Advanced Time-Lapse Microscopy in Mouse Embryo Culture: Protocols, 3D Imaging, and AI-Driven Analysis
This article provides a comprehensive overview of advanced time-lapse microscopy (TLM) applications in mouse embryo culture, a critical model for developmental biology and drug discovery.
Advanced Time-Lapse Microscopy in Mouse Embryo Culture: Protocols, 3D Imaging, and AI-Driven Analysis
Abstract
This article provides a comprehensive overview of advanced time-lapse microscopy (TLM) applications in mouse embryo culture, a critical model for developmental biology and drug discovery. It explores the foundational principles of morphokinetic analysis, detailing optimized protocols for lattice light-sheet and optical coherence microscopy to minimize photodamage while capturing high-resolution 3D data. The content covers practical methodologies for long-term imaging, troubleshooting common issues like embryo mounting and culture stability, and validates TLM through comparative analysis with conventional methods. Finally, it examines the emerging integration of artificial intelligence for automated embryo evaluation, offering researchers a validated framework to enhance reproducibility and predictive accuracy in preimplantation development studies.
Unveiling Embryonic Dynamics: Core Principles of Time-Lapse Morphokinetics
Defining Key Morphokinetic Parameters in Mouse Embryo Development
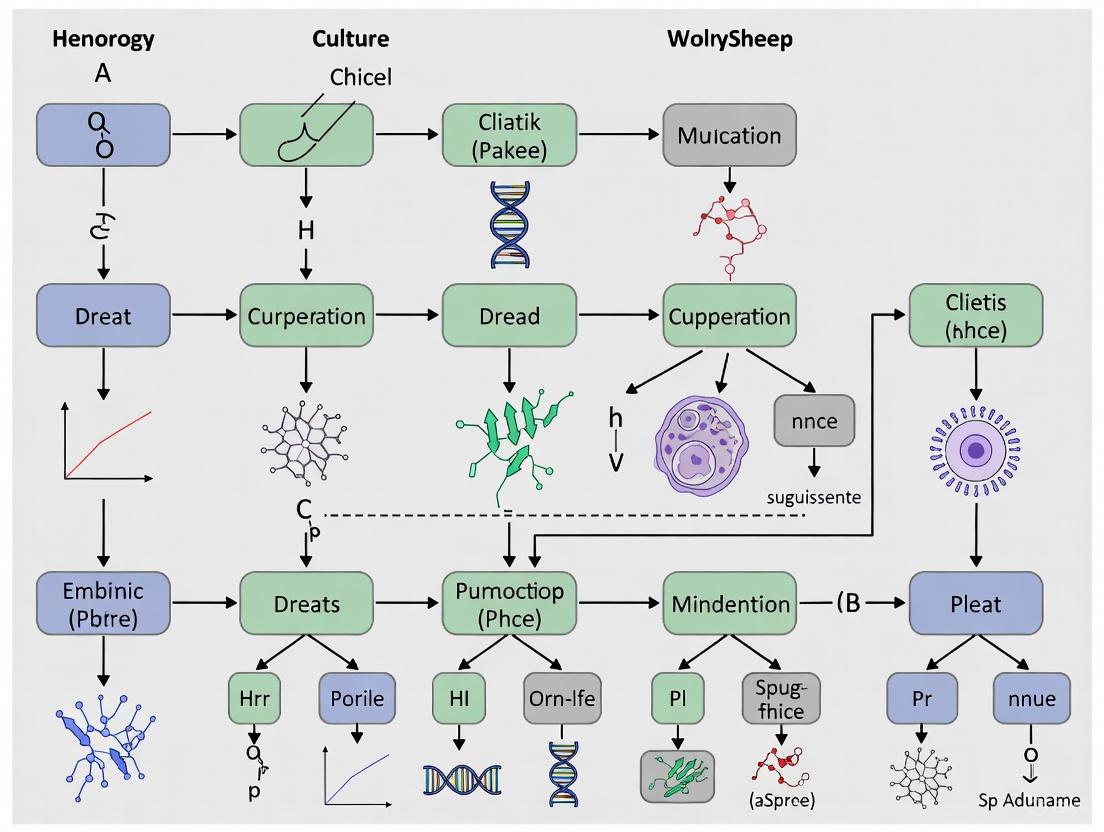
In the field of developmental biology and reproductive medicine, the non-invasive assessment of embryo quality is paramount for improving success rates in assisted reproductive technologies. Mouse embryos serve as a powerful model system due to their genetic and developmental similarity to humans [1]. The advent of time-lapse microscopy (TLM) has revolutionized this field by enabling the continuous monitoring of preimplantation development, allowing researchers to extract quantitative morphokinetic parameters that serve as predictive biomarkers of embryonic viability and developmental potential [2]. This Application Note details the key morphokinetic parameters defining mouse embryo development and provides standardized protocols for their measurement, presenting critical insights for researchers and drug development professionals working within the broader context of time-lapse microscopy mouse embryo culture research.
Key Morphokinetic Parameters in Mouse Embryo Development
Morphokinetics refers to the quantitative temporal analysis of the dynamic events that occur during embryonic development. The accurate measurement of these parameters provides a powerful, non-invasive method for evaluating embryo quality and predicting developmental outcomes.
Definition and Measurement of Core Parameters
Table 1: Core Morphokinetic Parameters in Mouse Embryo Development
| Developmental Stage | Morphokinetic Parameter | Symbol | Definition | Typical Time Range (hours post-insemination) | Significance |
|---|---|---|---|---|---|
| Fertilization | Time to pronuclei fading | tPNf | Time at which male and female pronuclei become no longer visible | ~6-24 h | Marks the end of the fertilization process and onset of first cleavage [2] |
| Cleavage Stages | Time to 2-cell stage | t2 | Time at which the first cleavage occurs, resulting in a 2-cell embryo | ~24-48 h | Initial cleavage event; delays can indicate suboptimal conditions [3] |
| Time to 3-cell stage | t3 | Time at which the embryo reaches the 3-cell stage | ~36-60 h | Asymmetric early cleavage [2] | |
| Time to 4-cell stage | t4 | Time at which the embryo reaches the 4-cell stage | ~38-62 h | Completion of second cleavage [2] | |
| Time to 5-cell stage | t5 | Time at which the embryo reaches the 5-cell stage | ~40-65 h | Start of more asynchronous divisions [2] | |
| Time to 8-cell stage | t8 | Time at which the embryo reaches the 8-cell stage | ~45-70 h | Precedes the onset of compaction [2] | |
| Compaction | Time to Morula | tM | Time at which compaction is initiated (>80% fading of cell membranes) | ~55-80 h | Critical for cell differentiation and formation of a cohesive cell mass [2] |
| Blastocyst Formation | Time to start of blastulation | tSB | Time at which a small pocket of blastocoel first becomes visible | ~70-90 h | Initiation of cavitation and blastocoel formation [2] |
| Time to Blastocyst | tB | Time at which the blastocoel fills ≥50% of the embryo volume | ~75-95 h | Formation of a true blastocyst [2] | |
| Time to Expanded Blastocyst | tEB | Time at which the blastocyst expands and the zona pellucida thins | ~80-100 h | Embryo preparation for hatching [2] |
The precise timing of these events is highly sensitive to both intrinsic factors (e.g., maternal age, genetic background) and extrinsic factors (e.g., culture conditions, oxygen tension) [2] [3]. For instance, the second (t2 to t3) and third (t3 to t4) embryonic cell cycles have been specifically indicated to be associated with blastocyst formation and subsequent hatching capability [1].
The Impact of Experimental Conditions on Morphokinetics
Morphokinetic parameters are not fixed but are influenced by the experimental environment. Oxygen concentration during culture is a critical factor. Studies have demonstrated that embryos cultured under atmospheric oxygen (~20%) show significant developmental delays compared to those cultured under physiological oxygen levels (~5%). Specifically, delays of 0.45 hours at the first cleavage and 0.84 hours at the second cleavage have been recorded, along with significantly prolonged second and third cell cycles [3].
Maternal aging is another key factor influencing developmental pace. Recent research utilizing machine learning for analysis revealed that embryos from aged maternal mice (10-14 months old) accelerated through cleavage stages from the 5-cell stage to the morula stage compared to embryos from young mothers (3-4 weeks old) [2]. This finding highlights how morphokinetics can capture subtle, phenotype-associated alterations in developmental programs.
Detailed Experimental Protocols
Protocol 1: Time-Lapse Imaging of Mouse Embryos using a Dual-Modality OCM/BF System
This protocol describes a method for acquiring time-lapse 3D Optical Coherence Microscopy (OCM) images with co-registered bright-field (BF) imaging, providing high-resolution structural and morphokinetic data [1].
The Scientist's Toolkit: Research Reagent Solutions
| Item Category | Specific Product/Model | Function/Application |
|---|---|---|
| Imaging System | Custom OCM/BF System with 20X objective [1] | Provides high-resolution (axial: ~2.1 μm, lateral: ~1.0 μm) 3D imaging of embryo microstructures. |
| Cell Culture Incubator | Heracell VIOS 160i [1] | Maintains optimal culture conditions (e.g., 37°C, 5% O₂, 6% CO₂) during long-term imaging. |
| Embryo Culture Dish | IVF store V005001 dish [1] | Specialized dish for culturing and imaging up to 25 embryos simultaneously. |
| Culture Medium | Potassium Simplex Optimization Medium (KSOM) [2] | A defined medium optimized for supporting preimplantation mouse embryo development in vitro. |
| Mouse Strain | C57Bl6/NJ [2] | A commonly used inbred strain for embryological studies due to well-characterized genetics. |
| Hormones for Superovulation | Pregnant Mare Serum Gonadotropin (PMSG) & Human Chorionic Gonadotropin (hCG) [2] | Used to hormonally stimulate female mice to produce a larger number of synchronized oocytes. |
Workflow Diagram: Dual-Modality Embryo Imaging Setup
Step-by-Step Procedure:
- Animal Preparation and Oocyte Harvest: Superovulate 6-week-old F1 hybrid (C57BL/6 × CBA/Ca) or C57Bl6/NJ female mice with intraperitoneal injections of 5 IU PMSG, followed by 5 IU hCG 48 hours later. Mate females with F1 males immediately after hCG injection. Harvest pronucleate oocytes approximately 16-18 hours post-hCG by dissecting the oviducts and collecting cumulus-oocyte complexes in HTF media. Remove cumulus cells enzymatically or mechanically [1] [2] [3].
- Embryo Culture and Loading: Wash fertilized zygotes and transfer them into a specialized embryo imaging dish (e.g., IVF store V005001) containing 20 µL of pre-equilibrated KSOM medium. Culture up to 25 embryos per dish under mineral oil to prevent evaporation [1] [2].
- System Setup and Imaging: Place the loaded imaging dish into the custom-built, compact dual-modality OCM/BF imaging system housed within the incubator. Set the environmental controls to maintain 37°C, 5% O₂, and 6% CO₂. Configure the automated acquisition software to capture co-registered 3D OCM and BF images at each well location every 10 minutes for the entire culture duration (up to 150 hours, until the hatched blastocyst stage) [1].
- Data Acquisition and Processing: The automated pipeline uses prior knowledge of well locations and image-guided auto-tracking to center each embryo for imaging. Acquired 3D OCM datasets provide structural details like nuclei location and size, while BF images offer conventional morphological context. Data is automatically transferred to a storage server for subsequent analysis [1].
Protocol 2: Morphokinetic Annotation and Analysis using Machine Learning
This protocol outlines the process for annotating developmental time points and applying machine learning algorithms to identify patterns and predict phenotypes based on morphokinetic data [2].
Workflow Diagram: Morphokinetic Analysis and ML Classification
Step-by-Step Procedure:
- Image Export and Annotation: Export time-lapse images from the culture system's software (e.g., EmbryoViewer) at the plane of best focus. A trained annotator, blinded to the experimental groups, then manually reviews the images and records the exact time (in hours post-insemination) at which each embryo reaches the 14 defined morphokinetic milestones: tPNf, t2, t3, t4, t5, t6, t7, t8, t9, tM, tSB, tB, and tEB [2].
- Data Preprocessing: Assemble the annotated timings into a structured data table. Exclude embryos that arrest prior to blastocyst formation from the machine learning analysis. Encode the outcome variable (e.g., Young vs. Aged maternal phenotype) as a binary variable (0/1) [2].
- Unsupervised Machine Learning: To explore inherent patterns without prior assumptions, use unsupervised clustering algorithms. The Normal Mixtures model, using the Expectation-Maximization (EM) algorithm, can be applied to the pooled morphokinetic data from all cohorts to identify distinct developmental clusters [2].
- Supervised Machine Learning: Split the curated dataset into a training set (70%) and a test set (30%). Train a supervised classification model, such as the Extreme Gradient Boosting (XGBoost) algorithm, on the training set using the morphokinetic time points as predictors and the maternal age group as the outcome. Perform hyperparameter tuning using 5-fold cross-validation on the training data to optimize model performance [2].
- Model Evaluation and Validation: Finally, evaluate the predictive performance of the tuned model on the held-out 30% test set. Report standard performance metrics such as accuracy, precision, and recall to validate the model's ability to differentiate embryos based on morphokinetic phenotypes [2].
Advanced Analytical Tools and Future Directions
Software for 3D Image Analysis
The analysis of complex 3D and 4D imaging data requires sophisticated software tools.
- GIANI (General Image Analysis of Nuclei-based Images): This open-source FIJI plugin is designed for automated segmentation of nuclei and cells in 3D microscopy images. It facilitates batch-processing and quantification of morphology and protein expression, and has been validated for use with mouse embryo datasets [4].
- IN Carta Image Analysis Software: A commercial platform that utilizes advanced artificial intelligence and deep learning (via its SINAP module) to solve complex image analysis problems. It provides user-friendly workflows for 2D, 3D, and 4D experiments, enabling robust segmentation and phenotypic analysis of complex structures like embryos [5].
Integration with Lineage Tracing and Molecular Biology
Future directions in morphokinetic research involve the integration of dynamic imaging with single-cell molecular biology. Techniques such as CRISPR-based DNA barcoding for lineage tracing allow for the reconstruction of cell lineage relationships over time, providing a historical record of cell divisions within an embryo [6] [7]. When combined with the morphological and temporal data provided by TLM, this creates a powerful multi-modal platform for understanding how cell lineage and signaling interactions (e.g., Notch and Wnt pathways) dictate cell fate, size asymmetry, and overall embryonic architecture [8]. This integrated approach is poised to significantly advance our understanding of the fundamental principles governing mammalian embryogenesis.
The Scientific Rationale for Continuous, Non-Invasive Embryo Monitoring
In the fields of developmental biology and drug discovery, the ability to accurately monitor embryonic development is paramount. Traditional embryo assessment methods rely on static observations at isolated time points, necessitating the removal of embryos from stable culture conditions and providing only limited snapshots of a dynamic process [9] [10]. Time-lapse microscopy (TLM) has emerged as a transformative technology that enables continuous, non-invasive monitoring of embryos throughout critical developmental stages without disturbing their culture environment [9] [11] [12]. This Application Note details the scientific rationale for implementing continuous embryo monitoring systems and provides detailed protocols for their application in mouse embryo research, specifically focusing on the quantification of cell cycle dynamics during preimplantation development.
The core advantage of TLM lies in its capacity to generate rich morphokinetic datasets that capture the precise timing of developmental events and subtle morphological changes invisible to conventional endpoint analysis [11]. This capability is particularly valuable for research aimed at understanding the fundamental mechanisms driving embryogenesis, as well as for toxicology and drug discovery screens where developmental abnormalities serve as critical indicators of compound effects [13] [14]. By maintaining optimal culture conditions while collecting temporal data, TLM bridges a critical methodological gap, enabling researchers to correlate dynamic cellular processes with functional outcomes.
Key Advantages Over Traditional Methods
The implementation of continuous monitoring addresses several significant limitations inherent in conventional embryo culture and assessment techniques.
Maintenance of Optimal Culture Conditions: Traditional morphological assessment requires removing embryos from incubators for daily observations under a microscope. This repeated movement exposes embryos to fluctuations in temperature, pH, and gas concentrations, potentially inducing cellular stress and compromising developmental potential [9] [10]. In contrast, TLM systems integrate microscopy within the incubator environment, eliminating the need for physical disturbance and maintaining culture stability throughout the experimental timeframe [12] [10].
Access to Dynamic Morphokinetic Parameters: Static evaluation provides isolated snapshots of development, missing critical transient events and the precise timing of cell divisions [10]. TLM enables the documentation of key developmental milestones, such as the exact timing of cleavage divisions, synchronicity of cell cycles, and occurrence of abnormal cleavage patterns like direct or reverse cleavage, which are associated with reduced embryo viability [11] [12]. This dynamic profiling facilitates the creation of predictive models of developmental competence.
Enhanced Objectivity and Reproducibility: Visual embryo grading by researchers is inherently subjective, leading to inter- and intra-observer variability [10] [15]. TLM provides an objective, permanent record of embryonic development, allowing for retrospective analysis and standardized assessment using defined algorithms, thereby enhancing the consistency and reproducibility of experimental findings [9] [10].
Table 1: Comparison of Embryo Monitoring Methodologies
| Feature | Conventional Static Monitoring | Time-Lapse Microscopy (TLM) |
|---|---|---|
| Culture Stability | Disrupted during removal for observation | Maintained uninterrupted |
| Data Type | Static snapshots at limited time points | Continuous, dynamic morphokinetics |
| Event Detection | Misses transient, rapid events | Captures timing of all visible events |
| Subjectivity | Higher (dependent on embryologist) | Lower (algorithm-based analysis) |
| Workflow Efficiency | Manual, time-consuming | Automated, high-throughput potential |
| Primary Application | Routine morphology assessment | Research, kinetic profiling, toxicology |
Quantitative Morphokinetic Parameters for Mouse Embryos
Continuous monitoring generates quantitative data on the timing of key developmental events. The following parameters are critical for evaluating the developmental kinetics and health of preimplantation mouse embryos in research settings.
Table 2: Key Morphokinetic Parameters in Mouse Preimplantation Development
| Developmental Stage | Parameter | Description | Research Significance |
|---|---|---|---|
| Cleavage Stages | t2-t8 | Time to reach 2-cell, 3-cell, ..., 8-cell stage | Indicates cleavage speed and synchrony |
| S1 (t3-t2) | Duration of 2-cell stage | Cell cycle regulation | |
| S2 (t4-t3) | Duration of 3-cell stage | Cell cycle regulation | |
| Compaction | tM | Time to start of compaction | Initiation of cell-cell adhesion |
| Cavitation | tC | Time to start of blastocoel formation | Initiation of fluid transport |
| Blastocyst | tB | Time to full blastocyst formation | Overall developmental competence |
| tEB | Time to expanding blastocyst | Developmental pace and health |
Protocol: Imaging and Quantifying CDK Activity in Single Mouse Embryo Cells
This protocol, adapted from recent methodology, details the steps for quantifying Cyclin-Dependent Kinase (CDK) activity dynamics in single cells of living mouse embryos using a fluorescent biosensor and time-lapse confocal microscopy [16] [17]. CDK activity is a fundamental regulator of the cell cycle, and understanding its dynamics is crucial for research on embryonic development and cell fate decisions.
Experimental Workflow
The diagram below illustrates the complete experimental workflow for this protocol, from embryo collection to data analysis.
Materials and Reagents
Table 3: Essential Research Reagent Solutions
| Item | Function/Description | Example/Note |
|---|---|---|
| Mouse Embryos | Research subject for pre-implantation development | Morula or blastocyst stage [16] [17]. |
| CDK Activity Biosensor | FRET-based or translocation biosensor to report CDK activity | Enables quantification of activity dynamics in single living cells [16]. |
| Culture Medium | Supports embryo development during imaging | e.g., KSOM or M16, pre-equilibrated [17]. |
| Imaging Chamber | Provides controlled environment for live imaging | Glass-bottom dish or specialized chamber maintaining 37°C, 5% CO2 [16]. |
| Confocal Microscope | High-sensitivity system for fluorescent time-lapse imaging | Must be equipped with environmental control [16] [17]. |
| Image Analysis Software | For segmentation and fluorescence quantification | e.g., Cellpose for segmentation [16], custom scripts for ratio calculation. |
Step-by-Step Procedure
Part I: Embryo Isolation and Culture
- Isolation: Collect morula or blastocyst stage embryos from pregnant mice using standard surgical and flushing techniques [17].
- Culture: Place embryos in pre-equilibrated KSOM medium under mineral oil in a standard incubator (37°C, 5% CO2) for a recovery period of 1-2 hours before imaging [17].
- Chamber Preparation: Transfer a small group of embryos (5-10) into a glass-bottom imaging chamber containing pre-warmed culture medium. Ensure the chamber is suitable for maintaining temperature and gas pH throughout the experiment.
Part II: Time-Lapse Confocal Microscopy Setup
- Microscope Configuration: Turn on the confocal microscope and its environmental chamber, allowing it to stabilize at 37°C and 5% CO2 for at least one hour prior to imaging.
- Biosensor Imaging: Set the appropriate laser lines and detection channels for the specific CDK biosensor being used (e.g., CFP and YFP for a FRET-based sensor) [16].
- Acquisition Parameters: Define the imaging protocol:
- Interval: Set acquisition every 15-20 minutes.
- Duration: Program the experiment to run for 12-48 hours to cover multiple cell cycles.
- Z-stacks: Acquire optical sections (e.g., 5-10 slices at 5-10µm intervals) to capture the entire volume of each embryo at each time point.
- Laser Power: Use the lowest possible laser intensity to minimize phototoxicity while achieving a sufficient signal-to-noise ratio.
Part III: Image Analysis and Data Quantification
- Cell Segmentation: Process the time-lapse image series using a segmentation tool like Cellpose [16] to automatically identify and outline individual nuclei and cell boundaries in each frame.
- Fluorescence Quantification: For each cell and time point, measure the mean fluorescence intensity of the biosensor in the nuclear region and the cytoplasmic region.
- CDK Activity Calculation: Compute the nuclear-to-cytoplasmic (N:C) ratio of the biosensor fluorescence for each cell over time. A decreasing N:C ratio typically indicates increasing CDK activity, as the biosensor is exported from the nucleus [16].
- Data Visualization and Analysis: Plot the CDK activity ratio over time for each cell lineage. These kinetic profiles can then be correlated with cell fate decisions and morphological events observed in the corresponding brightfield or phase-contrast channels.
Integration with Advanced Research Applications
The application of continuous embryo monitoring extends beyond basic phenotyping, providing powerful tools for advanced research applications.
Integration with Artificial Intelligence (AI)
The large, complex datasets generated by TLM are ideally suited for analysis with AI and machine learning. AI algorithms can be trained to identify subtle patterns in morphokinetic data that are imperceptible to the human eye, improving the predictive power of the technology [10] [15]. For instance, AI models like the Morphological Artificial Intelligence Assistance (MAIA) platform have been developed to provide objective, standardized embryo assessments, achieving significant accuracy in predicting clinical pregnancy outcomes in human IVF [15]. Similarly, AI systems have been successfully applied to automatically detect developmental abnormalities in zebrafish embryos with high accuracy, demonstrating the potential for automated high-throughput toxicity screening in drug discovery [14].
Application in Drug Discovery and Toxicology
Mouse embryonic stem (ES) cells and whole embryos are invaluable models for high-throughput drug screening and toxicity assessment [13]. Continuous monitoring allows researchers to observe the direct effects of chemical compounds on developmental progression in real time. Key applications include:
- Toxicity Assessment: Identifying compounds that cause developmental arrest, delays, or morphological abnormalities [13] [14].
- Differentiation Studies: Screening for small molecules that promote the differentiation of ES cells into specific terminal cell types (e.g., cardiomyocytes or neurons) by monitoring changes in morphology and motility [13].
- Phenotypic Screening: Enabling high-content screening based on complex phenotypic changes in developing embryos or differentiated cells, providing a more physiologically relevant model than immortalized cell lines [13].
Continuous, non-invasive embryo monitoring via time-lapse microscopy represents a significant methodological advancement for research in developmental biology and drug discovery. By maintaining optimal culture conditions and providing a comprehensive, objective record of developmental kinetics, TLM enables a deeper understanding of fundamental biological processes like cell cycle dynamics, lineage specification, and response to external compounds. The detailed protocol for quantifying CDK activity provided herein exemplifies the power of this approach to generate quantitative, single-cell resolution data in a living system. As the technology continues to evolve, particularly through integration with artificial intelligence, its role in accelerating basic research and improving the efficiency and predictive power of preclinical drug safety screening is poised to expand substantially.
Mouse Models as a Gateway to Understanding Mammalian Development and Infertility
Mouse models serve as an indispensable gateway to understanding the intricate processes of mammalian development and the pathological underpinnings of infertility. The genetic, physiological, and developmental similarities between mice and humans make them ideal model systems for investigating reproductive biology [18] [1]. Infertility affects a significant portion of the global population, with the World Health Organization suggesting approximately one in six (∼17.5%) adults experience infertility, with male factors being the sole or leading cause in 40–50% of cases [18]. Furthermore, sperm counts have declined by approximately 60% over the past 50-60 years, highlighting an urgent need to understand the causes behind these worrying trends [18].
Advanced imaging technologies and genetic tools have revolutionized our ability to interrogate developmental dynamics in real-time. The creation of genetically engineered mouse models has provided unprecedented insights into fundamental biological processes, from gametogenesis to early embryonic patterning [19] [20]. These models allow researchers to visualize and quantify molecular events at single-cell resolution throughout development, offering critical insights into the mechanisms that govern normal and pathological reproduction [16]. This document presents detailed application notes and experimental protocols that leverage mouse models to advance our understanding of mammalian development and infertility, with particular emphasis on methodologies applicable within the context of time-lapse microscopy mouse embryo culture research.
Application Notes: Insights from Advanced Mouse Models
Quantifying Single-Cell CDK Activity Dynamics in Early Embryogenesis
Background: Cyclin-dependent kinases (CDKs) are fundamental regulators of the cell cycle and embryonic development. Understanding their spatiotemporal activity dynamics at the single-cell level provides crucial insights into the coordination of cell division and differentiation during early embryogenesis.
Experimental Approach: A recent protocol detailed a comprehensive approach for imaging and quantifying CDK activity levels in single cells throughout different stages of pre-implantation mouse embryo development [16] [17]. This methodology utilizes fluorescent biosensors and time-lapse confocal microscopy to track the translocation of CDK activity reporters in living mouse embryos, requiring specialized expertise in both mouse embryology and computational analysis.
Key Findings and Significance:
- Lineage-Specific Dynamics: CDK activity dynamics were found to be lineage-specific, characterizing early mammalian development with distinct patterns in different cell lineages [16].
- Technical Innovation: The approach combines in vitro culture strategies optimized for live fluorescent confocal image acquisition with sophisticated computational pipelines for cell tracking and data visualization [16] [17].
- Quantitative Framework: This methodology establishes a quantitative framework for understanding how cell cycle regulation intersects with cell fate decisions, potentially revealing novel aspects of developmental competence and embryo viability.
Elucidating Sperm Function Through a Novel Transgenic Model
Background: The initial steps of fertilization involve complex molecular interactions between sperm and egg, many of which remain poorly understood. A key event is acrosome exocytosis, a process where enzymes are released from the sperm head to facilitate penetration of the egg's protective layers.
Experimental Approach: Researchers at the Baker Institute for Animal Health developed a transgenic mouse model with fluorescing markers that allow real-time visualization of calcium dynamics and acrosomal status in sperm [19]. This model employs proteins that glow red when inside the acrosome, while a green glow indicates a rise in intracellular calcium, which triggers exocytosis.
Key Findings and Significance:
- Gradual Exocytosis: Contrary to the traditional "all or nothing" acrosome reaction model, the data demonstrated that exocytosis occurs in a gradual, multi-step fashion within the sperm [19].
- Real-Time Visualization: The model enables separation and observation of different fertilization steps in real time, providing unprecedented temporal resolution of these critical events [19].
- Therapeutic Implications: Understanding the specific calcium channels involved in human sperm function could lead to the development of novel non-hormonal contraceptives and treatments for certain forms of male infertility [19].
Label-Free Embryo Quality Assessment Using Optical Coherence Microscopy
Background: Selecting high-quality embryos is essential for enhancing success rates in in vitro fertilization (IVF). Traditional methods rely on morphological evaluation at static time points or invasive genetic testing, which have limitations in predictive value and potential embryo safety.
Experimental Approach: A novel dual-modality imaging system combining bright-field (BF) and optical coherence microscopy (OCM) was developed for non-invasive, time-lapse 3D imaging of mouse embryo development from the one-cell stage to fully hatched blastocyst [1]. The compact system fits within a commercial incubator, maintaining appropriate culture conditions while acquiring images automatically every ten minutes for over 150 hours.
Key Findings and Significance:
- Microstructural Visualization: OCM provides high-resolution 3D imaging capability, revealing cellular and subcellular features including nuclei size, location, and organization during early cleavage stages [1].
- Blastocyst Evaluation: OCM enables detailed characterization of blastocyst structures, including the blastocoel cavity, inner cell mass (ICM), and trophectoderm (TE), allowing accurate grading according to established systems [1].
- Predictive Potential: The timing of the second and third embryonic cell cycles was indicated to be associated with blastocyst formation and hatching capability, suggesting early morphokinetic parameters may predict developmental potential [1].
Table 1: Key Application Areas of Mouse Models in Reproduction Research
| Application Area | Mouse Model Type | Key Measurable Parameters | Research Implications |
|---|---|---|---|
| Early Embryonic Development | Fluorescent biosensor models [16] | Single-cell CDK activity dynamics, cell cycle duration, lineage specification | Understanding cell fate decisions, developmental competence |
| Sperm Function & Fertilization | Transgenic sperm reporter models [19] | Calcium flux, acrosome exocytosis kinetics, motility parameters | Novel contraceptive targets, treatments for male infertility |
| Embryo Quality Assessment | Wild-type embryos with advanced imaging [1] | Cleavage timing, blastocyst formation rate, structural morphology | Improved IVF embryo selection criteria, viability prediction |
| Genetic Infertility Factors | Targeted knockout models [18] [20] | Spermatogenic arrest, meiotic defects, hormonal profiles | Identification of human infertility genes, pathological mechanisms |
Experimental Protocols
Protocol for Single-Cell CDK Activity Quantification in Live Mouse Embryos
This protocol details the methodology for quantifying cyclin-dependent kinase (CDK) activity levels in single cells throughout pre-implantation mouse embryo development using time-lapse microscopy of fluorescent biosensors [16].
Materials and Reagents
Table 2: Essential Research Reagents for CDK Activity Imaging
| Reagent/Material | Specification | Function/Application |
|---|---|---|
| Mouse Embryos | Expressing fluorescent CDK biosensor [16] | Primary biological material for live imaging |
| Culture Medium | Pre-equilibrated embryo culture medium | Supports embryo development during imaging |
| Imaging Dish | Commercial embryo imaging dish (e.g., IVF store V005001) [1] | Holds embryos for culture and microscopy |
| Fluorescent Confocal Microscope | With environmental chamber | Maintains physiological conditions during time-lapse |
| Image Analysis Software | Cellpose algorithm [16] | Automated cellular segmentation |
| Computational Pipeline | Custom scripts for data processing [16] | Quantifies CDK activity from imaging data |
Step-by-Step Methodology
Embryo Isolation and Culture:
- Isplicate morula and blastocyst stage embryos from superovulated mice using standard protocols [16].
- Transfer embryos to pre-equilibrated culture medium in a specialized imaging dish capable of maintaining up to 25 embryos [1].
- Place the dish in a microscope stage-top incubator maintaining 37°C, 5% O₂, and 6% CO₂ for the duration of imaging.
Time-Lapse Image Acquisition:
- Set up the confocal microscope for time-lapse acquisition with appropriate laser lines for exciting the fluorescent biosensor.
- Program an automated acquisition pipeline that includes well localization, image-guided auto-tracking, and auto-focusing to center embryo samples [1].
- Acquire co-registered 3D image stacks at regular intervals (e.g., every 10-15 minutes) throughout the desired developmental period (typically up to 150 hours) [1].
Image Processing and Data Analysis:
- Process acquired images using the Cellpose algorithm for cellular segmentation, which provides a generalist approach for accurate cell identification [16].
- Apply computational workflows to quantify CDK activity levels based on biosensor localization and fluorescence intensity.
- Perform single-cell tracking throughout different embryonic stages to correlate CDK activity dynamics with developmental transitions.
Data Visualization and Interpretation:
- Generate heat maps and kymographs to visualize CDK activity patterns across the embryo over time.
- Correlate CDK activity dynamics with key developmental events such as cell division, compaction, and lineage specification.
Protocol for Sperm Calcium Dynamics and Acrosome Exocytosis Imaging
This protocol utilizes a genetically engineered mouse model with fluorescing markers to visualize calcium dynamics and acrosomal status during the fertilization process [19].
Materials and Reagents
- Transgenic Mouse Model: Expressing red fluorescent protein in the acrosome and green calcium indicator in sperm head [19]
- Sperm Collection Medium: Appropriate buffer for maintaining sperm viability and function
- Imaging Chamber: Specialized chamber for visualizing sperm under microscopy
- Confocal Microscope: With capabilities for simultaneous dual-channel imaging
- Calcium Modulators: Pharmacological agents for manipulating calcium levels (inhibitors/activators)
- Image Analysis Software: For quantifying fluorescence intensity and kinetics
Step-by-Step Methodology
Sperm Sample Preparation:
- Collect sperm from the transgenic mouse model using standard dissection and collection techniques.
- Suspend sperm in appropriate medium that maintains functionality throughout the imaging period.
- Load sperm suspension into a specialized imaging chamber suitable for high-resolution microscopy.
Real-Time Imaging Setup:
- Set up the confocal microscope for simultaneous acquisition in red and green fluorescence channels.
- Focus on the sperm population in the imaging chamber, ensuring clear visualization of individual sperm heads.
- Begin time-lapse acquisition with appropriate temporal resolution (e.g., 1-5 second intervals) to capture rapid calcium dynamics.
Calcium Stimulation:
- Establish a baseline recording of fluorescence for 1-2 minutes before stimulation.
- Introduce appropriate stimuli to trigger calcium flux and acrosome exocytosis (e.g., progesterone or zona pellucida components).
- Continue imaging for sufficient time to capture the complete sequence of exocytosis events.
Data Analysis and Interpretation:
- Quantify changes in red (acrosome) and green (calcium) fluorescence intensity over time in individual sperm.
- Calculate kinetics parameters including time to calcium peak, rate of acrosome loss, and correlation between events.
- Compare patterns across sperm populations to determine heterogeneity in response.
Protocol for Label-Free Embryo Evaluation Using Time-Lapse OCM
This protocol describes the use of optical coherence microscopy (OCM) for non-invasive, 3D time-lapse imaging of mouse embryo development to assess embryo quality without labels [1].
Materials and Reagents
- Dual-Modality Imaging System: Combined OCM and bright-field system customized to fit inside an incubator [1]
- Embryo Imaging Dish: Commercial dish (e.g., IVF store V005001) capable of holding up to 25 embryos [1]
- Culture Medium: Pre-equilibrated medium supporting development to hatched blastocyst stage
- Automated Stage: 3-axis motorized sample stage for precise positioning
- Data Storage System: High-capacity storage server for large 3D time-lapse datasets
Step-by-Step Methodology
System Setup and Calibration:
- Ensure the dual-modality OCM/BF imaging system is properly installed within the incubator and environmental conditions are stabilized (5% O₂, 6% CO₂, 37°C) [1].
- Calibrate the OCM system to achieve optimal resolution (~2.1 μm axial, ~1.0 μm lateral in tissue).
- Verify the automated sample positioning and focusing systems are functioning correctly.
Embryo Loading and Initialization:
- Load one-cell stage mouse embryos into the specialized imaging dish with pre-equilibrated culture medium.
- Program the automated acquisition pipeline with knowledge of sample well locations.
- Initialize the time-lapse experiment with imaging intervals of approximately 10 minutes for over 150 hours of continuous monitoring [1].
Automated Time-Lapse Acquisition:
- For each time point, the system automatically moves each well to the imaging location using pre-programmed sample indexing.
- Perform image-guided auto-tracking and auto-focusing to center embryo samples.
- Acquire co-registered BF and 3D OCM images for each embryo at every time point.
- Transfer data automatically to storage servers for subsequent analysis.
Image Analysis and Morphokinetic Scoring:
- Reconstruct 3D structural information from OCM datasets at each time point.
- Track developmental events including cleavage timings, compaction, blastocoel formation, and hatching.
- Correlate early morphokinetic parameters (second and third cell cycles) with blastocyst formation and quality outcomes [1].
- Generate quantitative metrics of development dynamics for embryo quality assessment.
Discussion and Future Perspectives
The integration of advanced mouse models with cutting-edge imaging technologies represents a powerful paradigm for unraveling the complexities of mammalian development and infertility. The protocols detailed herein provide robust methodologies for quantifying molecular activity dynamics, cellular function, and structural development in real-time. These approaches offer significant advantages over traditional static endpoint analyses by capturing the temporal dimension of developmental processes.
Mouse models have been particularly instrumental in identifying genetic factors underlying infertility. Large-scale efforts like the Reproductive Genomics Program at the Jackson Laboratory have identified numerous genes essential for fertility, with over 400 genes yielding male infertility phenotypes when disrupted [18] [20]. These models demonstrate that spermatogenic failure represents the predominant outcome (50-60% of infertile KO phenotypes), while smaller subsets exhibit isolated sperm functional defects or endocrine abnormalities [18].
Future directions in this field will likely focus on increasing the resolution and dimensionality of data acquisition, while simultaneously developing more sophisticated computational tools for data integration and analysis. The combination of mouse models with multi-omics approaches and artificial intelligence-based image analysis promises to reveal unprecedented insights into the molecular networks governing reproduction. Furthermore, efforts to bridge species-specific differences between mice and humans will be essential for maximizing the translational impact of these findings in clinical infertility management.
As imaging technologies continue to advance and genetic tools become increasingly precise, mouse models will remain an essential gateway to understanding mammalian development and infertility, ultimately contributing to improved diagnostic and therapeutic strategies for human reproductive disorders.
The selection of high-quality embryos is fundamental to enhancing success rates in in vitro fertilization (IVF) and developmental biology research [1]. Traditional methods for evaluating embryo quality have relied heavily on morphological assessment at static time points, a approach that is inherently subjective and fails to capture the dynamic processes of preimplantation development [1]. This article details the application of advanced, non-invasive time-lapse microscopy techniques to overcome these historical limitations. By establishing dynamic developmental timelines, researchers can now correlate early embryonic events with ultimate developmental potential, thereby streamlining and improving embryo selection protocols within both clinical and research settings [1].
Application Notes: The Value of Time-Lapse 3D Imaging in Embryo Assessment
Limitations of Traditional Embryo Evaluation Methods
- Static Morphological Evaluation: Traditionally, embryo selection was based on microscopic images at static time points, requiring removal from the incubator and leading to subjective scoring and potential suboptimal culture conditions [1].
- Preimplantation Genetic Testing (PGT): While PGT directly links genetic abnormalities to quality, it involves trophectoderm or cell biopsy, an invasive process that may negatively impact IVF outcomes [1].
- Conventional Time-Lapse Imaging: Most systems acquire two-dimensional (2D) bright-field (BF) or dark-field images, which lack the three-dimensional (3D) structural information crucial for comprehensive analysis [1].
Advantages of Optical Coherence Microscopy (OCM) and Light-Sheet Imaging
Optical coherence microscopy (OCM) is a label-free, non-invasive imaging technique that combines the principles of optical coherence tomography (OCT) and confocal microscopy to provide 3D, high-resolution, volumetric imaging [1]. With a high numerical aperture objective lens, OCM provides micron-level resolutions capable of resolving cellular and subcellular features within living embryos [1]. The key benefits include:
- Non-Invasive 3D Structural Assessment: OCM reveals 3D morphological features, such as nuclei size and location, cleavage symmetry, and blastocoel cavity formation, without the need for staining or physical sectioning [1].
- Continuous Environmental Control: Compact imaging systems can be housed within standard incubators, maintaining critical temperature, gas, and humidity levels throughout the entire imaging period [1].
- Correlation of Early Events with Developmental Outcomes: Time-lapse OCM on mouse embryos indicates that the timing of the second and third embryonic cell cycles is associated with blastocyst formation and hatching capability [1].
Similarly, light-sheet microscopy is recognized for its ability to minimize phototoxicity and maximize signal intensity, making it suitable for long-term imaging of sensitive samples like regenerating plant roots and post-implantation mouse embryos [21] [22]. It illuminates a single plane of a sample at a time, enabling high time-resolution imaging with reduced light damage compared to confocal microscopy [21].
Table 1: Quantitative Comparison of Embryo Imaging Modalities
| Imaging Modality | Resolution (Lateral/Axial) | Imaging Depth | Speed | Phototoxicity | Key Applications |
|---|---|---|---|---|---|
| Bright-Field (BF) Microscopy | ~0.5-1.0 μm (diffraction-limited) | Shallow | High | Low | Basic morphology, cell counting [1] |
| Confocal Microscopy | High | Limited by scattering | Medium | High (with certain wavelengths) | High-resolution 2D/3D fluorescence imaging [21] |
| Optical Coherence Microscopy (OCM) | ~1.0 μm / ~2.1 μm [1] | ~1-2 mm in tissue | High | Low (label-free) | 3D microstructural analysis of live embryos [1] |
| Light-Sheet Microscopy | High | Good for cleared/small samples | Very High | Low | Long-term 4D imaging of development [21] [22] |
Experimental Protocols
Protocol 1: Time-Lapse 3D OCM of Mouse Embryos from One-Cell to Blastocyst
This protocol is adapted from the label-free evaluation of mouse embryo quality, enabling the acquisition of co-registered 3D OCM and bright-field images over the entire preimplantation period [1].
Equipment and Reagents
- Dual-Modality Imaging System: A custom OCM system with a broadband superluminescent diode and a 20X objective lens, combined with a BF imaging path, integrated into a compact prototype that fits inside an incubator [1].
- Incubator: Commercial incubator (e.g., Heracell VIOS 160i) capable of maintaining 5% O2 and 6% CO2 [1].
- Embryo Culture Dish: Commercial embryo imaging dish (e.g., IVF store V005001) capable of culturing and imaging up to 25 embryos per dish [1].
- Automated Stage: A 3-axis motorized sample stage for precise and efficient sample movement [1].
- Mouse Embryos: One-cell stage mouse embryos, an appropriate model due to genetic and developmental similarity with humans [1].
Procedure
- System Setup and Calibration: Place the dual-modality imaging system inside the incubator and allow it to equilibrate. Ensure the OCM system provides axial and lateral resolutions of approximately ~2.1 μm and ~1.0 μm in tissue, respectively [1].
- Embryo Loading: Transfer 8-12 one-cell stage mouse embryos into the wells of the embryo imaging dish containing pre-equilibrated culture medium [1].
- Automated Time-Lapse Imaging:
- Initiate Acquisition: Set the automated acquisition pipeline to capture images every ten minutes for over 150 hours [1].
- Sample Tracking: For each time point, the system automatically moves the target well to the imaging location using prior knowledge of the sample index, followed by image-guided auto-tracking and auto-focusing to center the embryo [1].
- Image Acquisition: Acquire co-registered BF and 3D OCM images for each embryo at each time point [1].
- Data Transfer: Automatically transfer the acquired data to a storage server for subsequent analysis [1].
- Data Analysis:
- Morphokinetic Analysis: Use the time-lapse data to track the timing of key developmental events (e.g., cleavage divisions, compaction, blastocyst formation, hatching).
- Structural Analysis: Utilize 3D OCM data to visualize and quantify microstructures such as nuclei count, zona pellucida thickness, inner cell mass (ICM) formation, and trophectoderm (TE) organization [1].
- Correlation with Outcomes: Correlate early morphokinetic parameters (e.g., duration of second and third cell cycles) with subsequent developmental outcomes like blastocyst formation and quality [1].
Protocol 2: Mounting and Long-Term Light-Sheet Imaging of Arabidopsis Roots (Adaptable for Embryos)
While developed for plant roots, the principles of this protocol for sample mounting and tracking in a light-sheet system are highly relevant for adapting to embryonic samples [21].
Equipment and Reagents
- Light-Sheet Microscope: A commercially available system (e.g., Mizar TILT system) or a custom-built setup [21].
- Chambered Cover Glass: 1-well chambered cover glass with optically clear sides (e.g., CellVis #C1-1.5H-N) [21].
- Mounting Media: Low melt agarose (e.g., VWR #89125-532) prepared in an appropriate culture medium (e.g., ½ MS salts for plants) and filter-sterilized for optical clarity [21].
- Tools: Sterile forceps (pointed and flat) and ultrathin scalpel blades [21].
Procedure
- Prepare Media Blankets:
- In a sterile environment, add ~5 mL of molten, filter-sterilized low melt agarose media to a chambered cover glass.
- Pipette off any surface bubbles and allow the media to solidify.
- Store the prepared blankets at 4°C in a closed sterile container for at least 24 hours before use to facilitate handling [21].
- Sample Mounting:
- For roots, a trench is cut into the pre-solidified media blanket, and the sample is placed inside. For embryos, a similar strategy can be employed by creating small wells or embedding embryos within a thin layer of agarose.
- Mount multiple samples to increase the chance of obtaining an ideal orientation, as some may become damaged or grow in a non-optimal direction [21].
- Imaging and Automated Tracking:
- Place the mounted sample into the light-sheet microscope chamber.
- Implement an automated tracking system (requiring no additional proprietary software) to keep the growing sample (e.g., a root tip or developing embryo) within the field of view over extended periods [21].
- Set imaging parameters (e.g., time resolution, z-stack depth) for multi-day time-lapse imaging.
The following workflow diagram illustrates the core steps of a generalized time-lapse microscopy experiment for developmental biology.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Time-Lapse Embryo Culture and Imaging
| Item | Function/Benefit | Example/Specification |
|---|---|---|
| Commercial Embryo Imaging Dish | Enables long-term culture and imaging of multiple embryos simultaneously without moving the dish; designed for optical clarity. | IVF store V005001 (capacity for 25 embryos) [1] |
| Low Melt Agarose | Used for embedding samples to provide structural support during imaging; low melting point minimizes heat damage to live samples. | VWR #89125-532; prepared at 2% w/v in culture medium [21] |
| Chambered Cover Glass | Provides an optically clear, sterile environment for sample mounting and high-resolution imaging. | CellVis #C1-1.5H-N (1 well) [21] |
| Filtered Culture Media | Ensures media is optically clear for light-sheet and other high-resolution microscopy techniques, reducing background noise. | Filter-sterilized using a 0.22 or 0.45 micron syringe filter [21] |
| Specific Gas Mixture | Critical for maintaining physiological conditions and embryo viability during extended culture. | 5% O2, 6% CO2, balanced N2 for mouse embryos [1] |
Data Interpretation: From 3D Volumes to Predictive Timelines
The power of time-lapse 3D imaging lies in translating acquired volumes into predictive metrics for embryo quality.
- Quantitative Structural Metrics: OCM allows for the quantification of the thickness and uniformity of the zona pellucida, the volume of the blastocoel cavity, and the compactness of the inner cell mass (ICM) [1]. These structural details are often indiscernible in 2D bright-field images but are critical for accurate grading according to established systems like Gardner's blastocyst grading system [1].
- Morphokinetic Parameters: The dynamic data enables the precise timing of key events. Research on mouse embryos indicates that the duration of the second and third embryonic cell cycles is predictive of successful blastocyst formation and hatching capability [1].
- Lineage Specification: During the blastocyst stage, OCM can differentiate the emerging lineages. The trophectoderm (TE) and ICM become distinct, and their morphological characteristics can be assessed over time, providing a non-invasive readout of developmental competence [1].
The diagram below summarizes the key developmental stages and quality indicators that can be monitored using these advanced imaging techniques.
The integration of non-invasive, high-resolution 3D imaging modalities like OCM and light-sheet microscopy with time-lapse culture systems represents a paradigm shift in developmental biology and assisted reproduction. This approach successfully overcomes the historical limitations of static snapshots by providing continuous, dynamic developmental timelines in a controlled environment. The protocols and application notes detailed herein provide a framework for researchers to implement these technologies, enabling the accurate correlation of early morphokinetic and structural events with embryo viability and developmental potential. This advancement holds significant promise for improving the efficiency and success of both basic research in embryonic development and clinical outcomes in assisted reproductive technologies.
From Theory to Bench: Optimized Protocols for High-Resolution Embryo Imaging
Within the context of a broader thesis on time-lapse microscopy for mouse embryo culture research, this application note provides a detailed protocol for embryo preparation and imaging. The ability to visualize morphogenetic and physiological processes in early post-implantation mouse embryos at high spatiotemporal resolution is crucial for understanding developmental mechanisms [23]. This protocol outlines optimized procedures for embryo isolation, mounting, and culture specifically tailored for time-lapse imaging applications, incorporating both advanced and accessible methodological approaches to accommodate varying laboratory resources and research objectives.
Materials and Equipment
Research Reagent Solutions
Table 1: Essential materials for embryo isolation, mounting, and culture
| Item | Function/Application | Specific Examples/Notes |
|---|---|---|
| Embryo Culture Medium | Supports embryo development during imaging | For 5.5 dpc embryos: 50% CMRL + 50% Knock Out Serum + L-Glutamine; For 6.5+ dpc: 50% DMEM-FluoroBrite with 50% rat serum [23] |
| Dissection Medium | Maintains embryo viability during isolation | M2 medium, warmed to room temperature [23] |
| Glass Capillaries | Mounting embryos for imaging | Commercially available capillaries with varying inner diameters; optimal diameter selected based on embryo stage and visibility requirements [24] |
| Imaging Chamber | Holds mounted embryos during time-lapse acquisition | 8-well chambered slides with specific assembly protocols [23] |
| Vacuum Grease | Creates barriers in imaging chambers | Used to create sealed environments without toxic effects on embryos [23] |
| Ops Culture Method | Alternative simple culture system | Uses sealed containers like PCR tubes or glass capillaries; doesn't require CO₂ incubator [24] |
Equipment Setup
Table 2: Imaging systems and environmental control options
| System Type | Key Features | Applications |
|---|---|---|
| Lattice Light-Sheet Microscope | Thin light-sheet from Bessel beams; minimal photodamage; high spatiotemporal resolution [23] | Visualizing highly dynamic, photo-sensitive processes; migration events; subcellular dynamics [23] |
| Dual-Modality OCM/BF System | Label-free; non-invasive; provides 3D high-resolution imaging; fits inside commercial incubator [1] | Long-term monitoring of embryo development; quantitative morphological assessment [1] |
| Simple Glass Capillary System | Low-cost; uses stereomicroscope, digital camera, thermoplate; minimal equipment requirements [24] | Preimplantation embryo analysis in resource-limited settings; developmental speed and morphology studies [24] |
Experimental Workflow
The following diagram illustrates the core pathways for preparing and imaging mouse embryos, integrating both conventional and simplified methods:
Step-by-Step Protocols
Institutional Permissions and Timed Mating
4.1.1 Regulatory Compliance: All animal experimentation procedures must be performed in full accordance with relevant institutional and national guidelines. Obtain necessary approvals from the relevant ethical review boards before initiating any experiments [23].
4.1.2 Timed Mating Setup:
- Plan experiments 7-10 days in advance to ensure availability of mice with desired reporter alleles.
- Pair stud males and females from desired strains near the start of the dark cycle in the facility.
- Check females each morning for vaginal plugs; noon on the day a plug is detected is designated 0.5 days post coitum (dpc).
- Use females between 6-18 weeks and males between 8-32 weeks for optimal litter sizes [23].
Embryo Isolation and Medium Preparation
4.2.1 Dissection Procedure:
- Isolate post-implantation mouse embryos (e.g., 5.5 dpc) using standardized dissection protocols.
- Perform dissections in pre-warmed M2 medium at room temperature (22°C) [23].
- Select healthy, undamaged embryos for live imaging experiments.
4.2.2 Embryo Culture Medium Preparation:
- Prepare 4mL of Embryo Culture Medium fresh for each experiment under sterile conditions.
- Combine 2mL CMRL with 2mL Knock Out Serum.
- Add 42μL of 200mM L-Glutamine (ensure complete dissolution to prevent imaging artifacts).
- Vortex to mix components thoroughly.
- Equilibrate medium in a humidified incubator at 37°C and 5% CO₂ for at least 1 hour with loosened screw cap to allow gas exchange [23].
CRITICAL: Since no antibiotics are used, maintain strict sterile technique throughout preparation to minimize infection risk.
Mounting Implement Preparation
4.3.1 Glass Capillary Pulling:
- Securely hold glass capillary at both ends and rotate horizontally over a Bunsen flame with a well-defined blue cone.
- When the center becomes malleable, remove from flame and pull evenly and quickly from both ends.
- Gently snap off the ends to isolate the central pulled region.
- Practice is required to determine appropriate pulling delay and speed to achieve desired capillary width for specific embryo stages [23].
4.3.2 Glass Capillary Selection (Simplified Method):
- Test commercially available glass capillaries with different inner diameters.
- Select capillaries with medium inner diameters that allow observation of several embryos simultaneously while maintaining focus clarity.
- Ensure chosen capillaries support high developmental rates to blastocyst stage comparable to control conditions (83.1-97.6% vs. 92.7% control) [24].
Imaging Chamber Assembly
4.4.1 Conventional Chamber Setup:
- Use an 8-chambered slide and fill the four end wells with 400μL of pre-equilibrated culture medium each.
- Fill a 5mL syringe with vacuum grease and attach a 200μL tip as a nozzle.
- Under a dissection microscope, extrude grease to create two barriers at the top and bottom of the central (unfilled) wells.
- Take care not to smear grease on the center of the well where imaging will occur [23].
4.4.2 Glass Capillary Method (Simplified):
- Prefill selected glass capillary with culture medium.
- Insert one-cell stage zygotes into capillary using a mouth pipette.
- Introduce air bubbles beside embryos inside capillary to help regulate embryo positions.
- Seal capillary ends with a burner, avoiding excessive heat near embryos.
- For improved clarity during imaging, place capillary in a dish of water or oil [24].
CRITICAL: Always handle chambers on a clean Petri dish lid to avoid scratching or dirtying the imaging surface.
Temperature Stabilization and Imaging
4.5.1 Temperature Optimization:
- For conventional systems, maintain stable incubator conditions at 37°C, 5% CO₂.
- For simplified system, use a thermoplate on a microscope for warming.
- Use a dish with a flat bottom without a rim for more effective temperature transmission.
- Conduct experiments in a small room with minimal temperature fluctuations [24].
4.5.2 Time-Lapse Imaging Parameters:
- For lattice light-sheet microscopy: Set imaging parameters to leverage fast dual-camera acquisition for capturing highly dynamic processes [23].
- For bright-field/OCM systems: Acquire images every 10 minutes for over 150 hours to monitor development from one-cell to hatched blastocyst [1].
- For simple capillary system: Capture images every 30 minutes for up to 5 days using a stereomicroscope and digital camera [24].
- Minimize light exposure by illuminating only during image acquisition periods.
Technical Specifications and Performance Metrics
Table 3: Quantitative performance data for different imaging approaches
| Parameter | Lattice Light-Sheet | OCM/BF Imaging | Simple Capillary Method |
|---|---|---|---|
| Temporal Resolution | Very high (fast dynamic processes) [23] | Moderate (10-min intervals) [1] | Lower (30-min intervals) [24] |
| Spatial Resolution | Unprecedented (subcellular) [23] | High (~1.0μm lateral, ~2.1μm axial) [1] | Standard microscope resolution [24] |
| Developmental Rate to Blastocyst | Suitable for concurrent imaging of several embryos [23] | Not specified | 81.5% (slightly lower than 95.8% control) [24] |
| Photodamage | Minimal [23] | Label-free, non-invasive [1] | Minimal with controlled light exposure [24] |
| Equipment Cost | Very high | High | Low |
Troubleshooting and Quality Control
6.1 Developmental Rate Optimization:
- If using the simplified capillary method with suboptimal developmental rates, ensure proper temperature stabilization using flat-bottom dishes and minimize room temperature fluctuations [24].
- Verify medium composition and equilibration for specific embryonic stages.
6.2 Imaging Artifact Reduction:
- Ensure complete dissolution of L-glutamine to prevent crystals that cause light refraction and scattering [23].
- For glass capillary methods, position embryos with air bubbles to prevent clustering and improve individual embryo tracking [24].
6.3 Blurriness in Capillary Imaging:
- When using glass capillaries, place the capillary in a dish of water or oil to improve optical clarity during imaging [24].
- Select capillaries with appropriate wall thickness and inner diameter for optimal focus.
Lattice Light-Sheet Microscopy (LLSM) represents a transformative advancement in fluorescence microscopy, enabling high-resolution volumetric imaging of dynamic biological processes with minimal photodamage. This technology utilizes a two-dimensional optical lattice of interfering Bessel beams to create an exceptionally thin light sheet, typically measuring between 0.4-1.0 μm in thickness [25]. Unlike point-scanning techniques like confocal microscopy that illuminate the entire sample thickness, LLSM employs selective plane illumination, thereby drastically reducing out-of-focus background signals, photobleaching, and phototoxicity [23] [25]. This combination of superior spatiotemporal resolution and gentle imaging conditions makes LLSM particularly valuable for studying delicate, dynamic processes in developmental biology, especially in pre-clinical research involving mouse embryo culture and organoid systems.
The fundamental advantage of LLSM lies in its ability to decouple the relationship between axial resolution and field of view that plagues conventional light-sheet microscopes using Gaussian beams [26]. Where Gaussian beams must become thicker to achieve longer propagation distances, lattice light-sheets maintain their thin profile over cellular dimensions, enabling high-resolution imaging across large volumes. Typical performance metrics for LLSM include lateral resolution of approximately 230-250 nm and axial resolution of 350-500 nm at imaging speeds of up to 100-200 frames per second [25] [27]. This unprecedented capability has opened new avenues for investigating morphological and physiological processes in four dimensions (three spatial dimensions plus time), particularly during the critical early post-implantation stages of mouse embryonic development when the anatomical axes, definitive germ layers, and first organs are established [23].
Technical Principles and Advantages
Fundamental Working Principles
LLSM generates its exceptional optical properties through a sophisticated optical arrangement that creates a two-dimensional lattice pattern at the sample plane. The system typically employs paired objectives arranged perpendicularly, with one objective delivering the lattice light-sheet and the other detecting emitted fluorescence [27]. The lattice pattern itself is formed from a massive parallel array of Bessel beams that mutually interfere to create an ultrathin light sheet [25]. Bessel beams belong to a special class of "non-diffracting" beams that maintain a narrow width over extended propagation distances due to self-interference effects, unlike Gaussian beams that rapidly diverge after focusing [25].
The LLSM platform offers two primary imaging modes: super-resolution structured illumination microscopy (SIM) and high-speed dithered mode [25]. In SIM mode, the system achieves enhanced resolution of approximately 150 nm laterally and 280 nm axially by collecting multiple images at each z-plane with different lattice orientations, though this comes at the cost of slower acquisition speeds (typically 4-second intervals for 3D volumes). In dithered mode, the two-dimensional lattice pattern is rapidly oscillated using a galvanometer to provide time-averaged uniform illumination, with only one image acquired per z-plane at remarkable speeds of up to 100-200 frames per second with resolutions of 230 nm laterally and 370 nm axially [25]. This dithered mode is particularly advantageous for capturing rapid biological processes like mitotic events and cellular migrations in developing embryos, as it minimizes photobleaching and phototoxicity while enabling data collection over thousands of time points.
Comparative Advantages Over Conventional Microscopy
The technical innovations in LLSM translate to significant practical advantages over conventional imaging approaches, particularly for sensitive, long-term live imaging applications such as mouse embryo culture:
Dramatically Reduced Phototoxicity and Photobleaching: By illuminating only the focal plane being imaged, LLSM limits photodamage to the specimen. This is crucial for extended time-lapse imaging of developing embryos, which are highly sensitive to light-induced stress [23] [25]. Studies have demonstrated the ability to capture high-resolution data over hundreds of volume acquisitions without detectable phototoxic effects, even on subsequent embryonic development [25] [28].
Superior Temporal Resolution for 3D Dynamics: The parallelized plane illumination and camera-based detection enable vastly faster 3D image acquisition compared to point-scanning methods. This allows researchers to completely sample entire embryonic volumes at sub-second intervals, capturing rapid cellular events and morphogenetic movements that were previously unobservable [23] [25].
Enhanced Spatial Resolution and Optical Sectioning: The ultrathin nature of the lattice light sheet provides exceptional axial resolution and optical sectioning capability. Compared to Gaussian light sheets of similar propagation length, square lattice light sheets demonstrate approximately 30% better axial resolution (1.62λ versus 1.83λ for Gaussian) with significantly less degradation along the beam propagation direction [26].
Flexibility in Fluorescent Probes: Unlike super-resolution techniques like STED and PALM that require special fluorophores or proteins, LLSM works with conventional fluorescent dyes and proteins, enabling broader application across existing transgenic models and multicolor imaging approaches [25].
Table 1: Quantitative Comparison of LLSM Imaging Modes and Performance Characteristics
| Parameter | High-Speed Dithered Mode | Super-Resolution SIM Mode | Conventional Confocal |
|---|---|---|---|
| Lateral Resolution | 230 nm | 150 nm | ~250 nm |
| Axial Resolution | 370 nm | 280 nm | ~600-700 nm |
| Volume Acquisition Speed | Up to 10 volumes/second | ~0.25 volumes/second | ~0.1-1 volumes/second |
| Photobleaching/Phototoxicity | Minimal | Moderate | High |
| Applicable Biological Processes | Fast cellular dynamics, migration | Subcellular structures, fine details | Fixed samples, less sensitive live samples |
Quantitative Performance Data
Rigorous characterization of LLSM performance reveals distinct advantages over alternative light-sheet modalities. Systematic comparisons between Gaussian, multi-Bessel square (MB-square), and hexagonal lattice light sheets demonstrate important trade-offs between resolution, propagation invariance, and optical sectioning capability [26].
When comparing light sheets with identical 20 μm propagation lengths, hexagonal lattice beams achieve the best axial resolution with a point spread function (PSF) having a full-width at half-maximum (FWHM) of 1.18λ, followed by MB-square lattices at 1.62λ, and Gaussian beams at 1.83λ [26]. This resolution advantage becomes even more pronounced away from the beam center. At a distance of 10 μm from the beam focus (the FWHM of the propagation profile), the axial FWHM values widen to 1.27λ for hexagonal lattices, 1.6λ for MB-square lattices, and 2.43λ for Gaussian beams [26]. This demonstrates the superior propagation invariance of lattice light sheets, maintaining consistent resolution across larger fields of view.
However, these resolution improvements come with a trade-off in excitation confinement. The same study quantified "optical sectioning" capability, defined as the width containing 63% of the cumulative intensity along the axial direction. Gaussian beams showed the best confinement at 0.84λ, compared to 1.87λ for MB-square lattices and 3.42λ for hexagonal lattices [26]. This decreased confinement translates to greater energy deposition in the sidelobes flanking the main beam, potentially increasing overall energy dose to the specimen despite the improved resolution characteristics.
Table 2: Quantitative Comparison of Light-Sheet Modalities (20 μm Propagation Length)
| Beam Type | Axial FWHM at Beam Focus (λ) | Axial FWHM at 10 μm from Focus (λ) | Optical Sectioning (63% Cumulative Intensity, λ) | Relative Energy in Sidelobes |
|---|---|---|---|---|
| Gaussian | 1.83 | 2.43 | 0.84 | Lowest |
| MB-Square Lattice | 1.62 | 1.60 | 1.87 | Moderate |
| Hexagonal Lattice | 1.18 | 1.27 | 3.42 | Highest |
| Flat-Top Beam | ~1.83 | ~1.90 | ~1.20 | Low-Moderate |
For developmental biology applications, these characteristics inform optimal beam selection. The hexagonal lattice provides superior resolution for structural studies where minimal phototoxicity is less critical, while MB-square lattices offer a favorable balance for long-term live imaging of delicate specimens like developing embryos, where both resolution and phototoxicity must be carefully managed.
Application Notes for Mouse Embryo Research
Specific Applications in Developmental Biology
LLSM has enabled unprecedented observation of key developmental processes in early mouse embryogenesis. Its combination of low phototoxicity and high spatiotemporal resolution makes it particularly valuable for capturing dynamic cellular behaviors during post-implantation stages, when the embryo undergoes dramatic morphogenetic transformations. Specific applications include:
Visualization of Anterior Visceral Endoderm (AVE) Migration: LLSM has been employed to image the migration of AVE cells, a critical process in establishing the anterior-posterior axis [23]. This technology has enabled characterization of how basal projections produced by migrating cells and overall migratory behavior are affected in mutants of the semaphorin-plexin pathway, providing mechanistic insights into this fundamental developmental process [23].
Analysis of Mitotic Processes and Spindle Dynamics: The high-speed volumetric imaging capabilities of LLSM allow tracking of growth at every spindle microtubule end and discrimination of individual chromosomes in living cells [25]. This has opened new possibilities for understanding how cells coordinate various mitotic events, including microtubule-kinetochore attachments, astral microtubule-cortex interactions, and chromosome movements during congression and separation.
Long-Term Imaging of Morphogenetic Events: LLSM supports continuous imaging of morphogenetic and physiological processes over extended durations (8+ hours), enabling complete documentation of key events such as germ layer formation, gastrulation, and early organogenesis [23]. The ability to concurrently image multiple embryos addresses the challenge of biological variability between embryos at these early stages by facilitating the construction of averaged cellular behavior maps [23].
Subcellular Process Visualization in 3D Context: With appropriate fluorescent labeling, LLSM can visualize subcellular events including actomyosin dynamics, membrane remodeling, chromosomal movements, and protein trafficking in specific regions or cells of the embryo [23]. This capability extends to multicellular embryo-like models and organoid systems, providing insights into how subcellular processes drive tissue-level morphogenesis.
Experimental Design Considerations
Successful implementation of LLSM for mouse embryo research requires careful experimental design:
Stage-Appropriate Culture Conditions: The protocol requires preparation of specialized Embryo Culture Medium, typically consisting of CMRL medium supplemented with KnockOut serum and L-glutamine for 5.5 days post coitum (dpc) embryos [23]. Later stage embryos (6.5+ dpc) may require different media formulations, such as a 1:1 mixture of DMEM-FluoroBrite and rat serum [23].
Genetic Background Selection: To ensure large litters with healthy embryos suitable for live imaging, females from outbred mouse strains like CD1 with genetically diverse backgrounds can be crossed with males homozygous for desired fluorescent reporters [23]. Alternatively, wildtype embryos can be used with live dyes that label membranes or nuclei.
Temporal Coordination: Timed matings must be planned 7-10 days in advance, with females in proestrus (exhibiting swollen, moist, pink vaginal openings) preferentially selected for mating to increase pregnancy success [23]. Embryonic age is typically calculated assuming noon on the day a vaginal plug is observed as 0.5 dpc.
Detailed Experimental Protocols
Embryo Isolation, Mounting and Culture Protocol
The following protocol outlines the essential steps for preparing and imaging post-implantation mouse embryos using LLSM, based on established methodologies [23]:
Step-by-Step Protocol:
Preparation and Equilibration of Media (Timing: 1.5 hours)
- Prepare 4 mL of Embryo Culture Medium fresh for each experiment in a laminar flow hood to minimize infection risk.
- Combine 2 mL of CMRL medium with 2 mL of KnockOut serum in a 15 mL tube.
- Add 42 μL of 200 mM L-glutamine (thawed completely to avoid crystalline artifacts).
- Vortex to mix components and equilibrate in a humidified incubator at 37°C and 5% CO₂ for at least 1 hour with the screw cap loosened to allow gas exchange.
- Warm M2 dissection medium (approximately 8 mL per litter) to room temperature.
Assembly and Pre-equilibration of Imaging Chamber (Timing: 20 minutes + 1 hour equilibration)
- Prepare mounting implements by heating glass capillaries over a Bunsen flame with a well-defined blue cone, rolling between fingers until the center becomes malleable.
- Remove from flame and pull evenly and quickly from both ends, then gently snap the pulled region to create fragments of appropriate length for embryo mounting.
- Take an 8-chambered slide and fill the four end wells with 400 μL of pre-equilibrated culture medium each.
- Using a syringe with vacuum grease and a 200 μL tip as a nozzle, extrude grease to make two barriers at the top and bottom of the central (unfilled) wells, taking care not to smear grease on the center of the well.
Embryo Mounting and Imaging (Timing: Variable based on experimental design)
- Isolate embryos at the desired developmental stage using standard dissection techniques in pre-warmed M2 medium.
- Select healthy, undamaged embryos for imaging and transfer to the imaging chamber using a mouth pipette or fine transfer pipette.
- Position embryos using the prepared glass capillaries in the central wells filled with culture medium.
- Set up LLSM imaging parameters appropriate for the experimental goals. For general morphogenetic studies, dithered mode with light-sheet thickness of 0.4-1.0 μm, z-steps of 0.3-0.5 μm, and time intervals of 30-120 seconds between volumes typically provide optimal results.
- Begin time-lapse acquisition, monitoring embryo health throughout the imaging session (up to 8 hours continuous imaging has been successfully demonstrated).
LLSM Setup and Image Acquisition Protocol
Critical Steps for Optimal Imaging:
Imaging Mode Selection: Choose between high-speed dithered mode for capturing rapid cellular dynamics or super-resolution SIM mode for structural details requiring higher resolution. For most mitotic processes and cell migrations in developing embryos, dithered mode provides sufficient resolution with minimal phototoxicity [25].
Lattice Pattern Optimization: Select appropriate lattice patterns based on resolution requirements and sample sensitivity. Square lattices provide a favorable balance between resolution and phototoxicity for most embryonic applications, while hexagonal lattices offer higher resolution at the cost of increased sidelobe energy and decreased optical sectioning [26].
Spatiotemporal Parameter Configuration: Set z-stack range to completely encompass the embryo or region of interest, with step size appropriate for the required axial resolution (typically 0.3-0.5 μm). Time intervals should be determined based on the dynamics of the process under study, with 30-120 seconds between complete volumes suitable for most morphogenetic events in early embryogenesis.
Environmental Control: Maintain temperature at 37°C throughout imaging using appropriate stage-top incubation systems. For extended imaging sessions beyond 4-5 hours, consider methods for maintaining pH and gas equilibrium, though the protocol described has successfully supported embryo development for up to 8 hours in the described culture medium [23].
The Scientist's Toolkit
Essential Research Reagent Solutions
Table 3: Key Research Reagents for LLSM of Mouse Embryos
| Reagent/Material | Function/Application | Specifications/Notes |
|---|---|---|
| CMRL Medium | Base component of embryo culture medium | Supplements with serum and glutamine |
| KnockOut Serum | Serum replacement in culture medium | Used at 50% concentration with CMRL |
| L-Glutamine | Essential amino acid supplement | Use 200 mM stock, ensure complete thawing |
| M2 Medium | Embryo dissection and manipulation | Room temperature for dissection |
| Matrigel | 3D culture matrix for certain applications | Used for neuronal cultures in some protocols |
| Fluorescent Reporters | Live imaging of specific structures | Transgenic lines or vital dyes |
| Glass Capillaries | Embryo mounting and positioning | Custom-pulled to appropriate diameters |
| Vacuum Grease | Creating barriers in imaging chambers | Prevents mixing between wells |
| Paraformaldehyde | Sample fixation for validation studies | 4% concentration in PBS |
Institutional and Safety Considerations
The protocol requires adherence to local institutional guidelines for laboratory safety and ethics. All animal experimentation should be performed in accordance with appropriate regulatory frameworks, such as the UK Animals (Scientific Procedures) Act 1986 for researchers in the United Kingdom, with approval from institutional ethical review processes and performed by licensed individuals [23]. Similar institutional permissions and approvals must be obtained from relevant local authorities before performing any experiments involving regulated species.
Data Processing and Analysis
LLSM generates substantial data volumes, easily reaching 100 GB to 1 TB for each time-lapse experiment, creating significant challenges in data management and analysis [25]. Effective processing pipelines typically include:
Data Preprocessing: Steps include background subtraction, flat-field correction, and channel registration for multi-color acquisitions. For dithered lattice images, processing may include deskewing to correct for the oblique light-sheet orientation relative to the detection axis.
Deconvolution: While LLSM provides excellent optical sectioning, deconvolution can further enhance image quality by reassigning out-of-focus light using measured or calculated point spread functions. The choice between measured and theoretical PSFs depends on the required accuracy and available resources.
Time-Lapse Registration: Embryos may undergo movement during extended imaging sessions, requiring volumetric registration to stabilize the time series for quantitative analysis of cellular dynamics.
Segmentation and Quantitative Analysis: Advanced computational tools are often necessary to extract quantitative information from the rich LLSM datasets. These may include automated cell segmentation, tracking of cellular trajectories, quantification of fluorescence intensities, and analysis of morphological dynamics.
The development of standardized computational workflows remains an active area of research, with emerging tools including machine learning approaches for identifying subcellular morphological motifs in 3D microscopy images [29]. These computational advances are crucial for fully leveraging the rich information content of LLSM datasets in developmental biology research.
Troubleshooting and Optimization
Successful implementation of LLSM for mouse embryo imaging often requires troubleshooting common challenges:
Poor Embryo Viability: If embryos show signs of degeneration during extended imaging sessions, verify culture medium composition and preparation, ensure complete gas equilibration, minimize temperature fluctuations, and reduce laser exposure by optimizing imaging parameters.
Suboptimal Image Quality: For images with poor signal-to-noise ratio, consider increasing camera exposure time (while respecting the dynamics of biological processes), optimizing laser power, selecting more appropriate lattice patterns, or implementing post-acquisition deconvolution.
Insufficient Resolution: If resolution requirements are not met, consider switching to super-resolution SIM mode (with acceptance of slower acquisition speeds), selecting higher resolution lattice patterns (e.g., hexagonal instead of square), or using detection objectives with higher numerical apertures.
Spatial Heterogeneity in Image Quality: For variations in image quality across the field of view, ensure proper alignment of the lattice light-sheet with the detection plane, consider implementing adaptive optics to correct for sample-induced aberrations [29], or utilize tiling strategies with recalibration between positions.
The exceptional capabilities of Lattice Light-Sheet Microscopy continue to expand the frontiers of live embryonic imaging, enabling unprecedented observation of developmental processes in their native three-dimensional context with minimal perturbation. As the technology becomes more accessible and computational analysis tools more sophisticated, LLSM promises to yield fundamental new insights into the complex choreography of mammalian embryogenesis.
Optical Coherence Microscopy (OCM) is emerging as a powerful, label-free imaging modality that combines the principles of optical coherence tomography and confocal microscopy to provide non-invasive, three-dimensional, high-resolution imaging of biological specimens [1]. For researchers in developmental biology and drug development, OCM offers significant advantages for the long-term study of dynamic processes in living tissues without the need for contrast agents or fluorescent labels that can potentially cause phototoxicity or alter biological function [30]. This application note details the methodology and quantitative benefits of implementing OCM for the structural assessment of mouse embryos, providing a framework for researchers to integrate this technology into their time-lapse microscopy workflows.
The capability of OCM to resolve cellular and subcellular features in living embryos over extended periods makes it particularly valuable for developmental studies. By enabling continuous volumetric imaging without compromising viability, OCM provides unprecedented access to the morphokinetic events that define normal and abnormal embryonic development [1]. When applied to mouse models—which exhibit significant genetic and developmental similarity with humans—OCM facilitates critical insights into mammalian development and potential teratogenic effects of pharmaceutical compounds [1] [31].
Key Quantitative Findings in Embryo Assessment
Research utilizing OCM for embryo evaluation has yielded significant quantitative metrics that correlate early developmental events with embryonic viability and developmental potential. The following table summarizes key temporal parameters identified through OCM analysis that serve as biomarkers for embryo quality assessment.
Table 1: Key morphokinetic parameters for embryo quality assessment identified via OCM
| Developmental Parameter | Correlation with Developmental Potential | Measurement Method |
|---|---|---|
| Timing of 2nd embryonic cell cycle | Correlates with blastocyst formation [1] | 3D OCM time-lapse imaging |
| Timing of 3rd embryonic cell cycle | Associated with hatching capability [1] | 3D OCM time-lapse imaging |
| Nuclei visualization capability | Accurate identification of cleavage and quantification of symmetry from 1-cell to 8-cell stage [1] | OCM en face slices and 3D rendering |
| Blastocyst expansion grading | Classification of embryos according to established Gardner grading system (Stages 1-6) [1] | OCM visualization of blastocoel cavity, ICM, and TE |
Beyond embryonic development, OCM has demonstrated significant diagnostic capabilities in tissue assessment, as evidenced by research on cervical tissue classification. The table below summarizes the performance metrics of a computer-aided diagnosis system using OCM images.
Table 2: Performance of OCM-based classification in tissue diagnostics
| Classification Task | Performance Metric | Result |
|---|---|---|
| Five-class cervical tissue classification | Accuracy | 88.3±4.9% [31] |
| Binary classification (low-risk vs. high-risk) | Area Under Curve (AUC) | 0.959 [31] |
| Binary classification | Sensitivity | 86.7±11.4% [31] |
| Binary classification | Specificity | 93.5±3.8% [31] |
| Human expert performance (high-risk identification) | Average Sensitivity | 80% [31] |
| Human expert performance (high-risk identification) | Average Specificity | 89% [31] |
Experimental Protocols
Integrated OCM Imaging System for Embryo Culture
Purpose: To enable long-term time-lapse OCM imaging of mouse embryos while maintaining optimal culture conditions essential for normal development [1].
Materials:
- Custom OCM system with broadband superluminescent diode source
- 20X objective lens (providing ~2.1 μm axial and ~1.0 μm lateral resolution in tissue)
- Commercial incubator (e.g., Heracell VIOS 160i) with gas control (5% O₂, 6% CO₂)
- Motorized 3-axis sample stage
- Embryo imaging dish (e.g., IVF store V005001) accommodating up to 25 embryos
- Environmental chamber maintaining 37°C temperature and high relative humidity
Procedure:
- System Configuration: Customize a compact OCM sample arm and bright-field (BF) imaging system to fit within the commercial incubator. Co-register BF and OCM imaging capabilities to provide complementary 2D and 3D information [1].
- Environmental Stabilization: Place the integrated imaging system inside the incubator and allow temperature and gas concentrations to stabilize to appropriate levels (37°C, 5% O₂, 6% CO₂) before introducing embryos [1].
- Embryo Loading: Transfer cultured mouse embryos at the one-cell stage to the specialized embryo imaging dish containing pre-equilibrated culture medium [1].
- Automated Imaging Setup: Program an automated acquisition pipeline to acquire co-registered BF and 3D OCM images at regular intervals (e.g., every 10 minutes) for extended durations (up to 150 hours) to cover development from one-cell stage to fully hatched blastocyst [1].
- Image Acquisition: For each time point, automatically move the sample well to the imaging location using prior knowledge of well positions, followed by image-guided auto-tracking and auto-focusing to center the embryo samples before acquiring volumetric OCM data [1].
- Data Management: Transfer acquired 3D OCM volumes to a dedicated storage server for subsequent analysis and archiving [1].
Embryo Mounting and Viability Maintenance
Purpose: To secure embryos for stable long-term imaging while minimizing mechanical stress and maintaining developmental potential.
Materials:
- Embryo Culture Medium (e.g., CMRL medium with Knock Out serum and L-Glutamine)
- M2 dissection medium
- Glass capillaries for mounting
- Diamond knife for capillary customization
- 8-chambered slides or specialized imaging dishes
Procedure:
- Medium Preparation: Prepare Embryo Culture Medium fresh for each experiment by combining basal medium with appropriate supplements and serum. Equilibrate in a humidified incubator at 37°C and 5% CO₂ for at least 1 hour before use, with loosened caps to allow gas exchange [23].
- Mounting Implement Fabrication: Using a Bunsen flame with a defined blue cone, heat glass capillaries evenly until malleable, then pull steadily to create fine tips appropriate for embryo staging. Use a diamond knife to score and break capillaries to appropriate lengths [23].
- Embryo Transfer: Following established dissection protocols for post-implantation mouse embryos, transfer embryos to the imaging chamber containing pre-equilibrated culture medium [23].
- Secure Mounting: Position embryos using fine implements to ensure stability during extended imaging sessions while minimizing physical constriction that could impair development [23].
- Environmental Protection: For open imaging chambers, create protective barriers using vacuum grease to minimize medium evaporation and maintain local humidity [23].
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful implementation of OCM for embryonic imaging requires specific materials and reagents to maintain embryo viability while acquiring high-quality volumetric data. The following table details essential components and their functions in the experimental workflow.
Table 3: Essential research reagents and materials for OCM embryo imaging
| Item | Function/Application | Specifications/Notes |
|---|---|---|
| Embryo Culture Medium | Supports embryo development during extended imaging sessions | CMRL medium with Knock Out serum; requires gas equilibration [23] |
| Specialized Imaging Dishes | Houses embryos during time-lapse imaging | Commercial dishes (e.g., IVF store V005001) can accommodate up to 25 embryos [1] |
| Glass Capillaries | Customized implements for embryo mounting | Pulled to appropriate fineness for embryo staging using heat [23] |
| Gas-Controlled Incubator | Maintains physiological conditions during imaging | Heracell VIOS 160i or equivalent; controls O₂ (5%), CO₂ (6%), temperature, humidity [1] |
| OCM System Components | Enables high-resolution 3D imaging | Broadband light source, 20X objective, motorized stage, interferometer [1] [30] |
Data Processing and Analysis Workflow
The volumetric data generated by OCM imaging requires specialized processing and analysis pipelines to extract meaningful biological insights. The following diagram illustrates the key stages in transforming raw OCM data into quantitative assessments of embryonic development.
Key Analysis Parameters
The OCM data processing pipeline enables quantification of specific structural and temporal parameters critical for embryonic assessment:
Cell Cycle Timing: Precise measurement of the duration between cleavage divisions, particularly the second and third embryonic cell cycles, which correlate with blastocyst formation and hatching capability [1].
Nuclear Dynamics: Tracking of nuclear size, position, and count during early development (1-cell to 8-cell stage), where OCM provides strong contrast between nuclei and cytoplasm [1].
Blastocyst Morphometrics: Quantitative assessment of blastocoel formation, inner cell mass (ICM) organization, and trophectoderm (TE) structure using the established Gardner grading system [1].
Zona Pellucida Characterization: Evaluation of zona pellucida thickness and uniformity throughout preimplantation development [1].
Optical Coherence Microscopy represents a transformative technology for label-free evaluation of embryonic development, providing unprecedented access to the three-dimensional structural dynamics of living embryos without compromising viability. The integration of OCM with advanced computational analysis methods creates a powerful platform for objective assessment of embryo quality, with significant implications for both basic developmental biology and pharmaceutical testing. As demonstrated in the referenced studies, the capability of OCM to identify critical morphokinetic parameters that predict developmental outcomes offers researchers a robust, non-invasive tool for quantifying embryonic development in longitudinal studies. The continued refinement of OCM technology and analysis protocols promises to further enhance our understanding of mammalian development and improve assessment protocols for developmental toxicity screening.
Application Notes for Time-Lapse Microscopy in Mouse Embryo Culture Research
Within the context of time-lapse microscopy for mouse embryo culture research, the selection of an appropriate nuclear DNA labeling strategy is paramount. Visualizing chromosomes and tracking cell lineages in living embryos requires methods that are highly efficient, minimally invasive, and compatible with long-term viability. The choice between electroporation, fluorescent dyes, and viral vectors involves critical trade-offs between transfection efficiency, cytotoxicity, and experimental timeline. This document provides a comparative analysis and detailed protocols for these key techniques, tailored specifically for researchers employing advanced imaging modalities like lattice light-sheet microscopy (LLSM) to study preimplantation development [23] [32].
Technical Comparison of Labeling Methods
The table below summarizes the core characteristics of the primary nuclear DNA labeling methods as applied to mouse embryo research.
Table 1: Strategic Comparison of Nuclear DNA Labeling Methods for Mouse Embryo Research
| Method | Key Principle | Typical Efficiency in Mouse Embryos | Relative Cytotoxicity / Impact | Ideal Use Case |
|---|---|---|---|---|
| mRNA Electroporation | Electrical pulses create transient pores in cell membranes, allowing mRNA encoding fluorescent histones (e.g., H2B-GFP) to enter cells [32]. | ~75% [32] | Low; no observed impact on cell number or lineage specification (trophectoderm/epiblast) when optimized [32]. | Long-term, high-resolution live imaging of cell divisions and lineage tracing [32]. |
| Fluorescent Dyes (e.g., DAPI) | Cell-permeant dyes bind stoichiometrically to DNA, often with AT-base pair preference [33]. | High for fixed samples; variable and lineage-specific in live embryos [32]. | High for live imaging; prolonged incubation can induce DNA damage and alter mitotic progression, compromising viability [32]. | End-point, fixed-sample counterstaining or very short-term live imaging with caution [33] [32]. |
| Viral Vectors (e.g., rAAV, Lentivirus) | Engineered viruses transduce cells to enable long-term expression of fluorescent protein transgenes [34] [35]. | Variable; AAV shown to be transient (≤24 hrs), Lentivirus prone to silencing in early embryos [32]. | Moderate; potential for immunogenicity and insertional mutagenesis, though AAV is considered safer [34]. | Stable, long-term transgene expression where electroporation is inefficient; less suitable for rapid, high-efficiency embryo labeling [34] [32]. |
Detailed Experimental Protocols
Protocol: mRNA Electroporation for Mouse Embryos
This protocol is optimized for introducing mRNA encoding fluorescently tagged histones (e.g., H2B-mCherry) into cleavage-stage or blastocyst-stage mouse embryos for live imaging [32] [36].
I. Reagent Solutions
- mRNA: Purified mRNA (e.g., H2B-mCherry, NLS-EGFP) at a concentration of 700-800 ng/µL in nuclease-free water [32].
- Electroporation Buffer: A low-conductivity HEPES-based buffer. The specific ionic composition (e.g., K⁺ vs. Mg²⁺) can be tuned to balance viability and efficiency [37].
- Embryo Culture Medium: Pre-equilibrated in a humidified incubator at 37°C and 5% CO₂ [23].
II. Step-by-Step Procedure
- Preparation: Set up the electroporator (e.g., a square-wave generator like BTX ECM 830) and 1-2 mm gap cuvettes. Prepare a working solution by mixing the mRNA into the electroporation buffer.
- Embryo Handling: Transfer 10-20 healthy mouse embryos into the mRNA/electroporation buffer solution in the cuvette. Ensure embryos are oriented to minimize blastomere fusion (e.g., for two-cell embryos, orient the contact surface between blastomeres horizontally to the electrodes) [36].
- Electroporation Pulse: Apply a single set of pulses. An example of an optimized condition is 20 V for 3 ms (on) / 97 ms (off), repeated 5 times [36].
- Post-Pulse Recovery: Immediately transfer the embryos from the cuvette into pre-warmed and equilibrated culture medium.
- Culture and Expression: Culture the embryos under standard conditions (37°C, 5% CO₂). Expression of the fluorescent protein can typically be assessed within a few hours.
III. Workflow Visualization
Protocol: DAPI Counterstaining for Fixed Samples
DAPI is ideal for end-point analysis to identify all nuclei after fixation [33].
I. Reagent Solutions
- DAPI Stock Solution: 5 mg/mL in deionized water or DMF. Aliquot and store at -20°C, protected from light.
- Working Solution: Dilute stock to 300 nM in Phosphate-Buffered Saline (PBS).
- Mounting Medium: Use an antifade reagent such as ProLong Gold or SlowFade Gold.
II. Step-by-Step Procedure
- Fixation and Permeabilization: Fix and permeabilize embryos using a standard protocol appropriate for your sample. Note: fixation is not required for DAPI staining but is typically done for immunostaining.
- Equilibration: Briefly equilibrate the sample in PBS.
- Staining: Add ~300 µL of the 300 nM DAPI staining solution to cover the sample. Incubate for 1-5 minutes at room temperature, protected from light.
- Rinsing: Rinse the sample several times with PBS to remove unbound dye.
- Mounting: Drain excess buffer and mount the sample using an antifade mounting medium. Seal the coverslip.
- Imaging: View using a fluorescence microscope with a UV or DAPI filter set [33].
The Scientist's Toolkit: Key Research Reagents
Table 2: Essential Reagents for Nuclear DNA Labeling in Embryo Research
| Reagent / Solution | Function / Application | Example & Notes |
|---|---|---|
| H2B-mCherry / NLS-EGFP mRNA | Encodes a fluorescent protein fused to a nuclear localization signal (NLS) or histone for specific nuclear labeling. | Critical for live imaging; allows direct visualization of chromosomes and mitosis [32]. |
| HEPES-based Electroporation Buffer | Provides a stable pH and controlled ionic environment during electroporation. | Low conductivity buffers can enhance efficiency and viability. Mg²⁺ can improve cell recovery [37]. |
| Square-Wave Electroporator | Instrument that delivers controlled electrical pulses to temporarily permeabilize cell membranes. | Enables highly efficient, non-viral delivery of nucleic acids into embryos [36]. |
| DAPI (4',6-diamidino-2-phenylindole) | A fluorescent dye that binds strongly to AT-rich regions in dsDNA. | Standard, high-contrast nuclear counterstain for fixed samples. Use with antifade mounting medium [33]. |
| Lattice Light-Sheet Microscope (LLSM) | Fluorescence microscope that illuminates a thin plane of the sample, minimizing photodamage. | Ideal for long-term, high-resolution live imaging of sensitive samples like embryos [23] [32]. |
| Embryo Culture Medium (e.g., CMRL-based) | Supports the development of mouse embryos ex vivo during and after manipulation. | Must be pre-equilibrated for gas and temperature; composition may vary by embryonic stage [23]. |
Strategic Decision Pathway
The following diagram outlines the decision-making process for selecting the optimal nuclear labeling strategy based on key experimental requirements.
Automated Data Acquisition Pipelines for Long-Term Imaging Inside Incubators
Within the field of time-lapse microscopy for mouse embryo culture research, the establishment of robust, automated data acquisition pipelines is fundamental to success. The ability to automatically and non-invasively monitor embryonic development from the one-cell stage to the fully hatched blastocyst over several days provides unparalleled insights into morphokinetic properties and developmental potential [38]. Such pipelines are critical for enhancing the success rates of in vitro fertilization (IVF) procedures by enabling the selection of high-quality embryos based on dynamic developmental events rather than static morphological snapshots [38] [39]. This document details the application notes and protocols for implementing these systems, framed within the context of a broader thesis on time-lapse microscopy in mouse embryo research.
System Configurations and Quantitative Comparisons
Selecting an appropriate imaging system involves balancing factors such as resolution, acquisition speed, cost, and compatibility with long-term embryo culture. The following systems have been successfully implemented for research involving mouse embryos and other live cells.
Table 1: Comparison of Imaging System Configurations for Long-Term Incubator Imaging
| System Feature | OCM & Bright-Field System [38] | Light-Sheet Fluorescence Microscopy [32] | Bio-DOME / Fluoro-DOME [40] |
|---|---|---|---|
| Microscopy Modality | Optical Coherence Microscopy (OCM) & Bright-Field | Light-Sheet Fluorescence Microscopy (LSFM) | Bright-Field & Fluorescence (Fluoro-DOME) |
| Key Advantage | Label-free 3D visualization of nuclei and microstructures; reveals cellular and subcellular features. | Minimal phototoxicity; suitable for long-term imaging of sensitive samples like human embryos. | Closed-loop, real-time optical control; extremely low-cost; open-source and modular. |
| Resolution (Axial/Lateral) | ~2.1 μm / ~1.0 μm (in tissue) | Not explicitly stated, but suitable for tracking chromosome segregation. | Resolution is sufficient for tracking collective cell behavior (e.g., wound healing). |
| Sample Throughput | Up to 25 embryos per commercial imaging dish. | Suitable for individual blastocysts. | Single field of view, but can be adapted. |
| Approximate Cost | Not stated (likely high-end). | Not stated (typically high-end). | £650 (Bio-DOME); <£1,000 (Fluoro-DOME). |
| Primary Application in Literature | Mouse embryo developmental staging and blastocyst quality assessment. | Live imaging of chromosome segregation errors in human and mouse blastocysts. | Long-term imaging and spatio-temporal optical control of living cells (e.g., wound healing, cancer cells). |
Beyond the systems above, confocal microscopy remains a powerful, though more phototoxic, option for in vivo imaging that provides optical sectioning for 3D reconstructions [41]. Its use in time-lapse imaging requires careful optimization of laser exposure to maintain sample viability over long periods.
Experimental Protocol: Automated Time-Lapse Imaging of Mouse Embryos
The following protocol describes the procedure for long-term, automated imaging of preimplantation mouse embryo development using a system such as the OCM and Bright-Field setup described in the search results [38].
Research Reagent Solutions and Essential Materials
Table 2: Essential Materials for Automated Embryo Time-Lapse Imaging
| Item | Function / Explanation | Example / Specification |
|---|---|---|
| Dual-Modality Imaging System | A compact system placed inside the incubator for uninterrupted culture. Provides co-registered 3D OCM and 2D bright-field data. | Custom OCM system with ~1.0 μm lateral resolution and integrated bright-field camera [38]. |
| Environmental Chamber | Maintains physiological conditions (temperature, CO₂, O₂, humidity) essential for normal embryo development during extended imaging. | Standard cell culture incubator (e.g., Heracell VIOS 160i) with gas control (5% O₂, 6% CO₂) [38]. |
| Embryo Imaging Dish | A specialized culture dish designed for imaging, allowing stable embryo positioning and media retention over many days. | Commercial embryo imaging dish (e.g., IVF store V005001), capacity for up to 25 embryos [38]. |
| Motorized Sample Stage | Enables automated, precise movement of the sample for sequential imaging of multiple embryos. | 3-axis motorized sample stage integrated with the acquisition software [38]. |
| Automated Acquisition Software | Orchestrates the entire pipeline: stage movement, auto-focusing, and image acquisition at set intervals. | Custom software with auto-tracking and auto-focusing capabilities [38]. |
Step-by-Step Procedure
System Setup and Calibration:
- Install the imaging system inside the environmental chamber. For custom systems like the DOME, this involves 3D printing the components (in ABS for stability) and assembling the imaging, projection, and computation modules [40].
- Power on the system and allow it to equilibrate to the incubator's internal temperature to avoid thermal drift during imaging.
- Calibrate the motorized stage to map the positions of all wells in the imaging dish.
Embryo Preparation and Loading:
- Obtain mouse zygotes or early-stage embryos following standard animal protocols and in vitro fertilization techniques.
- Culture embryos in pre-equilibrated medium under oil in the specialized embryo imaging dish.
- Carefully load the dish onto the pre-warmed motorized stage within the incubator.
Pipeline Configuration and Initiation:
- In the acquisition software, input the sample index and the known locations of the wells.
- Set the imaging parameters. For OCM, this includes the 3D scan settings. A time-lapse interval of every 10 minutes for over 150 hours has been successfully used to capture development from the one-cell stage to a hatched blastocyst [38].
- Initiate the automated acquisition pipeline. The software will execute a loop for each sample position: moving the well to the imaging location, performing image-guided auto-tracking and auto-focusing to center the embryo, and finally acquiring the co-registered BF and 3D OCM images [38].
Data Management and Processing:
- Transfer the acquired data to a storage server for subsequent analysis. The data volume from 3D time-lapses is substantial and requires adequate storage solutions.
- Use appropriate software for data analysis. For complex datasets, such as those tracing individual nuclei in 3D over time, semi-automated segmentation methods using customized deep learning models may be required [32].
Diagram 1: Automated data acquisition workflow for embryo imaging.
Application Note: Key Insights from Mouse Embryo Imaging
Implementing the described pipeline has yielded critical insights into early development. Time-lapse OCM imaging can detect structural features such as the precise timing of cleavage divisions, the compaction process, and the initiation of cavitation leading to blastocoel formation [38]. Furthermore, research indicates that the timing of the second and third embryonic cell cycles is correlated with successful blastocyst formation and its subsequent hatching capability [38]. This morphokinetic parameter, easily captured by an automated pipeline, serves as a non-invasive biomarker for embryo viability.
For studies requiring genetic or protein labeling, such as tracing cell lineages or monitoring gene expression, mRNA electroporation has been optimized as an efficient method for introducing labels like H2B-GFP into blastocyst-stage mouse embryos without significantly affecting development or lineage specification [32]. This allows for the combination of rich structural data from OCM with molecular specificity.
Diagram 2: Workflow for live imaging of nuclear dynamics in embryos.
Maximizing Viability and Data Quality: A Troubleshooting Guide for Robust Imaging
Critical Steps for Maintaining Physiological Culture Conditions Throughout Imaging
In time-lapse microscopy of mouse embryos, maintaining physiological culture conditions is not merely a supportive technique but a foundational aspect that determines the validity and reproducibility of experimental data. The period following implantation is marked by rapid, dynamic morphogenetic events that establish the embryonic axes, germ layers, and first organs [23]. These processes are exquisitely sensitive to environmental perturbations. Deviation from optimal conditions can induce cellular stress, alter developmental kinetics, and produce artifacts that confound the interpretation of high-resolution imaging data. Consequently, the protocols for embryo isolation, mounting, and culture are as critical as the imaging parameters themselves to ensure that the observed cellular behaviors genuinely reflect in vivo development.
This protocol provides a detailed framework for maintaining physiological integrity throughout the isolation and lattice light-sheet microscopy (LLSM) of post-implantation mouse embryos. By minimizing photodamage through selective plane illumination and providing a stable culture environment during extended imaging sessions, researchers can capture crucial developmental events with high spatial and temporal fidelity [23]. The approach described herein is also adaptable for use with stem cell-derived embryo models and organoids, broadening its applicability in developmental biology and drug discovery.
Establishing the Experimental Foundation: Isolation and Pre-Imaging Preparation
Institutional Permissions and Timed Mating
All experimentation must commence with the appropriate ethical and regulatory approvals. The procedures outlined require full accordance with relevant national legislation, such as the UK Animals (Scientific Procedures) Act 1986, and must be approved by an institutional ethical review process and performed under a project license by personal license holders [23].
- Timed Mating Setup: Plan experiments 7–10 days in advance. Pair stud males with females from outbred strains (e.g., CD1) near the start of the dark cycle in the animal facility. Using females in proestrus (identified by a swollen, moist, pink vaginal opening) increases the chance of successful pregnancy.
- Vaginal Plug Check: Check females early the next morning. The presence of a vaginal plug indicates mating, and noon on that day is designated as 0.5 days post coitum (dpc). Embryonic ages in this protocol are calculated from this point, though developmental stage variation can occur between and within litters.
Preparation and Equilibration of Culture Media
The preparation of dissection and culture medium is a critical step to ensure embryo viability. The following protocol is sufficient for imaging up to fifteen 5.5 dpc embryos for up to 8 hours.
Table 1: Composition of Embryo Culture Medium for Post-Implantation Mouse Embryos
| Component | Volume | Final Concentration/Purpose | Critical Notes |
|---|---|---|---|
| CMRL Medium | 2 mL | Base nutrient source | Prepare fresh for each experiment in a laminar flow hood. |
| KnockOut Serum | 2 mL | 50% concentration; provides essential growth factors | Ensures optimal support for embryonic development. |
| L-Glutamine (200 mM) | 42 µL | Essential amino acid for cell health | Aliquot and freeze; ensure all crystals are fully dissolved before use to prevent imaging artifacts [23]. |
Execution Steps:
- Clean the biosafety cabinet and all components with 70% ethanol.
- Combine 2 mL of CMRL, 2 mL of KnockOut Serum, and 42 µL of L-Glutamine in a 15 mL screw-cap tube.
- Vortex the mixture thoroughly and place it in a humidified incubator (37°C, 5% CO₂) for at least 1 hour before use, with the screw cap loosened to allow for gas exchange and proper equilibration [23].
- Warm an aliquot of M2 dissection medium (approximately 8 mL per litter) to room temperature (22°C).
For later-stage embryos (6.5+ dpc), alternative culture medium compositions, such as 50% DMEM-FluoroBrite with 50% rat serum, may be used.
Core Experimental Protocol: Mounting and Imaging with Physiological Maintenance
Assembly and Pre-equilibration of the Imaging Chamber
The imaging chamber must be assembled with care to prevent contamination and physical damage to the embryos.
Mounting Implement Preparation:
- Using a Bunsen flame with a well-defined blue cone, gently heat the center of a glass capillary by rolling it between your fingers over the flame.
- Once the center becomes malleable, remove it from the flame and pull both ends evenly and quickly. The speed of pulling determines the fineness of the tip.
- Gently snap the ends off the pulled central region and collect the pulled capillaries in a sample vial. Use a diamond knife to score and break the capillaries into fragments roughly the length of the imaging chamber's width. Practice is required to consistently produce capillaries of the appropriate width for different embryonic stages [23].
Chamber Assembly:
- Take an 8-chambered slide and fill the four end wells with 400 µL of pre-equilibrated culture medium each. This helps maintain humidity and minimize evaporation in the central imaging wells.
- Fill a 5 mL syringe with vacuum grease and attach a 200 µL pipette tip as a nozzle. Extrude the grease to create two barriers at the top and bottom of the central, unfilled wells, taking care not to smear grease in the center of the well where the embryo will be placed [23].
- Fill the central wells with pre-equilibrated Embryo Culture Medium and place the entire chamber slide back into the incubator for at least 1 hour to allow for temperature and pH stabilization before introducing the embryos.
Embryo Mounting and LLSM Imaging Parameters
Following the dissection of post-implantation mouse embryos (as detailed in Thowfeequ et al. and Stower et al. [23]), the mounting and imaging steps are crucial for maintaining viability.
Embryo Mounting:
- Transfer a healthy, undamaged embryo into the prepared central well of the imaging chamber.
- Using a fine pipette or needle, gently maneuver the embryo onto the tip of a previously prepared pulled glass capillary.
- Position the capillary with the attached embryo so that it is suspended in the medium, ensuring it is secure and will not move during imaging but is not compressed.
LLSM Imaging Setup:
- Minimizing Photodamage: Leverage the principle of selective plane illumination inherent to LLSM. The ZEISS LLSM L7 system generates a thin light-sheet from two-dimensional optical lattices of interfering Bessel beams, which provides high resolution with significantly lower illumination overhead compared to confocal microscopy, thereby reducing photobleaching and photodamage [23].
- Parameter Configuration: Set up imaging parameters, including exposure time, laser power, and z-stack interval, to balance signal-to-noise ratio with embryo viability. Fast dual-camera acquisition is essential for capturing highly dynamic cellular processes.
- Concurrent Imaging: A key advantage of this protocol is the ability to image several embryos simultaneously in a single chamber. This is vital for accounting for biological variability between embryos and building a robust, averaged map of cellular behaviors [23].
Table 2: Key Research Reagent Solutions for Embryo Culture and Imaging
| Reagent/Material | Function/Application | Specifications and Notes |
|---|---|---|
| CMRL Medium | Base nutrient solution for embryo culture | Used at 50% concentration with serum. |
| KnockOut Serum | Serum replacement providing essential components | Supports development of post-implantation embryos. |
| L-Glutamine | Essential amino acid for cellular metabolism | Must be fully dissolved to avoid light refraction artifacts during imaging [23]. |
| M2 Medium | Buffered salt solution for embryo dissection | Used at room temperature during isolation procedures. |
| Pulled Glass Capillaries | Physical support for mounting embryos | Fineness is tailored to embryonic stage; critical for stable, non-damaging immobilization [23]. |
| Vacuum Grease | Creates physical barriers in the imaging chamber | Prevents cross-contamination and medium evaporation between wells. |
Workflow and Data Processing
The entire process, from preparation to data output, must be executed as an integrated workflow where physiological maintenance is prioritized at every stage. The diagram below illustrates the critical path and decision points.
Post-Processing of LLSM Data
Following acquisition, the large 4D datasets generated by LLSM require specialized processing pipelines to prepare them for quantitative downstream analysis. This involves tasks such as:
- Deskewing and deconvolution to correct for the inherent light-sheet geometry and improve image clarity.
- Temporal and spatial registration to align images across time points and different channels.
- Segmentation and tracking of cells and subcellular structures over time to quantify dynamic behaviors such as migration speed, directionality, and morphological changes.
These processed data are essential for building accurate maps of cellular behavior and for characterizing how genetic or chemical perturbations affect key developmental processes [23].
The Scientist's Toolkit: Essential Materials for Success
Table 3: The Scientist's Toolkit: Key Equipment and Software
| Tool Category | Specific Example | Function in Protocol |
|---|---|---|
| Microscopy System | ZEISS LLSM L7 | Generates thin light-sheet for high-resolution, low-photodamage 4D imaging [23]. |
| Cell Culture Equipment | Humidified Incubator (37°C, 5% CO₂) | Maintains physiological temperature and pH for embryo culture before and during imaging [23]. |
| Sample Preparation | Pulled Glass Capillaries | Provides a stable, non-invasive mount for suspending embryos in the imaging chamber [23]. |
| Image Analysis Software | ImageJ / FIJI | Open-source platform for processing, analyzing, and visualizing 4D microscopy data [42]. |
Preventing and Identifying Photodamage and Phototoxicity in Sensitive Embryos
Live imaging of mouse embryos provides invaluable insights into developmental processes but introduces the significant risk of photodamage—light-induced cellular stress and toxicity that can compromise embryo viability and experimental results. Phototoxicity arises when the cumulative light exposure during microscopy triggers detrimental cellular responses, primarily through the generation of reactive oxygen species (ROS) and thermal damage [43]. For researchers conducting time-lapse microscopy on mouse embryos, mitigating this risk is paramount to obtaining physiologically relevant data while maintaining embryo developmental potential. This challenge is particularly acute in the context of a broader thesis on time-lapse microscopy of mouse embryo culture, where prolonged imaging sessions are necessary to capture key developmental events. This application note provides detailed protocols and analytical frameworks for identifying, quantifying, and preventing photodamage, enabling researchers to balance image quality with embryo health.
Mechanisms and Manifestations of Phototoxicity
Fundamental Photodamage Pathways
Photodamage in embryonic cells occurs through several interconnected mechanisms, with the primary pathway being multi-photon absorption processes. Under typical microscopy illumination conditions, photodamage in embryonic systems arises through both two- and three-photon absorption processes in a cumulative manner [44] [45]. When fluorescent molecules absorb photons, they enter an excited state and can transfer energy to molecular oxygen, generating highly reactive ROS that damage proteins, lipids, and DNA.
A second significant mechanism is thermal damage, particularly relevant with pulsed laser systems where localized heating can denature proteins and disrupt cellular structures. The production of reactive oxygen species is a key mechanism of phototoxicity during fluorescent observation, making antioxidant capacity a critical factor in embryonic resilience [43].
Observable Indicators of Embryo Photodamage
Identifying phototoxicity requires monitoring both morphological and developmental indicators:
- Developmental Arrest: Cessation of cleavage divisions or failure to progress to subsequent developmental stages.
- Cellular Morphology Changes: Membrane blebbing, cytoplasmic granulation, or abnormal nuclear condensation.
- Metabolic Dysfunction: Reduced mitochondrial activity or altered metabolic rates.
- Compromised Viability: Failure to implant or reach term development after transfer.
Table 1: Quantitative Indicators of Photodamage in Mouse Embryos
| Indicator Category | Specific Parameters | Acceptable Range | Phototoxic Threshold |
|---|---|---|---|
| Developmental Timing | Duration of 2nd cell cycle | 40-60 hours post-fertilization | >70 hours [1] |
| Blastocyst Formation | Formation rate by 100 h | >80% in controls | <60% [1] |
| Cellular Integrity | ROS levels (fluorescence) | <2x control levels | >3x control levels [43] |
| Gene Expression | Stress marker induction | <2x control | >3x control |
Microscope Technology Selection for Reduced Phototoxicity
Comparative Analysis of Imaging Modalities
Choosing appropriate microscopy technology is the first critical step in minimizing photodamage. Different imaging systems vary significantly in their illumination strategies and associated phototoxic potential:
Light-Sheet Microscopy: This technique illuminates only the focal plane being imaged, dramatically reducing total light exposure. Lattice light-sheet microscopy (LLSM) offers particularly advantageous characteristics for embryonic imaging, providing unprecedented spatial and temporal resolution while minimizing photodamage [23] [22]. The selective plane illumination characteristic of light-sheet imaging results in lower illumination overhead, leading to decreased photobleaching and increased viability compared to confocal microscopy or epi-illumination widefield approaches [23].
Multiphoton Microscopy: Utilizing near-infrared pulsed lasers, this approach limits excitation to the focal volume while reducing scattering in deeper tissues. Two-photon microscopy reduces photodamage since there is no absorption or fluorescence beyond the plane of focus, maintaining embryo viability especially over long imaging times [46]. For imaging depths exceeding 200 micrometers, two-photon excitation microscopy has demonstrated superior performance compared to single-photon approaches [43].
Confocal Microscopy: While providing excellent optical sectioning, point-scanning confocal systems typically expose the sample to higher total light doses. Single-photon excitation with laser scanning confocal microscopy may fail to visualize cells deeper than 50 μm from the embryo surface [43].
Table 2: Phototoxicity Comparison Across Imaging Modalities
| Microscopy Modality | Relative Phototoxicity | Optimal Application | Limitations |
|---|---|---|---|
| Widefield Epifluorescence | High | High-throughput, short-term | Whole-sample illumination |
| Laser Scanning Confocal | Medium-High | Fixed samples, high resolution | Point-scanning, photobleaching |
| Spinning Disk Confocal | Medium | Live cell imaging | Limited depth penetration |
| Two-Photon | Medium-Low | Deep tissue, live imaging | Expensive, complex alignment |
| Light-Sheet (LLSM) | Low | Long-term, sensitive samples | Sample mounting complexity |
Innovative Imaging Solutions
Recent technological advances have yielded new imaging solutions specifically designed to address phototoxicity challenges in embryonic systems:
Biaxial Light-Sheet Microscopy: This innovative approach integrates multidirectional selective plane illumination microscopy (mSPIM) with dual-view inverted SPIM (diSPIM) to achieve precise 3D rendering while minimizing light exposure. The system employs two multidirectional laser sheets introduced to the embryo from both sides and rotated by 90° for dual-axis acquisition, enabling high-resolution imaging with reduced phototoxic impact [43].
Dual-Modality Systems: Combined optical coherence microscopy (OCM) and bright-field imaging provides a label-free approach for monitoring embryo development. OCM can noninvasively provide three-dimensional high-resolution imaging of developing embryos, eliminating fluorophore-related phototoxicity entirely [1]. These systems can be compact enough to fit within standard incubators, maintaining optimal culture conditions throughout extended time-lapse experiments.
Experimental Optimization Framework
Parameter Optimization Strategy
Systematically optimizing imaging parameters represents the most effective approach for balancing signal-to-noise ratio with embryo viability:
Illumination Wavelength: Contrary to conventional wisdom, longer wavelengths do not necessarily imply reduced toxicity, emphasizing the wavelength-dependent phototoxicity effect [43]. Empirical testing of wavelength effects on specific embryonic stages is recommended.
Scan Speed: In light-sheet systems where a focused laser beam is scanned to create a pseudo-light sheet, scan speed critically influences phototoxicity. Research has demonstrated that scan speed plays a more critical role in reducing phototoxicity than irradiation intensity or the interval time between frames [43]. Faster scanning typically reduces dwell time and localized damage.
Pulse Duration and Power: For multiphoton systems, adjusting pulse duration can significantly impact the signal-to-damage ratio. Under illumination conditions typical for multiphoton imaging, general guidelines for improving the signal-to-damage ratio in two-photon or THG imaging can be achieved by adjusting the pulse duration and/or the imaging rate [44].
Temporal Sampling: The interval between image acquisitions should be optimized based on the biological process being studied. For many morphogenetic movements, intervals of 5-15 minutes provide sufficient temporal resolution while allowing cellular repair mechanisms to operate.
Environmental Control for Enhanced Viability
Maintaining embryo health during imaging requires meticulous environmental control beyond light optimization:
Temperature Stability: Implementing a two-layered incubator design effectively mitigates temperature fluctuations. A polycarbonate box surrounding the microscope system can block heat exchange between the room and chamber, maintaining temperature stability within ±0.1°C [43].
Humidity and Gas Control: Evaporation from small culture volumes can significantly impact osmolarity. The same two-layered incubator approach has proven more effective in inhibiting evaporation compared to commercialized incubators [43].
Physiological Media: Optimized culture media such as Embryo Culture Medium containing CMRL, KnockOut serum, and L-glutamine supports embryonic development during imaging sessions [23]. Antioxidant supplementation may further mitigate phototoxic effects.
Diagram 1: Phototoxicity mitigation requires integrated optimization across multiple experimental domains.
Protocol: Lattice Light-Sheet Microscopy of Post-Implantation Mouse Embryos
Embryo Isolation and Mounting
This protocol enables long-term imaging of post-implantation mouse embryos (5.5 dpc and later) with minimal photodamage, adapted from established methodologies [23] [22]:
Materials:
- Dissection microscope with dark-field illumination
- Pulled glass capillaries (1-2 mm diameter)
- Vacuum grease and 8-well chambered slides
- Embryo Culture Medium (CMRL + KnockOut serum + L-glutamine)
Procedure:
- Prepare Mounting Implements: Using a Bunsen flame, heat the center of a glass capillary while rolling it between your fingers. Once malleable, remove from flame and pull evenly and quickly from both ends. Snap the pulled region to create fine mounting capillaries.
- Assemble Imaging Chamber:
- Fill the four end wells of an 8-chambered slide with 400 μl of pre-equilibrated culture medium each.
- Using a syringe with a 200 μl tip nozzle, extrude vacuum grease to create two barriers at the top and bottom of the central unfilled wells, avoiding the center of the well.
- Mount Embryos:
- Transfer isolated 5.5 dpc embryos to the central well in a minimal volume of medium.
- Using fine forceps, carefully position each embryo within a pulled glass capillary fragment.
- Orient embryos to ensure optimal imaging access while minimizing mechanical constraint.
- Culture Stabilization: Place the assembled chamber in a humidified incubator at 37°C and 5% CO₂ for at least 1 hour before imaging to ensure temperature and pH equilibrium.
Microscope Configuration and Imaging Parameters
LLSM System Setup:
- Laser Power Calibration: Begin with minimal laser power (0.1-1% of maximum) and incrementally increase until acceptable signal-to-noise is achieved.
- Light-Sheet Alignment: Ensure the thin light-sheet derived from two-dimensional optical lattices of interfering Bessel beams is properly aligned with the detection objective.
- Detection Path Optimization: Adjust camera gain and binning settings to maximize detection efficiency rather than increasing illumination.
Safe Imaging Parameters for Long-Term Culture:
- Excitation Intensity: 0.5-5 μW/μm² (should support continuous imaging for 8-12 hours)
- Exposure Time: 10-50 ms per plane
- Z-stack Interval: 1-2 μm for entire embryo volume
- Temporal Resolution: 3-10 minutes between full volumes
- Total Acquisition Duration: 8-48 hours depending on developmental process
Protocol: Phototoxicity Assessment Using Embryonic Stem Cells
Quantitative Photodamage Evaluation
Before committing precious embryos to novel imaging conditions, this protocol uses embryonic stem cells (ESCs) as a phototoxicity biosensor [43]:
Materials:
- Mouse embryonic stem cell culture
- Propidium iodide (PI) solution (1 mg/mL)
- Control embryos for validation
- Confocal or light-sheet microscope system
Procedure:
- Cell Preparation: Culture ESCs under standard conditions until 70-80% confluency.
- PI Staining: Add propidium iodide to culture medium at 1:1000 dilution.
- Iterative Imaging:
- Define test imaging parameters (varying power, scan speed, duration)
- Acquire time-lapse sequences of ESC colonies
- Maintain control colonies in identical conditions without imaging
- Viability Quantification:
- Calculate PI-positive cell percentage after each parameter set
- Normalize to non-imaged controls
- Establish viability threshold (typically <10% increase in cell death)
- Embryo Validation: Apply optimized parameters to small cohort of embryos, assessing developmental progression and blastocyst formation rates.
Developmental Competence Assessment
The ultimate test of imaging conditions is the ability of embryos to develop normally following imaging:
- Culture Imaged Embryos for 24-48 hours beyond imaging endpoint
- Quantify Blastocyst Formation rates compared to non-imaged controls
- Perform Embryo Transfer of imaged blastocysts to assess in vivo developmental potential
- Monitor Post-Implantation Development through to live birth when possible
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Research Reagent Solutions for Phototoxicity Mitigation
| Reagent/Material | Function | Application Notes |
|---|---|---|
| CMRL Medium | Base culture medium | Specifically formulated for post-implantation embryo culture [23] |
| KnockOut Serum Replacement | Serum component | Defined formulation reduces batch variability |
| L-Glutamine Supplement | Metabolic support | Essential for embryo development; ensure complete dissolution [23] |
| Propidium Iodide | Viability staining | Cell-impermeant dye indicates membrane integrity loss [43] |
| Collagen I Gel | 3D embedding matrix | Provides structural support during imaging [43] |
| Vacuum Grease | Chamber sealing | Creates moisture barriers without toxic effects [23] |
| Pulled Glass Capillaries | Embryo mounting | Custom diameters accommodate different embryonic stages [23] |
Analytical Framework for Phototoxicity Identification
Multi-Parameter Assessment Matrix
Systematically evaluate potential photodamage using this structured approach:
Immediate Indicators (assessed during imaging):
- Abnormal cytoplasmic granulation
- Membrane blebbing or contraction
- Cessation of cytoplasmic streaming
- Altered mitotic timing
Short-Term Indicators (assessed 0-24 hours post-imaging):
- Cleavage arrest or delay
- Abnormal compaction patterns
- Failed blastocyst formation
- Reduced cell numbers in blastocysts
Long-Term Indicators (assessed 24+ hours post-imaging):
- Impaired lineage specification
- Reduced implantation potential
- Altered gene expression patterns
- Failure to reach term development
Troubleshooting Phototoxicity Issues
Diagram 2: Systematic troubleshooting pathway for addressing phototoxicity while maintaining image quality.
Successful long-term imaging of mouse embryos requires integrating phototoxicity mitigation as a fundamental consideration throughout experimental planning and execution. The strategies outlined herein—careful technology selection, systematic parameter optimization, environmental control, and rigorous assessment—enable researchers to capture crucial developmental events without compromising embryo viability. As imaging technologies continue advancing, particularly in light-sheet and multimodal approaches, the balance between image quality and physiological integrity becomes increasingly achievable. By adopting these application notes and protocols, researchers can design more robust time-lapse microscopy experiments within the broader context of mouse embryo culture research, generating reliable data that accurately reflects developmental processes unimpeded by photodamage artifacts.
Optimizing Mounting Techniques for Post-Implantation Stage Embryos
Within the broader scope of a thesis on time-lapse microscopy of mouse embryo culture, the precise mounting of post-implantation stage embryos represents a critical methodological cornerstone. The period following embryo implantation is marked by rapid morphogenetic events that establish the anatomical axes, germ layers, and first organs [23]. Capturing these dynamic processes at high spatial and temporal resolution requires imaging approaches that minimize photodamage while providing unprecedented clarity. Lattice light-sheet microscopy (LLSM) has emerged as a particularly powerful tool for this application, offering superior resolution compared to conventional light-sheet systems while maintaining the viability of delicate embryonic tissues through reduced illumination overhead [23]. This protocol details optimized mounting techniques specifically for post-implantation mouse embryos, enabling researchers to visualize and characterize the dynamic cellular behaviors driving early mammalian development.
Materials and Reagents
Research Reagent Solutions
Table 1: Essential reagents and materials for embryo mounting and imaging
| Item | Function/Application | Specifications/Notes |
|---|---|---|
| Embryo Culture Medium | Supports embryo viability during imaging | Composition: 50% CMRL, 50% Knock Out Serum, supplemented with L-Glutamine (42 μL of 200 mM per 4 mL) [23] |
| M2 Medium | Embryo dissection medium | Warmed to room temperature before use [23] |
| Glass Capillaries | Creating embryo mounting implements | Pulled to custom widths using a Bunsen flame; finer capillaries for earlier stage embryos [23] |
| 8-Chambered Slide | Imaging chamber | Central wells hold embryos; end wells filled with medium for humidity control [23] |
| Vacuum Grease | Creating barriers in imaging chamber | Prevents cross-contamination between wells without smearing on imaging area [23] |
| H2B-mCherry mRNA | Nuclear DNA labeling via electroporation | Enables chromosome tracking; 700-800 ng/μL concentration optimal [32] |
Protocol: Embryo Mounting for Lattice Light-Sheet Imaging
Preparation Phase
Institutional Permissions and Ethical Compliance
- Undertaking any experimental protocol requires adherence to local institutional guidelines for laboratory safety and ethics [23] [47].
- Obtain necessary approvals from relevant institutions before performing experiments involving regulated species [23].
Timed Mating and Embryo Isolation
- Plan timed matings 7-10 days in advance using females from outbred strains (e.g., CD1) crossed with males homozygous for desired fluorescent reporters [23].
- Check for vaginal plugs each morning; noon of the day a plug is detected is designated 0.5 days post coitum (dpc) [23].
- Isolate post-implantation embryos at desired stages (e.g., 5.5 dpc) using standard dissection techniques in pre-warmed M2 medium [23].
Media and Chamber Preparation
- Prepare Embryo Culture Medium fresh for each experiment under sterile conditions in a laminar flow hood [23].
- Equilibrate 4 mL of culture medium in a humidified incubator at 37°C and 5% CO₂ for at least 1 hour before use [23].
- For later stage embryos (6.5+ dpc), alternative culture medium compositions can be used (e.g., 50% DMEM-FluoroBrite with 50% rat serum) [23].
Mounting Implement Fabrication
Table 2: Capillary pulling parameters for different embryonic stages
| Embryonic Stage | Pulled Capillary Width | Pulling Technique | Application Notes |
|---|---|---|---|
| Early Post-Implantation (e.g., 5.5 dpc) | Finer capillaries | Faster pulling speed with shorter heating | Accommodates smaller embryo size |
| Later Stages (e.g., 6.5+ dpc) | Wider capillaries | Slower pulling speed with longer heating | Accommodates larger embryo size |
| Stem Cell-Derived Models/Organoids | Variable widths | Adjust parameters empirically | Dependent on sample dimensions |
Step-by-Step Capillary Preparation:
- Light a Bunsen flame with a well-defined blue cone [23].
- Hold a glass capillary securely at both ends and gently roll it while holding the center horizontally over the flame tip [23].
- Once the center becomes malleable, remove from the flame and pull evenly and quickly from both ends [23].
- After cooling, isolate the central pulled region by gently snapping the ends off [23].
- Using a diamond knife, mark a glass slide with guides roughly the width of the imaging chamber [23].
- Break the capillary into fragments of consistent length using the marks as guides [23].
Critical Notes: Practice is essential to determine the appropriate heating duration and pulling speed for achieving desired capillary widths. Proper technique ensures optimal embryo positioning without compression damage.
Imaging Chamber Assembly
Step-by-Step Chamber Assembly:
- Take an 8-chambered slide and fill the four end wells with 400 μL of pre-equilibrated culture medium each [23].
- Fill a 5 mL syringe with vacuum grease and attach a 200 μL tip as a nozzle, ensuring no air pockets remain [23].
- Under a dissection microscope, extrude grease to create two barriers at the top and bottom of the central (unfilled) wells [23].
- Take care not to smear grease on the center of the well where imaging will occur [23].
Critical Notes: Throughout assembly, handle the chamber on a clean Petri dish lid to prevent scratching or dirtying the bottom surface, which would compromise image quality.
Embryo Mounting and Positioning
- Transfer isolated embryos to the imaging chamber using a finely pulled glass pipette [23].
- Carefully orient embryos using the prepared glass capillaries to achieve optimal positioning for the structures of interest [23].
- Ensure embryos are securely positioned without constraining normal development or introducing mechanical stress [23].
- Multiple embryos can be mounted in the same chamber to increase data throughput and account for biological variability [23].
Nuclear Labeling for Lineage Tracing (Optional)
For studies requiring cell tracking and lineage analysis:
- Utilize mRNA electroporation for nuclear DNA labeling, optimized for blastocyst-stage embryos [32].
- Electroporate H2B-mCherry mRNA at concentrations of 700-800 ng/μL [32].
- Confirm labeling efficiency (approximately 75% in mouse embryos) without disruption to normal development or lineage specification [32].
Imaging Parameters and Data Processing
Microscope Configuration
- Utilize a ZEISS LLSM L7 system or equivalent lattice light-sheet microscope [23].
- Employ dual-camera acquisition for fast capture of highly dynamic processes [23].
- Set imaging parameters to balance spatial/temporal resolution with embryo viability [23].
Post-Processing Workflow
Following image acquisition:
- Process multi-view LSFM datasets using open-source software for computational reconstruction [47].
- Employ Fiji-based open-source software for quantification and visualization [47].
- Track cells at single-cell resolution using F-TGMM (Fuzzy-Temporal Gaussian Mixture Models) [47].
- Analyze migration patterns and morphodynamics to extract biologically meaningful data [47].
Expected Outcomes and Applications
Proper execution of this mounting protocol enables:
- High-resolution visualization of morphogenetic processes in post-implantation mouse embryos with minimal photodamage [23].
- Concurrent imaging of multiple embryos to account for biological variability and build comprehensive cellular behavior maps [23].
- Extension to stem cell-derived embryo models, organoids, and small organ explants with minor optimization [23].
- Characterization of dynamic cellular behaviors including cell migration, protrusion dynamics, and response to genetic perturbations [23].
This protocol has been successfully employed to image anterior visceral endoderm cell migration during anterior-posterior axis establishment and to characterize how basal projections and migratory behavior are affected in semaphorin-plexin pathway mutants [23].
Troubleshooting Guide
Table 3: Common issues and solutions in embryo mounting and imaging
| Problem | Potential Cause | Solution |
|---|---|---|
| Poor image quality | Grease smearing in imaging area | Carefully apply grease barriers without contacting center of well |
| Embryo damage during mounting | Overly aggressive handling | Use finer capillaries and gentle manipulation techniques |
| Developmental arrest | Phototoxicity or culture conditions | Optimize illumination intensity and ensure proper media equilibration |
| Insufficient labeling | Suboptimal electroporation parameters | Adjust mRNA concentration and electroporation settings |
| Imaging artifacts | L-glutamine crystals in medium | Ensure complete dissolution of L-glutamine before use |
Optimized mounting techniques for post-implantation stage embryos are fundamental for successful time-lapse microscopy in mouse embryo culture research. The protocol described herein leverages the advantages of lattice light-sheet microscopy to achieve high spatiotemporal resolution imaging while maintaining embryo viability. By carefully controlling mounting parameters, chamber conditions, and imaging settings, researchers can capture the dynamic cellular behaviors that drive early mammalian development. This methodology provides a robust foundation for investigating morphogenetic events, cellular dynamics, and the effects of genetic perturbations during the critical post-implantation period.
Addressing Data Artifacts and Managing Large, Multi-Dimensional Datasets
In time-lapse microscopy of mouse embryo culture, the transition from simple observation to high-resolution, long-term imaging generates vast and complex multi-dimensional datasets. This data deluge presents two fundamental challenges: the introduction of data artifacts from environmental instability and the logistical difficulty of storing and processing large image volumes. Artifacts, such as focus drift or pixel degradation, can compromise the integrity of delicate kinetic development data, while inefficient data management can stifle analysis and collaboration. This application note provides detailed protocols and data solutions to overcome these hurdles, ensuring the collection of high-fidelity, analytically ready data for developmental biology and pre-clinical drug discovery research.
Data Artifacts: Identification and Mitigation
Data artifacts in live-cell imaging are anomalies not reflecting the true biological state. For sensitive mouse embryo models, these can lead to misinterpretation of developmental kinetics.
Common Artifacts and Quantitative Impact
The following table summarizes frequent artifacts, their causes, and measurable effects on data quality.
Table 1: Common Data Artifacts in Mouse Embryo Time-Lapse Microscopy
| Artifact Type | Primary Cause | Effect on Data | Mitigation Strategy |
|---|---|---|---|
| Focus Drift | Thermal fluctuation in stage-top incubators [48] | Loss of sharpness; invalidated automated segmentation and tracking algorithms. | Use of a PID-controlled heating system with water jacket for stable thermal regulation [48]. |
| Pixel Intensity Degradation | Evaporation of culture medium due to improper humidity control [48] [49] | Altered contrast; inaccurate fluorescence quantification. | Maintain >90% relative humidity within a sealed mini-incubator or by layered mineral oil in microwells [50] [48]. |
| Aberrant Cell Morphology | pH shift from CO2 deviation [48] [49] | Compromised morphology-based embryo grading (e.g., cell symmetry, fragmentation). | Stable 5% CO2 delivery via a solenoid valve and sensor-based feedback system [48]. |
| Spatial Distortion | Optical aberrations from imaging through culture vessel plastic or air-liquid interfaces. | Inaccurate measurement of cell and embryo dimensions. | Use of open microwell platforms designed for high-resolution imaging [50]. |
Protocol: Validating Imaging System Environmental Control
This protocol ensures the imaging environment is stable prior to initiating a critical mouse embryo experiment.
Materials:
- On-stage mini-incubator (e.g., custom-built [48] or commercial)
- Calibrated external temperature logger
- pH sensor or phenol-red containing culture medium
- Time-lapse imaging system with environmental control (e.g., ImageXpress systems [49])
Method:
- System Calibration: Place the external temperature logger and pH sensor inside the empty mini-incubator chamber. Close the chamber and allow the system to activate.
- Stability Monitoring: Record temperature and pH (or take brightfield images of the phenol-red medium to assess color) every 15 minutes for 24 hours while the imaging system operates a mock time-lapse script.
- Data Analysis:
- Temperature Stability: Calculate the mean temperature and standard deviation. The system is validated if the mean is 37.0°C ± 0.2°C and no single measurement deviates by more than 0.5°C [48].
- pH Stability: Assess that the pH of the medium remains between 7.2 and 7.4 for the duration, with no observable color shift in phenol-red.
- Humidity Check: Weigh a small, open dish of pure water placed inside the chamber at the start and end of the 24-hour period. Evaporation should be less than 2% of the initial mass, confirming adequate humidity control.
Managing Multi-Dimensional Datasets
A single mouse embryo time-lapse experiment can generate terabytes of data, encompassing X, Y, Z (optical sections), time (T), and multiple fluorescence channels (C).
The Zarr Format for Cloud-Native Data
The Zarr storage protocol is an open-source, cloud-native format designed for efficient storage and access of large N-dimensional arrays [51].
- Chunked Storage: Data is broken into small, manageable "chunks" (e.g., 128x128x1x1x1 for X,Y,Z,T,C). Each chunk is compressed and stored as a separate object, enabling efficient parallel I/O and selective reading of data subsets without loading the entire dataset [51].
- Hierarchical Metadata: The data structure is self-describing, with metadata stored in simple JSON files (e.g.,
zarr.json), making datasets easily navigable and portable [51]. - Cloud-Optimized: This chunked structure is ideal for cloud object stores (e.g., Amazon S3), dramatically reducing network calls and enabling scalable, distributed analysis with tools like Dask and Xarray [51].
Protocol: Storing and Accessing Time-Lapse Data in Zarr
This protocol outlines converting a multi-dimensional image stack into a Zarr dataset for efficient analysis.
Materials:
- Raw image stack from time-lapse experiment (e.g., OME-TIFF format)
- Python environment with libraries:
zarr,xarray,dask, andbioformats-python
Method:
- Data Ingestion and Reorganization:
- Read the raw image file and its metadata. Extract key parameters: image dimensions (X, Y), number of Z-slices, timepoints, and channels.
- Reorganize the data into a logical 5-dimensional array structure (X, Y, Z, Time, Channel).
- Zarr Dataset Creation:
- Define a Zarr store, typically a directory on a local filesystem or a cloud storage bucket.
- Create a Zarr group to organize the data. Within this group, initialize a Zarr array with the correct 5D shape and data type.
- Define an appropriate chunk size. A good starting point is
(512, 512, 1, 1, 1)to keep chunks small for a single XY plane per timepoint and channel. - Choose a compressor, such as Blosc, for efficient lossless compression.
- Data Writing and Annotation:
- Write the reorganized 5D array into the Zarr array. The writing process will automatically handle compression and chunking.
- Use Xarray to create a
DataArrayfrom the Zarr array, labeling the dimensions and adding coordinates (e.g., timepoints in minutes, channel names). Store this as a dataset within the Zarr group. - Add custom attributes to the Zarr group, such as experimental conditions, embryo genotype, and culture parameters.
Integrated Experimental Workflow
The diagram below illustrates the complete, integrated workflow from embryo culture to data analysis, highlighting steps critical for artifact mitigation and efficient data management.
Integrated workflow for time-lapse microscopy of mouse embryos, highlighting artifact mitigation and data management.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for Mouse Embryo Time-Lapse Culture
| Item | Function/Application | Example/Note |
|---|---|---|
| Polydimethylsiloxane (PDMS) | Fabrication of open microwell platforms for embryo culture in defined sub-microliter volumes [50]. | Biocompatible polymer allowing for micropipette access and high-resolution imaging [50]. |
| Potassium Simplex Optimized Medium (KSOM) | A common culture medium for supporting preimplantation development of mouse embryos in vitro [50]. | Pre-equilibrated medium is used within the microwells [50]. |
| Mineral Oil | Layered on top of culture medium to prevent evaporation and spontaneously isolate individual culture microwells [50]. | Products like OVOIL-100 are specifically designed for embryo culture [50]. |
| CO₂-Independent Medium | Optional for short-term imaging outside a CO₂-controlled environment, though not recommended for long-term assays. | Requires validation for specific embryo models as pH may drift. |
| Live-Cell Fluorescent Dyes | For tracking dynamic processes like cell viability (e.g., calcein AM) or apoptosis [50] [49]. | Staining protocols must be optimized to avoid embryo toxicity. |
| 3D Collagen Matrix | For 3D culture assays, providing a more in vivo-like microenvironment for certain cell types, such as primary leukemia cells [52]. | Validated as an alternative to methylcellulose for colony-forming assays [52]. |
Balancing Imaging Frequency, Resolution, and Embryo Health
The application of time-lapse microscopy (TLM) in mouse embryo culture research provides an unparalleled opportunity to observe developmental dynamics. However, the optimal configuration of imaging parameters is critical, as the imaging process itself—specifically light exposure—can introduce stress, potentially compromising embryo health and developmental competence. This application note details protocols and experimental strategies for balancing the conflicting demands of high imaging frequency, high spatial resolution, and the maintenance of optimal embryo health. Adherence to these guidelines is essential for generating robust, reproducible data in developmental biology, toxicology, and drug discovery research using mouse models.
Quantitative Analysis of Imaging Parameters
The following tables summarize key quantitative data and their impacts on embryonic development, derived from current research. These parameters serve as a critical foundation for experimental design.
Table 1: Impact of Imaging Modalities and Parameters on Mouse Embryo Development
| Imaging Parameter / Modality | Typical Value / Specification | Documented Impact on Embryo Development / Capability |
|---|---|---|
| Bright-Field (BF) Time-Lapse | Interval: 10-30 minutes [1] [50] | Successfully supports culture from 2-cell to hatched blastocyst (89% rate); enables tracking of cell divisions and basic morphology [50]. |
| Optical Coherence Microscopy (OCM) | Axial Resolution: ~2.1 μm; Lateral Resolution: ~1.0 μm [1] | Enables 3D visualization of internal microstructures (e.g., nuclei, cavitation); label-free detection of blastocyst lineages (ICM/TE) [1]. |
| Dual-Modality (BF + OCM) | Interval: ~10 minutes for >150 hours [1] | Correlates early cell cycle timings (2nd, 3rd) with blastocyst formation potential; provides co-registered structural and kinetic data [1]. |
| Microwell Culture Volume | 393 nL [50] | Confines individual embryos for precise tracking; success rate comparable to conventional methods (89%) [50]. |
| Cleavage Timing Analysis | — | Predicts blastocyst competence with 94% sensitivity and 100% specificity in microwell culture [50]. |
Table 2: Developmental and Clinical Outcomes of Different Culture and Assessment Methods
| Method / Outcome | Key Findings | Reference |
|---|---|---|
| Undisturbed Culture (TLI) | Maintains stable temperature, pH, and gas levels; minimizes metabolic stress from environmental fluctuations [53] [10]. | |
| Static Assessment | Disrupts culture conditions; provides incomplete developmental data; introduces observer subjectivity [53] [10]. | |
| Conventional Microdrop | Larger microdrop sizes correlate with decreased blastocyst formation rates [50]. | |
| Live Birth Rate from Selected Blastocysts | 68% (15/22) of blastocysts and 40% (4/10) of cleavage-stage embryos selected via time-lapse developed into normal offspring after transfer [50]. |
Experimental Protocols
Protocol: Time-Lapse Imaging of Preimplantation Mouse Embryos Using a Microwell System
This protocol is adapted from a study that achieved an 89% success rate in culturing two-cell mouse embryos to hatched blastocysts [50].
I. Materials and Reagents
- Animals: Six-week-old ICR mice [50].
- Hormones: Pregnant Mare's Serum Gonadotropin (PMSG), Human Chorionic Gonadotropin (hCG) [50].
- Media: M2 medium (for collection), potassium simplex optimized medium (KSOM) (for culture) [50].
- Microwell Device: Fabricated from Polydimethylsiloxane (PDMS), with 70 microwells, each of 393 nL volume [50].
- Oil Overlay: Mineral oil (e.g., OVOILTM-100) [50].
- Time-Lapse Microscope: Equipped with a stage-top incubator maintaining 37°C, 5% CO₂, and high humidity [50].
II. Methods
- Embryo Collection:
- Superovulate female mice via intraperitoneal injection of 5 IU PMSG, followed by 5 IU hCG 42-48 hours later.
- House females with males overnight and check for vaginal plugs the following morning to confirm mating.
- Sacrifice plugged females 42-44 hours post-hCG injection by cervical dislocation.
- Flush oviducts with M2 medium to collect two-cell stage embryos [50].
Microwell Seeding and Culture Setup:
- Suspend embryos in ~150 μL of pre-equilibrated KSOM.
- Apply the embryo suspension onto the microwell device and allow embryos to sediment into the microwells by gravity.
- Carefully aspirate excess medium from the top surface of the device using a pipette.
- Layer 2.5 mL of sterile mineral oil over the device to prevent evaporation and isolate individual microwells.
- Transfer the device to the stage-top incubator for continuous culture without medium exchange [50].
Time-Lapse Imaging and Analysis:
- Program the microscope to capture images at multiple pre-defined locations every 30 minutes for up to 90 hours.
- Record the timing of key developmental events (e.g., cleavage divisions, compaction, blastulation, hatching).
- Use cleavage timings to distinguish blastocyst-competent from incompetent embryos. Embryos following normal development timings showed statistically significant differences from those that failed (p-value < 10⁻¹⁰) [50].
Protocol: Dual-Modality Time-Lapse Imaging (Bright-Field and OCM)
This protocol outlines the setup for high-resolution, label-free imaging of embryonic structures [1].
I. System Configuration:
- Integrate a compact OCM system with a co-registered bright-field imaging path into a commercial incubator (e.g., Heracell VIOS 160i).
- Use a broadband light source and a 20x objective lens to achieve micron-scale resolution.
- Employ a motorized stage for automated, sequential imaging of multiple embryos in a specialized dish [1].
II. Image Acquisition:
- Set the system to acquire co-registered 3D OCM stacks and BF images at intervals of approximately 10 minutes.
- Automate the acquisition pipeline using prior knowledge of sample locations followed by image-guided auto-tracking and auto-focusing [1].
III. Data Utilization:
- 3D Microstructure Analysis: Use OCM data to visualize nuclei, cell boundaries, initiation of cavitation, and differentiation of the inner cell mass (ICM) and trophectoderm (TE).
- Morphokinetic Correlation: Correlate the timing of the second and third embryonic cell cycles with blastocyst formation and quality outcomes [1].
Visualization of Workflows and Relationships
The following diagrams illustrate the core experimental workflow and the critical relationship between imaging parameters and experimental outcomes.
Experimental Workflow for Embryo TLM
Imaging Parameter Interplay
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Time-Lapse Mouse Embryo Culture
| Item | Function in Research | Application Note |
|---|---|---|
| KSOM Medium | A chemically defined, sequential or single culture medium that supports preimplantation development from zygote to blastocyst. | Suitable for extended culture in closed time-lapse systems without medium exchange [50]. |
| Mineral Oil | Provides an overlay to prevent evaporation and pH fluctuations in the culture medium; isolates individual culture wells. | Essential for maintaining medium stability in small-volume cultures like microwells [50]. |
| PDMS Microwell Device | A micro-engineered platform for individual embryo culture in defined, sub-microliter volumes. | Enables high-fidelity embryo tracking and eliminates confounding factors from group culture [50]. |
| Specialized Imaging Dish | A culture dish designed with multiple wells for holding individual embryos, compatible with high-resolution microscopy objectives. | Allows for simultaneous culture and time-lapse imaging of multiple embryos (e.g., up to 25) [1]. |
| Motorized Microscope Stage | Provides precise, automated movement for imaging multiple predefined positions over time. | Critical for high-throughput data acquisition from multiple embryos without manual intervention [1]. |
| Stage-Top Incubator | Maintains a stable environment (37°C, 5-6% CO₂, 5% O₂) directly on the microscope stage. | Enables continuous imaging without removing embryos from optimal culture conditions [1] [50]. |
Beyond Snapshots: Validating 3D Morphologies and AI for Predictive Embryology
Correlating 3D Morphological Parameters with Blastocyst Formation and Quality
Application Notes
The integration of three-dimensional morphological analysis represents a significant advancement in the assessment of blastocyst quality and developmental potential. Moving beyond traditional two-dimensional grading systems, 3D parameters provide a more comprehensive, objective, and quantitative framework for embryo evaluation, particularly within time-lapse microscopy mouse embryo culture research. These parameters have demonstrated significant correlations with key developmental outcomes, enabling more reliable prediction of blastocyst formation and quality.
Table 1: Key 3D Morphological Parameters and Their Correlation with Blastocyst Quality
| Parameter Category | Specific Parameter | Correlation with High-Quality Blastocysts | Biological Significance |
|---|---|---|---|
| Overall Blastocyst Morphology | Blastocyst Volume, Surface Area, Diameter [54] | Positive correlation [54] | Indicates overall expansion and developmental progression. |
| Blastocyst Surface Area/Volume Ratio [54] | Negative correlation [54] | Reflects compaction and structural efficiency. | |
| Blastocyst Cavity Volume [54] | Positive correlation [54] | Signifies proper blastocoel formation. | |
| Trophectoderm (TE) Quality | TE Surface Area, Volume, Cell Number [54] | Positive correlation [54] | Critical for implantation and placental development. |
| TE Density (TE cells/Blastocyst Surface Area) [54] | Positive correlation [54] | Indicates epithelial cohesion and cell density. | |
| Inner Cell Mass (ICM) Quality | ICM Shape Factor [54] | Negative correlation (closer to a sphere) [54] | Reflects ICM compaction and organizational integrity. |
| ICM Minor-to-Major Axis Ratio [54] | Not significantly correlated [54] | - | |
| ICM Surface Area/Volume Ratio [54] | Negative correlation with live birth [54] | Suggests a more compact, rounded ICM is superior. | |
| Spatial Relationships | Spatial Distance between ICM and TE [54] | Positive correlation with pregnancy [54] | May reflect proper lineage specification and positioning. |
| Number of TE cells in ICM quadrant [54] | Positive correlation with pregnancy [54] | Suggests coordinated development between ICM and TE. |
The transition from 2D to 3D analysis addresses critical limitations of conventional morphological assessment, which is often subjective and fails to fully capture spatial relationships [54]. Research on mouse models, utilizing advanced imaging techniques like Optical Coherence Microscopy (OCM), has been instrumental in validating these 3D parameters. For instance, time-lapse OCM can non-invasively reveal microstructures such as nuclei during early cleavage stages and clearly differentiate the ICM and trophectoderm (TE) lineages in the blastocyst, providing a direct means to quantify the parameters listed in Table 1 [1]. Furthermore, the timing of early embryonic cell cycles, such as the second and third cycles, has been observed to correlate with successful blastocyst formation, highlighting the link between morphokinetics and 3D morphological outcomes [1].
Artificial intelligence (AI) and deep learning algorithms are now being leveraged to automate the extraction and analysis of these 3D parameters. This automation standardizes embryo assessment, reduces subjectivity, and integrates complex multidimensional data to improve predictive power for developmental potential [54] [15] [55].
Experimental Protocols
Protocol for 3D Reconstruction of Blastocysts from Time-Lapse Imaging
This protocol details a non-invasive method for reconstructing 3D blastocyst structures directly from multi-focal images acquired using a standard time-lapse (TL) microscopy system, adapted for mouse embryo research [54].
I. Materials and Reagents
- Embryo Culture Medium: Pre-equilibrated mouse embryo culture medium.
- Time-Lapse Culture Dish: A specialized dish with individual wells for embryo culture (e.g., commercial embryo imaging dish) [1].
- Time-Lapse Microscopy System: An incubator-equipped TL system (e.g., EmbryoScopeⓇ, GeriⓇ) capable of automated multi-focal plane imaging.
II. Procedure
- Embryo Loading: After fertilization and culture to the desired pre-blastocyst stage, individually transfer mouse embryos into the wells of the time-lapse culture dish containing pre-equilibrated culture medium.
- Time-Lapse Program Setup:
- Place the loaded dish into the TL system, maintaining constant conditions of 37°C, 5-6% CO2, and 5% O2 [1].
- Configure the TL program to acquire images at multiple focal planes (Z-stacks) for each embryo at set time intervals (e.g., every 10-15 minutes) [54] [1].
- The imaging should continue from the initiation of culture until the fully expanded or hatched blastocyst stage (typically for over 90 hours for mouse embryos) [1].
- Image Acquisition: Allow the TL system to run automatically, collecting a sequential series of multi-focal images for each embryo over the culture period.
- 3D Model Reconstruction:
- Data Export: Upon completion of the culture period, export the complete set of multi-focal, time-stamped images for analysis.
- AI-Based Reconstruction: Process the image stacks using a dedicated 3D reconstruction algorithm. The algorithm aligns the multi-focal images and interpolates the data to generate a continuous 3D model for each time point [54].
- Parameter Quantification: The software automatically calculates the quantitative 3D morphological parameters (e.g., volume, surface area, cell numbers for ICM/TE) from the reconstructed models [54].
III. Validation
- Fluorescence Staining Verification: To validate the accuracy of the TL-based 3D reconstruction, a subset of blastocysts can be fixed and stained with fluorescent dyes (e.g., DAPI for nuclei, Phalloidin for actin) [54].
- Confocal Microscopy: Image the stained blastocysts using confocal microscopy to create a high-resolution 3D reference model using software like Imaris [54].
- Error Calculation: Compare the parameters (e.g., volume, cell number) obtained from the TL reconstruction with the "ground truth" values from fluorescence reconstruction to determine the relative error, which has been shown to be low (e.g., ~2% for surface area, ~4% for volume) [54].
Protocol for Label-Free 3D Evaluation Using Optical Coherence Microscopy (OCM)
This protocol uses OCM for high-resolution, label-free 3D imaging of mouse embryos, providing superior structural detail [1].
I. Materials and Reagents
- Custom OCM/BF System: A compact dual-modality OCM and bright-field (BF) imaging system designed to fit inside a standard cell culture incubator [1].
- Embryo Culture Dish: As in Protocol 2.1.
II. Procedure
- System Setup: Ensure the custom OCM/BF system is installed and calibrated within the incubator, maintaining culture-appropriate conditions (5% O2, 6% CO2) [1].
- Embryo Loading and Positioning: Load embryos into the culture dish and place it on the motorized stage inside the incubator.
- Automated Time-Lapse Imaging:
- Data Analysis:
- 3D Visualization: Use volume rendering software to visualize the 3D OCM data, which allows clear observation of nuclei in early stages and ICM/TE structure in blastocysts [1].
- Morphological and Morphokinetic Analysis: Manually or automatically analyze the 3D image stacks to determine both static 3D parameters (as in Table 1) and dynamic timings of developmental events (e.g., time to 2-cell, 3-cell, blastocyst formation) [1].
Experimental Workflow for 3D Analysis
Protocol for Quantifying Single-Cell CDK Activity in Live Mouse Embryos
This protocol complements morphological analysis by quantifying biochemical activity at the single-cell level, linking cell cycle dynamics to developmental potential [16] [17].
I. Materials and Reagents
- FRET-based CDK Biosensor: A genetically encoded fluorescent biosensor that changes fluorescence upon CDK activity changes [16].
- Confocal Microscope: Equipped with environmental control (temperature, CO2).
- Microinjection System: For pronuclear injection of the biosensor DNA/RNA into fertilized mouse zygotes.
II. Procedure
- Embryo Preparation and Injection: Microinject mouse zygotes with mRNA or DNA encoding the CDK activity biosensor [16].
- In Vitro Culture: Culture the injected embryos under oil in a glass-bottom dish suitable for confocal microscopy, maintaining optimal conditions.
- Time-Lapse Confocal Imaging:
- Place the dish on the confocal microscope stage with an environmental chamber.
- Acquire time-lapse images (e.g., every 15 minutes) at multiple z-planes as embryos develop from the one-cell to the blastocyst stage [16].
- Image Analysis and CDK Quantification:
- Cell Tracking: Use cell tracking software (e.g., Cellpose) to segment and track individual cells through divisions [16].
- FRET Ratio Calculation: For each cell and time point, calculate the FRET ratio (or biosensor readout), which serves as a proxy for CDK activity levels [16].
- Data Visualization: Correlate CDK activity dynamics with morphological events and eventual blastocyst quality.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for 3D Mouse Embryo Research
| Item | Function/Application | Examples / Notes |
|---|---|---|
| Time-Lapse Incubation System | Enables continuous, non-invasive imaging of embryo development under stable culture conditions. | EmbryoScopeⓇ, GeriⓇ [54] [15] |
| Optical Coherence Microscopy (OCM) | Provides high-resolution, label-free 3D imaging of embryonic microstructures (nuclei, cavities, ICM/TE). | Custom-built systems for incubator installation [1] |
| AI-Based 3D Reconstruction Software | Automates the creation of 3D models from multi-focal images and quantifies morphological parameters. | Custom algorithms [54], nnU-Net for segmentation [55] |
| Fluorescent Biosensors | Reports specific biochemical activities (e.g., CDK activity) in live cells via changes in fluorescence. | FRET-based CDK activity biosensors [16] |
| Confocal Microscope with Environmental Control | Captures high-resolution 3D fluorescence images of live embryos over time. | Essential for biosensor and validation studies [16] |
| Specialized Embryo Culture Dishes | Facilitates individual embryo tracking and high-quality imaging within the incubator. | Dishes with well-of-the-well design [1] |
3D Parameters Predicting Quality
The cultivation and selection of embryos with high developmental potential are pivotal for success in both clinical in vitro fertilization (IVF) and fundamental biological research [53] [56]. For decades, the assessment of embryo quality has relied on conventional static morphology, wherein embryos are removed from incubators and briefly examined under a microscope at set time points [53] [57]. This method, while practical, provides only limited "snapshots" of a dynamic developmental process. The advent of time-lapse imaging (TLI) systems introduces a paradigm shift, enabling continuous, non-invasive monitoring of embryo development within stable culture conditions [58] [10]. This analysis provides a detailed comparison of these two methodologies, framing the discussion within the context of mouse embryo culture research—a cornerstone model for mammalian developmental biology. It summarizes quantitative data, outlines essential experimental protocols, and visualizes key workflows to equip researchers and drug development professionals with the tools for implementing these technologies.
Comparative Data Analysis of Cultivation Systems
The core difference between the systems lies in their approach to culture and monitoring. Conventional systems use standard incubators for culture, requiring physical removal for periodic morphological assessment [58] [53]. In contrast, TLI systems integrate incubation with continuous imaging, capturing development without environmental disturbance [58] [10].
Table 1: Key Characteristics of Conventional Static Assessment and Time-Lapse Imaging
| Feature | Conventional Static Assessment | Time-Lapse Imaging (TLI) |
|---|---|---|
| Culture Environment | Stable in incubator, but disrupted during removal for assessment [53] | Uninterrupted, stable culture conditions within a dedicated incubator [58] [10] |
| Data Collection | Static "snapshots" at predefined daily time points [57] | Continuous, high-frequency imaging (e.g., every 5-20 minutes) [10] |
| Primary Outputs | Morphology at time of observation: cell number, symmetry, fragmentation [53] [57] | Morphokinetic parameters: precise timings of cleavages (t2, t3, t4, etc.), synchronicity of divisions, and dynamic anomaly detection [56] [10] |
| Handling & Workflow | Requires manual handling for each assessment, increasing workload and risk [53] | Minimal physical handling; embryos can be assessed remotely from image data [10] |
| Subjectivity | High reliance on embryologist's experience and subjective interpretation [53] [56] | Reduced subjectivity through quantitative data; potential for automated, AI-driven analysis [54] [10] |
Quantitative studies highlight the impact of these differences. A large randomized controlled trial in a clinical setting found that TLI use led to a significantly higher implantation rate for the first embryo transfer compared to standard incubators [58]. Furthermore, research utilizing TLI has identified specific morphokinetic abnormalities, such as direct and reverse cleavage, which are associated with a significantly adverse impact on implantation and are often missed in static assessment [58]. The limitations of static scoring are further exposed by time-lapse studies showing that key morphological markers, such as pronuclear patterns and embryo fragmentation, can change dramatically within short time intervals, making a single observation potentially misleading [57] [59].
Experimental Protocols for Mouse Embryo Culture and Analysis
The following protocols are adapted for post-implantation mouse embryo culture, a critical period for studying morphogenetic events.
Protocol 1: Lattice Light-Sheet Time-Lapse Imaging of Post-Implantation Mouse Embryos
This protocol leverages advanced lattice light-sheet microscopy (LLSM) for high-resolution, long-term imaging with minimal photodamage [23].
Application Note: Ideal for capturing highly dynamic and photo-sensitive processes, such as cell migration during anterior-posterior axis establishment or actomyosin dynamics [23].
Materials:
- Dissection Medium: M2 medium [23].
- Embryo Culture Medium: For 5.5 days post-coitum (dpc) embryos, use a 1:1 mixture of CMRL and Knock Out serum, supplemented with L-Glutamine. For later stages (6.5+ dpc), 50% DMEM-FluoroBrite with 50% rat serum can be used [23].
- Mounting Implements: Fine glass capillaries pulled to a width appropriate for the embryo stage [23].
- Imaging Chamber: An 8-chambered slide prepared with vacuum grease barriers [23].
Methodology:
- Embryo Isolation: Euthanize a timed-pregnant mouse at the desired gestation stage (e.g., 5.5-6.5 dpc). Dissect to isolate the uterus and carefully recover the embryos under a dissection microscope using fine tools [23].
- Mounting: Transfer an individual embryo into the central well of the pre-assembled imaging chamber, which is filled with pre-equilibrated culture medium. Use the prepared glass capillaries to gently position and immobilize the embryo for optimal imaging [23].
- Setting Imaging Parameters: Place the chamber on the LLSM stage. Set imaging parameters to minimize photodamage while ensuring sufficient resolution. For the ZEISS LLSM L7, this involves defining the lattice light-sheet properties and acquisition intervals [23].
- Data Acquisition and Processing: Initiate time-lapse acquisition. The system will capture multi-focal plane images at set intervals over several hours. Post-processing pipelines are then used to manage the large dataset, which may include 3D reconstruction and quantitative analysis of cellular behaviors [23].
Protocol 2: Conventional Static Assessment of Mouse Embryo Development
This traditional protocol relies on periodic removal of embryos from a standard incubator for morphological evaluation.
Application Note: A foundational method suitable for basic quality assessment when TLI equipment is unavailable, though it provides limited dynamic information [53] [57].
Materials:
- Standard CO₂ Incubator: Maintained at 37°C, 5-6% CO₂, and high humidity.
- Culture Dishes: 35-mm or 4-well dishes with appropriate embryo culture medium (sequential or single-step) [53] [60].
- Inverted Microscope: Equipped with differential interference contrast (DIC) or similar optics.
Methodology:
- Embryo Collection and Culture: Following fertilization or retrieval, pool embryos and culture them in groups in a standard incubator [53].
- Scheduled Static Observation: At predetermined time points (e.g., 24, 48, 72, and 96 hours post-fertilization), quickly remove the culture dish from the incubator.
- Morphological Scoring: Place the dish on the pre-warmed microscope stage. Under 200x or 400x magnification, score each embryo based on static criteria within a strict time limit (typically 2-5 minutes) to minimize environmental stress [57]. Key criteria include:
- Data Recording and Return to Culture: Record the morphological scores for each embryo and promptly return the culture dish to the incubator.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Key Research Reagent Solutions for Time-Lapse Embryo Culture
| Reagent/Material | Function | Application Notes |
|---|---|---|
| Single-Step Culture Medium | Supports uninterrupted embryo development from zygote to blastocyst with a constant nutrient composition [53] [60]. | Simplifies TLI workflow; based on the concept that the embryo self-regulates its nutrient uptake [53]. |
| CMRL + Knock Out Serum Medium | A defined culture medium formulation for supporting post-implantation mouse embryo culture ex vivo [23]. | Critical for advanced studies on gastrulation and early organogenesis; requires precise preparation and equilibration [23]. |
| L-Glutamine Supplement | An essential amino acid crucial for cellular metabolism and energy production [23]. | Must be properly aliquoted and thawed to prevent crystal formation, which can cause imaging artifacts [23]. |
| Fluorescent Reporter Mouse Lines | Genetically modified mice expressing fluorescent proteins (e.g., in actin, membranes, or nuclei) for live-cell imaging [23]. | Enables visualization of specific cellular and subcellular structures; outbred strains like CD1 can be used for larger litter sizes [23]. |
| Vacuum Grease & Glass Capillaries | Used for assembling custom imaging chambers and mounting embryos for light-sheet microscopy [23]. | Allows for immobilization of embryos without compromising viability; pulling fine capillaries requires practice [23]. |
The comparative analysis unequivocally demonstrates that time-lapse imaging transcends the capabilities of conventional static assessment by providing a dynamic, quantitative, and undisturbed view of embryonic development. While static morphology remains a valuable basic tool, TLI unveils a critical dimension of biological information through morphokinetic parameters and the detection of transient anomalies. For the field of mouse embryo research and drug development, the adoption of TLI, particularly when combined with advanced techniques like lattice light-sheet microscopy and 3D reconstruction, promises deeper insights into the fundamental mechanisms of mammalian development and the potential to more accurately evaluate the effects of genetic or pharmacological perturbations.
AI-Enhanced Morphokinetic Profiling for Objective Embryo Selection
Application Notes
The Technological Paradigm Shift in Embryo Assessment
The integration of artificial intelligence (AI) with time-lapse imaging (TLI) represents a transformative advancement in the field of embryo selection, moving beyond the limitations of traditional static morphological assessment. Conventional static evaluation involves removing embryos from stable culture conditions for brief microscopic examinations at predetermined time points, a process that disrupts culture stability, provides incomplete developmental data, and introduces significant embryologist subjectivity [61]. In contrast, TLI systems enable continuous, non-invasive monitoring of embryonic development within controlled incubator environments, maintaining precise temperature, pH, and gas levels while generating a comprehensive dynamic developmental timeline [61]. This technological foundation allows for the precise tracking of morphokinetic parameters—dynamic biomarkers of embryo viability that traditional methods cannot capture [61].
The combination of TLI with AI algorithms addresses a critical need in assisted reproductive technology (ART), where success rates remain approximately 30%, with most transferred embryos failing to implant in the uterus—a phenomenon that increases significantly with maternal age [62]. This low efficiency has been partly attributed to the subjectivity inherent in visual embryo assessment by embryologists [62]. AI-enhanced morphokinetic profiling offers a solution by providing data-driven, objective interpretation of embryo development status, morphology, and viability potential [62].
Performance Advantages of AI-Based Selection
Evidence from systematic comparisons demonstrates that AI systems consistently outperform clinical teams in embryo selection accuracy. When analyzing embryo morphology and predicting clinical outcomes, AI models achieve significantly higher accuracy rates compared to trained embryologists across multiple studies [62].
Table 1: Comparative Performance of AI vs. Embryologists in Embryo Selection
| Assessment Type | Input Data Used | AI Median Accuracy | Embryologists Median Accuracy |
|---|---|---|---|
| Embryo Morphology Grade | Images & Time-lapse | 75.5% (Range: 59-94%) | 65.4% (Range: 47-75%) |
| Clinical Pregnancy Prediction | Clinical Information | 77.8% (Range: 68-90%) | 64% (Range: 58-76%) |
| Combined Prediction | Images + Clinical Data | 81.5% (Range: 67-98%) | 51% (Range: 43-59%) |
The performance advantage of AI is particularly pronounced when integrating both morphokinetic data from time-lapse imaging and clinical patient information, where AI systems nearly double the accuracy of human embryologists (81.5% vs. 51%) [62]. This enhanced performance stems from AI's ability to integrate and analyze complex, multi-dimensional datasets that exceed human processing capabilities, identifying subtle patterns and correlations that may not be apparent through visual inspection alone.
Practical Implementation and System Considerations
Implementing AI-enhanced morphokinetic profiling requires specialized equipment and careful protocol optimization. Two primary commercial systems dominate the field:
Table 2: Commercial TLI Systems for AI-Enhanced Morphokinetic Profiling
| Feature | EmbryoScope (Vitrolife) | Early Embryo Viability Assessment - Eeva (Merck KGaA) |
|---|---|---|
| Core Technology | Integrated microscope & camera with continuous imaging in stable incubator | Automated algorithms for early-stage morphokinetic analysis (first 48 hours) |
| Primary Strength | Comprehensive morphokinetic profiling (t2 to tB); improves workflow standardization | Simplifies and standardizes selection; beneficial for labs with less embryology expertise |
| Key Algorithm | EmbryoScope+ using wide range of morphokinetic parameters | Viability score based on early cleavage events (e.g., first cytokinesis) |
| Main Limitation | Questionable consistent improvement in live birth rates over conventional methods; High cost | Limited predictive scope for blastocyst-stage outcomes; Limited algorithm generalizability |
For research settings, simplified methods like the Glass Capillary Time-lapse (GCT) observation method have been developed that don't require dedicated live-cell imaging machines [24]. This cost-effective approach utilizes sealed glass capillaries, a thermoplate, and a stereomicroscope with a digital camera, achieving 81.5% blastocyst development rates from mouse zygotes—slightly lower than dedicated systems but sufficient for analytical purposes [24]. The GCT method captures images every 30 minutes for up to 5 days, enabling comprehensive morphokinetic profiling without substantial equipment investment [24].
Integration with Functional Biological Metrics
Advanced applications combine AI-enhanced morphokinetic profiling with functional biological metrics, such as quantifying cyclin-dependent kinase (CDK) activity levels at single-cell resolution throughout pre-implantation development [16]. This integration requires specialized protocols for tracking the translocation of fluorescent-based reporters in living mouse embryos, involving:
- In vitro culture strategies enabling efficient live fluorescent confocal image acquisition
- Subsequent cell tracking throughout different embryonic stages
- Computational workflows for processing and visualizing the complex datasets obtained [16]
This multi-parameter approach links dynamic morphological patterns with fundamental cell cycle regulators, potentially enhancing predictive accuracy for developmental potential.
Experimental Protocols
Protocol 1: Mouse Embryo Culture and Time-Lapse Imaging Using the Glass Capillary Method
Background and Principles
The Glass Capillary Time-lapse (GCT) observation method provides a simplified, cost-effective alternative to commercial time-lapse systems for mouse embryo research [24]. This protocol enables continuous monitoring of preimplantation development from zygote to blastocyst stage while maintaining embryo viability, with proven compatibility with subsequent full-term development and production of healthy offspring [24].
Materials and Reagents
- Glass capillaries (Type D with medium inner diameter recommended based on developmental rate and visibility optimization) [24]
- Ops culture method components: Optimized CO2 containing (OptC) medium in sealed container [24]
- Thermoplate for maintaining stable temperature during imaging
- Stereomicroscope with simple digital camera
- Switch Bot Plug or similar timer device to control light emission
- Mineral oil for immersion
- Blackout material for microscope stage darkening
Step-by-Step Procedure
Capillary Preparation:
- Prefill selected glass capillary with OptC medium using fine pipetting system
- Introduce sequential air bubbles into capillary to create separated chambers for individual embryo positioning [24]
Embryo Loading:
- Collect one-cell stage zygotes from in vitro fertilization (IVF)
- Using mouth pipette, carefully insert zygotes into capillary chambers between air bubbles
- Regulate embryo positions to prevent clustering and ensure individual tracking [24]
Capillary Sealing:
- Carefully seal both ends of capillary using micro-burner
- Avoid excessive heating near embryo-containing regions [24]
Imaging Setup:
Time-Lapse Acquisition:
- Configure stereomicroscope with digital camera for image capture every 30 minutes
- Program Switch Bot Plug to activate light for only 1 minute during each capture interval to minimize photodamage [24]
- Continue imaging for up to 96 hours (to blastocyst stage) [24]
- Maintain darkening around microscope stage between captures
Data Collection and Analysis:
- Capture images continuously for up to 5 days
- Monitor developmental milestones: cleavage timing, blastocoel formation, and blastocyst hatching
- Track individual embryos throughout development using position mapping
Quality Control and Validation
- Compare developmental rates to control groups cultured in traditional CO2 incubators (expected: ~81.5% blastocyst rate vs. 95.8% in controls) [24]
- Validate system through embryo transfer experiments confirming normal full-term development potential [24]
- Ensure stable temperature maintenance throughout culture period
Protocol 2: Lattice Light-Sheet Microscopy for High-Resolution Post-Implantation Imaging
Background and Principles
Lattice light-sheet microscopy (LLSM) offers unprecedented spatial and temporal resolution for visualizing morphogenetic and physiological processes in post-implantation mouse embryos while minimizing photodamage [23]. This technology utilizes thin light-sheets derived from two-dimensional optical lattices of interfering Bessel beams, providing resolution advantages over conventional light-sheet approaches with reduced photobleaching and increased viability compared to confocal microscopy [23].
Materials and Reagents
- Embryo Culture Medium: 50% CMRL, 50% Knock Out serum, supplemented with 200mM L-Glutamine [23]
- M2 dissection medium warmed to room temperature
- Glass capillaries for mounting (pulled to appropriate fineness for embryo stage)
- Diamond knife for capillary cutting
- 8-chambered slides
- Vacuum grease for creating barriers
- Timed-pregnant mice (5.5-6.5 dpc for post-implantation stages)
Step-by-Step Procedure
Embryo Isolation:
- Sacrifice timed-pregnant mice at desired developmental stage (e.g., 5.5 dpc)
- Dissect uterine horns and isolate embryos using fine forceps
- Transfer to pre-equilibrated culture medium
Mounting Implement Preparation:
- Heat glass capillary over Bunsen flame with well-defined blue cone
- Rotate capillary continuously until center becomes malleable
- Remove from flame and pull evenly from both ends to create fine tip
- Break pulled capillaries into fragments of consistent length using diamond knife [23]
Imaging Chamber Assembly:
- Fill four end wells of 8-chambered slide with 400μl pre-equilibrated culture medium each
- Create grease barriers at top and bottom of central wells using syringe with fine tip
- Ensure no grease smearing in center of well where embryos will be positioned [23]
Embryo Mounting and Culture:
- Position embryos in central wells using prepared glass implements
- Arrange for optimal light-sheet exposure
- Maintain culture conditions at 37°C with 5% CO2 throughout preparation
LLSM Imaging Parameter Setup:
- Configure lattice light-sheet parameters for dual-camera acquisition
- Set spatial and temporal resolution appropriate for biological question
- Optimize for fast acquisition of highly dynamic processes in large 3D volumes [23]
Data Acquisition and Processing:
- Acquire time-lapse sequences capturing morphogenetic events
- Process data through established pipelines for downstream analysis
- Concurrently image multiple embryos to account for biological variability [23]
Applications and Considerations
This protocol enables high-resolution visualization of dynamic cellular behaviors including:
- Cell migration patterns (e.g., anterior visceral endoderm migration)
- Actomyosin dynamics and membrane trafficking
- Signal propagation in specific embryonic regions
- Characterization of mutant phenotypes affecting morphogenesis [23]
Protocol 3: AI Model Training and Validation for Morphokinetic Profiling
Background and Principles
AI-enhanced embryo selection requires rigorous training and validation protocols to ensure robust performance across diverse embryo populations. The integration of time-lapse morphokinetic data with AI algorithms addresses the critical limitation of human subjectivity in traditional embryo assessment [62]. Successful implementation requires large, annotated datasets and careful validation against meaningful clinical outcomes.
Data Requirements and Preparation
Input Data Types:
- Time-lapse image sequences (every 5-20 minute intervals)
- Morphokinetic parameters (t2, t3, tB cleavage timings)
- Static morphological images at key developmental stages
- Clinical information (maternal age, ovarian response, fertility diagnosis) [62]
Dataset Characteristics:
- Minimum of thousands of embryo development sequences
- Balanced representation of implantation success and failure cases
- Multi-center collaboration to ensure algorithm generalizability [61]
AI Training Protocol
Data Preprocessing:
- Standardize image quality and resolution across datasets
- Time-align development sequences using fertilization as reference point
- Annotate developmental milestones according to standardized criteria
Feature Extraction:
- Automated extraction of morphokinetic parameters (cleavage timings, synchronization)
- Quantitative morphological features (cell size, symmetry, fragmentation patterns)
- Temporal patterns and developmental trajectories
Model Architecture Selection:
- Convolutional Neural Networks (CNNs) for image sequence analysis
- Recurrent Neural Networks (RNNs) for temporal pattern recognition
- Hybrid architectures combining image and clinical data processing [62]
Training and Validation:
- Implement k-fold cross-validation to prevent overfitting
- Validate against blinded test sets not used during training
- Compare performance against embryologist assessments using same ground truth [62]
Outcome Correlation:
- Train against clinically relevant endpoints: implantation, ongoing pregnancy, live birth
- Avoid over-reliance on intermediate outcomes like blastocyst formation [62]
Performance Validation
- Benchmarking: Compare AI performance against experienced embryologists using standardized datasets [62]
- Clinical Validation: Prospective evaluation in real-world clinical settings
- Generalizability Testing: Validate across multiple clinics with varying patient populations and protocols [61]
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents and Materials for AI-Enhanced Morphokinetic Profiling
| Item | Function/Application | Specifications/Considerations |
|---|---|---|
| EmbryoScope+ System (Vitrolife) | Comprehensive morphokinetic profiling with integrated incubation | Maintains optimal temperature, pH, gas levels; Proprietary algorithms analyze developmental milestones [61] |
| Eeva System (Merck KGaA) | Early-stage viability assessment | Focuses on first 48-hour development; Generates viability score based on early cleavage events [61] |
| Glass Capillaries (Type D) | Simplified time-lapse imaging without dedicated equipment | Medium inner diameter optimal for development rate and visibility; Compatible with Ops culture method [24] |
| Ops Culture Medium | Embryo culture without CO2 incubator | Optimized CO2 pressure in sealed container; Enables simplified imaging setups [24] |
| Lattice Light-Sheet Microscope | High-resolution imaging of post-implantation embryos | Minimal photodamage; Fast 3D volume acquisition; Suitable for dynamic morphogenetic events [23] |
| CDK Activity Reporters | Cell cycle monitoring in single cells | Fluorescent-based reporters track CDK activity dynamics; Reveals lineage-specific patterns [16] |
| Cellpose Algorithm | Automated cellular segmentation | Generalist algorithm for embryonic cell segmentation; Compatible with various imaging modalities [16] |
Technical Considerations and Implementation Challenges
Algorithm Generalizability and Validation
A significant challenge in AI-enhanced morphokinetic profiling is the limited generalizability of proprietary algorithms across different laboratory environments and patient populations [61]. Performance variability stems from differences in:
- Laboratory-specific protocols and culture conditions
- Patient population characteristics and fertility diagnoses
- Imaging parameter settings and quality standards [61]
Multicenter collaboration and external validation are crucial for developing robust algorithms that maintain performance across diverse clinical settings [61]. Furthermore, many existing AI models focus on locally generated databases and lack sufficient external validation, limiting their broad applicability [62].
Clinical Outcome Correlation
While AI systems demonstrate strong predictive accuracy for morphological assessment, there remains debate about their ability to consistently improve ultimate clinical outcomes. Large trials and meta-analyses reveal no universal improvement in live birth rates compared to conventional methods, highlighting the complex relationship between morphokinetic parameters and embryonic developmental potential [61]. To enhance clinical relevance, developers should shift focus from predicting implantation likelihood to forecasting more meaningful endpoints like ongoing pregnancy or live birth [62].
Cost-Benefit Analysis
The implementation of AI-enhanced TLI systems requires substantial financial investment for equipment acquisition and maintenance [61]. Researchers and clinicians must carefully consider the cost-benefit ratio in their specific context, particularly given the variable clinical utility across different patient populations [61]. While these technologies offer operational efficiencies and reduced embryologist subjectivity, their value in significantly improving IVF outcomes appears context-dependent rather than universal [61].
The use of mouse models has long been a cornerstone of biomedical research, providing critical insights into mammalian development and disease mechanisms. However, a significant translational gap often exists between findings in mouse models and human applications. Recent advances in live imaging technologies are now bridging this divide by enabling direct observation of dynamic biological processes across species. These technologies provide unprecedented access to morphogenetic events and cellular behaviors that were previously inaccessible, particularly during early post-implantation stages when critical developmental events occur [23]. By establishing parallel methodologies for mouse and human stem cell-derived models, researchers can now perform direct cross-species comparisons at cellular and subcellular resolutions, accelerating the translation of basic research findings into clinical applications and drug development pipelines.
Key Live-Imaging Technologies and Their Applications
Lattice Light-Sheet Microscopy (LLSM) for Embryonic Development
Lattice light-sheet microscopy represents a transformative approach for imaging post-implantation mouse embryos, offering unprecedented spatial and temporal resolution while minimizing photodamage to delicate living samples [23]. This technology utilizes a thin light-sheet derived from two-dimensional optical lattices of interfering Bessel beams, enabling rapid, high-resolution imaging of highly dynamic and photosensitive processes.
The key advantages of LLSM for embryonic imaging include:
- Reduced photobleaching and increased viability compared to confocal microscopy or epi-illumination approaches
- Capacity for concurrent imaging of multiple embryos, essential for accounting for biological variability between specimens
- Capacity to visualize subcellular events including actomyosin dynamics, membrane dynamics, chromosomal dynamics, and signal propagation
This protocol has been successfully employed to image migration of anterior visceral endoderm cells necessary for establishing the anterior-posterior axis, allowing characterization of how basal projections and overall migratory behavior are affected in genetic mutants [23].
Quantitative Phase Imaging with Digital Holographic Microscopy
Digital holographic microscopy (DHM) provides a label-free, non-invasive approach for quantitative phase imaging of living cells [63]. Unlike conventional microscopic techniques that require exogenous contrast agents, DHM combines interferometric technique with modern CCD sensors and image processing systems to provide quantitative measurements of cellular physical properties.
The Live-cell Analysis Framework (LAF) builds upon slightly off-axis holographic imaging to enable:
- Automated cell segmentation for extracting valid cellular regions
- Determination of physical and morphological properties including area, perimeter, irregularity, volume, and dry mass
- Long-term time-lapse investigations for quantitative monitoring of dynamic changes in cell morphology, motility, and proliferation
This framework is particularly valuable for drug development applications where non-destructive, long-term monitoring of cellular responses to compounds is essential [63].
Fluorescent Biosensors for Kinase Activity Monitoring
The combination of fluorescent-based reporters with time-lapse microscopy enables quantification of cyclin-dependent kinase (CDK) activity levels in single cells throughout different stages of pre-implantation embryo development [16]. This specialized approach requires expertise in both mouse embryology and computational analysis, but provides unparalleled insights into cell cycle dynamics during early mammalian development.
Table 1: Comparison of Live-Imaging Technologies for Mouse Embryo Research
| Technology | Spatial Resolution | Temporal Resolution | Key Applications | Advantages |
|---|---|---|---|---|
| Lattice Light-Sheet Microscopy | High (subcellular) | High (fast processes) | Morphogenetic events, cell migration, axial patterning | Minimal photodamage, multi-embryo imaging, high viability |
| Digital Holographic Microscopy | Cellular level | Medium to high | Cellular dry mass, morphology, motility | Label-free, non-invasive, quantitative phase imaging |
| Fluorescent Confocal Microscopy | High (subcellular) | Medium | CDK activity dynamics, cell cycle progression | Molecular specificity, well-established protocols |
Experimental Protocols for Cross-Species Live Imaging
Mouse Embryo Isolation and Culture for Lattice Light-Sheet Imaging
Objective: To isolate, mount, and culture post-implantation mouse embryos for time-lapse imaging using lattice light-sheet microscopy.
Materials and Reagents:
- Embryo Culture Medium (4 mL per experiment: 2 mL CMRL + 2 mL Knock Out serum + 42 μl of 200 mM L-Glutamine)
- M2 dissection medium (∼8 mL per litter)
- 8-chambered slides
- Glass capillaries for mounting
- Vacuum grease for barriers
Protocol Steps:
Timed mating setup: Pair stud males and females from desired strains, checking for vaginal plugs each morning. Noon of the day a plug is observed is designated 0.5 days post coitum (dpc) [23].
Medium preparation: Prepare Embryo Culture Medium fresh for each experiment in a laminar flow hood to minimize infection risk. Equilibrate in a humidified incubator at 37°C and 5% CO₂ for at least 1 hour before use [23].
Embryo isolation: At desired developmental stage (5.5-6.5 dpc), dissect embryos in room-temperature M2 medium using fine tools to maintain embryo integrity.
Mounting implement preparation: Create pulled glass capillaries using a Bunsen flame, breaking them into fragments roughly the same length as the imaging chamber width [23].
Chamber assembly:
- Fill the four end wells of an 8-chambered slide with 400 μl of pre-equilibrated culture medium each
- Create two grease barriers at the top and bottom of the central wells using vacuum grease
- Carefully position embryos in the central wells using pulled glass capillaries
Microscope setup: Configure LLSM imaging parameters appropriate for the experimental objectives, balancing resolution and speed with viability concerns.
Critical Considerations:
- For later stage embryos (6.5+ dpc), alternative Embryo Culture Medium compositions may be required (e.g., 50% DMEM-FluoroBrite with 50% rat serum)
- Practice capillary pulling to achieve appropriate widths for different embryonic stages
- Always handle chambers on a clean Petri dish lid to avoid scratching or dirtying the imaging surface
Cross-Species Validation Using Neuroimaging Intermediate Phenotypes
Objective: To identify neuroimaging patterns that serve as intermediate phenotypes linking genetic and stress factors to depression-related behaviors across rodent models and humans.
Experimental Framework:
Animal models: Utilize both genetic (P11 knockout mice) and environmental stress (chronic unpredictable mild stress in rats) models of depression [64].
Behavioral assessments: Perform sucrose preference test (SPT), forced swim test (FST), and open field test (OFT) to quantify depression-like behaviors [64].
MRI acquisition: Acquire whole-brain fMRI data using a gradient-echo echo-planar imaging sequence to measure amplitude of low-frequency fluctuations (ALFF) [64].
Human participant cohorts: Recruit participants with depression and healthy controls across multiple independent sites for cross-validation [64].
Machine learning approaches: Apply t-distributed Stochastic Neighbor Embedding (t-SNE) and agglomerative hierarchical clustering to identify neuroimaging subtypes of depression [64].
Cross-species validation: Linear regression to determine which neuroimaging features predict core depression symptoms across species [64].
Key Findings: This approach has demonstrated that distinct subcortical-sensorimotor neuroimaging patterns predict anhedonia in both rodent models and human depression subtypes, validating their utility as robust intermediate phenotypes [64].
Visualization of Experimental Workflows
Lattice Light-Sheet Microscopy Workflow
Cross-Species Neuroimaging Validation Pipeline
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 2: Essential Research Reagents for Cross-Species Live Imaging Studies
| Reagent/Material | Function | Application Notes |
|---|---|---|
| CMRL Medium | Base component of embryo culture medium | Provides essential nutrients and salts for embryo viability during imaging |
| Knock Out Serum | Serum component for culture medium | Supports embryo development; requires careful preparation to minimize infection risk |
| L-Glutamine | Nutrient supplement | Must be fully dissolved to prevent imaging artifacts from light refraction |
| M2 Dissection Medium | Buffer for embryo isolation | Maintains physiological conditions during dissection procedures |
| Glass Capillaries | Embryo mounting implements | Must be pulled to appropriate width for specific embryonic stages |
| Vacuum Grease | Creation of imaging barriers | Prevents medium leakage without contaminating imaging field |
| Fluorescent Reporters | Monitoring molecular activity | Enable tracking of CDK activity and other signaling events in live embryos |
Data Analysis and Interpretation Frameworks
Quantitative Morphological Analysis
The Live-cell Analysis Framework (LAF) provides a systematic approach for extracting quantitative data from live-cell images [63]. This framework includes:
- Automated cell segmentation to extract valid cellular regions from phase images
- Calculation of physical parameters including area, perimeter, irregularity, and volume
- Determination of cellular dry mass through surface integral of optical phase shift
These quantitative measurements enable researchers to move beyond qualitative observations to precise, reproducible characterization of cellular responses to genetic manipulations or pharmacological treatments.
Cross-Species Neuroimaging Analysis
The integration of rodent and human neuroimaging data requires specialized analytical approaches [64]:
- Amplitude of low-frequency fluctuations (ALFF) analysis to identify characteristic neural activity patterns
- Unsupervised machine learning to identify neuroimaging subtypes without a priori hypotheses
- Polygenic risk score analysis to connect genetic susceptibility with neuroimaging findings
- Metabolite data integration to link imaging findings with biochemical pathways
This comprehensive analytical framework has revealed distinct subtypes of depression with different etiological underpinnings, demonstrating the power of cross-species validation approaches [64].
Applications in Drug Development and Precision Medicine
The methodologies described herein have significant implications for drug development and precision medicine:
- Improved predictive validity of animal models through cross-species validation
- Identification of biomarkers for patient stratification in clinical trials
- Non-invasive monitoring of treatment responses in both animal models and human patients
- Accelerated translation of basic research findings into clinical applications
By establishing robust cross-species methodologies, researchers can bridge the gap between mouse models and human applications, ultimately leading to more effective and targeted therapeutic interventions.
Within time-lapse microscopy research utilizing mouse embryo cultures, three-dimensional (3D) reconstruction has emerged as a powerful tool for capturing comprehensive morphological data. These reconstructions allow for the quantitative analysis of dynamic developmental processes. However, the reliability of all subsequent biological interpretations hinges on the accuracy of the digital models. This protocol details a method for establishing the validity of 3D reconstructions generated from time-lapse (TL) imaging by using fluorescence staining as a reference standard. This process is critical for ensuring that quantitative measurements of embryo morphology, such as volume and surface area, faithfully represent the biological specimen.
Materials and Methods
Research Reagent Solutions
The following table catalogues the essential materials required for the execution of this validation protocol.
Table 1: Essential Research Reagents and Materials
| Item | Function in the Protocol | Specific Example / Note |
|---|---|---|
| Mouse Embryos | Biological specimens for 3D reconstruction and validation. | Use strains with fluorescent reporters or wild-type for dye labeling [23]. |
| Time-Lapse Incubator | Maintains physiological conditions for embryo development during multi-focal image capture. | Provides environmental control (e.g., 5% O₂, 6% CO₂) [1]. |
| Embryo Culture Medium | Supports embryo development during extended time-lapse imaging. | e.g., CMRL supplemented with Knock Out serum and L-Glutamine [23]. |
| Fixative Solution | Preserves embryo structure post time-lapse imaging for subsequent staining. | Typically paraformaldehyde. |
| Fluorescent Dyes | Labels specific cellular structures for high-resolution reference imaging. | Dyes for nucleus, trophectoderm, cell membrane, and inner cell mass (ICM) [54]. |
| Mounting Medium | Embeds stained embryos for imaging, often with anti-fading agents. | |
| Lattice Light-Sheet Microscope | Generates high-resolution, low-photodamage 3D reference images via fluorescence. | Uses thin light-sheet from Bessel beams for optimal resolution [23]. |
Experimental Workflow for Validation
The validation of 3D reconstructions involves a parallel processing pipeline where the same set of embryos undergoes both non-invasive time-lapse imaging and high-resolution fluorescence imaging, followed by a quantitative comparison of the resulting models. The workflow is designed to minimize handling and maximize comparability between the two modalities.
Detailed Protocol Steps
Embryo Preparation and Time-Lapse Imaging
Culture mouse embryos following standard protocols in a time-lapse incubator system. Acquire multi-focal plane image stacks using the TL system at the desired developmental stage. These images are the input for the 3D reconstruction algorithm, which generates the test model whose accuracy is to be validated [54] [1]. Ensure imaging parameters are documented.
Fluorescence Staining and Reference Image Acquisition
Following the completion of TL imaging, fix the embryos. Perform fluorescent staining to label key cellular structures such as the nucleus, trophectoderm (TE), cell membrane, and inner cell mass (ICM) [54]. Acquire high-resolution 3D image stacks of the stained embryos using a reference imaging modality, such as lattice light-sheet microscopy (preferred for its low photodamage and high resolution) or confocal microscopy [23]. Reconstruct the 3D model from these fluorescence images, which will serve as the ground truth for validation.
Image Processing and Quantitative Comparison
Co-register the 3D model generated from TL images with the model generated from fluorescence staining. This aligns the two models in the same spatial coordinate system. Subsequently, calculate key morphological parameters from both models and compute the relative error for each parameter [54]. The formula for relative error is: Relative Error = |(Value_TL - Value_Fluorescence)| / Value_Fluorescence * 100%
Results and Data Analysis
The core of the validation lies in the quantitative comparison of morphological parameters derived from both imaging modalities. The following table summarizes example validation data, demonstrating the level of accuracy that can be achieved.
Table 2: Quantitative Comparison of 3D Morphological Parameters from Time-Lapse and Fluorescence Reconstructions
| 3D Morphological Parameter | Relative Error (Mean ± SD) | Commentary on Validation Outcome |
|---|---|---|
| Blastocyst Surface Area | 2.13% ± 1.63% | High accuracy, validating use for surface morphology studies. |
| Blastocyst Volume | 4.03% ± 2.24% | Good accuracy, suitable for volumetric growth analysis. |
| Blastocyst Diameter | 1.98% ± 1.32% | Excellent agreement with the reference standard. |
| ICM Surface Area | 4.83% ± 6.26% | Moderate accuracy; caution advised for fine ICM surface details. |
| ICM Volume | 6.64% ± 12.83% | Higher variability; TL is a good estimator but less precise for small volumes. |
| TE Cell Number | 10.00% ± 8.73% | Most challenging parameter; may require manual verification. |
Data adapted from a large-scale validation study comparing TL and fluorescence reconstructions [54].
Discussion
The validation data confirm that 3D reconstructions from time-lapse microscopy can achieve high accuracy for overall embryo morphology, such as surface area, volume, and diameter. This establishes confidence in using these parameters for quantitative analyses in mouse embryo research. However, parameters pertaining to smaller or more complex structures, like the ICM volume and TE cell count, show higher relative errors. This underscores the importance of context when interpreting data; while TL-based reconstructions are excellent for gross morphological assessment, high-resolution fluorescence may remain necessary for specific, fine-detail analyses.
This protocol provides a robust framework for researchers to establish the accuracy of their 3D reconstruction pipelines, thereby strengthening the foundation for subsequent biological conclusions drawn from quantitative morphological data in time-lapse mouse embryo studies.
Conclusion
Time-lapse microscopy has fundamentally transformed the study of mouse embryogenesis, providing an unparalleled, dynamic view of developmental processes. The integration of advanced techniques like lattice light-sheet and optical coherence microscopy allows for the continuous, high-resolution capture of critical morphokinetic events and 3D structures with minimal intervention. When combined with emerging AI-driven analysis, these technologies offer a powerful, objective framework for predicting embryo viability and understanding the mechanisms underlying developmental defects. Future research will focus on refining multi-modal imaging, standardizing AI algorithms across laboratories, and further bridging insights from mouse models to improve clinical outcomes in human assisted reproduction and developmental disease modeling. The ongoing synthesis of high-dimensional imaging data with molecular biology promises to unlock new frontiers in regenerative medicine and personalized drug development.
References
- 1. Label-free evaluation of mouse embryo quality using time- ... [https://www.nature.com/articles/s42003-025-08044-5]
- 2. Machine learning in time-lapse imaging to differentiate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11180621/]
- 3. Time-lapse analysis of mouse embryo development in ... [https://www.sciencedirect.com/science/article/abs/pii/S1472648310002889]
- 4. GIANI – open-source software for automated analysis of 3D ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9189431/]
- 5. IN Carta Image Analysis Software [https://www.moleculardevices.com/products/cellular-imaging-systems/high-content-analysis/in-carta-image-analysis-software]
- 6. Genetic Tools for Cell Lineage Tracing and Profiling ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11034889/]
- 7. Lineage tracing: technology tool for exploring the ... [https://stemcellres.biomedcentral.com/articles/10.1186/s13287-020-01941-y]
- 8. Cell lineage-resolved embryonic morphological map ... [https://www.nature.com/articles/s41467-025-58878-0]
- 9. Embryo selection: the role of time-lapse monitoring - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4290130/]
- 10. Time-Lapse Imaging in IVF: Bridging the Gap Between ... [https://www.mdpi.com/1422-0067/26/19/9609]
- 11. Morphokinetic analysis of in vitro fertilization outcomes [https://www.sciencedirect.com/science/article/pii/S0015028223006180]
- 12. Comparison of pregnancy outcomes using a time-lapse ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8176153/]
- 13. Embryonic stem cell application in drug discovery - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4009932/]
- 14. AI Accelerates Drug Discovery: New Model Automates ... [https://cispa.de/en/sivaprasad-anomaly-detection]
- 15. MAIA platform for routine clinical testing: an artificial ... [https://www.nature.com/articles/s41598-025-17755-y]
- 16. Protocol for imaging and quantifying single-cell CDK ... [https://pubmed.ncbi.nlm.nih.gov/40924577/]
- 17. Protocol for imaging and quantifying single-cell CDK ... [https://www.sciencedirect.com/science/article/pii/S266616672500471X]
- 18. translating mouse knockout models of human male infertility [https://pmc.ncbi.nlm.nih.gov/articles/PMC12558365/]
- 19. Mouse model may help explain, treat infertility [https://news.cornell.edu/stories/2024/01/mouse-model-may-help-explain-treat-infertility]
- 20. Reproductive Genomics Program: Mutant Models for Infertility [https://www.jax.org/research-and-faculty/resources/reproductive-genomics-resource]
- 21. A long term time lapse microscopy technique for ... [https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2025.1601397/full]
- 22. Protocol for lattice light-sheet time-lapse imaging of early ... [https://pubmed.ncbi.nlm.nih.gov/41138190/]
- 23. Protocol for lattice light-sheet time-lapse imaging of early post ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12593599/]
- 24. Time-lapse observation of mouse preimplantation embryos ... [https://www.nature.com/articles/s41598-023-47017-8]
- 25. Imaging mitotic processes in three dimensions with lattice ... [https://link.springer.com/article/10.1007/s10577-021-09656-3]
- 26. A quantitative analysis of various patterns applied in lattice ... [https://www.nature.com/articles/s41467-022-32341-w]
- 27. The Applications of Lattice Light-Sheet Microscopy for ... [https://www.mdpi.com/2072-666X/10/9/599]
- 28. 3i Lattice Light-sheet Microscope [https://microscopy.jhmi.edu/equipment/3i-lattice-light-sheet-microscope/]
- 29. News | Fiolka Lab | UT Southwestern, Dallas, Texas [https://labs.utsouthwestern.edu/fiolka-lab/news]
- 30. Serial optical coherence microscopy for label-free ... [https://www.nature.com/articles/s41598-020-63460-3]
- 31. Computer-Aided Diagnosis of Label-Free 3-D Optical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6724217/]
- 32. Live imaging of late-stage preimplantation human embryos ... [https://www.nature.com/articles/s41587-025-02851-1]
- 33. DAPI Counterstaining Protocols [https://www.thermofisher.com/us/en/home/references/protocols/cell-and-tissue-analysis/dapi-protocol/basic-dapi-counterstaining-protocols.html]
- 34. Transfection types, methods and strategies: a technical review [https://pmc.ncbi.nlm.nih.gov/articles/PMC8067914/]
- 35. AAV vector development, back to the future [https://www.sciencedirect.com/science/article/pii/S1525001625002734]
- 36. Efficient genome editing of two-cell mouse embryos via ... [https://www.nature.com/articles/s41598-024-81198-0]
- 37. The effects of electroporation buffer composition on cell ... [https://www.nature.com/articles/s41598-020-59790-x]
- 38. Label-free evaluation of mouse embryo quality using time ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12000469/]
- 39. Time-lapse imaging systems for embryo incubation and ... [https://pubmed.ncbi.nlm.nih.gov/40730404/]
- 40. Long-term imaging and spatio-temporal control of living ... [https://link.springer.com/article/10.1007/s12213-024-00165-0]
- 41. Time-lapse confocal imaging helps to reveal a secret ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12277207/]
- 42. Protocol for the quantitative characterization of cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12336826/]
- 43. Trans-scale live-imaging of an E5.5 mouse embryo using ... [https://www.life-science-alliance.org/content/8/3/e202402839]
- 44. Mitigating phototoxicity during multiphoton microscopy of live... [https://pubmed.ncbi.nlm.nih.gov/25111506/]
- 45. [PDF] Mitigating Phototoxicity during Multiphoton... | Semantic Scholar [https://www.semanticscholar.org/paper/Mitigating-Phototoxicity-during-Multiphoton-of-Live-D%C3%A9barre-Olivier/96624479115b66b81c103958874631f1d2fb0672]
- 46. Two-Photon Microscopy Applications [https://www.news-medical.net/life-sciences/Two-Photon-Microscopy-Applications.aspx]
- 47. 4D light sheet imaging, computational reconstruction, and ... [https://www.sciencedirect.com/science/article/pii/S2666166724006804]
- 48. Facilitating long-term cell examinations and time-lapse ... [https://www.nature.com/articles/s41598-024-52866-y]
- 49. Live Cell Imaging, Live Cell Applications, Time-Lapse ... [https://www.moleculardevices.com/applications/live-cell-imaging]
- 50. Microwells support high-resolution time - lapse imaging and... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4417014/]
- 51. Fundamentals: What Is Zarr? A Cloud-Native Format for ... [https://earthmover.io/blog/what-is-zarr/]
- 52. Leveraging automated time-lapse microscopy coupled with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11879803/]
- 53. Time-lapse technology for embryo culture and selection [https://pmc.ncbi.nlm.nih.gov/articles/PMC7720962/]
- 54. Timelapse-based 3D reconstruction of blastocysts reveals ... [https://www.nature.com/articles/s41746-025-02028-9]
- 55. Automatic Human Embryo Volume Measurement in First ... [https://www.jmir.org/2025/1/e60887]
- 56. Research progress of time-lapse imaging technology and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10519478/]
- 57. Which morphological scoring system is relevant in human ... [https://www.sciencedirect.com/science/article/abs/pii/S0143400411002669]
- 58. Comparison of embryo implantation potential between time ... [https://www.sciencedirect.com/science/article/abs/pii/S1472648322004291]
- 59. Which morphological scoring system is relevant in human ... [https://pubmed.ncbi.nlm.nih.gov/21782239/]
- 60. Considerations Regarding Embryo Culture Conditions [https://pmc.ncbi.nlm.nih.gov/articles/PMC6000787/]
- 61. Time-Lapse Imaging in IVF: Bridging the Gap Between ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12524532/]
- 62. Embryo selection through artificial intelligence versus ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10426717/]
- 63. Live-cell analysis framework for quantitative phase imaging ... [https://www.frontiersin.org/journals/photonics/articles/10.3389/fphot.2022.1083139/full]
- 64. Bridging animal models and humans: neuroimaging as ... [https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-025-03850-4]