytotoxic effects | Cytotoxicity evaluation in QC [2] |
Signaling Pathways in Organoid Maturation and Functional Development
The maturation of cerebral organoids and the emergence of functional electrical activity are governed by complex signaling pathways that coordinate neuronal development, circuit formation, and network refinement. Understanding these molecular pathways provides critical insights for both quality assessment and targeted maturation protocols.
The synergistic integration of advanced maturation benchmarking with comprehensive electrophysiological profiling represents a paradigm shift in cerebral organoid quality control. This multidimensional assessment approach addresses the fundamental reproducibility challenges that have limited the broader adoption of organoid technologies in both basic research and translational applications [2] [8]. By establishing standardized, quantitative frameworks that span structural, cellular, molecular, and functional dimensions, researchers can now more reliably distinguish biologically significant phenotypes from technical variability, enhancing the validity of disease modeling and drug screening applications [23] [2].
Looking forward, the continued refinement of these assessment frameworks will be essential as organoid technologies evolve toward greater physiological complexity through approaches such as vascularization, assembloid formation, and enhanced cellular diversity [23] [62]. The incorporation of advanced functional analyses, including more sophisticated MEA systems capable of three-dimensional recording and high-content calcium imaging, will further strengthen the validation of organoid models [60] [61]. Ultimately, these standardized assessment protocols provide the foundation for realizing the full potential of cerebral organoids as human-relevant platforms for understanding brain development, modeling neurological disorders, and accelerating the discovery of novel therapeutics [23] [5].
Scalability and Industrial Applicability for High-Throughput Screening
High-Throughput Screening (HTS) represents a foundational technology in modern drug discovery, enabling the rapid automated testing of hundreds of thousands of chemical or biological compounds against therapeutic targets [63]. This paradigm has largely replaced traditional "trial and error" approaches, allowing pharmaceutical companies and research institutions to accelerate early-stage research, reduce costs, and increase the likelihood of discovering new therapies [64] [65]. The global HTS market, valued at US$28.8 billion in 2024 and projected to reach US$50.2 billion by 2029, reflects its critical role in contemporary life sciences [65].
At its core, HTS leverages robotics, automated liquid handling, miniaturized assays, and advanced data analytics to identify potential drug candidates with unprecedented efficiency [63] [66]. The technology can process over 10,000 compounds per day—a dramatic improvement over traditional methods that might manage only 100 samples weekly [66]. This scalability has made HTS indispensable not only for drug discovery but also for toxicology, genomic and functional screening, and biologics development [64].
Scaling HTS Technologies: Platforms and Methodologies
Evolution of HTS Formats and Throughput
The scalability of HTS platforms has evolved significantly through miniaturization and automation, progressing from 96-well plates to 384-well and 1536-well formats that dramatically increase throughput while reducing reagent consumption [64]. This progression enables testing of increasingly large compound libraries while conserving valuable biological samples and chemical reagents.
Ultra-High Throughput Screening (uHTS) represents the current pinnacle of this scaling, capable of testing millions of compounds daily through advanced microfluidics and high-density microwell plates with volumes as small as 1-2μL [64] [65]. The transition to uHTS necessitates substantial infrastructure investments, including sophisticated robotic systems, specialized detection technologies, and advanced data management capabilities [64].
Table 1: Comparison of HTS Platform Capabilities and Scalability
| Attribute | Traditional HTS | uHTS | Industrial Application Considerations |
|---|---|---|---|
| Throughput | Up to 100,000 compounds/day | >300,000 compounds/day | Enables screening of diverse chemical libraries in practical timeframes |
| Format Complexity | 96-, 384-well plates | 1536-well plates, nanofluidic chips | Miniaturization reduces reagent costs but increases technical complexity |
| Automation Level | Robotic liquid handling | Fully integrated robotic systems | High initial investment offset by long-term efficiency gains |
| Data Generation | Terabytes per run | Petabyte-scale outputs | Requires sophisticated bioinformatics and cloud computing infrastructure |
| Technical Expertise | Moderate | Advanced | Demands specialized personnel for operation and data interpretation |
Advanced Detection Technologies for Scaled Applications
HTS detection methodologies have evolved to meet the demands of scaled screening campaigns. Fluorescence-based techniques remain predominant due to their high sensitivity, diverse available fluorophores, and adaptability to miniaturized formats [64] [67]. However, recent advances have expanded the technological toolbox to include luminescence, absorbance, mass spectrometry, and nuclear magnetic resonance approaches [64].
For cell-based HTS—particularly relevant for neuropharmacology—high-content screening (HCS) platforms combine automated microscopy with multiparametric image analysis to extract rich phenotypic information from each well [68]. These systems can investigate numerous cellular features simultaneously, including morphology, protein translocation, and viability markers, providing deeper biological insights alongside primary screening data [68].
Figure 1: High-Throughput Screening Workflow. This standardized process enables scalable drug discovery through automation and miniaturization.
Quality Control Frameworks for Complex Cellular Models
The Challenge of Organoid Variability in Screening
The adoption of three-dimensional cerebral organoids as physiologically relevant models for neuroscience research represents a significant advancement over traditional two-dimensional cultures [2]. These complex structures recapitulate aspects of human brain development, offering unprecedented opportunities for studying neurodevelopmental disorders, neurodegenerative diseases, and neurotoxicity [2] [8]. However, their inherent variability in morphology, size, cellular composition, and cytoarchitectural organization presents substantial challenges for HTS applications where reproducibility is essential [2].
This variability stems from the stochastic nature of stem cell differentiation and spontaneous self-organization processes, resulting in inconsistencies that compromise experimental reproducibility [2]. Without standardized quality assessment, organoid-based screening campaigns risk generating irreproducible data with poor translational potential—a critical concern for industrial drug discovery programs [2] [8].
Standardized QC Framework for Cerebral Cortical Organoids
To address these challenges, recent research has established a comprehensive Quality Control (QC) framework specifically designed for 60-day cortical organoids [2] [8]. This hierarchical scoring system evaluates five critical criteria:
- Morphology: Assessing compactness, border integrity, and absence of cysts through non-invasive imaging
- Size and Growth Profile: Ensuring proper developmental dynamics and appropriate dimensions
- Cellular Composition: Verifying presence of expected neural populations (neural progenitors, neurons, astrocytes)
- Cytoarchitectural Organization: Evaluating structural integrity and organization of neural layers
- Cytotoxicity: Detecting DNA damage and cell viability impairment [2]
The framework employs a two-stage assessment strategy with initial non-invasive QC (morphology and size) for pre-study organoid selection, followed by comprehensive post-study evaluation incorporating all criteria [2]. This approach efficiently excludes low-quality organoids before resource-intensive screening while providing standardized quality metrics for data interpretation.
Figure 2: Hierarchical Quality Control Framework for Cerebral Organoids. This standardized assessment ensures only high-quality organoids proceed to screening applications.
Experimental Protocols for HTS Hit Validation
Triage Strategies for Hit Confirmation
The transition from primary screening to validated hits requires rigorous experimental triage strategies to eliminate false positives and identify high-quality bioactive compounds [68]. This process begins with dose-response confirmation of primary hits, followed by specialized counter-screens and orthogonal assays that address specific artifact mechanisms [68].
Key components of this triage cascade include:
- Counter Screens: Designed to identify compounds that interfere with assay technology rather than the biological target, addressing issues such as autofluorescence, signal quenching, singlet oxygen quenching, and reporter enzyme modulation [68]
- Orthogonal Assays: Confirm bioactivity using different readout technologies or assay conditions, ensuring observed effects reflect genuine biological activity rather than assay-specific artifacts [68]
- Cellular Fitness Assays: Evaluate general toxicity or harm to cells, distinguishing specific bioactivity from nonspecific cytotoxic effects [68]
These validation steps are particularly crucial for complex cell-based systems like cerebral organoids, where multiple confounding factors can contribute to false positive signals [68] [2].
Specialized Methodologies for Organoid-Based Screening
Screening campaigns utilizing cerebral organoids require adaptation of standard HTS protocols to accommodate the complexity and three-dimensional nature of these models [2]. Essential methodological considerations include:
Sample Preparation and Handling
- Gentle dissociation protocols that preserve cellular integrity while enabling single-cell analysis
- Specialized fixation and sectioning techniques for 3D structures
- Permeabilization methods that ensure uniform antibody penetration for immunohistochemistry
Image Acquisition and Analysis
- Automated microscopy systems with enhanced depth of field for 3D samples
- Multiplexed fluorescent staining protocols for simultaneous evaluation of multiple cell types
- High-content analysis algorithms capable of extracting quantitative data from complex organoid architectures
Viability and Cytotoxicity Assessment
- Adaptation of standard viability assays (CellTiter-Glo, MTT) for 3D culture systems
- Specialized dyes for evaluating membrane integrity (TO-PRO-3, YOYO-1) in thick tissues
- Multiparametric cytotoxicity assessment using high-content imaging [68] [2]
Essential Research Reagent Solutions
The successful implementation of scalable HTS platforms, particularly for complex models like cerebral organoids, depends on specialized research reagents and materials. The table below details critical components and their functions in HTS workflows.
Table 2: Essential Research Reagent Solutions for HTS with Complex Cellular Models
| Reagent Category | Specific Examples | Function in HTS Workflow | Considerations for Scalability |
|---|---|---|---|
| Compound Libraries | ChemBridge, ChemDiv, National Cancer Institute libraries | Provide diverse chemical matter for screening campaigns | Quality control, structural diversity, and drug-like properties critical for success [63] |
| Cell Culture Platforms | Transwell HTS Systems, UniMat permeable membrane platforms | Support 3D organoid growth with enhanced uniformity and maturity | Geometrical constraints improve reproducibility; permeability supports nutrient exchange [63] [69] |
| Detection Reagents | Fluorescent dyes (DAPI, MitoTracker), luminescent substrates (CellTiter-Glo) | Enable quantitative measurement of biological activities | Compatibility with automation, stability under screening conditions, minimal interference [68] |
| Assay Kits | LDH cytotoxicity assays, caspase activity kits, metabolic flux assays | Provide standardized protocols for specific biological readouts | Validation in miniaturized formats, batch-to-batch consistency, well-characterized performance [68] |
| Extracellular Matrices | Matrigel, synthetic hydrogels, self-assembling peptides | Support 3D architecture and cell-matrix interactions | Lot-to-lot consistency, definition, and scalability of production [2] [69] |
Comparative Analysis of HTS Platforms for Organoid Research
Platform Performance Metrics
The selection of appropriate HTS platforms for cerebral organoid research requires careful consideration of multiple performance parameters. The table below provides a comparative analysis of currently available technologies.
Table 3: Performance Comparison of HTS Platforms for Cerebral Organoid Applications
| Platform Type | Max Throughput (compounds/day) | 3D Compatibility | Data Richness | Specialized Requirements | Best-Suited Applications |
|---|---|---|---|---|---|
| Conventional HTS | 10,000-100,000 | Moderate (adapted protocols) | Low to moderate (single endpoints) | Standard liquid handling robotics | Primary screening of large compound libraries |
| uHTS | >300,000 | Low (geometric constraints) | Low (single endpoints) | Specialized nanofluidics, high-density detection | Massive library screening, fragment-based discovery |
| High-Content Screening | 1,000-50,000 | High (imaging-based) | High (multiparametric) | Automated microscopy, advanced image analysis | Phenotypic screening, mechanism of action studies |
| Organoid-Specialized Platforms | 5,000-20,000 | High (optimized for 3D) | Moderate to high | Specialized culture vessels (UniMat), perfusion systems | Disease modeling, neurotoxicity assessment [2] [69] |
Industrial Applicability and Scalability Assessment
Translation of HTS technologies from academic research to industrial applications demands robust, reproducible systems capable of operating at scale. The UniMat platform represents a significant advancement, demonstrating improved uniformity and maturity in kidney organoid production while maintaining compatibility with standard culture workflows [69]. This platform incorporates 3D geometrically-engineered permeable membranes that provide physical constraints for uniformity while permitting unhindered nutrient exchange—a critical combination for scalable organoid production [69].
For cerebral cortical organoids, the implementation of standardized QC frameworks enables objective quality assessment essential for industrial adoption [2] [8]. By establishing quantitative thresholds for key quality parameters, these systems reduce observer bias and enhance reproducibility across batches and research groups—fundamental requirements for pharmaceutical screening campaigns [2].
The integration of advanced HTS technologies with physiologically relevant cerebral organoid models represents a powerful approach for neuroscience drug discovery. However, realizing the full potential of this combination requires strategic implementation of quality control frameworks and careful platform selection based on specific research objectives.
Successful scaling of HTS for complex cellular models depends on:
- Standardized quality assessment protocols that ensure model system reproducibility
- Appropriate technology matching between screening platforms and biological questions
- Rigorous hit validation strategies that address the specific challenges of complex phenotypic screens
- Specialized reagent solutions optimized for 3D culture systems and automated workflows
As the field continues to evolve, emerging innovations in AI-driven screening design, organoid engineering, and microfluidic integration promise to further enhance the scalability and industrial applicability of HTS platforms. These advancements will progressively address current limitations in throughput, reproducibility, and physiological relevance, strengthening the role of HTS as a cornerstone technology for neuroscience drug discovery.
Conclusion
The development of a standardized, hierarchical QC framework for cerebral cortical organoids marks a significant advancement toward achieving reproducibility and reliability in organoid-based research. By integrating both non-invasive initial screening and comprehensive final assessment, this methodology provides a robust system for classifying organoid quality, effectively addressing the critical challenge of variability that has hindered broader adoption. The framework's successful validation through controlled stress tests confirms its sensitivity and reliability. Looking forward, the integration of functional maturity assessments—such as electrophysiological profiling with high-density MEAs and advanced maturity benchmarking—will be crucial for creating organoids that more fully recapitulate adult brain functionality. This evolution in quality control promises to accelerate the translation of cerebral organoid technology, enhancing its value in disease modeling, neurotoxicity testing, and the drug discovery pipeline, ultimately bridging the gap between in vitro models and human neuropathology.
References
- 1. Human brain organoids: an innovative model for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12417440/]
- 2. Towards a quality control framework for cerebral cortical ... [https://www.nature.com/articles/s41598-025-14425-x]
- 3. Quantitative profiling of human brain organoid cell diversity ... [https://www.sciencedirect.com/science/article/pii/S2211124725009398]
- 4. Deciphering brain organoid heterogeneity by identifying key ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12488937/]
- 5. New Quality Control Framework for Cerebral Organoids ... [https://www.pcrm.org/news/innovative-science/new-quality-control-framework-cerebral-organoids-can-help-scale-their]
- 6. Brain Organoid Computing – an Overview [https://arxiv.org/html/2503.19770v1]
- 7. Rigor and reproducibility in human brain organoid research [https://www.sciencedirect.com/science/article/pii/S2213671124001140]
- 8. Towards a quality control framework for cerebral cortical ... [https://netri.com/towards-a-quality-control-framework-for-cerebral-cortical-organoids-netris-new-publication-in-nature-scientific-reports/]
- 9. Examining the Market for Organoid Culture Systems in 2025 [https://eureka.patsnap.com/report-examining-the-market-for-organoid-culture-systems-in-2025]
- 10. Validation of non-destructive morphology-based selection ... [https://www.sciencedirect.com/science/article/pii/S2213671124002674]
- 11. MOrgAna: accessible quantitative analysis of organoids ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8451065/]
- 12. Protocol for generating human cortical organoids enriched ... [https://www.sciencedirect.com/science/article/pii/S2666166724004453]
- 13. Viability Analysis and High-Content Live-Cell Imaging for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10216787/]
- 14. Protocol for drug screening of patient-derived tumor ... [https://www.sciencedirect.com/science/article/pii/S2666166722002878]
- 15. Evaluation Models & Applications of Drug Neurotoxicity [https://neuros.creative-biolabs.com/evaluation-models-and-applications-of-drug-neurotoxicity.htm]
- 16. Regulatory momentum builds for organoid neurotoxicity ... [https://www.fiercebiotech.com/sponsored/regulatory-momentum-builds-organoid-neurotoxicity-testing]
- 17. Organoid intelligence for developmental neurotoxicity testing [https://www.frontiersin.org/journals/cellular-neuroscience/articles/10.3389/fncel.2024.1480845/full]
- 18. Neural organoids as advanced tools for neurotoxicity modeling [https://pmc.ncbi.nlm.nih.gov/articles/PMC12281010/]
- 19. Everything You Always Wanted to Know About Organoid ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9233279/]
- 20. Mapping the Trajectory of Organoid Research: Insights into ... [https://www.sciencedirect.com/science/article/pii/S2666138125000179]
- 21. Emerging brain organoids: 3D models to decipher, identify ... [https://www.sciencedirect.com/science/article/pii/S2452199X25000258]
- 22. Brain organoid protocols and limitations [https://www.frontiersin.org/journals/cellular-neuroscience/articles/10.3389/fncel.2024.1351734/full]
- 23. Navigating Brain Organoid Maturation: From Benchmarking ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12383812/]
- 24. Mapping the future of organoids [https://www.roche.com/stories/mapping-the-future-of-organoids]
- 25. Towards a quality control framework for cerebral cortical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12340003/]
- 26. A beginner's guide on the use of brain organoids for ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-023-03302-x]
- 27. Organoid assessment technologies - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC10731122/]
- 28. Minimum Information about Organoid Research (MIOR) [https://www.sciopen.com/article/10.26599/CO.2025.9410020]
- 29. 2025 Trends: Organoids [https://www.genengnews.com/topics/genome-editing/2025-trends-organoids/]
- 30. Noninvasive, label-free image approaches to predict ... [https://www.nature.com/articles/s41598-024-66591-z]
- 31. Morphological heterogeneity description enabled early and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8801074/]
- 32. Breakthroughs and challenges of organoid models for ... [https://www.nature.com/articles/s41420-025-02505-w]
- 33. Brain organoids: a new paradigm for studying human ... [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2025.1699814/full]
- 34. A simplified protocol for the generation of cortical brain ... [https://www.frontiersin.org/journals/cellular-neuroscience/articles/10.3389/fncel.2023.1114420/full]
- 35. Human cerebral organoids maintain integrity and viability ... [https://www.sciencedirect.com/science/article/pii/S2949921625000213]
- 36. Brain Organoids or Cerebral Derived from hPSCs Organoids [https://www.stemcell.com/technical-resources/area-of-interest/organoid-research/neural-organoids/overview.html]
- 37. Morphodynamics of human early brain organoid ... [https://www.nature.com/articles/s41586-025-09151-3]
- 38. Brain organoids: building higher-order complexity and neural ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12276974/]
- 39. Challenges of Organoid Research - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC10559943/]
- 40. Reliability of high-quantity human brain organoids for ... [https://www.nature.com/articles/s41467-024-55226-6]
- 41. Why Do Organoids Fail to Grow After Passaging? [https://www.antbioinc.com/blogs/technical-articles/why-do-organoids-fail-to-grow-after-passaging]
- 42. Quantitative profiling of human brain organoid cell diversity ... [https://pubmed.ncbi.nlm.nih.gov/40864552/]
- 43. Organoid Culture Guide [https://www.atcc.org/resources/culture-guides/organoid-culture-guide]
- 44. A mouse organoid platform for modeling cerebral cortex ... [https://www.sciencedirect.com/science/article/abs/pii/S1534580725005015]
- 45. Electrophysiological Maturation of Cerebral Organoids ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7562943/]
- 46. Electrophysiological Properties of Human Cortical Organoids [https://pmc.ncbi.nlm.nih.gov/articles/PMC8888527/]
- 47. Brain organoid maturation and implantation integration ... [https://www.sciencedirect.com/science/article/pii/S2090123224003783]
- 48. A feedback-driven brain organoid platform enables ... [https://www.sciencedirect.com/science/article/pii/S2542660525001854]
- 49. AI-enabled organoids: Construction, analysis, and ... [https://www.sciencedirect.com/science/article/pii/S2452199X23002761]
- 50. AI in Biotech: 2025 Trends, Discoveries, and Game-Changing ... [https://blackthorn.ai/blog/ai-in-biotech-trends-discoveries-and-game-changing-technologies/]
- 51. Inside ELRIG's Drug Discovery 2025 [https://www.drugtargetreview.com/article/190237/inside-elrigs-drug-discovery-2025/]
- 52. Cross-site reproducibility of human cortical organoids ... [https://www.sciencedirect.com/science/article/pii/S2213671124002170]
- 53. Harnessing artificial intelligence for brain disease: advances ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12336123/]
- 54. High-throughput Assays for Superoxide and Hydrogen ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4047388/]
- 55. Validation of non-destructive morphology-based selection ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11589179/]
- 56. Validation of non-destructive morphology-based selection ... [https://pubmed.ncbi.nlm.nih.gov/39393360/]
- 57. Seeing is believing: Leveraging defining morphological ... [https://www.cira.kyoto-u.ac.jp/e/pressrelease/news/241202-100000.html]
- 58. Three-Dimensional Morphological Characterisation of ... [https://www.mdpi.com/2674-1172/4/1/1]
- 59. Human organoids: New strategies and methods for ... [https://www.sciencedirect.com/science/article/pii/S0092867422008443]
- 60. Electrophysiological Analysis of Brain Organoids [https://www.frontiersin.org/journals/neuroscience/articles/10.3389/fnins.2020.622137/full]
- 61. Review The emergence of electrical activity in human brain ... [https://www.sciencedirect.com/science/article/pii/S221367112500236X]
- 62. Emerging approaches to enhance human brain organoid ... [https://www.sciencedirect.com/science/article/pii/S096289242400254X]
- 63. What Is High Throughput Screening? [https://www.corning.com/worldwide/en/products/life-sciences/resources/stories/at-the-bench/what-is-high-throughput-screening-and-how-is-it-evolving.html]
- 64. High-Throughput Screening - Drug Discovery [https://www.technologynetworks.com/drug-discovery/articles/high-throughput-screening-principles-applications-and-advancements-405121]
- 65. How Is High Throughput Screening Transforming Drug ... [https://www.marketsandmarkets.com/blog/HC/high-throughput-screening]
- 66. High-Throughput Screening (HTS): A Simple Guide ... [https://www.chemcopilot.com/blog/hts-material-discovery]
- 67. High-Throughput Screening Platforms in the Discovery of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7926814/]
- 68. Brief Guide: Experimental Strategies for High-Quality Hit ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8293735/]
- 69. Scalable production of uniform and mature organoids in a ... [https://www.nature.com/articles/s41467-024-53073-z]