Automated RNAscope on Ventana DISCOVERY: A Complete Guide for Robust, High-Throughput RNA ISH
This article provides a comprehensive resource for researchers, scientists, and drug development professionals implementing automated RNAscope in situ hybridization on the Roche Ventana DISCOVERY ULTRA platform.
Automated RNAscope on Ventana DISCOVERY: A Complete Guide for Robust, High-Throughput RNA ISH
Abstract
This article provides a comprehensive resource for researchers, scientists, and drug development professionals implementing automated RNAscope in situ hybridization on the Roche Ventana DISCOVERY ULTRA platform. It covers the foundational principles of the technology, detailed methodological workflows for various assay types (HRP, AP, Duplex, and BaseScope), essential troubleshooting and optimization strategies for challenging samples, and rigorous validation protocols. The guide also explores the role of automated RNAscope as a powerful complementary and primary tool for biomarker research and diagnostic development, highlighting its advantages in specificity, sensitivity, and single-cell resolution within morphological context.
Understanding Automated RNAscope: Principles and Assay Selection on the DISCOVERY ULTRA
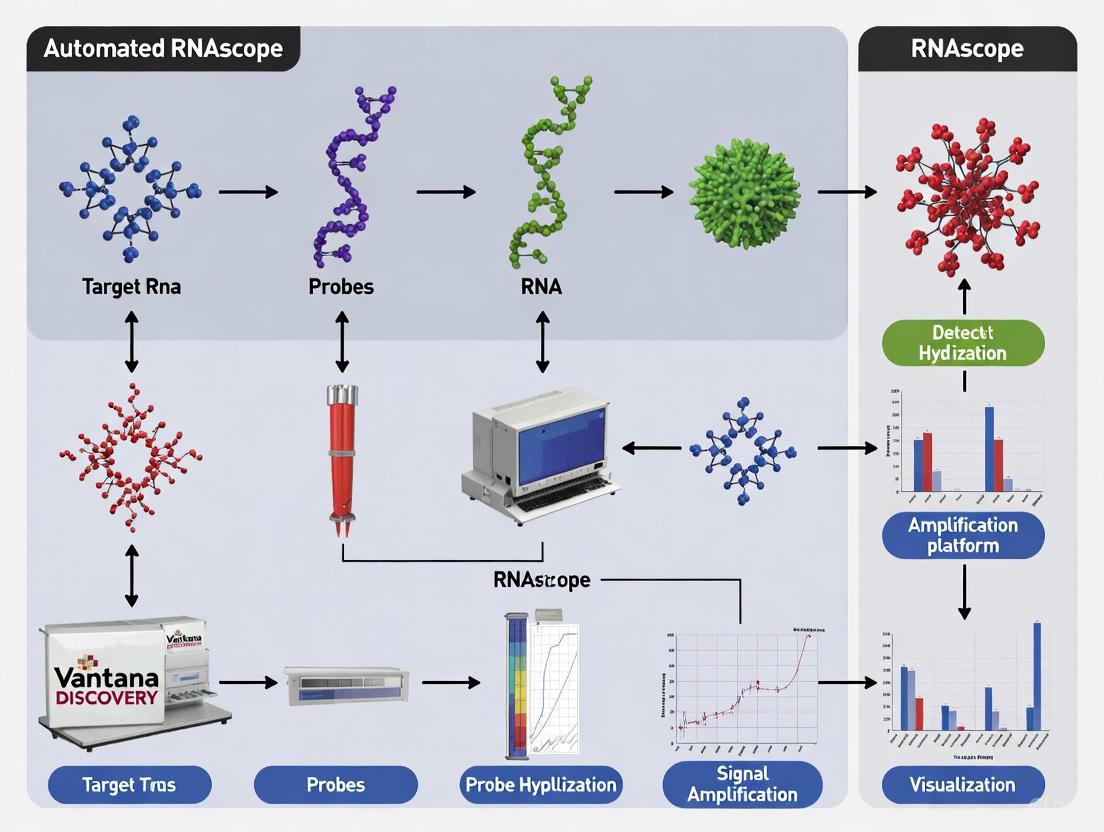
The accurate detection of RNA within its native morphological context is crucial for advancing biomarker research and molecular diagnostics. Traditional RNA in situ hybridization (ISH) techniques have been hampered by significant limitations, including insufficient sensitivity for low-abundance transcripts and high background noise from non-specific probe binding [1] [2]. The RNAscope technology, launched in 2012, represents a paradigm shift in RNA ISH by introducing a novel probe design and signal amplification system that simultaneously achieves single-molecule sensitivity and exceptional specificity [1]. This application note details the core technology behind RNAscope, with a specific focus on its application within automated platforms like the Roche Ventana DISCOVERY ULTRA system, providing researchers and drug development professionals with the protocols and data necessary to implement this powerful technique effectively.
Core Technology Principles: The ZZ Probe Design and Amplification Cascade
The foundational innovation of RNAscope lies in its proprietary double Z (ZZ) probe design and subsequent hybridization-based signal amplification. This system is engineered to amplify target-specific signals while effectively suppressing background noise.
The Double Z (ZZ) Probe Architecture
Each RNAscope probe pair is meticulously designed with the following components [1] [3]:
- Target Binding Sequence: The lower "arm" of each Z consists of an 18-25 base pair region that is complementary to the target RNA of interest.
- Spacer Sequence: A linker that connects the target-binding sequence to the tail sequence.
- Tail Sequence: The upper "arm" of each Z, featuring a 14-base tail. Two such tails from a contiguous probe pair form a unique 28-base binding site for the preamplifier molecule.
A set of approximately 20 such ZZ probe pairs is designed to hybridize along a ~1 kilobase region of the target RNA [1] [4]. This multi-probe approach provides robustness, as the detection of a single RNA molecule requires only three ZZ probe pairs to bind successfully, making the assay tolerant to partial RNA degradation or variable target accessibility [3] [4].
The Signal Amplification Cascade
Signal generation follows a sequential, hybridization-mediated cascade that results in profound signal amplification without propagating non-specific background [1] [3]:
- Preamplifier Binding: The 28-base site formed by a bound ZZ probe pair is recognized and bound by a preamplifier molecule.
- Amplifier Binding: Each preamplifier contains multiple binding sites (typically 20) for amplifier molecules.
- Label Probe Binding: Each amplifier, in turn, contains numerous binding sites (also typically 20) for enzyme-conjugated (e.g., HRP or AP) or fluorescently tagged label probes.
This cascade can theoretically yield up to 8,000 labels for each target RNA molecule, providing the high sensitivity required for single-molecule detection [1]. The requirement for two independent probes to bind in tandem for initiation makes nonspecific amplification statistically improbable, ensuring high specificity.
The following diagram illustrates the key steps of the RNAscope ZZ probe design and signal amplification process:
Figure 1: The RNAscope ZZ Probe Design and Signal Amplification Cascade. Two Z probes must bind contiguously to the target RNA to form a binding site for the preamplifier. Sequential hybridization of amplifiers and numerous label probes then generates a powerful, specific signal for visualization.
Performance Data and Validation
The performance of the RNAscope technology is demonstrated by its high sensitivity and specificity, which have been validated against established gold-standard methods across numerous studies.
Table 1: Performance Characteristics of RNAscope Technology
| Performance Metric | Description | Experimental Validation |
|---|---|---|
| Sensitivity | Single-molecule visualization; requires only 3 ZZ probe pairs for detection [3] [4]. | Enables detection of low-abundance transcripts (e.g., 3-15 copies/cell like POLR2A) [5]. |
| Specificity | Double-Z probe design prevents amplification of non-specific signals [1] [3]. | High concordance with qPCR and RT-qPCR (81.8–100%) [2]. |
| Single-Molecule Quantification | Each punctate dot represents a single RNA molecule [3] [6]. | Scoring system (0-4) based on dots per cell correlates directly with transcript abundance [6] [5]. |
| Compatibility with Archival Tissues | Short target hybridization regions (40-50 bases) work effectively with partially degraded RNA in FFPE samples [1] [4]. | Successful detection in FFPE tissues fixed according to ASCO/CAP guidelines (10% NBF for 6–72 hours) [1]. |
A systematic review of 27 studies concluded that RNAscope is a "highly sensitive and specific method" with a high concordance rate compared to PCR-based techniques and DNA ISH, confirming its reliability for clinical diagnostic research [2]. Furthermore, a 2021 study on Mantle Cell Lymphoma demonstrated that RNAscope provided reliable quantification of SOX11 mRNA levels that correlated well with IHC and RT-qPCR, while also revealing a significant correlation between TP53 mutations and low SOX11 expression [7].
Automated Protocol for Ventana DISCOVERY ULTRA Platform
Integrating RNAscope with the Roche Ventana DISCOVERY ULTRA platform standardizes the workflow, reduces hands-on time, and minimizes inter-user variability, making it ideal for high-throughput biomarker research [6] [5]. The following protocol is optimized for this automated system.
Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for Automated RNAscope on Ventana DISCOVERY ULTRA
| Reagent/Material | Function | Specific Recommendation / Kit |
|---|---|---|
| RNAscope VS Assays | Automated assay kits for Ventana systems. | RNAscope VS Universal HRP or AP Assay for singleplex detection [8]. |
| Target Probes | Hybridize to specific RNA target. | Predesigned >12,000 probes; custom probes for novel targets (>300 bp) [4]. |
| Control Probes | Assess assay performance and tissue RNA quality. | Positive: PPIB (moderate expression), UBC (high). Negative: bacterial dapB [6] [5]. |
| DISCOVERY Wash Buffer | Stringency washes to reduce background. | DISCOVERY 1X SSC Buffer only, diluted 1:10 [5]. |
| Chromogenic Substrates | Visualize RNA signals. | DAB (Brown), Fast Red (Red), or Green HRP [8] [9]. |
| Superfrost Plus Slides | Tissue adhesion. | Required to prevent tissue detachment during stringent assay steps [5]. |
Detailed Automated Workflow Protocol
The entire process is executed on the Ventana DISCOVERY ULTRA instrument according to a pre-loaded staining protocol. Key steps and parameters are detailed below.
Day 0: Slide Preparation (Manual)
- Sectioning: Cut formalin-fixed, paraffin-embedded (FFPE) tissue sections at 5 μm thickness [6].
- Mounting: Mount sections on Superfrost Plus slides [5].
- Baking: Bake slides for 1 hour at 60°C to ensure tissue adhesion.
Day 1: Automated Run on DISCOVERY ULTRA
- Deparaffinization and Dehydration: The instrument automatically performs this using onboard solvents [6].
- Target Retrieval (Pretreatment):
- Solution: Ventana Discovery RiboWash Buffer or similar.
- Condition: 24 minutes at 97°C for tissue sections [6]. This heat-induced epitope retrieval step unmasks the target RNA.
- Protease Digestion:
- Condition: 16 minutes at 37°C [6]. This step permeabilizes the tissue, allowing probe access to the RNA.
- Probe Hybridization:
- Condition: 2 hours at 43°C [6].
- Action: The target-specific ZZ probes are hybridized to the RNA.
- Signal Amplification: The instrument automatically executes a series of stringent washes and applies the preamplifier, amplifier, and HRP- or AP-conjugated label probes as per the RNAscope VS assay kit protocol [8] [6].
- Chromogenic Detection:
- Action: Apply chromogenic substrate (e.g., DAB for HRP, Fast Red for AP) to develop the signal. Each dot represents a single RNA molecule.
- Counterstaining and Coverslipping: The instrument performs counterstaining (e.g., hematoxylin) and mounting.
Critical Troubleshooting and Optimization Notes:
- Instrument Maintenance: Regular decontamination every three months is crucial to prevent microbial growth in fluidic lines [5].
- Pretreatment Optimization: For over- or under-fixed tissues, adjustment of protease treatment duration may be necessary. Always validate with control probes [5].
- Software Settings: Ensure the "Slide Cleaning" option is disabled in the Ventana protocol, as it can interfere with the assay [5].
Data Analysis and Scoring Guidelines
Analysis of RNAscope results focuses on quantifying the punctate dots, which correspond directly to individual RNA molecules.
Table 3: Standardized Scoring System for RNAscope Assay Results [6] [5]
| Score | Dots per Cell (Criteria) | Interpretation |
|---|---|---|
| 0 | No staining or <1 dot per 10 cells | Negative |
| 1 | 1-3 dots per cell | Low expression |
| 2 | 4-9 dots per cell, very few clusters | Moderate expression |
| 3 | 10-15 dots per cell, <10% in clusters | High expression |
| 4 | >15 dots per cell, >10% in clusters | Very high expression |
For quantitative analysis, automated image analysis software such as HALO or QuPath can be used to count dots on a cell-by-cell basis across whole slides or defined regions of interest, providing robust and reproducible quantitative data [6] [2].
The core ZZ probe design and multiplex amplification strategy of RNAscope technology provide an unparalleled combination of sensitivity, specificity, and morphological context for RNA analysis. Its seamless integration into automated platforms like the Roche Ventana DISCOVERY ULTRA standardizes the workflow, enhances reproducibility, and enables high-throughput spatial gene expression analysis. This makes it an indispensable tool for researchers and drug development professionals working to validate novel RNA biomarkers and further the development of molecular diagnostics and targeted therapies.
The integration of the RNAscope in situ hybridization (ISH) technology with the Roche Ventana DISCOVERY ULTRA platform represents a significant advancement in spatial biology, enabling fully automated, quantitative RNA analysis within intact cells and tissues. This automated partnership addresses critical challenges in biomedical research by providing single-cell resolution with spatial and morphological context, which is often lost in bulk tissue analysis techniques like qPCR [6]. The automation standardizes the complex RNA ISH process, minimizing inter-user variability and allowing for high-throughput sample processing—key requirements for both biomarker research and diagnostic assay development [6] [10].
This application note details the implementation, optimization, and analytical validation of RNAscope assays on the DISCOVERY ULTRA system. We provide detailed protocols and data to guide researchers and drug development professionals in deploying this powerful integrated platform for their spatial transcriptomics workflows.
RNAscope Assay Principle
The RNAscope ISH assay is based on a patented signal amplification and background suppression technology that represents a major advance over traditional RNA ISH methods [5]. The core technology employs a unique double Z (ZZ) probe design, which enables high-specificity detection by requiring two adjacent probe pairs to bind in tandem for signal amplification to proceed [6]. This design minimizes non-specific off-target signals, a common limitation of traditional ISH.
The assay visualizes target RNAs as discrete, punctate dots, where each dot corresponds to an individual RNA molecule, allowing for single-molecule sensitivity and direct quantification of transcript abundance at the single-cell level [6]. The automated version maintains this high signal-to-noise ratio with little to no background staining while providing the consistency and reproducibility required for research and potential diagnostic applications [6].
Ventana DISCOVERY ULTRA Platform
The DISCOVERY ULTRA is a fully automated IHC/ISH staining system designed for research use. Its key features that enable robust RNAscope integration include:
- Independent Slide Drawers: 30 individual reaction chambers allow different protocols (including both IHC and ISH), detection chemistries, and temperatures to run simultaneously without cross-interference [11].
- Open Reagent System: User-fillable dispensers and barcode-driven protocols provide flexibility for using RNAscope reagents while maintaining traceability and reducing staining errors [11].
- Software Flexibility: The Universal Procedure Software supports up to nine sequential detection steps and allows manual touchpoints at multiple stages, enabling complex multiplexing experiments [11].
- Precise Temperature Control: Independent temperature control for each slide drawer (including specific requirements for RNAscope protease steps at 40°C) ensures optimal hybridization conditions [5] [11].
Table 1: RNAscope Assay Options Available on the DISCOVERY ULTRA Platform
| Assay Type | Detection Options | Chromogens Used | Reaction Type | Ideal For |
|---|---|---|---|---|
| RNAscope VS Universal HRP | Chromogenic/Fluorescent | DAB, Purple, Teal, Green, FAM, FITC, Red610, Rhodamine, Cy5, DCC | Singleplex | High throughput routine applications [8] |
| RNAscope VS Universal AP | Chromogenic | Fast Red | Singleplex | High throughput routine applications [8] |
| RNAscope VS Duplex Assay | Chromogenic | DAB & Fast Red, Teal & Fast Red, Green & Fast Red | Duplex | Co-localization studies to map co-expression of two targets [8] |
| BaseScope VS Assay - RED | Chromogenic | Fast Red | Singleplex | Detection of splice variants, exon junctions, and short targets (50-300bp) [8] |
Figure 1: Generalized RNAscope workflow on the DISCOVERY ULTRA platform, showing key stages from sample pretreatment through detection and analysis. Specific timing and temperatures may vary based on assay and sample type.
Materials and Methods
Essential Research Reagent Solutions
Table 2: Key Research Reagent Solutions for RNAscope on DISCOVERY ULTRA
| Item | Function | Specific Recommendations |
|---|---|---|
| Control Probes | Assess sample RNA quality and assay performance | Positive: PPIB, POLR2A, or UBC [5] [12]; Negative: Bacterial dapB [5] [12] |
| Control Slides | Verify proper assay conditions | Human Hela Cell Pellet (Cat. No. 310045) or Mouse 3T3 Cell Pellet (Cat. No. 310023) [12] |
| Sample Preparation | Ensure RNA preservation and accessibility | Fixation in fresh 10% NBF for 16-32 hours; 5μm FFPE sections on Superfrost Plus slides [5] [12] |
| Detection Kits | Chromogenic or fluorescent signal generation | RNAscope VS Universal HRP, AP, or Duplex Assays specifically validated for DISCOVERY ULTRA [8] |
| Wash Buffers | Maintain proper stringency and pH | DISCOVERY 1X SSC Buffer only (diluted 1:10); RiboWash Buffer diluted 1:10 [5] |
| Mounting Media | Preserve staining and enable visualization | Xylene-based media (CytoSeal XYL) for Brown assay; EcoMount or PERTEX for Red and 2-plex assays [5] |
Automated Protocol for RNAscope on DISCOVERY ULTRA
Sample Preparation Guidelines
Proper sample preparation is critical for successful RNAscope staining:
- Fixation: Fix tissues in fresh 10% neutral-buffered formalin (NBF) for 16-32 hours at room temperature [12]. Avoid over-fixation beyond 32 hours as it may reduce RNA accessibility.
- Embedding and Sectioning: Process fixed tissues through graded ethanol and xylene, then infiltrate with paraffin. Cut 5μm (±1μm) sections and mount on Fisher Scientific SuperFrost Plus slides to prevent tissue detachment during the assay [5] [12].
- Slide Storage: Analyze specimens within 3 months of sectioning when stored at room temperature with desiccant [12].
Automated Staining Procedure
The following protocol is adapted for the DISCOVERY ULTRA system:
Slide Baking and Deparaffinization:
Target Retrieval:
Protease Digestion:
- Protease treatment for 16 minutes at 37°C [6].
- This step permeabilizes the tissue to enable probe access while maintaining RNA integrity.
Probe Hybridization:
Signal Amplification and Detection:
- Follow the RNAscope amplification steps as specified in the user manual.
- Apply chromogenic detection using reagents specified for your assay type (DAB for brown, Fast Red for red, or multiplex combinations) [8].
Counterstaining and Mounting:
Quality Control and Validation
Always run appropriate controls with each assay batch:
- Positive Control: Housekeeping genes (PPIB, POLR2A, or UBC) verify RNA integrity and successful assay performance. Successful staining should yield a PPIB/POLR2A score ≥2 or UBC score ≥3 [5] [12].
- Negative Control: Bacterial dapB should generate minimal background (score <1) in properly fixed tissue [5] [12].
- Instrument Maintenance: Perform Ventana instrument decontamination every three months to prevent microbial growth in fluid lines. Replace all bulk solutions with recommended buffers before running RNAscope assays [5].
Results and Discussion
Performance Validation and Scoring
The RNAscope assay uses a semi-quantitative scoring system based on the number of punctate dots per cell rather than signal intensity. This approach correlates directly with transcript copy numbers, enabling accurate assessment of gene expression levels [5] [12].
Table 3: RNAscope Scoring Guidelines for Quantitative Assessment
| Score | Criteria | Interpretation |
|---|---|---|
| 0 | No staining or <1 dot/10 cells | Negative/Negligible expression |
| 1 | 1-3 dots/cell | Low expression level |
| 2 | 4-9 dots/cell; none or very few dot clusters | Moderate expression |
| 3 | 10-15 dots/cell; <10% dots in clusters | High expression |
| 4 | >15 dots/cell; >10% dots in clusters | Very high expression |
Validation studies demonstrate that the automated RNAscope platform yields a high signal-to-noise ratio with minimal background staining, comparable to manual assay performance [6]. Quantitative analysis of housekeeping genes across multiple experiments and reagent lots shows excellent consistency and reproducibility, with PPIB signals consistently scoring ≥2 and dapB background scores remaining <1 in properly qualified samples [6].
Advanced Applications and Recent Developments
Multiplex RNA Detection
The RNAscope VS Duplex Assay enables simultaneous detection of two RNA targets within the same tissue section using different chromogens (e.g., DAB and Fast Red) [8]. This capability is particularly valuable for co-localization studies, such as mapping ligand-receptor interactions or identifying cell subtypes based on multiple RNA markers.
For 2-plex assays, Channel C1 target probes are Ready-To-Use (RTU), while Channel C2 probes are shipped as 50X concentrated stock. The target probes must be in different channels, and there must be a C1 probe in the mixture. If no C1 target probe is included, a "Blank Probe - C1" (Cat. No. 300041) can be used to maintain the proper probe ratio [5].
Protease-Free Workflows for Multiomics
A significant recent advancement is the development of RNAscope protease-free assays on the DISCOVERY ULTRA platform [13]. This innovation enables:
- Superior protein co-detection: Preservation of protease-sensitive protein epitopes for simultaneous RNA and protein detection in the same tissue section.
- Enhanced tissue morphology: Improved preservation of tissue architecture and histology for better cell segmentation and spatial analysis.
- Comprehensive biomarker validation: Simultaneous assessment of RNA and protein biomarkers in their native spatial context.
This protease-free workflow is particularly valuable for therapeutic development applications, including cancer research, gene therapy, and mechanism of action studies [13].
Figure 2: Protease-free multiomics workflow enabling simultaneous detection of RNA and protein biomarkers on the same tissue section, preserving both RNA integrity and protease-sensitive protein epitopes.
Specialized Applications in Therapeutic Development
The integrated RNAscope-DISCOVERY ULTRA platform supports diverse research applications:
- Gene Therapy Development: Visualization of AAV capsid biodistribution and transgene expression [14].
- Oligonucleotide Therapeutics: Detection of synthetic small RNAs (ASOs, siRNAs, miRNAs) using miRNAscope and RNAscope Plus assays [15].
- Immuno-oncology: Multiplex detection of immune cell markers and checkpoint inhibitors like PD-L1 [6].
- Biomarker Validation: Quantitative assessment of candidate biomarkers in archival specimens with spatial context [6] [10].
Troubleshooting and Optimization
Addressing Common Challenges
Weak or No Signal:
High Background:
- Confirm negative control (dapB) shows minimal staining (score <1).
- Use fresh reagents, including ethanol and xylene [5].
- Ensure adequate washing stringency with properly diluted buffers.
Tissue Detachment:
Optimization for Suboptimal Samples
For tissues not fixed according to recommended guidelines:
Adjust Pretreatment Conditions:
- For over-fixed tissues: Increase ER2 (target retrieval) time in 5-minute increments and protease time in 10-minute increments while maintaining standard temperatures [5].
- For under-fixed tissues: Optimize protease treatment duration to balance RNA accessibility with preservation of tissue integrity.
Instrument-Specific Optimization:
The integration of RNAscope technology with the Ventana DISCOVERY ULTRA platform provides researchers with a robust, automated solution for spatial transcriptomics that delivers single-cell resolution with preserved morphological context. This partnership addresses key challenges in reproducibility, throughput, and analytical precision that have traditionally limited the application of RNA ISH in both basic research and drug development.
The platform's flexibility—supporting chromogenic and fluorescent detection, multiplexing, and combined RNA-protein analysis—makes it suitable for diverse applications from biomarker discovery to therapeutic efficacy assessment. Recent innovations, including protease-free workflows, further expand its utility for comprehensive multiomics analyses.
Following the detailed protocols, quality control measures, and optimization strategies outlined in this application note will enable researchers to consistently generate high-quality, quantitative spatial gene expression data to advance their research programs.
Automated RNA in situ hybridization (ISH) has become a cornerstone of spatial biology, enabling researchers to visualize gene expression within the morphological context of tissues. For laboratories utilizing the Roche Ventana DISCOVERY ULTRA platform, the RNAscope VS series offers a suite of robust, standardized assays designed for high-throughput analysis. These assays leverage patented signal amplification and background suppression technology to achieve single-molecule sensitivity in formalin-fixed, paraffin-embedded (FFPE) tissues without requiring RNA-free environments [16] [17] [6]. This application note provides a detailed comparative analysis of four primary RNAscope VS assays—Universal HRP, Universal AP, Duplex, and BaseScope—to guide researchers in selecting and optimizing the appropriate method for their experimental goals in biomarker research and drug development.
Assay Comparison and Selection Guide
The selection of an appropriate RNAscope assay depends on several factors, including the number of targets, target size, expression levels, and desired detection output. The table below provides a systematic comparison of the four main VS assays to inform your experimental design.
Table 1: Comprehensive Comparison of RNAscope VS Assays for the DISCOVERY ULTRA System
| Assay Feature | RNAscope VS Universal HRP | RNAscope VS Universal AP | RNAscope VS Duplex | BaseScope VS Assay |
|---|---|---|---|---|
| Detection Method | Chromogenic/Fluorescent [8] | Chromogenic [8] | Chromogenic [17] [8] | Chromogenic [8] |
| Primary Chromogen | Diaminobenzidine (DAB) [16] | Fast Red [16] | DAB & Fast Red; Teal & Fast Red; Green & Fast Red [17] [8] | Fast Red [8] |
| Plexing Level | Singleplex [8] | Singleplex [8] | Duplex (2-plex) [17] [8] | Singleplex [8] |
| Target Sequence Length | 300-1000 bp [8] | 300-1000 bp [8] | 300-1000 bp [8] | 50-300 bp [8] |
| Key Benefit | Robust, sensitive, permanent stain; most widely used [8] | High contrast against pigmented backgrounds [16] | Simultaneous detection of two RNA targets [17] [8] | Specific detection of short targets (e.g., splice variants) [8] |
| Ideal Application | High-throughput routine applications [8] | Tissues with high melanin or background (e.g., lung, liver) [16] | Co-localization studies (e.g., ligand-receptor pairs) [17] [8] | Splice variants, exon junctions, highly homologous genes [8] |
Assay Selection Guidelines
- For Novel Targets or Routine High-Throughput Screening: The Universal HRP (DAB) assay is the standard for molecular pathology diagnostics. Its results are robust, permanent, and easily visualized under a standard bright-field microscope, making it ideal for archiving [16] [8].
- For Tissues with Endogenous Background: The Universal AP (Fast Red) assay is superior for highly pigmented tissues like melanoma, liver, or retina. The red chromogen provides a high-contrast signal against the tissue background and hematoxylin counterstain [16].
- For Co-localization Studies: The Duplex Assay is designed to simultaneously detect two distinct RNA species within the same cell. For optimal results, assign the lower expressed target to the C2 (AP/Fast Red) channel and the higher expressed target to the C1 (HRP/DAB) channel to leverage the brightness of the red chromogen [17].
- For Challenging Targets: The BaseScope VS Assay is specifically engineered for short RNA targets, including splice variants, exon junctions, and highly homologous gene family members, which are not accessible with full-length probe designs [8].
Core Experimental Protocols
This section outlines the critical procedural steps for the automated RNAscope VS assays on the Roche DISCOVERY ULTRA platform. The workflow is highly standardized but requires careful attention to sample preparation and pretreatment.
Universal Workflow for Sample Preparation
Proper sample preparation is the most critical factor for a successful RNAscope experiment. Consistent fixation and processing are paramount.
- Tissue Fixation and Embedding: For FFPE tissues, fix samples in 10% neutral-buffered formalin (NBF) for 16–32 hours at room temperature. Under-fixation or over-fixation can compromise RNA integrity and accessibility. After fixation, tissues should be dehydrated and embedded in paraffin using a standard automated processor [12].
- Sectioning: Cut tissue sections at a thickness of 5 μm (±1 μm). For optimal adhesion, use Fisher Scientific SuperFrost Plus slides and air-dry the sections before baking at 60°C for 1-2 hours prior to the automated run [12].
- Controls: Always include a species-specific positive control probe (e.g., PPIB, POLR2A) and a negative control probe (bacterial dapB) to verify assay performance and RNA quality [12].
Automated Staining Protocol for RNAscope VS Universal HRP Assay
The following protocol details the steps automated by the DISCOVERY ULTRA system for the Universal HRP assay, which serves as the foundation for other VS assays [16] [6].
Table 2: Required Reagents for RNAscope VS Universal HRP Workflow
| Component | Source | Product Name / Description | Ordering Code Example |
|---|---|---|---|
| Detection Kit | Roche | mRNA DAB Detection Kit | 06614353001 [16] |
| Sample Prep Kit | Roche | mRNA Sample Prep Kit | 08127166001 [16] |
| Probe Amplification Kit | Roche | mRNA Probe Amplification Kit | 06614337001 [16] |
| Target Probes | ACD | RNAscope 2.5 VS Target Probes | Catalog or Made-to-Order [16] |
| Control Probes | ACD | RNAscope 2.5 VS Control Probes | Species-specific [16] |
| Universal Reagent Kit | ACD | RNAscope VS Universal HRP Reagent Kit (Brown) | - [16] |
Step-by-Step Automated Protocol:
- Baking & Deparaffinization: Slides are baked (32 min at 37°C) and deparaffinized on the instrument [6].
- Target Retrieval: Tissue sections are subjected to heat-induced epitope retrieval for 16-24 minutes at 97°C using Roche's proprietary buffer [6].
- Protease Digestion: Protease treatment is applied for 16 minutes at 37°C to permeabilize the tissue and expose target RNA [6].
- Probe Hybridization: Target-specific ZZ probes are hybridized for 2 hours at 43°C [16] [6].
- Signal Amplification: A series of amplifier molecules are hybridized in a cascade to achieve substantial signal amplification. This proprietary process creates a polymer that can be visualized with chromogenic development [6].
- Chromogenic Detection: The HRP enzyme is used to catalyze the deposition of the DAB chromogen, resulting in a permanent brown precipitate at the site of each target RNA molecule [16].
- Counterstaining and Coverslipping: Tissues are counterstained with hematoxylin and coverslipped for permanent preservation [16].
Protocol Modifications for Other VS Assays
- For Universal AP Assay: Substitute the HRP-based detection kits with the mRNA RED Detection Kit (Roche #07099037001) and the RNAscope VS Universal AP Reagent Kit from ACD. The workflow remains identical, but the final detection uses Fast Red to produce a red signal [16].
- For Duplex Assay: The protocol involves a sequential detection process. The first target (C1) is detected with an HRP-based chromogen (e.g., DAB, Teal, or Green), followed by the detection of the second target (C2) with an AP-based Fast Red chromogen. The assay requires the RNAscope VS Duplex Reagent Kit and Roche's mRNA Duplex AMP kit (#08127174001) [17].
- For BaseScope Assay: The protocol is similar but optimized for shorter probes, using the BaseScope VS Reagent Kit and Roche's mRNA RED Detection Kit [8].
The Scientist's Toolkit: Essential Research Reagent Solutions
A successful automated RNAscope experiment requires a precise set of reagents and components. The following table details the essential materials for setting up these assays.
Table 3: Key Research Reagent Solutions for RNAscope VS Assays
| Item Category | Function and Importance | Specific Examples and Part Numbers |
|---|---|---|
| Core Detection Kits (Roche) | Enable the chromogenic visualization of the hybridized probes. The choice of kit determines the color of the final signal. | - mRNA DAB Detection Kit (06614353001) [16]- mRNA RED Detection Kit (07099037001) [16]- mRNA Teal/Green/Purple HRP Kits [16] [8] |
| Sample & Amplification Kits (Roche) | Prepare the tissue for hybridization and amplify the signal from the probe system. | - mRNA Sample Prep Kit (08127166001) [16]- mRNA Probe Amplification Kit (06614337001) [16]- mRNA Duplex AMP kit (08127174001) [17] |
| ACD Probe Sets | Target-specific reagents that are the foundation of assay specificity. "ZZ" probe design ensures single-molecule sensitivity. | - RNAscope 2.5 VS Target Probes [16]- Positive Control Probes (e.g., Hs-PPIB) [12]- Negative Control Probes (dapB) [12] |
| ACD Universal Reagent Kits | Contain the universal amplifier molecules and enzymes required for the signal amplification cascade specific to each assay type. | - RNAscope VS Universal HRP Reagent Kit (Brown) [16]- RNAscope VS Universal AP Reagent Kit (Red) [16]- RNAscope VS Duplex Reagent Kit [17] |
Workflow and Signaling Pathway Visualizations
The unique "ZZ" probe design and subsequent amplification steps are fundamental to the RNAscope technology's high sensitivity and low background. The following diagram illustrates this proprietary signaling pathway.
Diagram 1: RNAscope Signal Amplification Pathway.
The automated workflow on the DISCOVERY ULTRA integrates these biochemical steps into a seamless, hands-off procedure, as summarized below.
Diagram 2: Automated RNAscope VS Workflow on DISCOVERY ULTRA.
Data Interpretation and Analysis
Accurate interpretation of RNAscope results is based on a semi-quantitative scoring system that focuses on counting discrete, punctate dots, each representing a single RNA molecule.
- Scoring Guidelines: Evaluate the number of dots per cell, not the signal intensity [12].
- Score 0: No staining or <1 dot per 10 cells.
- Score 1: 1-3 dots per cell.
- Score 2: 4-10 dots per cell, very few clusters.
- Score 3: >10 dots per cell, <10% positive cells have dot clusters.
- Score 4: >10 dots per cell, >10% positive cells have dot clusters [6].
- Quality Control: A successful experiment requires a positive control (e.g., PPIB) score of ≥2 and a negative control (dapB) score of <1 [12].
- Quantitative Analysis: For advanced quantification, the HALO image analysis platform or other open-source software can be used to automatically count transcripts and cells, providing highly reproducible quantitative data [8] [6].
The RNAscope VS assays for the Roche DISCOVERY ULTRA platform provide a powerful and flexible suite of tools for precise spatial gene expression analysis in a standardized, high-throughput format. The choice between HRP, AP, Duplex, and BaseScope assays is dictated by the specific experimental question, whether it involves routine single-target detection, overcoming tissue background challenges, mapping cellular interactions through co-localization, or identifying elusive splice variants. By adhering to the recommended protocols for sample preparation, utilizing the appropriate controls, and applying the correct scoring methodology, researchers can reliably generate high-quality, publication-ready data that advances our understanding of gene expression in the context of tissue morphology and cellular heterogeneity.
The Ventana (Roche) DISCOVERY ULTRA platform represents a transformative automated system for advanced RNA in situ hybridization (ISH) analysis, enabling researchers to perform sophisticated spatial biology investigations with high throughput and exceptional reproducibility. Through ACD's RNAscope and BaseScope assays optimized for this platform, researchers can achieve single-molecule sensitivity in detecting RNA biomarkers within the morphological context of formalin-fixed, paraffin-embedded (FFPE) tissues. This automation significantly standardizes the complex processes of biomarker co-detection, splice variant analysis, and therapeutic oligonucleotide validation, making these advanced techniques accessible for routine laboratory applications. The integrated workflows on this system provide robust solutions for critical research areas including biomarker development, therapeutic efficacy assessment, and mechanism of action studies across diverse fields such as oncology, neuroscience, and gene therapy [8] [13].
The platform's versatility supports multiple detection modalities including chromogenic and fluorescent outputs, simultaneous RNA-RNA multiplexing, and integrated RNA-protein co-detection. Recent advancements include protease-free pretreatment reagents that preserve protease-sensitive epitopes, enabling superior protein detection alongside RNA targets without compromising antigen integrity. This capability is particularly valuable for comprehensive spatial multiomics approaches that require concurrent assessment of multiple biomarker classes within the same cellular context [13] [18]. The automation of these complex procedures ensures staining consistency and reduces technical variability, which is essential for quantitative image analysis and valid comparative studies across large sample sets.
Automated Assay Portfolio for High-Throughput Applications
Comprehensive Assay Comparison
The automated RNAscope platform offers multiple specialized assays tailored to distinct research applications, each with optimized chemistry for specific detection requirements. The selection criteria encompass factors such as target type, multiplexing needs, detection method, and application objectives.
Table 1: Automated RNAscope and BaseScope Assays on the DISCOVERY ULTRA Platform
| Assay Name | Detection Options | Chromogen Used | Reaction Type | Key Benefit | Ideal Applications |
|---|---|---|---|---|---|
| RNAscope VS Universal HRP | Chromogenic/Fluorescent | DAB, Purple, Teal, Green, FAM, FITC, Red610, Cy5 | Singleplex | Robust, sensitive, permanent stain; most widely used | High throughput routine applications |
| RNAscope VS Universal AP | Chromogenic | Fast Red | Singleplex | Robust, sensitive, permanent stain | High throughput routine applications |
| RNAscope VS Duplex | Chromogenic | DAB & Fast Red, Teal & Fast Red, Green & Fast Red | Singleplex, Duplex | Simultaneous detection of two RNA targets | Co-localization studies to map co-expression |
| BaseScope VS Assay - RED | Chromogenic | Fast Red | Singleplex | Specific detection of splice variants and short targets | Splice variants, exon junctions, short targets (50-300bp) |
All listed assays are compatible with archival FFPE specimens and can be quantitatively analyzed through visual inspection, HALO software, or any open-source image analysis software. Each kit typically accommodates 60 tissue sections (20mm × 20mm), making them suitable for medium-to-high throughput study designs [8].
Specialized Assay Workflows
Beyond the core RNAscope assays, specialized workflows address emerging research needs. The RNAscope Plus small RNA-RNA Assay enables detection of one small RNA (17-50 nucleotides) such as ASOs, miRNAs, or siRNAs, plus up to three mRNA targets in intact tissues. This capability is particularly valuable for therapeutic development, allowing researchers to simultaneously monitor oligonucleotide biodistribution and its functional effects on target gene expression [19] [15].
For integrated multi-omics approaches, the platform supports combined ISH-IHC workflows using translucent chromogens that enable clear visualization of both RNA and protein targets within the same tissue section. The recent introduction of protease-free methods further enhances these co-detection applications by preserving protein epitopes that might be damaged by conventional protease treatments [13] [18]. This advancement is particularly beneficial for detecting proteins with protease-sensitive epitopes while maintaining optimal RNA signal intensity.
High-Throughput Biomarker Screening Protocols
Automated Co-Detection Workflow for RNA and Protein Biomarkers
The integration of RNAscope ISH with immunohistochemistry (IHC) on the DISCOVERY ULTRA platform enables simultaneous visualization of RNA and protein biomarkers with single-cell resolution. This protocol leverages the newly developed protease-free pretreatment (VS PretreatPro) to preserve protein epitopes while allowing efficient RNA target accessibility.
Protocol Steps:
- Tissue Section Preparation: Cut FFPE tissue sections at 4-5μm thickness and mount on charged slides. Bake slides at 60°C for 1 hour to ensure proper adhesion.
- Deparaffinization and Pretreatment: Perform automated deparaffinization using EZ Prep solution at 75°C. Apply VS PretreatPro protease-free reagent for 8-16 minutes at 37°C [13].
- Probe Hybridization: Apply target-specific RNAscope probes (designed against 300-1000bp target sequences) and incubate for 2 hours at 40°C. For multiplex protein detection, apply primary antibodies diluted in antibody diluent simultaneously with RNA probes.
- Signal Amplification: Execute the RNAscope signal amplification steps using the VS Universal HRP or AP assay components with the following sequence:
- AMP 1 incubation: 20 minutes at 40°C
- AMP 2 incubation: 20 minutes at 40°C
- AMP 3 incubation: 20 minutes at 40°C
- Chromogen Development: For HRP-based detection, apply DAB chromogen for 10 minutes at room temperature. For AP-based detection, apply Fast Red for 10 minutes at room temperature. For fluorescent detection, apply appropriate fluorophores [8] [18].
- Counterstaining and Mounting: Apply hematoxylin counterstain for 10-20 seconds, followed by bluing reagent. Dehydrate through graded alcohols, clear in xylene, and mount with permanent mounting medium.
Troubleshooting Notes:
- For optimal RNA-protein co-detection, titrate both RNA probes and antibodies to determine ideal concentrations that maximize signal while minimizing background.
- When using translucent chromogens, ensure proper filter settings on brightfield microscopes to distinguish colorimetric signals.
- For quantitative analysis, include appropriate positive and negative control tissues in each run to validate staining performance [13] [20].
Quantitative Image Analysis Framework
High-throughput biomarker screening requires robust quantification methodologies to extract meaningful biological insights from stained tissues. The Professional Assay Services team at ACD utilizes multiple approaches for quantitative assessment:
Table 2: Quantitative Image Analysis Methods for RNAscope Data
| Analysis Method | Description | Applications | Output Metrics |
|---|---|---|---|
| Semi-quantitative Scoring | Visual assessment by trained pathologists | Rapid screening, quality control | Expression intensity (0-3+), distribution pattern |
| Visual H-Scoring | Semi-quantitative assessment incorporating intensity and percentage of positive cells | Biomarker validation, expression level comparison | H-score (0-300) |
| Digital Image Analysis with HALO | Automated quantification using Indica Labs HALO software | High-throughput studies, precise spatial quantification | RNA transcripts per cell, positive cell percentage, spatial distribution patterns |
| Spatial Biology Analysis | Assessment of cellular neighborhood and spatial relationships | Tumor microenvironment, host-response interactions | Spatial coordinates, cell-cell proximity, cluster analysis |
These analytical approaches can be applied to various research contexts including oncology biomarker validation, neuroscience applications, cell and gene therapy development, and single-cell RNAseq validation [20]. The integration of board-certified pathologist review with computational analysis provides both morphological context and quantitative rigor.
Splice Variant Detection Using BaseScope Technology
Principles of Splice Variant Analysis
Splice variants resulting from alternative splicing of exons in pre-mRNA significantly expand the functional complexity of the genome, with specific variants playing important roles in human diseases, particularly cancer and neurological disorders [21]. The detection of these variants within tissue context has been historically challenging due to the limited unique sequence space distinguishing different isoforms. The BaseScope Assay addresses this challenge by employing proprietary ZZ probes specifically designed to span exon-exon junctions, ensuring specific detection of mature mRNA variants rather than pre-mRNA or other similar sequences [22].
This technology enables:
- Specific detection of any exon junction with single-molecule sensitivity
- Analysis of broad sample types including FFPE and fresh frozen tissues
- Precise localization of cells expressing specific splice variants within complex tissue architectures
- Detection of short targets (50-300bp) including differentially included exons, splice junctions, and mutations within highly homologous gene families [22]
The ability to visually identify specific splice variants at cellular resolution provides critical insights into tumor heterogeneity, therapy resistance mechanisms, and tissue-specific isoform expression patterns that are often obscured in bulk sequencing approaches.
Experimental Protocol for Splice Variant Detection
The BaseScope assay on the DISCOVERY ULTRA platform follows an optimized workflow for specific detection of splice variants in FFPE tissues:
Probe Design Strategy:
- Identify the specific exon junction of interest (e.g., exon 13/15 junction for METΔ14 detection)
- Design a single ZZ probe (~50 bases) spanning the precise junction region
- Include control probes for:
- A constitutive exon junction present in all transcripts (e.g., exon 12/13 for MET)
- The wild-type specific junction (e.g., exon 14/15 for MET) [22]
Staining Protocol:
- Tissue Preparation: Cut FFPE sections at 4-5μm thickness. Bake at 60°C for 1 hour.
- Deparaffinization and Pretreatment: Deparaffinize with EZ Prep at 75°C. Perform target retrieval with appropriate retrieval solution followed by protease digestion (Protease III for 15-30 minutes at 40°C).
- Probe Hybridization: Apply BaseScope probe solution and incubate for 2 hours at 40°C.
- Signal Amplification: Perform sequential amplifier incubation:
- AMP 1: 20 minutes at 40°C
- AMP 2: 20 minutes at 40°C
- AMP 3: 20 minutes at 40°C
- AMP 4: 15 minutes at 40°C
- AMP 5: 30 minutes at 40°C
- AMP 6: 15 minutes at 40°C
- Detection: Develop with Fast Red chromogen for 10 minutes at room temperature.
- Counterstaining: Counterstain with hematoxylin, dehydrate, clear, and mount [22].
Validation and Controls:
- Include cell lines with known splice variant status as controls (e.g., H596 for METΔ14, A549 for MET wild-type)
- Run all three probe sets (common, wild-type-specific, variant-specific) in parallel on adjacent sections
- Correlate staining patterns with known expression profiles from orthogonal methods
- For novel splice variants, validate specificity using CRISPR-engineered cell lines with and without the specific junction [22]
Applications in Cancer Research
The BaseScope assay has been successfully applied to detect clinically relevant splice variants in cancer research. For example, in non-small cell lung cancer, the detection of MET exon 14 skipping (METΔ14) has therapeutic implications, as these tumors may respond to MET inhibitors. The assay enables:
- Specific identification of METΔ14 positive tumor cells within heterogeneous tissue sections
- Assessment of tumor heterogeneity in splice variant expression
- Correlation of variant expression with morphological features and protein expression
- Analysis of archival tissues for retrospective biomarker studies [22]
Similar approaches can be applied to other therapeutic targets such as EGFR, ERBB2, and AR splice variants, providing a powerful tool for biomarker discovery and validation in the era of precision oncology.
Research Reagent Solutions
The successful implementation of automated RNAscope and BaseScope assays relies on a comprehensive suite of specialized reagents and tools optimized for the DISCOVERY ULTRA platform.
Table 3: Essential Research Reagents for Automated RNAscope Applications
| Reagent/Category | Function | Application Notes |
|---|---|---|
| RNAscope VS Universal HRP/AP Assays | Core detection reagents for single-plex RNA detection | Available in 60-test kits; compatible with FFPE tissues |
| BaseScope VS Assay - RED | Specific detection of splice variants and short targets | Designed for targets 50-300bp; uses Fast Red chromogen |
| RNAscope Plus smRNA-RNA Assay | Simultaneous detection of 1 small RNA + 3 mRNAs | Enables ASO/miRNA/siRNA detection with mRNA targets |
| miRNAscope Assay | Detection of small RNAs (17-50 nt) | Optimized for ASOs, miRNAs, siRNAs |
| VS PretreatPro | Protease-free pretreatment reagent | Preserves protease-sensitive protein epitopes in co-detection |
| Target Probes | Target-specific ZZ probes | Designed against 300-1000bp for RNAscope; 50-300bp for BaseScope |
| HALO Software | Quantitative image analysis | Enables transcript counting, spatial analysis, and multiplex data quantification |
| Translucent Chromogens | Enzyme substrates for detection | Enable multiplexing with minimal spectral overlap |
These reagents form the foundation of robust automated ISH workflows, with extensive validation supporting their performance across diverse tissue types and research applications [8] [19] [18].
Workflow Visualization
Automated RNAscope Workflow Diagram
The workflow illustrates the integrated process for automated RNA in situ hybridization on the DISCOVERY ULTRA platform, highlighting key decision points for specialized applications including RNA-protein co-detection, small RNA analysis, and splice variant detection. The standardized protocol begins with tissue preparation and progresses through hybridization, amplification, and detection stages, with branching paths accommodating specific research needs. This automation ensures consistent staining quality essential for quantitative spatial biology applications [8] [13] [18].
Splice Variant Detection Strategy
The splice variant detection workflow outlines the systematic approach for identifying and validating specific exon junctions using the BaseScope assay. The process begins with target identification focusing on the specific exon junction of interest, followed by proprietary ZZ probe design that spans the precise junction region. A comprehensive control strategy employing three parallel probe sets (common, wild-type-specific, and variant-specific) ensures accurate interpretation of staining patterns. This method enables specific detection of mature mRNA variants rather than pre-mRNA, providing crucial information about functional isoform expression within the tissue morphological context [22].
Executing Flawless Runs: Automated RNAscope Workflows and Multiplexing Applications
Automated RNAscope in situ hybridization (ISH) on the Roche Ventana DISCOVERY ULTRA platform represents a significant advancement in molecular pathology, enabling robust, high-throughput detection of RNA biomarkers within intact tissue architecture. This standardized protocol leverages the proprietary RNAscope technology, which utilizes a novel double Z (ZZ) probe design for exceptional specificity and sensitivity, allowing for single-molecule detection at single-cell resolution [23]. Automation on the Ventana platform minimizes operational variability, enhances reproducibility, and streamlines complex workflows, making it an indispensable tool for translational research and drug development. The following sections provide a detailed application note and protocol, from initial setup to final chromogenic detection, specifically framed within the context of automated biomarker analysis for research purposes.
Key Research Reagent Solutions
The successful execution of the automated RNAscope assay depends on a suite of specialized reagents. The table below catalogs the essential materials and their functions within the workflow.
Table 1: Essential Research Reagents for Automated RNAscope Assays
| Reagent Solution | Function / Description |
|---|---|
| RNAscope VS Universal HRP Assay | A widely used chromogenic assay for singleplex RNA detection, offering robust, sensitive, and permanent staining [8]. |
| RNAscope VS Universal AP Assay | A chromogenic assay utilizing an alternative enzyme (alkaline phosphatase) for detection, with Fast Red as a common chromogen [8]. |
| RNAscope VS Duplex Assay | Enables simultaneous chromogenic detection of two distinct RNA targets within the same sample, ideal for co-localization studies [8]. |
| BaseScope VS Assay - RED | Designed for the specific detection of short RNA targets, such as splice variants, exon junctions, and highly homologous gene families [8]. |
| ZZ Probe Pairs | The core of RNAscope technology; these proprietary probe pairs provide signal amplification while suppressing background, ensuring high specificity [23]. |
| Protease-Free Reagents | Newer workflow options that allow for detection of proteins with protease-sensitive epitopes when performing RNA-protein co-detection [13]. |
| Chromogens (e.g., DAB, Fast Red) | Enzyme substrates that produce a visible, precipitating color at the target site. DAB yields a brown precipitate, while Fast Red yields a red precipitate [8]. |
Assay Selection and Quantitative Comparison
Choosing the appropriate assay format is critical for addressing specific research questions. The Roche DISCOVERY ULTRA system supports multiple RNAscope assay types, each with distinct advantages. The following table summarizes the key characteristics and applications of the primary assay formats to guide selection.
Table 2: Quantitative Comparison of Automated RNAscope Assay Formats on the DISCOVERY ULTRA
| Parameter | RNAscope VS Universal HRP | RNAscope VS Universal AP | RNAscope VS Duplex | BaseScope VS Assay |
|---|---|---|---|---|
| Detection Options | Chromogenic & Fluorescent [8] | Chromogenic [8] | Chromogenic [8] | Chromogenic [8] |
| Common Chromogen | Diaminobenzidine (DAB) [8] | Fast Red [8] | DAB & Fast Red [8] | Fast Red [8] |
| Reaction Type | Singleplex [8] | Singleplex [8] | Singleplex, Duplex [8] | Singleplex [8] |
| Key Benefit | Robust, sensitive, permanent stain; most widely used [8] | Robust, sensitive, permanent stain [8] | Simultaneous detection of two RNA targets [8] | Detection of splice variants, exon junctions, and short targets [8] |
| Ideal Application | High-throughput routine applications [8] | High-throughput routine applications [8] | Co-localization studies (e.g., ligand-receptor mapping) [8] | Short targets (<300 bp), complex gene families [8] |
| Target Length | 300-1000 bp [8] | 300-1000 bp [8] | 300-1000 bp [8] | 50-300 bp [8] |
| Sections per Kit | 60 (20 mm x 20 mm) [8] | 60 (20 mm x 20 mm) [8] | 60 (20 mm x 20 mm) [8] | 60 (20 mm x 20 mm) [8] |
Detailed Automated Workflow Protocol
This section outlines the comprehensive, step-by-step protocol for performing an automated RNAscope assay, from pre-run preparation to post-staining analysis.
Pre-Run Preparation and Slide Loading
Step 1: Tissue Section Preparation. Cut formalin-fixed, paraffin-embedded (FFPE) tissue sections at 4-5 μm thickness using a standard microtome. Mount sections on positively charged glass slides to ensure adherence during the rigorous automated protocol. Bake the mounted slides at 60°C for 60 minutes to secure the tissue.
Step 2: Deparaffinization and Dehydration. Load the baked slides onto the Ventana DISCOVERY ULTRA instrument. The automated run begins with a standard deparaffinization series using EZ Prep solution (Ventana) or xylene and ethanol, depending on the laboratory's standard operating procedure and reagent compatibility.
Step 3: Heat-Induced Epitope Retrieval. After deparaffinization, the slides are subjected to a controlled heating step in a proprietary cell conditioning solution (Ventana). This step is crucial for breaking cross-links formed during fixation and exposing the target RNA for probe hybridization. A typical retrieval condition is 95-100°C for 8-16 minutes, though this can be optimized for specific tissues.
Protease Digestion and Target Retrieval
Step 4: Protease Treatment. Following epitope retrieval, the tissue is digested with a specific protease enzyme (e.g., Protease 3 or Protease PLUS, Ventana) for 15-30 minutes at 37°C. This step permeabilizes the tissue and further unveils the target RNA sequences. Note: For workflows involving co-detection of proteins with protease-sensitive epitopes, newer protease-free reagent sets should be employed to preserve antigen integrity [13].
In Situ Hybridization and Amplification
Step 5: Probe Hybridization. Apply the specific RNAscope ZZ probe pair, diluted in a proprietary hybridization buffer, to the tissue section. The instrument then incubates the slides at 40°C for 2 hours. This controlled temperature allows for specific binding of the probe pairs to the target RNA sequence.
Step 6: Signal Amplification. The RNAscope assay employs a multi-step amplification process that occurs automatically within the instrument:
- Amp 1: Hybridized probes are sequentially bound by pre-amplifier and amplifier molecules.
- Amp 2-6: A series of amplifier molecules (e.g., AMP 2, AMP 3, etc.) build a complex on the initial ZZ probe structure.
- Label Probe: The final amplifier is conjugated with an enzyme label, either Horseradish Peroxidase (HRP) or Alkaline Phosphatase (AP), depending on the selected assay (see Table 2).
This cascade results in a massive amplification of the signal at the site of each target RNA molecule, enabling single-molecule sensitivity [23].
Chromogenic Detection and Counterstaining
Step 7: Chromogen Application. For HRP-based assays, the substrate Diaminobenzidine (DAB) is applied, which produces a brown, permanent precipitate upon enzymatic reaction [8]. For AP-based assays, Fast Red is a common chromogen, producing a red precipitate [8]. The development time is carefully controlled by the instrument to ensure optimal signal-to-noise ratio.
Step 8: Counterstaining and Coverslipping. Following chromogenic development, the slides are automatically counterstained with hematoxylin to provide nuclear context. The run concludes with the application of a mounting medium and a glass coverslip, resulting in slides ready for microscopic evaluation.
Diagram 1: Automated RNAscope workflow overview.
The RNAscope Technology Mechanism
The unparalleled specificity of the RNAscope assay is rooted in its unique probe design and amplification strategy. The core of this technology is the double Z (ZZ) probe, which consists of two separate probe strands that must bind adjacent to each other on the target RNA for amplification to initiate. This paired binding requirement effectively prevents non-specific hybridization and background signal. If only one Z probe binds, no amplification complex can form. Upon successful dual hybridization, a series of pre-designed amplifiers build a branching tree-like structure. The final step attaches an enzyme label (HRP or AP) to this complex, which, when exposed to its chromogenic substrate, generates a visible, localized signal dot representing a single RNA molecule [23].
Diagram 2: RNAscope mechanism of action for specific signal amplification.
Data Analysis and Quantitation Methods
Following the automated staining process, analysis is performed to extract quantitative and qualitative data from the samples.
Visual Inspection and Scoring: The initial assessment involves a pathologist or trained researcher using a standard bright-field microscope to evaluate staining distribution, intensity, and sub-cellular localization. A semi-quantitative H-score or a simple dot-counting method can be employed.
Digital Image Analysis: For robust, reproducible quantitation, slides are digitized using a whole-slide scanner. The resulting digital images are then analyzed with specialized software such as HALO or other open-source image analysis platforms [8]. These tools can automatically count individual RNA dots (signals) per cell, calculate the percentage of positive cells, and assess signal intensity within the context of the tissue morphology. This objective, high-content data is crucial for rigorous biomarker validation and research reproducibility.
In the era of precision medicine, understanding the complex spatial relationships between RNA transcripts and proteins within the tissue microenvironment has become crucial for biomarker discovery and therapeutic development. Traditional techniques that analyze bulk tissue mask critical cell-to-cell variations and fail to preserve valuable morphological context. The advent of automated, multiplexed in situ hybridization technologies, particularly on platforms like the Roche Ventana DISCOVERY ULTRA, has revolutionized our ability to extract rich biological information from single tissue sections while maintaining spatial architecture. This application note details streamlined strategies for implementing duplex RNA detection and RNA-protein co-localization within the framework of automated RNAscope assays, empowering researchers to unlock deeper insights from precious samples.
Core Principles of RNAscope Technology
The RNAscope platform employs a unique signal amplification strategy that enables single-molecule RNA detection at the cellular level with high specificity and sensitivity. The technology uses paired "Z"-shaped probes designed to bind adjacent target sequences on the RNA of interest [6] [24]. Each probe contains a tail region that serves as a binding site for pre-amplifier molecules. Only when both probes hybridize correctly in tandem can the subsequent signal amplification cascade proceed, dramatically reducing non-specific background binding that plagues traditional in situ hybridization methods [6].
This proprietary design is particularly suited for detecting partially degraded RNA in formalin-fixed, paraffin-embedded (FFPE) tissue samples, making it invaluable for clinical research applications [24]. The recent automation of this technology on platforms like the Ventana DISCOVERY ULTRA has standardized and simplified the process, enabling higher throughput and improved reproducibility for complex multiplexing applications [8] [6].
Visualization of RNAscope Mechanism
The following diagram illustrates the proprietary RNAscope probe design and signal amplification mechanism:
Automated Assay Configuration Options
The table below summarizes the primary RNAscope assay options available for automated multiplexing on the DISCOVERY ULTRA platform:
| Assay Type | Detection Method | Targets Detected | Primary Applications | Key Benefit |
|---|---|---|---|---|
| RNAscope VS Universal HRP | Chromogenic/Fluorescent | Single RNA target | High-throughput routine applications | Robust, sensitive, permanent stain [8] |
| RNAscope VS Universal AP | Chromogenic (Fast Red) | Single RNA target | High-throughput routine applications | Robust, sensitive, permanent stain [8] |
| RNAscope VS Duplex | Chromogenic (DAB & Fast Red) | Two RNA targets simultaneously | Co-localization studies to map co-expression [8] | Simultaneous detection of two RNA targets [8] |
| BaseScope VS | Chromogenic (Fast Red) | Short targets (~50-300 bp) | Splice variants, exon junctions, highly homologous genes [8] | Specific detection of challenging targets [8] |
| Integrated Co-detection | Chromogenic/Fluorescent | RNA and protein combinations | Cell-type specific gene expression with protein markers [25] | Simultaneous examination of RNA and protein [25] |
Experimental Protocols
Protocol 1: Automated Duplex RNA Detection on DISCOVERY ULTRA
This protocol enables simultaneous detection of two different RNA targets within the same tissue section using the RNAscope VS Duplex Assay.
Materials and Reagents
- RNAscope VS Duplex Assay Kit (Cat. #)
- Target-specific probe pairs (designed for each RNA target)
- DISCOVERY ULTRA Wash Buffer
- Protease solution
- DAB and Fast Red detection kits
- FFPE tissue sections (5 μm thickness)
Methodology
Slide Preparation: Bake FFPE tissue sections for 32 minutes at 37°C on the instrument, followed by deparaffinization using the integrated protocol [6].
Target Retrieval: Perform heat-induced epitope retrieval for 16-24 minutes at 97°C, depending on tissue type [6].
Protease Digestion: Apply protease treatment for 16 minutes at 37°C to permeabilize tissue and enhance probe accessibility [6].
Probe Hybridization: Hybridize target-specific probe pairs for 2 hours at 43°C. The duplex assay utilizes specially designed probe sets that enable simultaneous detection of two distinct RNA targets [8] [6].
Signal Amplification: Execute the RNAscope amplification steps according to the automated protocol. The proprietary amplification system builds detectable signals on the paired Z-probes [6].
Chromogenic Detection:
- Apply Fast Red chromogen for the first target
- Apply DAB chromogen for the second target
- Counterstain as appropriate [8]
Image Acquisition and Analysis: Scan slides using a digital pathology scanner and quantify signals using image analysis software such as HALO, which can distinguish and count individual RNA dots for each target [8] [6].
Protocol 2: RNA-Protein Co-detection Workflow
This integrated workflow enables researchers to simultaneously examine RNA expression and protein localization within the same tissue section, providing insights into cellular sources of secreted proteins and correlating transcript and protein expression patterns.
Materials and Reagents
- RNAscope Multiplex Fluorescent V2 Assay reagents
- Primary antibodies against target proteins
- Species-appropriate secondary antibodies or direct conjugates
- Protease solution
- Signal amplification components
- FFPE or fresh frozen tissue sections
Methodology
Sample Preparation: Fix and permeabilize tissue sections following standard protocols optimized for both RNA and protein preservation [25].
Protein Detection (First Round):
- Apply primary antibodies against the target protein
- Detect using chromogenic or fluorescent methods compatible with subsequent RNA ISH steps
- Image the protein signal if using sequential detection method [24]
RNA In Situ Hybridization:
- Perform target retrieval (15 minutes at 100°C for manual assays)
- Apply protease treatment (15 minutes at 40°C)
- Hybridize RNAscope probes targeting specific transcripts (2 hours at 40°C)
- Complete the RNAscope amplification steps [25]
Simultaneous Detection: For fully automated co-detection on the DISCOVERY ULTRA, optimize the antibody application and RNA ISH steps within a single automated run to minimize hands-on time and maximize reproducibility [24].
Signal Visualization: Use compatible chromogenic or fluorescent detection systems that allow clear distinction between RNA and protein signals.
Image Analysis: Employ multi-spectral imaging and advanced analysis software to quantify co-localization of RNA and protein signals within individual cells [24].
Workflow for RNA-Protein Co-detection
The following diagram outlines the integrated workflow for simultaneous RNA and protein detection:
Research Reagent Solutions
Essential materials and tools for implementing automated multiplexing assays:
| Reagent/Tool | Function | Application Notes |
|---|---|---|
| RNAscope Probe Sets | Target-specific oligonucleotide pairs | Designed for 300-1000 bp targets; 18-25 bp binding regions with 14 bp amplifier binding sites [6] |
| BaseScope Probe Sets | Detection of short RNA targets | Optimized for 50-300 bp targets; ideal for splice variants and highly homologous sequences [8] |
| Protease Solution | Tissue permeabilization | Critical for probe accessibility; optimized concentration and timing prevent over-digestion [6] |
| Chromogenic Substrates | Visual signal generation | DAB (brown), Fast Red (red), and additional colors enable multiplex detection [8] |
| HALO Image Analysis Software | Quantitative signal analysis | Automated dot counting and co-localization analysis for objective quantification [8] [6] |
| Multispectral Imaging Scanners | Signal capture and separation | Instruments like Akoya Polaris exclude autofluorescence for cleaner multiplex data [24] |
Data Analysis and Quantification
Ensuring Assay Quality and Reproducibility
Robust quality control measures are essential for generating reliable multiplexing data. The RNAscope platform incorporates built-in controls, including:
- Positive Control Probe (PPIB): Validates RNA integrity and overall assay performance
- Negative Control Probe (dapB): Assesses non-specific background staining
- Sample Qualification Criteria: PPIB scores ≥2 and dapB scores ≤1 indicate acceptable sample quality [6]
Quantitative analysis of RNAscope signals can be performed using automated image analysis algorithms. Studies have demonstrated excellent lot-to-lot consistency and run-to-run reproducibility, with statistical analysis showing no significant differences in signal counts across multiple reagent lots and experiments (P >0.05 by ANOVA) [6].
Discussion
Applications in Biomarker Research
The ability to perform automated, multiplexed RNA and protein detection opens new avenues in biomarker discovery and validation. Key applications include:
- Tumor Microenvironment Characterization: Simultaneous identification of immune cell transcripts and corresponding protein markers to map cellular interactions in cancer [24]
- Therapeutic Target Validation: Co-localization studies to verify that protein targets are expressed in cells containing the corresponding transcripts
- Host-Pathogen Interactions: Detection of viral RNA alongside host response proteins in infectious disease research [6]
- Drug Mechanism Studies: Tracking changes in both transcript and protein expression in response to therapeutic interventions
Implementation Considerations
Successful implementation of automated multiplexing strategies requires careful planning:
Sample Quality: Ensure proper tissue fixation (24 hours in 10% NBF recommended) and processing to preserve both RNA integrity and protein epitopes [6]
Assay Design: Select appropriate probe pairs and antibody combinations based on expression levels and cellular localization
Validation: Include appropriate controls and establish quantification benchmarks before running precious samples
Image Analysis: Implement standardized algorithms for consistent signal quantification across multiple experiments
Automated multiplexing technologies on platforms like the Ventana DISCOVERY ULTRA have significantly lowered the barrier to implementing sophisticated duplex RNA detection and RNA-protein co-localization assays. The RNAscope platform provides the sensitivity and specificity required for single-molecule detection while maintaining morphological context that is essential for understanding biological complexity. By following the optimized protocols and quality control measures outlined in this application note, researchers can reliably extract rich, multi-parameter data from individual tissue sections, accelerating biomarker discovery and therapeutic development programs.
For researchers and drug development professionals implementing automated RNAscope on the Roche Ventana DISCOVERY ULTRA platform, selecting the appropriate detection method represents a critical decision point that directly impacts experimental outcomes. The choice between chromogenic and fluorescent detection affects not only the visualization of RNA targets within the spatial context of tissues but also influences multiplexing capabilities, quantification potential, and compatibility with downstream analysis workflows. The Ventana DISCOVERY ULTRA system offers both chromogenic and fluorescent detection pathways through specialized reagent kits designed specifically for automated staining [8]. This guide provides a practical framework for selecting and implementing these detection options, focusing on the technical considerations most relevant to automated spatial biology research. Understanding the capabilities and limitations of each method enables researchers to optimize their experimental designs for robust, reproducible results that advance drug discovery and development pipelines.
Available Detection Options & Their Characteristics
Comprehensive Comparison of Detection Methods
The Roche Ventana DISCOVERY ULTRA platform supports multiple RNAscope detection strategies through dedicated assay kits. The RNAscope VS Universal HRP Assay offers the most flexibility, supporting both chromogenic and fluorescent detection, while the RNAscope VS Universal AP Assay and BaseScope VS Assay are dedicated chromogenic options for specific applications [8]. The selection criteria should be driven by experimental goals, including the number of targets, required contrast with tissue background, quantification needs, and compatibility with existing laboratory infrastructure.
Table 1: Detection Options for Automated RNAscope on Ventana DISCOVERY ULTRA
| Assay Name | Detection Type | Available Chromogens/Dyes | Plexing Level | Key Benefit | Ideal Application |
|---|---|---|---|---|---|
| RNAscope VS Universal HRP Assay | Chromogenic & Fluorescent | DAB, Purple, Teal, Green, FAM, FITC, Red610, Rhodamine, Cy5, DCC [8] | Singleplex | Maximum flexibility; most widely used | High throughput routine applications; studies requiring either chromogenic or fluorescent output |
| RNAscope VS Universal AP Assay | Chromogenic | Fast Red [8] | Singleplex | Bright color stains with high contrast to background | Tissues with endogenous background (melanin in skin, liver, lung from smokers) |
| RNAscope VS Duplex Assay | Chromogenic | DAB & Fast Red, Teal & Fast Red, Green & Fast Red [8] | Duplex | Simultaneous detection of two RNA targets | Co-localization studies to map co-expression of two targets (e.g., secreted ligand and its receptor) |
| BaseScope VS Assay - RED | Chromogenic | Fast Red [8] | Singleplex | Specific detection of short targets (50-300 bp) | Detection of splice variants, exon junctions, short insertions/deletions |
Chromogenic vs. Fluorescent Detection: Core Principles
Chromogenic detection generates a permanent, precipitate-based signal visible under standard bright-field microscopy, while fluorescent detection emits light at specific wavelengths when excited by appropriate light sources [1]. The fundamental difference in signal generation translates to distinct practical implications for automated RNAscope applications. Chromogenic methods, utilizing enzymes such as horseradish peroxidase (HRP) or alkaline phosphatase (AP) with substrates like 3,3'-diaminobenzidine (DAB) or Fast Red, produce insoluble precipitates that remain stable for years, making them ideal for clinical archives or long-term studies [26] [8]. Fluorescent detection, employing tyramide signal amplification (TSA) with fluorophores like FAM, FITC, or Cy5, provides superior sensitivity and is essential for multiplexing applications [8].
The RNAscope technology itself employs a unique "double-Z" probe design strategy that enables simultaneous signal amplification and background suppression, achieving single-molecule visualization while preserving tissue morphology [1]. This core technology functions with both chromogenic and fluorescent detection systems on automated platforms, ensuring high sensitivity and specificity regardless of the detection method selected.
Diagram 1: RNAscope Signal Detection Pathways. The core RNAscope technology utilizes a proprietary double-Z probe design that hybridizes to target RNA, followed by signal amplification through preamplifier, amplifier, and label probe binding. The detection pathway then diverges into chromogenic or fluorescent options depending on the experimental requirements.
Decision Framework: Selecting the Optimal Detection Method
Application-Driven Selection Criteria
The choice between chromogenic and fluorescent detection should be guided by specific experimental requirements rather than personal preference or laboratory convention. Chromogenic detection excels in several scenarios: for single-plex or duplex studies where permanent archival of samples is required; when working with tissues exhibiting high autofluorescence; in laboratories with only bright-field microscopy capabilities; and for clinical pathology applications where alignment with traditional IHC workflows is beneficial [26] [8]. The RNAscope VS Universal HRP Assay with DAB provides a robust, sensitive, permanent stain that is the most widely used RNAscope assay for high-throughput routine applications [8].
Fluorescent detection offers distinct advantages for multiplexing applications, superior sensitivity for low-abundance targets, and compatibility with quantitative image analysis. The RNAscope VS Universal HRP Assay supports fluorescent detection with multiple dye options including FAM, FITC, Red610, Rhodamine, and Cy5 [8]. Fluorescent detection is ideally suited for co-localization studies requiring multiple targets, quantitative analysis using automated image analysis platforms, and when working with thick tissue sections that benefit from optical sectioning capabilities of fluorescence microscopy [27].
Experimental Workflow Integration
Diagram 2: Detection Method Decision Framework. The selection between chromogenic and fluorescent detection methods should be guided by multiple experimental factors including target characteristics, tissue type, available equipment, and desired outputs, leading to optimized applications for each pathway.
Protocols for Automated Detection on Ventana Systems
Automated RNAscope Workflow on DISCOVERY ULTRA
The automated RNAscope workflow on the Ventana DISCOVERY ULTRA platform standardizes the in situ hybridization process, reducing variability and increasing reproducibility across experiments. While the specific protocol details vary based on the selected assay kit and detection method, the fundamental workflow follows a consistent pattern: sample preparation, pretreatment, hybridization, signal amplification, detection, and counterstaining [1] [8]. For FFPE tissues—the most common sample type for automated RNAscope—the process begins with baking and deparaffinization, followed by target retrieval to undo cross-links from formalin fixation [1].
A critical step in the automated workflow is protease treatment, which permeabilizes the tissue to enable probe access. The appropriate protease strength must be matched to the tissue type and fixation method [28]. Following protease treatment, the target probes are hybridized, and the signal amplification system builds the detection complex. The detection step then diverges based on the selected method: chromogenic detection involves enzyme-substrate reactions, while fluorescent detection employs fluorophore-conjugated labels [8]. The entire process is optimized for the DISCOVERY ULTRA system, with each kit supporting 60 tissue sections (20mm × 20mm) per kit [8].
Ventana DISCOVERY ULTRA Protocol Table
Table 2: Automated RNAscope Protocol Framework for Ventana DISCOVERY ULTRA
| Step | Process | Chromogenic Detection | Fluorescent Detection | Notes |
|---|---|---|---|---|
| 1 | Sample Preparation | FFPE sections (5μm) baked & deparaffinized | FFPE sections (5μm) baked & deparaffinized | Standard FFPE processing; ensure section adhesion |
| 2 | Target Retrieval | Citrate buffer, boiling (100-103°C), 15 min [1] | Citrate buffer, boiling (100-103°C), 15 min | Critical for FFPE samples; reverses cross-links |
| 3 | Protease Treatment | Protease Plus, 40°C, 30 min [1] | Protease III, 40°C, 30 min | Enzyme strength varies by tissue and fixation |
| 4 | Probe Hybridization | Target probes in hybridization buffer, 40°C, 3 hr [1] | Target probes in hybridization buffer, 40°C, 3 hr | Automated probe application by instrument |
| 5 | Signal Amplification | Preamplifier (30 min) → Amplifier (15 min) → Label Probe (15 min) [1] | Preamplifier (30 min) → Amplifier (15 min) → Label Probe (15 min) | Sequential hybridizations at 40°C |
| 6 | Detection | HRP/AP + Chromogen (DAB/Fast Red), 10-15 min | Fluorophore conjugate (TSA system), 10-15 min | Multiple color options available for each [8] |
| 7 | Counterstaining | Hematoxylin for chromogenic [1] | DAPI for fluorescent | Provides cellular context |
| 8 | Coverslipping | Non-aqueous mounting medium | Aqueous mounting medium (anti-fade) | Preserves signal integrity |
Quantitative Analysis & Data Interpretation
Image Analysis and Quantification Approaches
Both chromogenic and fluorescent RNAscope data can be quantitatively analyzed using increasingly sophisticated computational tools. The discrete punctate nature of RNAscope signals—with each dot theoretically representing a single RNA molecule—enables precise quantification at the cellular level [29] [1]. For chromogenic signals, analysis typically involves semi-quantitative scoring systems (0-5 scale) performed by trained pathologists, or digital image analysis using software such as HALO that quantifies signal area and intensity [20] [8]. For fluorescent signals, automated quantification is more straightforward, with software platforms capable of identifying individual transcripts and calculating transcript counts per cell [30].
Advanced analysis approaches include multiplex co-localization analysis, where the spatial relationship between multiple RNA targets is quantified, and spatial biology analysis, which maps transcript distribution within tissue architectures [20]. The open-source software QuPath has emerged as a powerful tool for automated quantification of RNAscope signals, particularly for fluorescent multiplex assays, offering cell detection algorithms and interactive machine learning for accurate segmentation of complex tissues [30].
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Essential Research Reagent Solutions for Automated RNAscope
| Reagent/Material | Function | Example Products | Application Notes |
|---|---|---|---|
| RNAscope Assay Kits | Core detection reagents | RNAscope VS Universal HRP Assay; RNAscope VS Universal AP Assay [8] | Kit selection determines detection options (chromogenic/fluorescent) |
| Target Probes | Gene-specific detection | C1, C2, C3, C4 channel probes [30] | Channel designation enables multiplexing; must match assay type |
| Protease Reagents | Tissue permeabilization | Protease Plus, Protease III, Protease IV [28] | Strength varies; selection critical for signal-to-noise optimization |
| Target Retrieval Reagents | Antigen unmasking | RNAscope Target Retrieval [28] | Essential for FFPE tissues; reverses formalin cross-links |
| Chromogenic Substrates | Enzyme-based signal generation | DAB, Fast Red, Green, Teal, Purple [8] | Produces permanent, precipitative signals for brightfield microscopy |
| Fluorophore Conjugates | Fluorescent signal generation | FAM, FITC, Red610, Rhodamine, Cy5 [8] | Enables multiplexing; compatible with TSA amplification |
| Counterstains | Nuclear visualization | Hematoxylin (chromogenic), DAPI (fluorescent) [1] [30] | Provides cellular context; must not interfere with signal detection |
| Mounting Media | Slide preservation | Non-aqueous (chromogenic), Aqueous anti-fade (fluorescent) | Critical for signal preservation, especially for fluorescent assays |
| Image Analysis Software | Quantification & analysis | HALO, QuPath, any open-source software [20] [8] [30] | Enables automated, high-throughput quantification of signals |
Troubleshooting & Optimization Strategies
Addressing Common Detection Challenges
Successful implementation of automated RNAscope on the Ventana platform requires anticipation of potential technical challenges and implementation of appropriate optimization strategies. Excessive background often results from inadequate protease optimization or insufficient washing between amplification steps. For chromogenic detection, endogenous enzyme activity (peroxidases or phosphatases) can cause background, which is typically blocked by included reagents [28]. Weak signal intensity may indicate over-fixed tissue, suboptimal protease treatment, or probe degradation. Running appropriate controls is essential for troubleshooting: the housekeeping gene UBC (ubiquitin C) assesses RNA integrity, while the bacterial gene dapB serves as a negative control [1].
For fluorescent detection, additional considerations include tissue autofluorescence, which can be mitigated using specialized imaging techniques or signal validation approaches, and fluorophore bleaching, which requires anti-fade mounting media and proper storage. Multiplex fluorescent assays require careful validation of each channel individually to confirm specificity and avoid bleed-through between channels [27]. The automated nature of the Ventana DISCOVERY ULTRA platform minimizes many procedural variabilities, but initial validation of each target and tissue type remains essential for generating reliable, reproducible data.
Optimization Workflow Diagram
Diagram 3: Troubleshooting and Optimization Workflow. Common issues in automated RNAscope detection require targeted optimization strategies, with validation using appropriate positive and negative controls essential for confirming assay performance.
The transition from manual to digital analysis of RNAscope assays represents a critical advancement in biomarker research, particularly within automated workflows like those on the Ventana DISCOVERY platform. This application note provides a detailed protocol for performing both traditional manual scoring and objective, high-content digital quantification using the HALO image analysis platform. By comparing established manual guidelines with the capabilities of HALO digital modules, we demonstrate how researchers can achieve reproducible, single-cell resolution data for gene expression analysis, enabling more robust and standardized outcomes in therapeutic development.
Manual RNAscope Scoring Guidelines
The RNAscope assay employs a unique probe design and signal amplification strategy that allows for the visualization of target RNAs as punctate dots, where each dot represents an individual RNA molecule [6]. The manual scoring of these signals is a foundational skill for researchers.
The Traditional 5-Tier Scoring System
Manual interpretation of RNAscope results relies on a standardized scoring system that categorizes signal abundance and distribution into five distinct grades. This system requires visualization at specific magnifications to accurately assess dot count and clustering [6].
Table 1: Standard RNAscope Manual Scoring Guidelines
| Score | Staining Description | Dot Count per Cell | Dot Clusters | Visualization Magnification |
|---|---|---|---|---|
| 0 | No staining or negligible staining | <1 dot per 10 cells | Not applicable | 40x |
| 1 | Faint staining visible | 1-3 dots | Very few | 20x-40x |
| 2 | Moderate staining with good visibility | 4-10 dots | Very few | 20x-40x |
| 3 | Strong staining | >10 dots | <10% of positive cells | 20x |
| 4 | Very strong, abundant staining | >10 dots | >10% of positive cells | 20x |
For a sample to be considered of acceptable quality, the positive control probe (e.g., for the housekeeping gene PPIB) should typically yield a score of 2 or higher, while the negative control probe (e.g., bacterial gene dapB) should yield a background score of 1 or lower [6].
Digital Quantification with the HALO Platform
Digital image analysis with the HALO platform brings objective, quantitative data to RNA in situ hybridization, enabling a new generation of applications based on single-cell analysis [31].
HALO ISH and FISH Modules
The HALO platform offers purpose-built modules for RNAscope analysis. The ISH module is designed for brightfield images and can simultaneously analyze up to three chromogenic or silver-labelled DNA or RNA ISH probes on a cell-by-cell basis [32]. The FISH module performs analogous functions for fluorescent assays, capable of analyzing an unlimited number of fluorescent nucleic acid probes [33]. Both modules measure spot numbers and area per cell and compartment, and calculate H-scores for each probe.
Key Analytical Outputs of HALO
- Quantitative and Reproducible Spot Counting: Automated counting of RNA molecules represented as punctate dots across entire tissue sections [31].
- Cell-by-Cell Expression Profiles: Analysis of expression data at single-cell resolution while maintaining the spatial and morphological context [31] [6].
- H-Scores and Histograms: Automatic generation of traditional pathology scores like H-scores and graphical representation of data distributions [31].
- Spatial Analysis: With the Spatial Analysis module, users can perform proximity analysis, nearest neighbor analysis, infiltration analysis, and generate density heatmaps to understand cellular relationships [31].
The workflow below illustrates the integrated process of automated RNAscope staining on the Ventana platform followed by digital analysis with HALO.
Comparative Analysis: Manual vs. Digital Scoring
The transition from manual interpretation to digital quantification represents a significant evolution in RNAscope data analysis, with each method offering distinct advantages.
Table 2: Comparison of Manual Scoring and HALO Digital Quantification
| Analysis Characteristic | Manual Scoring | HALO Digital Quantification |
|---|---|---|
| Objectivity | Subjective, based on pathologist's interpretation | Objective, algorithm-driven analysis [31] |
| Throughput | Low to moderate, time-consuming for large studies | High, rapid whole-slide analysis with batch processing [31] [33] |
| Data Granularity | Categorical (0-4) | Continuous numerical data (exact dot counts, intensities) [6] |
| Spatial Context | Maintained but qualitatively assessed | Quantitatively maintained with spatial analysis modules [31] |
| Reproducibility | Variable, inter-observer discrepancies likely | High, consistent parameters applied across all samples [31] |
| Multiplexing Capacity | Limited by human perception | Robust analysis of multiplex assays (up to 12-plex with HiPlex) [33] |
| Output Format | Score designation | Cell-by-cell data, summary statistics, H-scores, histograms [31] |
| Training Required | Extensive pathology expertise | Minimal, user-friendly interface [33] |
A key advantage of HALO digital quantification is its ability to generate continuous, rather than categorical, data outputs. This allows for more sensitive detection of subtle expression changes and robust statistical analysis. The software can process millions of cells across entire tissue sections while maintaining an interactive link between quantitative data and its morphological context [33].
Experimental Protocols
Protocol: Automated RNAscope on Ventana DISCOVERY ULTRA
The following protocol has been validated for the Ventana DISCOVERY ULTRA platform [6]:
Sectioning and Setup
- Cut formalin-fixed, paraffin-embedded (FFPE) tissue sections at 5μm thickness.
- Bake sections for 32 minutes at 37°C on the instrument.
- Deparaffinize on the instrument using standard protocols.
Target Retrieval
- Perform target retrieval for 16 minutes at 97°C for cell pellets or 24 minutes at 97°C for tissues using appropriate retrieval buffers.
Protease Treatment
- Apply protease treatment for 16 minutes at 37°C to permeabilize tissues.
Probe Hybridization
- Hybridize with RNAscope probes for 2 hours at 43°C.
- Follow with RNAscope signal amplification steps according to manufacturer specifications.
Chromogenic Detection
- Perform DAB chromogenic detection using Ventana detection reagents.
- Counterstain as appropriate, then coverslip.
Protocol: HALO Image Analysis for RNAscope Assays
For analyzing RNAscope assays with the HALO platform [31] [33] [32]:
Image Import and Quality Control
- Import whole slide images in compatible formats (SVS, MRXS, NDPIS, CZI, etc.).
- Annotate regions of interest using freehand or selection tools.
Module Selection and Setup
- Select the appropriate HALO module (ISH for brightfield, FISH for fluorescence).
- For brightfield ISH analysis, configure color deconvolution parameters to isolate chromogenic signals.
Nuclear Segmentation
- Utilize pre-trained AI-based networks for optimal nuclear segmentation.
- Adjust segmentation parameters using real-time tuning features for live feedback.
Spot Detection and Quantification
- Set parameters for spot detection, including minimum and maximum dot size and intensity thresholds.
- Define cellular compartments (nuclear, cytoplasmic) for subcellular localization analysis.
Phenotype Assignment (for multiplex assays)
- Use the intuitive phenotype editor to assign cell types based on marker expression patterns.
- Visualize phenotypes using real-time tuning features.
Analysis Execution and Validation
- Execute analysis on single images or use batch processing for high-throughput studies.
- Validate results by visually comparing image markups with raw data, toggling cell populations.
Data Export and Visualization
- Export cell-by-cell data, summary statistics, or customized reports.
- Generate publication-quality figures with the figure maker feature.
- Perform spatial analysis using heat maps, proximity analysis, or infiltration analysis.
The Scientist's Toolkit: Research Reagent Solutions
Successful RNAscope analysis and HALO quantification requires specific reagents and tools. The following table details essential materials and their functions.
Table 3: Essential Research Reagents and Tools for RNAscope with HALO Analysis
| Reagent/Tool | Function/Description | Example Applications |
|---|---|---|
| RNAscope Probe | Target-specific oligonucleotide pairs designed to bind adjacent sequences on target RNA [6] | Hs-PPIB (positive control), Hs-TBP (target testing), dapB (negative control) [6] |
| Ventana Reagent Buffers | Automated platform-compatible buffers for target retrieval, protease digestion, and washing | Target retrieval (ER2), protease digestion, and wash buffers optimized for Ventana systems [6] |
| Chromogenic Detection Kits | Enzyme-chromogen systems for signal visualization (DAB, Fast Red) | Single-plex (DAB) and duplex (Fast Red + DAB) detection [6] |
| HALO ISH/FISH Module | Purpose-built software modules for spot quantification and cellular analysis | RNAscope brightfield (ISH) and fluorescent (FISH) assay quantification [31] [32] |
| HALO AI Add-on | Pre-trained deep learning networks for optimized nuclear and membrane segmentation | Enhanced segmentation accuracy in challenging tissues [33] |
| Positive Control Tissues | Tissues with known expression patterns for assay validation | Qualification of assay performance across experiments [6] |
The integrated workflow from automated staining to digital analysis creates a robust pipeline for biomarker research. The schematic below details the decision pathway for appropriate analysis selection based on experimental parameters.
The integration of automated RNAscope technology on the Ventana DISCOVERY platform with HALO digital image analysis creates a powerful, standardized pipeline for gene expression analysis. While manual scoring provides an essential framework for understanding RNAscope signals, digital quantification with HALO offers unprecedented objectivity, reproducibility, and depth of data extraction. By following the detailed protocols and guidelines outlined in this application note, researchers can confidently implement both analytical approaches, selecting the most appropriate method based on their specific research questions and throughput requirements. This comprehensive approach to post-assay analysis ensures robust, publication-quality data that advances biomarker discovery and therapeutic development.
Solving Common Challenges: Troubleshooting and Optimization for Peak Assay Performance
For researchers and drug development professionals utilizing automated RNAscope on the Ventana DISCOVERY platform, robust and reproducible results are paramount. The success of this advanced in situ hybridization technology hinges on pre-analytical steps. Proper sample preparation—encompassing fixation, processing, and sectioning—is the foundation for preserving RNA integrity and tissue morphology, enabling sensitive and specific single-molecule RNA detection within an authentic histological context. This application note details the critical best practices to ensure your samples are optimally prepared for automated RNAscope analysis.
The Impact of Sample Preparation on RNAscope Results
The quality of sample preparation directly dictates the outcome of the RNAscope assay by influencing two key factors: RNA accessibility and RNA integrity. The conditions of fixation and processing determine whether the target RNA is available for probe hybridization, while also protecting it from degradation.
- Under-fixation: This occurs with shorter fixation times or lower temperatures than recommended. It results in inadequate tissue preservation, leading to protease over-digestion during the assay pretreatment. The consequence is a loss of RNA and poor tissue morphology [34].
- Over-fixation: Fixing tissues for longer than the recommended window causes excessive cross-linking. This makes the tissue impermeable, resulting in protease under-digestion. The probes cannot effectively access the target RNA, leading to low signal and a poor signal-to-background ratio, despite excellent tissue morphology being preserved [34].
- Delayed Fixation: Any delay between tissue collection and immersion in fixative allows endogenous RNases to degrade RNA, producing lower signal or no signal in the final assay [34].
Best Practices for Fixation
Fixation is the most critical step in stabilizing the tissue and its RNA content. Adherence to the following protocol is essential for optimal results on the Ventana DISCOVERY system and other automated platforms.
Recommended Fixation Protocol
Table 1: Optimal Fixation Parameters for FFPE Tissues
| Parameter | Recommended Specification |
|---|---|
| Fixative | Fresh 10% Neutral Buffered Formalin (NBF) [34] [12] |
| Fixation Time | 16–32 hours (24 hours ± 8 hours) at Room Temperature [34] [12] |
| Fixation Temperature | Room Temperature (do not fix at 4°C) [34] [35] |
| Tissue Thickness | 3–4 mm blocks to ensure complete fixative penetration [12] |
Protocol Details
- Fixative Selection: While 10% NBF is strongly recommended, 4% Paraformaldehyde (PFA) prepared in RNase-free PBS is an acceptable alternative for fixed-frozen tissues [36]. It is crucial to use fresh fixative to ensure consistency and efficacy.
- Tissue Handling: Tissues should be dissected and placed into a generous volume of fixative (approximately 10:1 fixative-to-tissue ratio) immediately after collection to prevent RNA degradation [34] [1].
Figure 1: The direct impact of fixation quality on RNAscope assay outcomes.
Tissue Processing and Embedding
Following fixation, tissues must be processed into Formalin-Fixed, Paraffin-Embedded (FFPE) blocks using standardized protocols.
- Dehydration and Clearing: Fixed tissues are dehydrated in a graded series of ethanol, followed by clearing in xylene [12] [37].
- Paraffin Infiltration and Embedding: Tissues are infiltrated with melted paraffin. The paraffin-embedding temperature should be held at no more than 60°C to prevent RNA degradation [12]. The use of an automated processor is recommended for consistency.
Sectioning and Slide Storage
Proper sectioning and slide storage are vital to prevent RNA loss and ensure tissue adhesion during the automated assay procedure.
Table 2: Sectioning and Storage Guidelines by Sample Type
| Sample Type | Recommended Section Thickness | Slide Type | Storage Conditions After Sectioning |
|---|---|---|---|
| FFPE Tissue | 5 ± 1 μm [12] [6] | SuperFrost Plus [12] [38] | With desiccant, at room temperature, for up to 3 months [12] [35] |
| Fixed-Frozen Tissue | 7–15 μm [12] [36] | SuperFrost Plus [36] | At -80°C in an airtight container for up to 3 months [35] |
| Fresh-Frozen Tissue | 10–20 μm [12] | SuperFrost Plus [12] | At -80°C in an airtight container for up to 3 months [35] |
Protocol Details
- Slide Preparation: After sectioning, FFPE slides should be air-dried and then baked at 60°C for 1–2 hours prior to the RNAscope assay to ensure tissue adhesion [12].
- Critical Note: The use of Fisher Scientific SuperFrost Plus slides is mandatory for all tissue types. Other slide types may result in tissue loss during the rigorous protocol steps [12] [38] [36].
Quality Control and Sample Qualification
Before running precious experimental samples on the Ventana DISCOVERY system, it is imperative to qualify the sample RNA quality and optimize pretreatment conditions if the preparation history is unknown or suboptimal [34] [38].
Control Probes and Scoring
A minimum of three slides per sample should be run: one for the target probe, one for a positive control probe, and one for a negative control probe [35].
- Positive Control Probes: Species-specific probes for housekeeping genes assess tissue RNA integrity. Common choices include:
- Negative Control Probe: The bacterial dapB gene should not generate signal in properly fixed tissue. A score of <1 indicates low to no background [12] [38] [1].
Table 3: RNAscope Semi-Quantitative Scoring Guidelines
| Score | Staining Criteria | Interpretation |
|---|---|---|
| 0 | No staining or <1 dot per 10 cells | Negative |
| 1 | 1-3 dots per cell (visible at 20-40X magnification) | Low |
| 2 | 4-9 dots per cell, very few dot clusters | Moderate |
| 3 | 10-15 dots per cell, & <10% dots are in clusters | High |
| 4 | >15 dots per cell, & >10% dots are in clusters | Very High |
Scoring is based on the number of punctate dots per cell, not signal intensity [38].
Pretreatment Optimization for Automated Platforms
If control probe results are suboptimal (low positive signal or high background), pretreatment conditions on the automated system may require optimization.
- For Ventana DISCOVERY ULTRA/XT: Adjust the "Pretreat 2" (boiling) and/or protease treatment times as outlined in the user manual for over- or under-fixed tissues [38].
- For Leica BOND RX: The standard pretreatment is 15 minutes Epitope Retrieval 2 (ER2) at 95°C and 15 minutes Protease at 40°C. For over-fixed tissues, increase ER2 time in 5-minute increments and Protease time in 10-minute increments (e.g., 20 min ER2, 25 min Protease) [38].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Key Materials and Reagents for RNAscope Assay
| Item | Function | Note |
|---|---|---|
| SuperFrost Plus Slides | Ensures tissue adhesion throughout the rigorous assay procedure. | Mandatory; other slides may cause tissue loss [12] [38]. |
| HybEZ Oven | Provides precise temperature (40°C) and humidity control during manual hybridization steps. | Required for manual assay performance [38] [35]. |
| ImmEdge Hydrophobic Barrier Pen | Creates a barrier to retain reagents over the tissue section. | The only pen recommended to maintain a barrier throughout the procedure [38] [35]. |
| RNAscope Control Probes (PPIB, dapB) | Qualifies sample RNA integrity and assay performance. | Essential for interpreting experimental results [12] [38]. |
| RNAscope 2.5 LS Reagent Kit | Contains all reagents for the detection assay on automated platforms. | Specifically designed for automated staining systems [37]. |
| Fresh 10% NBF, Ethanol, Xylene | Standard reagents for fixation, dehydration, and deparaffinization. | Always use fresh reagents for optimal results [38]. |
Meticulous sample preparation is not merely a preliminary step but a critical determinant of success for automated RNAscope assays. Strict adherence to the prescribed protocols for fixation in fresh 10% NBF for 16–32 hours, controlled processing and embedding, and precise sectioning on recommended slides establishes the foundation for reliable, high-quality data. By integrating rigorous quality control using positive and negative controls, researchers can confidently utilize the full power of the Ventana DISCOVERY platform to achieve sensitive, specific, and quantifiable in situ RNA analysis for drug development and biomarker research.
For researchers utilizing the Ventana DISCOVERY platform for RNAscope assays, control probes provide an essential toolkit for diagnosing assay performance and ensuring reliable gene expression data. The positive control probes—PPIB, POLR2A, and UBC—represent housekeeping genes with varying expression levels, while the negative control probe dapB detects a bacterial gene that should not be present in mammalian tissues [38] [39]. Proper interpretation of these controls is fundamental to distinguishing technical artifacts from biological findings, particularly in automated environments where standardized protocols are critical for reproducible results. Systematic analysis of control probe performance enables researchers to qualify their tissue samples, verify RNA integrity, and optimize pretreatment conditions for different tissue types and fixation protocols [37].
Control Probe Profiles and Expected Results
Characteristics of Individual Control Probes
Each control probe serves a distinct purpose in assay validation, with expression characteristics tailored to specific diagnostic functions.
Table 1: Control Probe Characteristics and Interpretation Guidelines
| Probe Name | Function | Expression Level | Expected Result | Failed Assay Indicator |
|---|---|---|---|---|
| dapB | Negative Control | Bacterial gene | Score <1: No or minimal background staining [38] [39] | Score ≥1: Excessive background or non-specific signal [38] |
| PPIB | Positive Control | Low-copy (10-30 copies/cell) [38] [39] | Score ≥2: Relatively uniform signal [38] [39] | Score <2: Poor RNA quality or suboptimal protease treatment [38] |
| POLR2A | Positive Control | Low-copy (5-15 copies/cell) [38] [39] | Score ≥2: Relatively uniform signal [38] [39] | Score <2: Poor RNA quality or suboptimal protease treatment [38] |
| UBC | Positive Control | High-copy [38] [39] | Score ≥3: Relatively uniform signal [38] [39] | Score <3: Poor RNA quality or suboptimal protease treatment [38] |
Establishing a Baseline with Control Slides
Before evaluating experimental samples, researchers should establish performance benchmarks using control slides provided by ACD (Human Hela Cell Pellet, Cat. No. 310045; Mouse 3T3 Cell Pellet, Cat. No. 310023) [38]. These standardized controls enable researchers to verify that the entire RNAscope workflow—from reagent preparation to final detection—is functioning optimally before proceeding with precious experimental samples [38] [37]. On the Ventana DISCOVERY system, this includes confirming that instrument maintenance has been performed, bulk solutions have been replaced with appropriate buffers, and the slide cleaning option has been disabled in the software settings [38].
Diagnostic Framework for Troubleshooting Assay Issues
Systematic Approach to Control Probe Interpretation
A structured diagnostic workflow enables efficient problem identification. The following diagram illustrates the decision-making process for interpreting control probe results on the Ventana DISCOVERY platform:
Troubleshooting Common Issues on Ventana DISCOVERY Systems
High Background (dapB Score ≥1)
When the negative control probe shows elevated signal, several technical factors should be investigated:
- Protease Optimization: On the Ventana DISCOVERY ULTRA platform, adjust the VS Protease treatment times. For over-fixed tissues, increase protease incubation incrementally [38] [39].
- Instrument Maintenance: Perform the decontamination protocol every three months to prevent microbial growth in fluidic lines. Replace all bulk solutions with recommended buffers before running the RNAscope assay [38].
- Reagent Preparation: Warm probes and wash buffer at 40°C to resolubilize any precipitation that occurs during storage [38].
Weak Target Signal (PPIB/POLR2A Score <2 or UBC Score <3)
Inadequate signal from positive controls indicates issues with RNA accessibility or quality:
- Epitope Retrieval Optimization: Adjust RNAscope VS Universal Target Retrieval v2 conditions. For under-fixed tissues, increase retrieval time or temperature incrementally [38] [39].
- RNA Quality Assessment: Check tissue collection and fixation procedures. Tissues should be fixed in fresh 10% NBF for 16-32 hours [38]. For frozen tissues, ensure proper snap-freezing protocols were followed to prevent RNA degradation [40].
- Protease Treatment: Increase protease digestion times for over-fixed tissues to enhance RNA accessibility [38].
Irregular Staining Patterns
Non-uniform staining across tissue sections suggests variable tissue processing:
- Fixation Consistency: Ensure consistent fixation times and formalin penetration throughout all tissue samples [37].
- Section Quality: Use Superfrost Plus slides to prevent tissue detachment and ensure even reagent coverage [38] [39].
- Hybridization Conditions: Maintain optimum humidity and temperature during hybridization steps using the HybEZ system [38].
Experimental Protocols for Control Probe Analysis
Automated RNAscope Protocol for Ventana DISCOVERY Systems
The following protocol has been optimized specifically for control probe analysis on the Ventana DISCOVERY platform:
Table 2: Automated RNAscope Protocol for Ventana DISCOVERY Systems
| Step | Reagent/Process | Conditions | Purpose | Quality Control Check |
|---|---|---|---|---|
| Pretreatment | Baking & Deparaffinization | 32 min at 37°C [6] | Remove paraffin, adhere tissue | Visualize tissue integrity after deparaffinization |
| Target Retrieval | 16-24 min at 97°C [6] | Unmask target RNA | Adjust time based on fixation level | |
| Protease Treatment | 16 min at 37°C [6] | Permeabilize tissue | Critical step - optimize for each tissue type | |
| Hybridization | Probe Hybridization | 2 hr at 43°C [6] | Target-specific binding | Verify probe mixture temperature at 40°C before use [38] |
| Signal Amplification | AMP1-AMP7 | Standard conditions [37] | Signal buildup | Perform all steps in sequence - omitting any step results in no signal [38] |
| Detection | DAB Reaction | Standard conditions [37] | Chromogenic detection | Monitor development time consistency |
| Counterstaining | Hematoxylin [37] | Nuclear visualization | Dilute Gill's Hematoxylin 1:2 for optimal results [38] |
Sample Qualification Workflow
Before running experimental samples, implement this sample qualification protocol:
- Section Preparation: Cut 5μm sections from FFPE blocks and mount on Superfrost Plus slides [37].
- Control Probe Selection: Include PPIB, POLR2A, or UBC based on target expression level, plus dapB negative control [38] [37].
- Automated Processing: Run slides on Ventana DISCOVERY following the protocol in Table 2.
- Scoring and Interpretation:
- Pretreatment Optimization: If controls fail, adjust target retrieval and protease times incrementally [38].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Essential Research Reagents for Automated RNAscope
| Reagent/Equipment | Function | Specific Recommendation |
|---|---|---|
| Control Probes | Assay qualification | PPIB, POLR2A, UBC (positive); dapB (negative) [38] [39] |
| Microscope Slides | Tissue adhesion | Fisherbrand Superfrost Plus slides [38] [39] |
| Hydrophobic Barrier Pen | Reagent containment | ImmEdge Hydrophobic Barrier Pen (Vector Laboratories) [38] |
| Mounting Media | Slide preservation | Cytoseal or other xylene-based mounting medium for Brown assay [39] |
| Automation Platform | Standardized processing | Ventana DISCOVERY ULTRA with RNAscope-specific protocol [6] |
| Image Analysis Software | Signal quantification | HALO, QuPath, or Aperio algorithms [40] [37] |
Advanced Interpretation and Normalization Strategies
Quantitative Analysis of Control Probes
While the semi-quantitative scoring system provides a rapid assessment tool, advanced image analysis enables more precise quantification:
- Digital Dot Counting: Use HALO or QuPath software to perform automated dot counting per cell [40] [41]. This approach is particularly valuable for establishing baseline expression levels of control probes in different tissue types.
- Threshold Establishment: Derive mRNA signal thresholds using negative control samples to establish statistically valid cutoffs for positive signal detection [40].
- Spatial Analysis: Assess control probe distribution patterns across different tissue regions to identify potential spatial biases in staining [41].
Normalization Approaches
For rigorous quantification, implement these normalization strategies:
- Housekeeper Ratio: Express target signal as a ratio of PPIB or POLR2A signal to control for variations in RNA quality between samples.
- Background Subtraction: Subtract dapB signal from target signal to account for non-specific background [41].
- Cell-type Specific Normalization: For heterogeneous tissues, normalize target expression to control probe expression within specific cell populations [41].
The proper implementation of these control probe interpretation strategies on the Ventana DISCOVERY platform ensures generation of reliable, reproducible RNAscope data essential for confident decision-making in drug development and biomarker research.
The successful application of the automated RNAscope in situ hybridization (ISH) assay on the Ventana DISCOVERY platform hinges on optimal tissue pretreatment. This step is critical for achieving the delicate balance between exposing target RNA and preserving tissue morphology and RNA integrity. While standard protocols exist for ideally prepared tissues, researchers frequently encounter suboptimal samples in drug development and biomedical research. This Application Note provides detailed guidance and validated experimental protocols for optimizing protease and retrieval conditions to ensure reliable, high-quality RNA detection in such challenging samples within the context of an automated RNAscope workflow on Ventana DISCOVERY systems.
The Critical Role of Pretreatment in Automated RNAscope
The RNAscope assay is a sophisticated RNA ISH technology that enables single-molecule detection of RNA with single-cell resolution within an intact morphological context [6]. Its proprietary double Z (ZZ) probe design and signal amplification system allow target RNAs to be visualized as distinct, punctate dots, where each dot represents an individual RNA molecule [6] [41].
In the automated workflow on the Ventana DISCOVERY system, pretreatment serves two essential functions:
- Breaking protein cross-links formed during formalin fixation to make target RNA accessible to probes.
- Controlling background staining by managing tissue permeability without destroying the target RNA or compromising tissue architecture.
For tissues fixed under suboptimal conditions (e.g., over-fixed, under-fixed, or with alternative fixatives), the standard pretreatment conditions often require adjustment to maximize signal-to-noise ratio. As emphasized by experts, "determining the optimal RNAscope pretreatment protocol for each tissue type you have" is fundamental, and "once this is established, it's not going to change" [42].
Establishing Baselines and Assessing Tissue Quality
Before optimizing for suboptimal tissues, it is crucial to establish baseline performance with properly controlled tissues and confirm that the standard RNAscope protocol is functioning correctly.
Standard Protocol for Reference
The standard automated RNAscope pretreatment protocol on the Ventana DISCOVERY ULTRA platform for well-fixed FFPE tissues typically includes [6]:
- Baking: 32 minutes at 37°C (on-instrument)
- Deparaffinization: Standard protocols on the DISCOVERY system
- Target Retrieval: 16-24 minutes at 97°C using appropriate retrieval buffer
- Protease Treatment: 16 minutes at 37°C with RNAscope Protease
Essential Control Probes
Always implement a systematic approach using control probes to validate your results [12]:
- Positive Control: Housekeeping genes (PPIB, POLR2A, or UBC) confirm RNA integrity and assay performance. Successful staining should achieve a PPIB/POLR2A score ≥2 or UBC score ≥3 [12].
- Negative Control: Bacterial DapB gene assesses non-specific background staining. A score of <1 is acceptable [12].
- Interpretation: Score the number of dots per cell rather than signal intensity, as dots correlate directly to RNA copy numbers [12] [41].
Table 1: RNAscope Semi-Quantitative Scoring Guidelines for Dot Counting
| Score | Description | Dots per Cell |
|---|---|---|
| 0 | No staining or minimal | <1 dot per 10 cells |
| 1 | Low expression | 1-3 dots per cell |
| 2 | Moderate expression | 4-10 dots per cell, very few clusters |
| 3 | High expression | >10 dots per cell, <10% cells with clusters |
| 4 | Very high expression | >10 dots per cell, >10% cells with clusters |
Optimization Strategies for Suboptimal Tissues
When standard pretreatment conditions yield suboptimal results with challenging tissues, systematic optimization of protease and retrieval conditions is necessary.
Protease Optimization
Protease treatment is perhaps the most critical variable for suboptimal tissues, as it directly controls tissue permeability and accessibility of target RNA.
Common Scenarios and Solutions:
- Over-fixed Tissues: Extended formalin fixation creates excessive cross-linking, requiring more aggressive protease treatment. Bradley Spencer-Dene's team found it "necessary to dilute the RNAscope pretreatment 3 (protease plus) so you might also need to consider optimizing this protease digestion step" [42].
- Under-fixed Tissues: Inadequate fixation may make tissues more susceptible to protease damage, requiring reduced protease concentration or duration.
- Alternative Fixatives: Tissues fixed with non-formalin fixatives often require completely customized protease conditions.
Experimental Protocol: Protease Titration
- Prepare a dilution series of the RNAscope Protease (e.g., 1:2, 1:1, 2:1 dilution factor relative to standard concentration).
- Maintain constant incubation parameters (16 minutes at 37°C) initially, varying only concentration.
- If further optimization is needed, maintain the optimal concentration and vary incubation time (±5 minutes).
- Evaluate results based on positive control signal intensity and tissue morphology preservation.
Target Retrieval Optimization
Target retrieval reverses formaldehyde cross-links and is particularly important for RNA that is difficult to access.
Key Optimization Variables:
- Duration: Standard is 16-24 minutes at 97°C [6]. For over-fixed tissues, extend duration incrementally by 5-8 minutes. For under-fixed tissues, reduce duration.
- Temperature: While typically fixed at 97°C, some challenging targets may benefit from slight temperature adjustments if the platform allows.
- Buffer Composition: Different retrieval buffers (e.g., citrate-based, EDTA-based) may be tested for specific challenges.
Experimental Protocol: Retrieval Optimization
- Establish a duration gradient (e.g., 12, 16, 20, 24 minutes) at constant temperature (97°C).
- Process all slides with identical subsequent steps and protease conditions.
- Evaluate based on signal intensity of positive control and tissue morphology.
Table 2: Troubleshooting Guide for Suboptimal Tissues
| Tissue Issue | Protease Adjustment | Retrieval Adjustment | Expected Outcome |
|---|---|---|---|
| Over-fixed (>48 hours) | Increase concentration 1.5-2x or extend time | Extend time by 5-8 minutes | Improved target accessibility |
| Under-fixed (<12 hours) | Decrease concentration 0.5-0.75x | Reduce time by 3-5 minutes | Better morphology preservation |
| Decalcified Tissue | Increase concentration 2-2.5x; test protease plus | Extend time by 8-12 minutes | Restoration of RNA detection |
| Old Archives (>5 years) | Increase concentration 1.2-1.5x | Standard or slight extension | Enhanced signal recovery |
| Alternative Fixatives | Require empirical determination; start with 0.5x and titrate | Test multiple durations | Customized optimal conditions |
Integrated Workflow for Method Development
Developing an optimized pretreatment protocol requires a systematic approach. The following diagram illustrates the decision-making workflow for establishing the optimal conditions.
The Scientist's Toolkit: Essential Research Reagent Solutions
Successful optimization requires specific reagents and controls. The following table details essential materials for pretreatment optimization on the Ventana DISCOVERY platform.
Table 3: Research Reagent Solutions for RNAscope Pretreatment Optimization
| Reagent/Category | Function in Optimization | Application Notes |
|---|---|---|
| RNAscope Protease Reagents | Enzymatically digests proteins obscuring RNA targets | Critical titration variable; multiple concentrations often required [42] |
| Target Retrieval Reagents | Breaks protein cross-links from formalin fixation | Duration and temperature optimization needed for suboptimal tissues [6] |
| Control Probes (PPIB, POLR2A) | Assess RNA integrity and assay performance | Must yield score ≥2 for valid results [12] |
| Negative Control Probe (DapB) | Measures non-specific background | Must yield score <1 for acceptable background [12] |
| RNAscope VS Red & Fluorescent Kits | Signal detection systems | Compatible with subsequent IHC; Fast Red is naturally fluorescent [42] |
| DISCOVERY Reagent Solutions | Platform-specific compatibility | Ventana DISCOVERY system reagents ensure automated workflow integrity [23] |
Case Studies and Expert Insights
Dual ISH-IHC Applications
Kristie Wetzel at Novartis developed a dual ISH-IHC protocol for identifying cells producing secreted proteins. Her team faced significant challenges with protein stability affected by RNAscope pretreatments. "It took us a number of weeks and a number of iterations before we really got the protocol optimized," highlighting that even with individual optimized protocols, "you're still going to need to tweak and optimize once you combine them" [42].
Challenging Antibody Targets
Bradley Spencer-Dene's work revealed that "there are some possible issues with antibodies that normally require trypsin digestion, e.g., F4/80. We found that this antibody didn't work so you may need to include a trypsin digestion step after ISH at start of IHC protocol" [42]. This demonstrates that some targets require additional customization beyond standard protease treatment.
Subcellular Localization
For investigators studying RNA in specific subcellular compartments, pretreatment optimization is particularly crucial. As noted in analysis guidelines, "data analysis can be challenging due to 2D representation of a 3 dimensional structure," making proper tissue preservation through careful pretreatment essential for accurate interpretation [41].
Optimizing pretreatment conditions for suboptimal tissues in automated RNAscope on the Ventana DISCOVERY platform requires a systematic, iterative approach centered on titrating protease and retrieval parameters. By establishing robust baseline performance with control probes, methodically adjusting key variables, and validating against both signal quality and morphological preservation, researchers can overcome the challenges posed by variably fixed tissue specimens. The protocols and guidelines presented here provide a roadmap for developing reliable, reproducible RNA detection methods that maintain the sophisticated single-molecule sensitivity of the RNAscope technology while accommodating real-world tissue heterogeneity.
For researchers utilizing automated RNAscope in situ hybridization (ISH) on the Ventana DISCOVERY ULTRA platform, rigorous instrument maintenance is not ancillary—it is a fundamental prerequisite for assay success. Automated RNAscope represents a significant advancement over traditional RNA ISH, enabling single-molecule RNA visualization within intact cells while preserving tissue morphology [43]. However, the sensitivity of this signal amplification and background suppression technology means that even minor deviations in fluidic path cleanliness or buffer composition can compromise results. This application note provides detailed protocols for buffer management and systematic decontamination, framed within the broader thesis that proactive maintenance is integral to achieving the precision, reproducibility, and sensitivity required in therapeutic development workflows.
Critical Maintenance Protocols
Regular maintenance of the Ventana DISCOVERY ULTRA system is essential to prevent microbial contamination and ensure reagent integrity, which directly impacts the performance of sensitive RNAscope assays.
Buffer Management System
Proper buffer management is crucial for maintaining the analytical integrity of RNAscope assays. The following protocols must be meticulously followed:
- Buffer Specification: Use only DISCOVERY 1X SSC Buffer for RNAscope assays. This buffer must be diluted 1:10 prior to adding it to the optional bulk buffer container [38].
- Buffer Prohibition: Do not use the Benchmark 10X SSC Buffer with RNAscope assays, as it is not compatible and may lead to suboptimal results or assay failure [38].
- RiboWash Buffer: The RiboWash Buffer must be diluted 1:10 in the RiboWash bulk container only [38].
- Bulk Solution Replacement: All bulk solutions should be replaced with the recommended buffers before running the RNAscope assay. The internal reservoir must be purged several times with the appropriate buffer to ensure complete replacement [38].
Systematic Decontamination
Preventing microbial contamination in the instrument's fluidic lines is critical for maintaining assay performance and instrument reliability:
- Decontamination Frequency: A comprehensive decontamination protocol must be performed every three months to prevent microbial growth in the fluidic lines [38].
- Qualified Service: This decontamination must be performed by a Ventana/Roche Diagnostics representative, ensuring the procedure is conducted with the appropriate protocols and solutions [38].
- Post-Cleaning Protocol: If water is used to clean the instrument, ensure all residual water is replaced with the appropriate buffers by purging the system several times [38].
Table: Essential Maintenance Schedule for Ventana DISCOVERY ULTRA with RNAscope Assays
| Maintenance Activity | Frequency | Key Specifications | Responsibility |
|---|---|---|---|
| Buffer System Replacement | Before RNAscope runs | Use DISCOVERY 1X SSC Buffer (diluted 1:10); Avoid Benchmark 10X SSC Buffer | Researcher/Lab Technician |
| Fluidic Line Decontamination | Every 3 months | Full system decontamination to prevent microbial growth | Ventana/Roche Representative |
| Post-Cleaning Buffer Purging | After any water cleaning | Purge internal reservoir several times with appropriate buffers | Researcher/Lab Technician |
Instrument Maintenance Workflow
The following diagram illustrates the integrated maintenance workflow for the Ventana DISCOVERY ULTRA system when running RNAscope assays, emphasizing the relationship between different maintenance activities and their frequency.
Maintenance Workflow for RNAscope Assays
Research Reagent Solutions
The following table details essential materials and reagents required for proper maintenance and operation of the Ventana DISCOVERY ULTRA system for RNAscope assays.
Table: Essential Research Reagent Solutions for Ventana DISCOVERY ULTRA Maintenance
| Reagent/Buffer | Specification | Function in RNAscope Assay |
|---|---|---|
| DISCOVERY 1X SSC Buffer | Must be diluted 1:10 before use | Primary wash buffer for removing unbound probes and reagents during ISH procedure [38] |
| RiboWash Buffer | Must be diluted 1:10 in dedicated bulk container | Specialized wash solution optimized for RNAscope assay steps [38] |
| Decontamination Solutions | Proprietary solutions applied by Ventana representative | Eliminates microbial contamination in fluidic lines to prevent assay interference [38] |
| Hydrophobic Barrier Pen | ImmEdge Pen (Vector Laboratories) only | Maintains hydrophobic barrier throughout procedure to prevent tissue drying [38] |
Experimental Maintenance Protocol
This detailed methodology provides a step-by-step approach to maintaining the Ventana DISCOVERY ULTRA system for optimal RNAscope performance.
Pre-Run Buffer Management Protocol
- Buffer Preparation: Dilute DISCOVERY 1X SSC Buffer 1:10 with nuclease-free water in a clean container [38].
- Bulk Container Cleaning: Rinse the bulk solution containers thoroughly with nuclease-free water.
- System Purging: Replace all bulk solutions with recommended buffers and purge the internal reservoir several times with the appropriate buffer to ensure complete replacement of previous solutions [38].
- Buffer Prohibition Check: Confirm that Benchmark 10X SSC Buffer is not present in the system or available for use with RNAscope assays [38].
Quarterly Decontamination Protocol
- Schedule Planning: Plan decontamination service every three months, coordinating with your Ventana/Roche Diagnostics representative [38].
- Pre-Service Preparation: Run appropriate cleaning cycles as recommended in the user manual prior to the service visit.
- Professional Decontamination: The Ventana representative will perform the complete decontamination protocol using specialized solutions to address microbial growth in fluidic lines [38].
- Post-Decontamination Validation: After service, run control RNAscope assays using positive and negative control probes (e.g., PPIB and dapB) to verify system performance [38].
Software Configuration for RNAscope
- Slide Cleaning Setting: Deactivate the "Slide Cleaning" option in the instrument software [38].
- Temperature Verification: Confirm that hybridization temperatures are set according to the RNAscope user manual specifications without adjustment [38].
- Version-Specific Settings: For software version 2.0, note that fully automated settings are applicable only for brain and spinal cord samples [38].
Maintaining the Ventana DISCOVERY ULTRA system with disciplined buffer management and regular decontamination is not merely operational but foundational to research integrity. For scientists advancing RNA therapeutics and biomarker discovery, these protocols ensure that the sophisticated sensitivity of RNAscope technology delivers on its promise of single-molecule detection within precious clinical and preclinical samples. In the context of automated RNAscope research, such rigorous instrument stewardship transforms maintenance from routine procedure to critical scientific differentiator, enabling reliable detection of everything from splice variants to oligonucleotide therapeutics with the spatial context essential for mechanistic understanding.
Within the context of automated RNAscope on the Ventana DISCOVERY research platform, achieving precise image analysis is paramount for accurate biomarker research and diagnostic assay development. The power of this technology lies in its ability to provide single-cell RNA expression resolution within its morphological context [6]. However, this precision is critically dependent on optimal assay performance. Inconsistent staining, tissue artifacts, and signal heterogeneity can introduce significant analytical pitfalls, potentially compromising data integrity and reproducibility. This application note provides detailed methodologies to identify, troubleshoot, and mitigate these common challenges, ensuring reliable and quantifiable results for researchers and drug development professionals. The following guide leverages the latest technical protocols and troubleshooting recommendations to support robust experimental outcomes.
Common Image Analysis Pitfalls and Their Origins
Successful interpretation of RNAscope data requires an understanding of potential artifacts. The semi-quantitative nature of the assay means that analytical errors can directly lead to erroneous biological conclusions. The primary pitfalls stem from sample preparation, staining procedures, and signal interpretation.
Saturated and Over-Stained Signals
Saturation occurs when the signal intensity exceeds the detectable dynamic range of the imaging system, resulting in a loss of quantitative information. This often manifests as large, coalesced dots where individual RNA transcripts can no longer be distinguished and counted.
- Causes: The primary cause is excessive protease treatment, which over-exposes target RNA, leading to unnaturally high signal density [38] [44]. Alternatively, overly extended chromogenic development times can cause dots to enlarge and merge. On automated Ventana systems, deviations from the recommended protease or target retrieval times for a specific tissue type are a frequent culprit.
- Impact on Analysis: Signal saturation prevents accurate transcript quantification. A score of "4" requires distinguishable dots and clusters; saturation obscures this, making it impossible to determine whether a score of 3 or 4 is appropriate, thereby skewing gene expression calculations.
Tissue and Sample Preparation Artifacts
The integrity of the tissue sample and its preparation is the foundation of a successful RNAscope assay. Artifacts introduced at these stages can render even a perfectly executed hybridization unreadable.
- Tissue Detachment: This occurs when the tissue section lifts off the slide during the rigorous assay procedure. The use of non-recommended slides, such as those other than Superfrost Plus, is a common cause [38]. An inadequate hydrophobic barrier from using a barrier pen other than the ImmEdge Pen can also lead to drying and tissue loss [38] [44].
- Morphological Damage: Over-fixation (exceeding 32 hours in 10% NBF) can make tissues brittle and compromise RNA integrity, while under-fixation fails to preserve morphology and RNA [38]. Over-digestion with protease can destroy tissue architecture, creating holes and degrading the cellular context necessary for analysis.
- High Background Noise: Non-specific, diffuse staining that obscures the specific punctate signal can be caused by under-fixation, insufficient protease digestion (preventing probe access), or the use of old or degraded reagents [44].
Heterogeneous and Suboptimal Signals
A more subtle class of pitfalls involves inconsistent staining across the tissue, leading to challenges in scoring and interpretation.
- Signal Heterogeneity: True biological variation must be distinguished from technical artifacts. Uneven staining can result from inconsistent protease activity across the tissue, incomplete reagent coverage during manual pipetting, or uneven heating during hybridization or retrieval steps [38].
- Weak or Absent Signal: A lack of expected signal can stem from RNA degradation in poorly preserved samples, inadequate protease treatment (hiding the target RNA), or under-retrieval [44]. Incorrect probe handling, such as failure to warm precipitated probes to 40°C before use, can also lead to failure [38].
- False Positives: Signal from the negative control probe (e.g., dapB) indicates non-specific binding. This is often due to over-retrieval or over-digestion, which creates sticky sites for nonspecific probe binding [6].
Detailed Protocols for Mitigation and Optimization
A systematic approach to troubleshooting is essential. The following protocols provide a step-by-step guide to diagnosing and resolving the pitfalls described above, with a focus on the Ventana DISCOVERY platform.
Protocol: Systematic Troubleshooting of Staining Issues
This workflow is designed to methodically identify the root cause of common staining problems.
Diagram Title: Root Cause Analysis for RNAscope Staining Issues
Procedure:
- Initiate with Controls: Always begin troubleshooting by running the assay with positive control probes (e.g., PPIB or UBC) and the negative control probe (dapB) on the problematic tissue [38] [44].
- Evaluate Control Results:
- Scenario A (Poor PPIB/UBC, Good dapB): If the positive control staining is weak (PPIB score <2, UBC score <3) and the negative control is clean (dapB score <1), the issue likely lies with sample or RNA quality. Proceed to "Optimize Pretreatment" (Protocol 3.2).
- Scenario B (Good PPIB/UBC, Poor dapB): If the positive control is strong but the negative control shows high background (dapB score >1), the problem is excessive background. This is primarily a pretreatment issue. Proceed to "Optimize Pretreatment" (Protocol 3.2).
- Scenario C (Good Controls, Poor Target): If control stains are optimal but the target probe signal is weak, the issue may be specific to the target probe or detection reagents. Check that the probe was warmed to 40°C to dissolve precipitates and verify the expiration dates and correct order of all detection reagents [38] [44].
- Implement Corrections: Based on the diagnosis, follow the targeted optimization protocols below.
- Verify Resolution: Repeat the assay with the optimized conditions and re-evaluate the control probes to confirm the issue is resolved.
Protocol: Optimizing Pretreatment for Automated Platforms
Pretreatment (target retrieval and protease digestion) is the most critical step to balance signal and background. The following table summarizes optimization parameters for the Ventana DISCOVERY ULTRA system.
Table 1: Pretreatment Optimization Guide for Ventana DISCOVERY ULTRA
| Tissue Condition | Target Retrieval (VS Universal Target Retrieval v2) | Protease (Protease 3 or VS Protease) | Expected Outcome |
|---|---|---|---|
| Standard (24h NBF) | 16-24 min at 97°C [6] | 16 min at 37°C [6] | Strong signal, low background |
| Over-fixed (>32h NBF) | Increase time in 5 min increments [38] | Increase time in 10 min increments [38] | Restores signal accessibility |
| Under-fixed | Decrease time slightly | Decrease time slightly | Reduces background & morphology damage |
| High Background | Maintain or slightly decrease time | Decrease time (primary adjustment) [44] | Suppresses non-specific signal |
| Weak/No Signal | Increase time (primary adjustment) [44] | Increase time (primary adjustment) [44] | Enhances target exposure |
Procedure:
- Baseline Assessment: Start with the standard pretreatment conditions for your tissue type as listed in the user manual.
- Diagnose: Use the control probes to determine if the issue is weak signal (increase retrieval/protease) or high background (decrease protease).
- Iterate Systematically: Adjust only one parameter at a time (e.g., increase protease by 10 minutes) while keeping other variables constant. Use a test slide with known positive and negative controls to evaluate the effect.
- Validate: Once optimal conditions are found, run the target probe to confirm performance. Document the final validated protocol for consistent future use.
Protocol: Quantitative Signal Scoring and Interpretation
Accurate scoring is the final, critical step in analysis. The RNAscope assay is scored based on the number of distinct punctate dots per cell, not the intensity of the stain [38] [44] [6].
Table 2: RNAscope Semi-Quantitative Scoring Guidelines [38] [44]
| Score | Criteria (Dots Per Cell) | Interpretation |
|---|---|---|
| 0 | No staining or <1 dot/10 cells | Negative |
| 1 | 1-3 dots/cell | Low expression |
| 2 | 4-9 dots/cell; few/no clusters | Moderate expression |
| 3 | 10-15 dots/cell; <10% in clusters | High expression |
| 4 | >15 dots/cell; >10% in clusters | Very high expression |
Scoring Procedure:
- Microscope Setup: Perform scoring at 20x-40x magnification.
- Field Selection: Systematically scan the entire tissue section. Avoid edges or obvious damaged areas unless they are the region of interest.
- Dot Counting: For each representative field of view, count the number of dots in at least 10-50 cells. Do not count clustered dots as individual transcripts; a cluster is scored as a single transcription site.
- Assign a Score: Calculate the average dots per cell for the sample and assign the corresponding score based on Table 2. The sample is considered qualified for analysis if the positive control (PPIB) scores ≥2 and the negative control (dapB) scores <1 [44].
The Scientist's Toolkit: Essential Research Reagent Solutions
The following reagents and materials are critical for the successful execution of the automated RNAscope assay on the Ventana DISCOVERY system.
Table 3: Essential Research Reagent Solutions for Automated RNAscope
| Item | Function/Description | Example/Catalog Recommendation |
|---|---|---|
| Positive Control Probes | Verify sample RNA integrity & assay performance. PPIB (low-copy) and UBC (high-copy) are standards. | Hs-PPIB (Cat. #313909), Hs-UBC [44] [6] |
| Negative Control Probe | Assess background and non-specific binding. Should yield minimal signal. | dapB (Cat. #312039) [44] [6] |
| Superfrost Plus Slides | Provide superior tissue adhesion to prevent detachment during stringent assay steps. | Fisher Scientific [38] [44] |
| ImmEdge Hydrophobic Barrier Pen | Creates a resilient barrier to contain reagents and prevent tissue drying. | Vector Laboratories (Cat. # H-4000) [38] |
| Ventana DISCOVERY Wash Buffer | Used for slide washing. Must be the correct formulation for RNAscope. | DISCOVERY 1X SSC Buffer, diluted 1:10 [38] |
| Assay-Specific Mounting Medium | Preserves staining and enables visualization. Critical for image clarity and longevity. | Xylene-based for Brown assay; EcoMount/PERTEX for Red assay [38] [44] |
Navigating the pitfalls of image analysis in automated RNAscope requires a disciplined, systematic approach centered on rigorous validation and controlled optimization. By adhering to the detailed protocols for troubleshooting, pretreatment optimization, and quantitative scoring outlined in this document, researchers can significantly enhance the reliability and reproducibility of their gene expression data. The consistent use of recommended controls and reagents is not optional but foundational to generating high-quality, publication-ready results that robustly support biomarker discovery and diagnostic development on the Ventana DISCOVERY platform.
Establishing Confidence: Validation, Comparative Analysis, and Clinical Diagnostic Potential
The integration of automated RNAscope in situ hybridization (ISH) on the Ventana DISCOVERY platform represents a significant advancement for gene expression analysis in clinical and research settings. This technological synergy offers the potential for highly sensitive and localized detection of mRNA molecules within intact tissue architectures [45] [46]. However, the translation of this potential into clinically reliable data requires a rigorous validation framework based on the Clinical Laboratory Improvement Amendments (CLIA) guidelines. CLIA regulations establish quality standards for laboratory testing performed on human specimens to ensure accurate, reliable, and reproducible results for diagnosis, prevention, or treatment of disease [47] [48]. For researchers and drug development professionals, adherence to these guidelines is not merely a regulatory hurdle but a fundamental component of robust assay development, ensuring that data generated for biomarker discovery and therapeutic development is of the highest quality and integrity.
The CLIA framework encompasses the total testing process (TTP), which extends beyond the assay itself to include pre-analytic, analytic, and post-analytic phases [48]. This end-to-end approach is crucial because errors can occur at any stage, from specimen collection to result reporting. For RNAscope assays, which are often employed to identify patient populations for clinical trials or to validate pharmacodynamic biomarkers, a CLIA-compliant validation provides the necessary foundation for scientific and regulatory confidence [49].
Core CLIA Validation Parameters for RNAscope Assays
CLIA guidelines mandate the verification of several key analytical performance characteristics. For a quantitative or semi-quantitative RNAscope assay with digital image analysis, the following parameters must be systematically established.
Specificity
Specificity confirms that the assay detects the intended mRNA target without cross-reacting with similar sequences. A successful validation, as demonstrated for a DKK1 RNAscope assay, must show that signal is localized to tumor cells with minimal to no detection in non-target cells [49]. Furthermore, probe design is critical; proprietary algorithms enable the design of probes targeting unique sequence regions, which minimizes the risk of cross-reactivity with other family members, such as DKK2, DKK3, and DKK4 [49].
Sensitivity
Sensitivity defines the lowest level of target mRNA that can be reliably detected. The RNAscope technology is exquisitely sensitive, capable of detecting a single RNA molecule per cell, with each dot representing an individual mRNA molecule [49]. During validation, sensitivity is confirmed by detecting a dynamic range of expression, including cells with only a single dot [49]. The lower limit of detection (LOD) should be empirically determined.
Accuracy
Accuracy measures the agreement between the assay result and a reference method or known truth. This can be demonstrated through multiple approaches:
- Correlation with orthogonal methods: A strong correlation (e.g., Spearman’s rho = 0.86) between RNAscope results and RNA-Seq data from the same cell lines provides powerful evidence of accuracy [49].
- Comparison with IHC: While IHC detects protein, consistency between mRNA and protein expression patterns supports accuracy, though discrepancies can occur due to post-transcriptional regulation [49].
- Use of control cell lines: Staining of cell lines with known expression levels (high, medium, low, and negative) confirms the assay detects the expected expression range [49].
Precision
Precision, or reliability, assesses the agreement between repeated measurements of the same sample. CLIA guidelines, detailed in documents like EP05, provide standardized experimental designs for evaluating precision [50]. For a single-site validation, a common approach is a 20-day precision experiment incorporating two runs per day and two replicates per run to capture within-run, between-run, and between-day variability [50]. The use of a digital image analysis (DIA) algorithm for quantification significantly improves precision by removing pathologist bias and providing reproducible counts of mRNA dots per cell [49].
Table 1: Key Analytical Performance Parameters for CLIA-Compliant RNAscope Assay Validation
| Performance Parameter | Definition | Validation Approach for RNAscope | Acceptance Criterion Example |
|---|---|---|---|
| Specificity | Ability to detect the intended target without cross-reactivity. | Probe design to unique sequences; stain cell lines expressing related genes; assess signal localization in tissue. | No staining in negative control cell lines; specific cellular staining pattern [49]. |
| Sensitivity | Lowest amount of target mRNA that can be reliably detected. | Determine Lower Limit of Detection (LOD) using low-expressing cell lines; confirm detection of single RNA molecules. | Detection of cells with a single dot; LOD established with a specific cell line [49]. |
| Accuracy | Agreement with a reference method or known value. | Correlation with RNA-Seq or qPCR data; comparison with IHC on serial sections; use of well-characterized cell pellets. | Spearman's correlation coefficient >0.8 with RNA-Seq data [49]. |
| Precision | Agreement between repeated measurements. | 20-day precision study with multiple runs and replicates; use of digital image analysis for quantification. | Coefficient of variation (CV) for H-score < a pre-defined limit (e.g., 15%) [50] [49]. |
The Validation Workflow: A Phase-Based Approach
A CLIA-compliant validation follows the entire testing pathway. The diagram below outlines the core workflow for validating and running an automated RNAscope assay.
Pre-Analytic Phase
The pre-analytic phase is the most vulnerable part of the testing process and requires strict control [48]. For RNAscope, this begins with proper specimen collection and fixation. Adherence to a standardized protocol is critical; for example, fixing tissue for 72 hours at room temperature in 10% Neutral Buffered Formalin (NBF) is a recommended practice to preserve RNA integrity [42]. Tissues are then processed into Formalin-Fixed Paraffin-Embedded (FFPE) blocks [45] [49]. Laboratories must have written procedures for specimen acceptance and rejection, monitoring indicators such as RNA integrity, which can be assessed with a control probe for a ubiquitously expressed gene like Peptidylprolyl Isomerase B (PPIB), and background signal, assessed with a negative control probe targeting a bacterial gene like Dihydrodipicolinate Reductase (dapB) [49].
Analytic Phase
The analytic phase covers the testing process itself. For automated RNAscope, this involves running the optimized assay on the Ventana DISCOVERY Ultra platform, which provides significant advantages for repeatability, reduction in variability, and flexibility of workflows [45] [46]. Following the RNAscope protocol to the letter is strongly recommended for success [42]. The analytic phase also includes digital image analysis to quantify mRNA expression, for example, by generating an H-score [49]. Finally, a pathologist reviews the DIA output for final acceptance, ensuring the result is biologically and technically sound [49].
Post-Analytic Phase
In the post-analytic phase, results are reported and stored. CLIA requires that laboratories have a written quality assurance program and accurately transmit test results [48]. This includes procedures for reporting critical values and protecting patient data per HIPAA regulations. Furthermore, proficiency testing is mandated, where laboratories analyze external samples to verify their testing accuracy [47] [48]. For novel assays, if commercial proficiency panels are unavailable, laboratories should work with providers to create suitable reagents [51].
Experimental Protocol: A Template for Validation
The following protocol is adapted from a validated DKK1 RNAscope CISH assay for gastric cancer and can be tailored for other targets on the Ventana DISCOVERY platform [49].
Initial Assay Setup and Optimization
- Control Selection: Identify and procure control cell lines with a range of target expression (high, medium, low, negative) using databases like the Cancer Cell Line Encyclopedia (CCLE). Generate a Formalin-Fixed Paraffin-Embedded Cell Pellet Array (CPA) from these lines [49].
- Probe Design: Submit at least 300 base pairs of unique target gene sequence to a proprietary probe design provider (e.g., ACD) for RNAscope probe synthesis [52].
- Tissue Optimization: Determine the optimal pretreatment conditions (especially protease digestion time) for each primary tissue type under investigation [42].
- Orthogonal Confirmation: Verify the expression levels in control cell lines using an orthogonal method, such as qPCR or RNA-Seq, to establish a ground truth for accuracy assessments [49].
The Precision Evaluation Experiment
CLIA guidelines recommend a rigorous precision study. The standard design for a single-site validation is summarized below.
Table 2: Experimental Design for a 20-Day Precision Study as per CLSI EP05 Guidelines [50]
| Factor | Specification | Rationale |
|---|---|---|
| Total Duration | 20 separate days | Captures day-to-day and environmental variability. |
| Runs per Day | 2 independent runs | Captures between-run variability. |
| Replicates per Run | 2 replicates | Captures within-run variability. |
| Sample Types | At least 2: one with high expression and one with low/normal expression. | Characterizes precision across the assay's measuring interval. |
| Reagent Lot | A single lot for the entire study. | Isolates precision from lot-to-lot variation. |
| Personnel | May involve multiple trained operators. | Reflects real-world laboratory conditions. |
| Data Analysis | Variance Component Analysis (VCA) to quantify sources of variation and calculate total CV. | Provides a verifiable and detailed precision profile. |
Digital Image Analysis Algorithm Validation
If a DIA algorithm is used for quantification, it must be validated alongside the assay.
- Algorithm Training: Train the algorithm to identify tumor cells and quantify the target mRNA signal (e.g., dots per cell).
- Correlation with Manual Scoring: Compare the digital H-scores with manual pathologist scores from a set of training samples to ensure strong correlation and accuracy [49].
- Precision: Demonstrate that the DIA algorithm produces reproducible results when the same slide is re-analyzed.
The Scientist's Toolkit: Essential Research Reagent Solutions
The successful implementation of a validated RNAscope assay relies on a set of key reagents and controls.
Table 3: Essential Research Reagent Solutions for RNAscope Validation
| Reagent / Solution | Function | Example & Notes |
|---|---|---|
| Target Probe | Detects the specific mRNA of interest. | Custom-designed probes (e.g., RNAscope DKK1 probe); designed to a unique 300 bp sequence [52] [49]. |
| Positive Control Probe | Verifies sample RNA integrity. | Probe for a housekeeping gene like PPIB; adequate signal (e.g., ≥ 4 dots/cell) indicates acceptable RNA quality [49]. |
| Negative Control Probe | Assesses background/non-specific staining. | Probe for a bacterial gene like dapB; should show no detectable signal, confirming assay specificity [49]. |
| Control Cell Pellet Array (CPA) | Serves as a built-in control for assay performance. | FFPE block containing cell lines with known expression levels; run with each batch to monitor sensitivity and specificity [49]. |
| Ventana DISCOVERY Reagents | Enables automated staining. | Proprietary buffers, amplification reagents, and detection kits (e.g., RNAscope VS Red) optimized for the platform [45] [42]. |
| Digital Image Analysis Software | Provides objective, quantitative readout. | Software (e.g., QuPath) and custom algorithms to identify tumor cells and count mRNA dots, generating an H-score [49]. |
Implementing a CLIA-compliant validation framework for automated RNAscope assays is a critical investment in data quality and reliability. By systematically addressing specificity, sensitivity, accuracy, and precision across the pre-analytic, analytic, and post-analytic phases, researchers and drug developers can build a robust foundation for their scientific inquiries. The automation offered by the Ventana DISCOVERY platform, coupled with the quantitative power of digital image analysis, creates a powerful tool for biomarker discovery and validation. Adherence to this rigorous framework ensures that results are not only scientifically valid but also maintain the quality standards required for informing clinical development decisions.
Immunohistochemistry (IHC) remains one of the most widely used techniques for detecting protein expression in situ, playing a crucial role in both research and clinical diagnostics. However, significant challenges regarding antibody specificity and reproducibility have been widely documented in the scientific literature. A publication in Science highlighted the inherent problems and pitfalls associated with antibody use, revealing that these reagents often lack proper validation [53]. This validation deficit forces researchers to perform their own independent antibody testing, consuming substantial time, financial resources, and precious samples, often while yielding questionable results [53]. The core issue stems from a fundamental limitation: IHC detects antibody binding but cannot independently verify that this binding corresponds to the intended target protein.
The emergence of RNA in situ hybridization (ISH) technologies, particularly the RNAscope platform, provides a powerful orthogonal method for validating antibody-based protein detection. This Application Note explores the comparative performance of RNAscope and IHC, with a specific focus on resolving antibody discrepancies and validating protein expression within the context of automated workflows on the Ventana DISCOVERY research platform. By employing a method that detects mRNA expression—an entirely different biomolecule than the target protein—researchers can independently verify whether the spatial and quantitative expression patterns observed with IHC genuinely reflect target gene activity.
Comparative Analysis: RNAscope vs. IHC for Diagnostic and Research Biomarkers
Analytical Performance in Detecting Urothelial Carcinoma Markers
A rigorous 2022 study directly compared RNAscope and IHC for detecting UPK2, a specific marker for urothelial carcinoma (UC), in 219 patient samples [54]. The results provide valuable insights into the relative performance of these two methodologies.
Table 1: Comparison of UPK2 Detection by RNAscope vs. IHC in Urothelial Carcinoma
| UC Tissue Type | Number of Cases | POSitivity Rate (IHC) | POSitivity Rate (RNAscope) | P-value | Correlation (R value) |
|---|---|---|---|---|---|
| All UC Tissues | 219 | 62.6% | 68.0% | P = 0.141 | R = 0.441 |
| Conventional Bladder UC | 127 | 68.5% | 72.4% | P = 0.511 | Not specified |
| Variant Bladder UC | 45 | 35.6% | 53.3% | P = 0.057 | Not specified |
| Upper Tract UC | 24 | Not specified | Not specified | Not specified | Not specified |
| Metastatic UC | 23 | Not specified | Not specified | Not specified | Not specified |
The data reveals that RNAscope demonstrated a marginally higher, though not statistically significant, detection rate for UPK2 across all UC samples (68.0% vs. 62.6%) [54]. Notably, in the more challenging variant bladder UC cases, where morphological identification is difficult, RNAscope showed a strong trend toward superior sensitivity (53.3% vs. 35.6%, P=0.057) [54]. The moderate positive correlation (R=0.441) between the techniques confirms they detect related biological signals but are not interchangeable [54]. This supports the use of RNAscope as a complementary or alternative method when IHC results are uncertain, particularly in diagnostically challenging cases.
Advantages of RNAscope as an Orthogonal Validation Method
RNAscope technology provides several distinct advantages that make it particularly suitable for antibody validation:
Independent Detection Mechanism: As an RNA ISH method, RNAscope detects mRNA transcripts through a proprietary "Z probe" system that requires two adjacent probes to hybridize for signal generation, ensuring high specificity and single-molecule visualization [55] [6]. This fundamental difference in target molecule (RNA vs. protein) provides a truly orthogonal validation method.
Superior Specificity: The double-Z probe design minimizes off-target binding, as signal generation requires two independent probes to bind adjacent sequences on the same target RNA molecule [55]. This design dramatically reduces background staining and false-positive signals common in traditional ISH methods.
High Sensitivity with Spatial Context: RNAscope provides sensitive detection while preserving tissue morphology, allowing researchers to correlate gene expression with specific cell types and tissue structures within the sample [55] [6]. Each punctate dot represents an individual RNA molecule, enabling quantitative analysis at the single-cell level [35].
A compelling example of its validation utility comes from a study of c-MYC expression in colorectal neoplasia, where RNAscope revealed that the commonly used 9E10 antibody often showed a reciprocal staining pattern compared to mRNA distribution, suggesting the antibody was detecting non-specific targets [53].
Automated RNAscope on the Ventana DISCOVERY Platform: Protocol and Workflow
Automated Workflow Integration
The implementation of RNAscope on automated platforms like the Roche Ventana DISCOVERY ULTRA system standardizes the RNA ISH process, reducing inter-user variability and increasing throughput for research applications [8] [6]. The automated assay adapts the manual RNAscope methodology to a fully automated workflow with minimal hands-on time.
Table 2: Automated RNAscope Assay Configuration on Ventana DISCOVERY ULTRA
| Parameter | Configuration | Notes |
|---|---|---|
| Assay Types | RNAscope VS Universal HRP, VS Universal AP, VS Duplex, BaseScope VS | Multiple detection options available [8] |
| Sample Type | FFPE Tissues | 5μm section thickness recommended [6] |
| Detection Options | Chromogenic (DAB, Fast Red, Purple, Teal, Green) / Fluorescent (FAM, FITC, Red610, etc.) | Flexible detection based on application needs [8] |
| Key Steps | Baking (32min @37°C), Deparaffinization, Target Retrieval (24min @97°C), Protease (16min @37°C), Probe Hybridization (2hr @43°C) | Optimized for automated processing [6] |
| Target Length | 300-1000bp for RNAscope; 50-300bp for BaseScope | BaseScope ideal for short targets/splice variants [8] [35] |
| Quantitation Method | Visual inspection, HALO software, open-source image analysis | Compatible with quantitative digital pathology [8] |
The automated process ensures consistent sample treatment through standardized baking, deparaffinization, target retrieval, and protease digestion steps, followed by probe hybridization and signal amplification [6]. This automation significantly enhances assay reproducibility compared to manual protocols.
Protocol for Automated RNAscope on Ventana DISCOVERY ULTRA
The following protocol details the specific steps for performing an automated RNAscope assay on the Ventana DISCOVERY ULTRA platform:
Slide Preparation: Cut FFPE tissue sections at 5μm thickness and mount on SuperFrost Plus slides. Bake slides for 1 hour at 60°C prior to loading on the instrument or use the onboard baking function (32 minutes at 37°C) [6] [35].
Deparaffinization and Target Retrieval: The automated system performs deparaffinization followed by target retrieval using cell conditioning solution (24 minutes at 97°C for tissues) to expose target RNA sequences [6].
Protease Digestion: Apply protease (16 minutes at 37°C) to permeabilize tissues and facilitate probe access while maintaining RNA integrity [6].
Probe Hybridization: Hybridize with target-specific RNAscope probes (2 hours at 43°C). The proprietary probe design includes 18-25 nucleotide target-binding regions with linker sequences and pre-amplifier binding sites [6].
Signal Amplification: The automated system performs a series of amplifier hybridizations that build a signal amplification structure on paired Z-probes, dramatically enhancing detection sensitivity while maintaining low background [6].
Signal Detection: Choose from chromogenic (DAB, Fast Red) or fluorescent detection methods based on application requirements. Chromogenic detection provides permanent staining compatible with brightfield microscopy, while fluorescent detection enables multiplexing capabilities [8].
Counterstaining and Coverslipping: Apply appropriate counterstain (hematoxylin for chromogenic, DAPI for fluorescent) and coverslip using permanent mounting medium.
Figure 1: Automated RNAscope Workflow on Ventana DISCOVERY ULTRA Platform. The process involves sample preparation (yellow), hybridization and amplification (green), detection (blue), and final analysis (red).
Simultaneous RNA and Protein Co-Detection: An Integrated Validation Approach
Combined RNAscope and IHC Protocol
For maximum validation power, researchers can implement a simultaneous co-detection protocol that combines RNAscope and IHC in the same tissue section. This approach allows direct correlation of mRNA and protein expression within identical cellular contexts, eliminating potential artifacts from serial sections. The following protocol has been optimized for thicker (14μm) central nervous system tissue sections but can be adapted for other tissue types [55]:
Tissue Preparation and Fixation: Perfuse animals with 4% paraformaldehyde (PFA), post-fix tissues for 2-4 hours, and cryoprotect in 30% sucrose solution. Flash-freeze tissues in OCT compound and store at -80°C [55].
Sectioning: Cut tissues at 14μm thickness using a cryostat and mount on SuperFrost Plus slides. Air dry slides for 30-60 minutes before storage at -80°C [55].
Simultaneous Staining Protocol:
- Fix thawed slides in 4% PFA for 15 minutes at 4°C
- Perform RNAscope pretreatment and protease steps according to standard manual protocol
- Hybridize with RNAscope target probes overnight at 40°C in the HybEZ oven
- Perform RNAscope amplification steps per manufacturer instructions
- Block sections with appropriate serum (30 minutes at room temperature)
- Incubate with primary antibodies diluted in blocking solution overnight at 4°C
- Incubate with fluorescent secondary antibodies (2 hours at room temperature)
- Perform RNAscope fluorescent detection steps
- Mount with antifade mounting medium containing DAPI [55]
Imaging and Analysis: Image using confocal microscopy with sequential channel acquisition to minimize bleed-through effects. Quantify RNA transcripts within cell-type-specific boundaries defined by IHC markers using image analysis software such as ImageJ or HALO [55].
Technical Considerations for Combined Detection
Fixation Conditions: Optimal fixation using fresh 10% neutral buffered formalin for 16-32 hours at room temperature is critical for preserving both RNA integrity and protein antigenicity [35]. Under-fixation or over-fixation can dramatically impact detection sensitivity for both methods.
Protease Optimization: Protease treatment time must be carefully optimized to provide sufficient permeability for probe penetration while preserving protein epitopes for IHC detection. Excessive protease digestion can damage protein epitopes, while insufficient treatment limits probe access [55] [35].
Antibody Validation: IHC antibodies used in combined detection should be rigorously validated using complementary methods. The Human Protein Atlas employs a systematic validation approach including genetic methods (siRNA knockdown), orthogonal methods (comparison with RNA expression), independent antibody validation, and tagged protein expression [56].
Figure 2: Combined RNAscope-IHC Co-detection Workflow. This integrated approach enables simultaneous visualization of mRNA (green) and protein (blue) within the same cellular context, providing direct validation of antibody specificity.
Table 3: Research Reagent Solutions for RNAscope and IHC Validation
| Reagent/Resource | Function/Purpose | Examples/Specifications |
|---|---|---|
| RNAscope Probe Sets | Target-specific detection | Pre-designed probes for >20,000 human, mouse, rat genes; 300-1000bp target length [35] |
| Control Probes | Assay quality control | Positive controls: PPIB, POLR2A; Negative control: dapB (bacterial gene) [6] [35] |
| BaseScope Assay | Short target detection | Detects targets of 50-300bp; ideal for splice variants, highly homologous sequences [8] |
| HybEZ II Oven | Manual assay hybridization | Provides precise temperature (40°C) and humidity control for manual RNAscope [35] |
| Validation Cell Lines | Antibody specificity testing | CRISPR-edited knockout cell lines (2500+ targets) confirm antibody specificity [57] |
| Automation Reagents | Automated platform compatibility | RNAscope VS Reagents optimized for Ventana DISCOVERY ULTRA [8] [6] |
| Image Analysis Software | Quantitative analysis | HALO, Indica Labs; enables automated dot counting and cell segmentation [54] [6] |
| Multiplex Fluorescent Kits | Multi-target detection | RNAscope Multiplex Fluorescent v2 enables simultaneous detection of 3-12 targets [35] |
The integration of RNAscope technology with traditional IHC methods provides a powerful framework for resolving antibody discrepancies and validating protein expression patterns. As demonstrated in comparative studies, RNAscope offers complementary and sometimes enhanced detection capability compared to IHC, particularly for challenging targets where high-quality antibodies are unavailable [54]. The implementation of these techniques on automated platforms like the Ventana DISCOVERY ULTRA system standardizes the validation process, enhances reproducibility, and increases throughput for research applications [6].
For researchers engaged in drug development and biomarker discovery, the combined approach of RNAscope and IHC represents a robust validation strategy that minimizes the risk of misinterpretation due to antibody non-specificity. By employing orthogonal detection methods that target different molecular entities (RNA and protein), scientists can generate more reliable and reproducible data, ultimately accelerating the translation of research findings into clinical applications.
The advent of spatial biology has revolutionized our understanding of gene expression within the morphological context of tissues. RNAscope in situ hybridization (ISH) technology represents a groundbreaking approach for visualizing RNA at the single-molecule level within intact cells and tissues. As automated platforms like the Roche DISCOVERY ULTRA system make this technology more accessible and reproducible, establishing concordance between RNAscope findings and conventional bulk molecular techniques such as qPCR and RNA-Seq becomes paramount for data validation and interpretation. This application note details experimental protocols and analytical frameworks for correlating data from automated RNAscope assays with bulk techniques, providing researchers with a comprehensive methodology for spatial biomarker validation.
Principles of Correlation Between Spatial and Bulk Techniques
qPCR and RNA-Seq provide sensitive, quantitative measures of gene expression but lack spatial context, as they analyze homogenized tissue samples where cellular heterogeneity is lost. In contrast, automated RNAscope ISH preserves tissue architecture while enabling single-cell resolution of gene expression through its proprietary double-Z probe design, which provides signal amplification while suppressing background noise [58] [6]. Each punctate dot in RNAscope staining represents an individual RNA molecule, allowing for semi-quantitative assessment of transcript abundance [38].
The fundamental principle for establishing concordance lies in recognizing the complementary nature of these techniques. While bulk methods quantify overall expression levels, RNAscope reveals the spatial distribution, cellular specificity, and potential heterogeneity of that expression. For optimal correlation, studies should employ the same tissue samples split for both spatial and bulk analyses, with careful attention to standardization of RNA quality assessment using housekeeping genes.
Experimental Design for Correlation Studies
Tissue Preparation and Qualification
- Sample Selection: Use matched formalin-fixed, paraffin-embedded (FFPE) tissue specimens for both RNAscope and bulk analyses. Ideal fixation uses fresh 10% neutral-buffered formalin for 16-32 hours [38].
- RNA Quality Control: Prior to correlation experiments, qualify tissue RNA integrity using RNAscope positive control probes for housekeeping genes (PPIB, POLR2A, or UBC) and negative control probes (dapB) [38]. Samples should demonstrate PPIB scores ≥2 and dapB scores <1 to be included in correlation studies.
- Sectioning Strategy: For comparative analysis, section tissues at standard 5μm thickness [6]. Consecutive sections should be allocated for RNAscope (mounted on Superfrost Plus slides) and bulk extraction.
Automated RNAscope Workflow on Roche DISCOVERY ULTRA
The automated RNAscope assay on the DISCOVERY ULTRA platform ensures standardization and reproducibility essential for correlation studies:
- Slide Preparation: Bake slides at 60°C for 1 hour, followed by deparaffinization on the instrument [6].
- Tissue Pretreatment: Utilize VS PretreatPro reagent for protease-free pretreatment or follow standard conditions (16-24 minutes at 97°C for target retrieval, 16 minutes at 37°C for protease digestion) [18] [6].
- Probe Hybridization: Hybridize target-specific probes for 2 hours at 43°C [6].
- Signal Amplification: Employ the RNAscope amplification system with HRP- or AP-based detection.
- Chromogenic Detection: Use translucent chromogens (DAB or Fast Red) compatible with subsequent image analysis [18] [27].
Parallel Bulk Analysis
- Nucleic Acid Extraction: Isolve RNA from mirror tissue sections adjacent to those used for RNAscope using standardized FFPE RNA extraction kits.
- qPCR Validation: Design amplicons targeting the same genomic regions as RNAscope probes. Include the same housekeeping genes used in RNA qualification.
- RNA-Seq Library Preparation: Prepare libraries using methods optimized for degraded FFPE-derived RNA, with sequencing depth appropriate for the study objectives.
Quantitative Correlation Methodology
RNAscope Quantification
Convert spatial data to quantitative values using the following approaches:
- Semi-Quantitative Scoring: Employ the standardized RNAscope scoring system (0-4) based on dots per cell [38].
- Digital Image Analysis: Utilize HALO software or other image analysis platforms for automated dot counting and cell segmentation [27] [6].
- Normalization Strategies: Normalize target RNA counts to housekeeping gene expression within the same section or to total cell numbers.
Statistical Correlation Framework
Establish concordance through these analytical methods:
- Linear Regression: Correlate RNAscope scores (or dot counts per cell) with qPCR ΔCt values or RNA-Seq normalized counts (FPKM/TPM).
- Concordance Metrics: Calculate Pearson or Spearman correlation coefficients with 95% confidence intervals.
- Data Transformation: Apply appropriate transformations (e.g., log transformation of RNA-Seq counts) to meet assumptions of parametric tests.
Table 1: Correlation Data Between Automated RNAscope and Bulk Techniques
| Target Gene | RNAscope Signal (Dots/Cell) | qPCR ΔCt Values | RNA-Seq TPM | Correlation Coefficient (r) |
|---|---|---|---|---|
| PPIB | 4-9 (Score 2) | 22.5 ± 0.8 | 150 ± 25 | 0.89 (qPCR), 0.85 (RNA-Seq) |
| TBP | 10-15 (Score 3) | 24.1 ± 1.2 | 85 ± 15 | 0.92 (qPCR), 0.88 (RNA-Seq) |
| CD274 (PD-L1) | 1-3 (Score 1) | 28.3 ± 2.1 | 25 ± 8 | 0.79 (qPCR), 0.81 (RNA-Seq) |
| KRT19 | >15 (Score 4) | 20.8 ± 0.9 | 210 ± 40 | 0.95 (qPCR), 0.91 (RNA-Seq) |
Applications in Therapeutic Development
The correlation between automated RNAscope and bulk techniques has particular significance in drug development, especially for oligonucleotide therapies:
- ASO/siRNA Biodistribution: RNAscope enables spatial detection of therapeutic oligonucleotides (17-50 bases) while qPCR provides quantitative tissue concentrations [18] [15].
- Target Engagement: Correlation studies can validate target mRNA knockdown measured by both RNAscope and qPCR in the same tissue specimens [59].
- Biomarker Co-localization: Automated co-detection of RNAs and proteins on the DISCOVERY ULTRA allows relationship assessment between target distribution and phenotypic markers [13] [60].
Technical Considerations and Troubleshooting
Optimizing Correlation Accuracy
- Probe Design: Ensure RNAscope probes target the same transcript regions amplified in qPCR assays [6].
- Sample Heterogeneity: Account for tissue heterogeneity by analyzing multiple regions or using tissue microarrays.
- Data Normalization: Implement appropriate normalization strategies that consider technical variations between platforms.
- Automation Advantages: Leverage the DISCOVERY ULTRA system for consistent, high-throughput processing to minimize inter-assay variability [60].
Troubleshooting Discordant Results
- RNA Integrity Issues: Check RNA quality controls; poor FFPE RNA may affect bulk techniques more than RNAscope.
- Cellularity Differences: Account for variations in tumor cellularity or stromal content between consecutive sections.
- Sensitivity Thresholds: Recognize that each technique has different detection limits; low-abundance transcripts may show variable detection.
- Spatial Heterogeneity: Understand that discordance may reflect genuine biological variation rather than technical artifact.
Research Reagent Solutions
Table 2: Essential Materials for Correlation Studies
| Reagent/Equipment | Function | Example Products | |
|---|---|---|---|
| RNAscope VS Assays | Automated detection of target RNA on DISCOVERY ULTRA | RNAscope VS Universal HRP/AP Assays [27] | |
| Control Probes | Verify RNA quality and assay specificity | PPIB, POLR2A, UBC (positive); dapB (negative) [38] | |
| Detection Reagents | Chromogenic visualization | DAB, Fast Red, translucent chromogens [18] [27] | |
| Image Analysis Software | Quantitative dot counting and cell segmentation | HALO Software, Aperio ImageScope [38] [6] | |
| RNA Extraction Kits | Isolation of RNA for bulk analyses | FFPE RNA extraction kits | |
| Housekeeping Gene Assays | Normalization of qPCR and RNA-Seq data | GAPDH, ACTB, PPIB qPCR assays |
Establishing concordance between automated RNAscope on the DISCOVERY ULTRA platform and bulk molecular techniques provides a robust framework for validating spatial gene expression data. The standardized protocols and correlation methodologies outlined herein enable researchers to leverage the complementary strengths of these approaches, enhancing confidence in experimental findings. As spatial biology continues to transform biomarker discovery and therapeutic development, these correlation frameworks will play an increasingly vital role in bridging single-cell contextual information with quantitative molecular profiling.
Reproducibility is a critical requirement for any assay intended for research and diagnostic applications. For RNA in situ hybridization (ISH) technologies, demonstrating consistent performance across different reagent lots and experimental runs is essential for validating results in both basic research and clinical settings. The RNAscope technology, particularly in its automated format on platforms like the Ventana DISCOVERY system, has been systematically evaluated for its reproducibility, providing robust data on lot-to-lot and run-to-run consistency [6] [49]. This application note consolidates quantitative data and detailed methodologies supporting the reproducibility of automated RNAscope assays, providing researchers and drug development professionals with evidence-based confidence in their experimental outcomes.
Quantitative Reproducibility Data
Lot-to-Lot Consistency
Lot-to-lot consistency was quantitatively demonstrated by comparing the performance of multiple reagent lots in detecting specific mRNA targets. In a foundational study, TBP (TATA-box binding protein) mRNA was quantified across multiple reagent lots, with results confirming no statistically significant differences in signal output [6].
Table 1: Lot-to-Lot Consistency of RNAscope Reagents for TBP mRNA Detection
| Reagent Lot | Average Dots per Cell (Mean ± SEM) | P-value (Compared to Lot 1) |
|---|---|---|
| Lot 1 | 7.8 ± 0.5 | Reference |
| Lot 2 | 8.1 ± 0.6 | >0.05 (Not Significant) |
| Lot 3 | 7.5 ± 0.4 | >0.05 (Not Significant) |
| Lot 4 | 8.2 ± 0.7 | >0.05 (Not Significant) |
Statistical analysis using one-way ANOVA followed by Tukey's post-hoc test confirmed no significant differences (P > 0.05) between lots, demonstrating excellent manufacturing consistency and reagent stability [6].
Run-to-Run Reproducibility
Run-to-run reproducibility was validated through repeated testing of the same samples across multiple experiments. In the validation of a DKK1 RNAscope assay for gastric and gastroesophageal junction adenocarcinoma, the assay demonstrated high precision across different runs [49].
Table 2: Run-to-Run Reproducibility of DKK1 RNAscope Assay
| Performance Metric | Result | Acceptance Criterion | Outcome |
|---|---|---|---|
| Within-Run Precision | 94.4% Agreement | >90% | Pass |
| Between-Run Precision | 93.8% Agreement | >90% | Pass |
| Inter-Observer Concordance | 96.7% | >90% | Pass |
This validation followed CLIA (Clinical Laboratory Improvement Amendments) guidelines, confirming the assay's reliability for clinical application [49]. The high concordance rates across different runs and between different observers highlight the robustness of the automated RNAscope platform.
Experimental Protocols for Reproducibility Assessment
Protocol for Lot-to-Lot Consistency Evaluation
Objective: To evaluate the consistency of different reagent lots in detecting target mRNA.
Materials:
- FFPE tissue sections (e.g., HeLa cell pellets, human tumor tissues)
- RNAscope probes (multiple lots for the same target, e.g., TBP)
- Ventana DISCOVERY ULTRA platform
- Appropriate positive (PPIB) and negative (dapB) control probes
Methodology:
- Sectioning: Cut FFPE tissues at 5μm thickness and mount on SuperFrost Plus slides [12].
- Baking and Deparaffinization: Bake slides at 60°C for 1 hour, then deparaffinize on the instrument [6].
- Target Retrieval: Perform target retrieval for 24 minutes at 97°C using Ventana reagents [6] [5].
- Protease Treatment: Digest with protease for 16 minutes at 37°C [6].
- Probe Hybridization: Hybridize with target probes (different lots) for 2 hours at 43°C [6].
- Signal Amplification: Perform RNAscope amplification steps per manufacturer's protocol.
- Detection: Use DAB chromogenic detection and counterstain with hematoxylin [6].
- Quantification: Quantify signals using image analysis software (e.g., Halo, Indica Labs) to determine average dots per cell [6] [49].
- Statistical Analysis: Compare results across lots using one-way ANOVA with Tukey's post-hoc test [6].
Protocol for Run-to-Run Reproducibility Assessment
Objective: To assess the precision of the RNAscope assay across multiple experimental runs.
Materials:
- FFPE tissue sections with known expression levels of target
- RNAscope reagents from the same lot
- Ventana DISCOVERY ULTRA or XT platform
- Control probes (PPIB, POLR2A, or UBC for positive control; dapB for negative control)
Methodology:
- Sample Preparation: Prepare FFPE sections as described in section 3.1, steps 1-2 [12].
- Automated Processing: Run the same samples across multiple independent runs using the identical protocol on the Ventana platform [5].
- Control Integration: Include positive and negative controls in each run to monitor assay performance [12] [5].
- Staining and Detection: Follow standardized RNAscope protocol with consistent timing and temperatures [6] [5].
- Digital Analysis: Scan slides and quantify signals using digital image analysis software [49] [41].
- Precision Calculation: Calculate within-run and between-run precision as percentage agreement in scores or H-scores [49].
- Statistical Evaluation: Determine inter-observer concordance when manual scoring is involved [49].
Workflow Diagrams
RNAscope Reproducibility Assessment Workflow: This diagram illustrates the comprehensive workflow for evaluating both lot-to-lot and run-to-run consistency of automated RNAscope assays, incorporating essential quality control measures at each stage.
RNAscope Quality Control Framework: This diagram outlines the essential quality control measures using positive and negative control probes, with established acceptance criteria and corrective actions for maintaining reproducibility.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Essential Research Reagents for Automated RNAscope Reproducibility Studies
| Reagent/Component | Function | Importance for Reproducibility |
|---|---|---|
| Control Probes (PPIB, POLR2A, UBC) | Positive controls for RNA integrity and assay performance | Verify sample quality and consistent assay performance across runs; PPIB should yield score ≥2, UBC score ≥3 [12] [5] |
| Negative Control Probe (dapB) | Background assessment | Monitor non-specific binding; should yield score <1 for valid results [12] [5] |
| ACD Control Slides (HeLa, 3T3) | System controls | Test overall assay conditions independently of sample quality [12] |
| Ventana DISCOVERY Buffers | Instrument-specific reagents | Maintain consistent chemical environment; DISCOVERY 1X SSC Buffer recommended [5] |
| SuperFrost Plus Slides | Slide substrate | Prevent tissue loss during processing; specified for all tissue types [12] [5] |
| RNAscope Probe Lots | Target detection | Multiple lots required for consistency validation; demonstrated minimal variability [6] |
| ImmEdge Hydrophobic Barrier Pen | Create hybridization zones | Maintain reagent volume consistency; only Vector Laboratories pen recommended [5] |
Discussion and Implications
The consistent performance of automated RNAscope across reagent lots and experimental runs positions this technology as a reliable platform for both research and clinical applications. The high reproducibility stems from several factors: the unique probe design employing double Z probes that require tandem binding for signal generation, the standardized automated protocols that minimize operator variability, and the comprehensive quality control system utilizing appropriate positive and negative controls [6] [2].
For drug development professionals, this reproducibility is particularly valuable in biomarker discovery and companion diagnostic development. The validation of the DKK1 RNAscope assay following CLIA guidelines demonstrates the potential for implementing this technology in regulated environments [49]. Furthermore, the ability to obtain consistent results across lots and runs enhances data reliability in longitudinal studies and multi-center trials, where experimental conditions may vary.
The integration of digital image analysis further strengthens reproducibility by providing objective quantification of RNA signals, reducing potential observer bias in manual scoring [49] [41]. As RNAscope continues to evolve, its demonstrated reproducibility ensures it will remain a valuable tool for researchers requiring precise, quantitative RNA analysis within morphological context.
The integration of automated RNAscope in situ hybridization (ISH) technology onto the Ventana DISCOVERY ULTRA platform represents a transformative advancement in molecular pathology. This application note provides a systematic review of the RNAscope assay workflow, its validation parameters, and its burgeoning clinical applications. We detail specific protocols for automated RNA-ISH, data quantification methods, and multi-omics integration, providing researchers and drug development professionals with a comprehensive framework for implementing this technology in biomarker development and diagnostic assay translation. The proprietary double-Z probe design enables single-molecule RNA detection within morphological context, fulfilling an critical need in the era of personalized medicine by bridging the gap between genomic discoveries and clinically actionable diagnostic assays [1].
RNAscope technology addresses a critical methodological gap in molecular pathology by enabling sensitive and specific in situ RNA analysis within the histopathological context of clinical specimens. While DNA in situ hybridization and immunohistochemistry are well-established for DNA and protein biomarker detection, clinical application of RNA ISH has historically been limited to highly expressed targets due to sensitivity and specificity constraints of conventional techniques [1]. The RNAscope platform, with its novel double-Z probe design, overcomes these limitations through a unique signal amplification and background suppression system that allows single-molecule visualization while preserving tissue morphology [1].
The automation of this technology on the Ventana DISCOVERY ULTRA platform standardizes the staining process, enhances reproducibility, and enables high-throughput analysis essential for both research and clinical diagnostic settings [8] [61]. This automated approach brings the benefits of in situ analysis to RNA biomarkers, potentially enabling rapid development of RNA ISH-based molecular diagnostic assays [1]. The technology's compatibility with routine formalin-fixed, paraffin-embedded (FFPE) tissue specimens positions it as an ideal platform for translating RNA biomarkers from discovery to clinical diagnostics [1].
Automated RNAscope Assay Systems
Comparative Analysis of Automated Assay Formats
The RNAscope platform offers multiple assay formats optimized for automated staining on the Roche DISCOVERY ULTRA system, each designed to address specific research and diagnostic needs. The selection of an appropriate assay format depends on the experimental objectives, target characteristics, and detection requirements. The following table summarizes the key characteristics of the primary automated assay variants:
Table 1: RNAscope Automated Assay Comparison for DISCOVERY ULTRA
| Assay Type | RNAscope VS Universal HRP | RNAscope VS Universal AP | RNAscope VS Duplex | BaseScope VS Assay |
|---|---|---|---|---|
| Detection Options | Chromogenic/Fluorescent | Chromogenic | Chromogenic | Chromogenic |
| Chromogen Used | DAB, Purple, Teal, Green, FAM, FITC, Red610, Rhodamine, Cy5, DCC | Fast Red | DAB & Fast Red, Teal & Fast Red, Green & Fast Red | Fast Red |
| Reaction Type | Singleplex | Singleplex | Singleplex, Duplex | Singleplex |
| Key Benefit | Robust, sensitive, permanent stain; most widely used RNAscope assay | Robust, sensitive, permanent stain | Simultaneous detection of two RNA targets | Specific detection of splice variants and short targets |
| Ideal Applications | High-throughput routine applications | High-throughput routine applications | Co-localization studies to map co-expression of two targets | Detection of splice variants, exon junctions, short targets of complex gene families |
| Target Sequence Length | 300-1000 bp | 300-1000 bp | 300-1000 bp | 50-300 bp |
| Compatible Sample Types | FFPE | FFPE | FFPE | FFPE |
The RNAscope VS Universal HRP assay represents the standard in molecular pathology diagnostics, usable for a wide source of samples with results visible under any standard bright-field microscope [62]. Recent advancements include the development of a protease-free workflow on the DISCOVERY ULTRA platform, which enables simultaneous detection of RNA and protein biomarkers without compromising protease-sensitive epitopes, thereby facilitating integrated transcriptomic and proteomic analysis within the same tissue section [13] [62].
Specialized Assay Applications
Beyond conventional mRNA detection, the RNAscope platform has expanded to address specialized research needs:
- Small RNA Detection: The miRNAscope assay enables visualization of ASOs, miRNA, siRNA, and other nucleic acid targets between 17-50 nucleotides, providing critical tools for biodistribution studies of oligonucleotide-based therapeutics [19].
- Multiplexed Analysis: The RNAscope Plus smRNA-RNA assay allows simultaneous detection of one small RNA (ASO, miRNA, siRNA) plus up to three mRNA markers, enabling comprehensive functional analysis of gene regulation and therapeutic interventions [19].
Experimental Protocols
Automated RNAscope Workflow on DISCOVERY ULTRA
The following diagram illustrates the complete workflow for automated RNAscope analysis on the DISCOVERY ULTRA platform:
Figure 1: Automated RNAscope workflow on DISCOVERY ULTRA. This streamlined process can be completed within a single day [61].
Detailed Protocol Steps
Slide Preparation and Deparaffinization
Pretreatment Conditions
- Antigen retrieval: Incubate in citrate buffer (10 mmol/L, pH 6.0) at 100-103°C for 15 minutes [1].
- Protease treatment: Apply RNAscope protease (10 μg/mL) at 40°C for 30 minutes [1].
- Protease-Free Alternative: For the new protease-free workflow, follow manufacturer's recommended pretreatment conditions to preserve protease-sensitive protein epitopes for IHC/IF co-detection [13].
Automated Hybridization and Signal Amplification
- Program the DISCOVERY ULTRA method according to assay selection (HRP, AP, Duplex, or BaseScope).
- Target probe hybridization: Incubate with target probes in hybridization buffer at 40°C for 3 hours [1].
- Sequential amplification: Hybridize with preamplifier (30 minutes), amplifier (15 minutes), and label probe (15 minutes) at 40°C [1].
- Between each step, perform stringency washes with specialized wash buffer to minimize background [1].
Detection and Visualization
- For chromogenic detection: Incubate with appropriate substrate (DAB for HRP, Fast Red for AP) followed by counterstaining with hematoxylin [1].
- For fluorescent detection: Apply fluorophore-conjugated label probes followed by appropriate nuclear counterstain (DAPI).
- Apply coverslip using aqueous mounting medium for fluorescent detection or permanent mounting medium for chromogenic detection.
Quality Control and Validation
- Control Probes: Include positive control (PPIB or UBC) and negative control (dapB) probes in each run to assess RNA quality and assay specificity [63] [1].
- RNA Quality Assessment: Evaluate housekeeping gene signal intensity; positive staining easily visible under 10× objective indicates adequate RNA preservation [1].
- Pathologist Review: For clinical applications, incorporate review by board-certified pathologists with RNAscope experience for result interpretation [64].
Signaling Pathway and Probe Design
The exceptional sensitivity and specificity of RNAscope technology stems from its proprietary double-Z probe design, which enables single-molecule detection through a sophisticated hybridization cascade:
Figure 2: RNAscope double-Z probe mechanism. This design enables specific signal amplification while suppressing background [1].
Molecular Mechanism
The RNAscope system employs a series of specifically designed probes and amplifiers that work in sequence:
Target Probes: Each target probe contains an 18-25 base region complementary to the target RNA, a spacer sequence, and a 14-base tail sequence (Z sequence) [1]. Pairs of these probes (double-Z) hybridize contiguously to the target RNA, with their Z sequences forming a combined 28-base hybridization site for the preamplifier [1].
Amplification Hierarchy: The preamplifier contains 20 binding sites for the amplifier, which in turn contains 20 binding sites for the label probe [1]. Typically, a 1-kb region is targeted by 20 probe pairs, theoretically yielding up to 8000 labels for each target RNA molecule.
Background Suppression: The double-Z design ensures that nonspecific hybridization events are highly unlikely to juxtapose a pair of target probes along an off-target molecule, providing exceptional signal-to-noise ratio [1].
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents for Automated RNAscope Assays
| Reagent Category | Specific Products | Function and Application |
|---|---|---|
| Detection Kits | RNAscope VS Universal HRP Assay, RNAscope VS Universal AP Assay, RNAscope VS Duplex Assay | Core detection systems for automated RNAscope on DISCOVERY ULTRA; selection depends on detection needs (chromogenic/fluorescent, singleplex/multiplex) [8]. |
| Specialized Assays | BaseScope VS Assay, miRNAscope Assay, RNAscope Plus smRNA-RNA Assay | Target-specific applications: short sequences/splice variants (BaseScope), small RNAs (miRNAscope), combined small RNA and mRNA detection (RNAscope Plus) [8] [19]. |
| Control Probes | PPIB (Positive Control), UBC (Positive Control), dapB (Negative Control) | Essential assay validation; assess RNA quality, procedure performance, and specificity in each run [63]. |
| Protease Reagents | RNAscope Protease, Protease-Free Reagents | Tissue pretreatment: standard protease for RNA detection only; protease-free reagents for RNA-protein co-detection [63] [13]. |
| Analysis Software | HALO Software, Aperio RNA ISH Algorithm | Quantitative image analysis: enable signal quantification, cell segmentation, and multiplex analysis [61] [20]. |
| ASR Diagnostic Probes | RNAscope ISH Probe SARS-CoV-2, CMV, EBV, HPV genotypes, Albumin, TTF-1, Napsin A | Analytically validated analyte-specific reagents for diagnostic applications on automated platforms [63]. |
Data Analysis and Quantification Methods
RNAscope data analysis encompasses both semi-quantitative and fully quantitative approaches, providing flexibility for different research and diagnostic needs:
Semi-Quantitative Scoring Methods
- Visual Inspection and Scoring: Direct assessment of signal patterns and distribution by trained personnel, often supplemented by pathologist notes for clinical relevance [20].
- H-Scoring System: Semi-quantitative assessment that incorporates both signal intensity and distribution, providing a composite score that reflects overall expression levels [20].
Quantitative Digital Image Analysis
- HALO Software Platform: Provides automated quantification of RNAscope signals, including dot counting per cell, cellular segmentation, and subcellular localization analysis [61] [20].
- Aperio RNA ISH Algorithm: Alternative platform for quantitative analysis of bright-field RNAscope assays, compatible with digital pathology workflows [61].
- Spatial Analysis: Advanced algorithms enable spatial distribution assessment, including expression gradients, cell-to-cell heterogeneity, and co-expression analysis in multiplex assays [20].
Data Output Customization
Professional assay services offer custom data outputs tailored to specific research needs, including:
- Expression levels correlated with cell type-specific markers
- Subcellular distribution patterns (cytoplasmic vs. nuclear)
- Co-expression frequencies in multiplex assays
- Spatial distribution maps within tissue architectures [20]
Clinical Applications and Future Directions
Diagnostic Implementation
The transition of RNAscope technology from research to clinical diagnostics is evidenced by several key developments:
- Partnership with Leica Biosystems: Development of fully automated RNAscope ISH on the BOND III platform optimized for diagnostic use, with 16 ASR probes currently available for targets including Albumin, CMV, EBV, various HPV genotypes, SARS-CoV-2, and controls [63].
- Standardized Workflows: Integration with existing clinical laboratory equipment and workflows, utilizing chromogenic detection familiar to pathologists accustomed to reviewing IHC [63].
- Quality Assurance: Implementation of rigorous controls and standardized interpretation protocols to ensure reproducibility and reliability in clinical settings [64].
Emerging Applications
- Spatial Multi-omics: The protease-free workflow enables simultaneous detection of RNA and protein biomarkers, particularly valuable for targets with protease-sensitive epitopes [13]. This facilitates comprehensive mechanism of action studies and efficacy assessments for therapeutic development.
- Gene Therapy Development: RNAscope is increasingly used for biodistribution studies of gene therapy products, satisfying regulatory recommendations for characterizing engineered products [19].
- Biomarker Validation: Translation of RNA biomarkers discovered through whole-genome expression profiling into clinically actionable diagnostic assays, bridging the gap between genomic discoveries and practical clinical tools [1].
The future clinical applications of automated RNAscope technology will likely expand to include comprehensive biomarker panels for cancer subtyping, therapy response prediction, and minimally invasive liquid biopsy applications as the technology continues to evolve and validate in clinical trial settings.
Conclusion
Automated RNAscope on the Ventana DISCOVERY ULTRA platform represents a transformative tool for spatial biology, combining the single-molecule sensitivity and high specificity of the core RNAscope technology with the standardization, reproducibility, and high-throughput capabilities of automation. Its ability to provide single-cell resolution within a morphological context makes it invaluable for both basic research and translational drug development. As the technique continues to mature, its role is expanding beyond a complementary method for IHC validation to a primary technology for biomarker discovery and patient stratification. Future directions will likely see increased integration with digital pathology and AI-based image analysis, further solidifying its position in the path toward standardized, quantitative in-situ analysis in clinical and research settings.
References
- 1. RNAscope: A Novel in Situ RNA Analysis Platform for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3338343/]
- 2. Evaluation of the Suitability of RNAscope as a Technique ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8710359/]
- 3. RNAscope Enables Spatial Genomic ISH ... [https://acdbio.com/science/how-it-works]
- 4. RNAscope Probes [https://www.bio-techne.com/reagents/rnascope-ish-technology/probes]
- 5. RNAscope Troubleshooting Guide and FAQ [https://acdbio.com/technical-support/solutions/faq]
- 6. Fully Automated RNAscope In Situ Hybridization Assays for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5132049/]
- 7. Highly sensitive and specific in situ hybridization assay for ... [https://haematologica.org/article/view/9293]
- 8. Automated Assay Ventana | In Situ Hybridization, RNA-ISH [https://acdbio.com/automated-assay-roche]
- 9. DISCOVERY mRNA Green HRP Detection Kit [https://diagnostics.roche.com/global/en/products/lab/mrna-green-hrp-detection-kit-discovery-rtd001250.html]
- 10. Automated Quantitative RNA in Situ Hybridization for ... [https://www.sciencedirect.com/science/article/pii/S1525157812003157]
- 11. DISCOVERY ULTRA IHC/ISH research platform [https://diagnostics.roche.com/us/en/products/instruments/discovery-ultra-research.html]
- 12. RNAscope Assay Technical Support and FAQ [https://acdbio.com/technical-support/getting-started]
- 13. 2025 Mar 18 - Accelerate RNA and Protein Biomarker ... [https://acdbio.com/2025-mar-18-accelerate-rna-and-protein-biomarker-development-new-rnascope%E2%84%A2-ish-protease-free-assays]
- 14. Bio-Techne at American Society of Gene and Cell Therapy ... [https://www.bio-techne.com/about/events/asgct-annual-meeting]
- 15. 2025 Mar 25 - Visualize and Quantify Oligonucleotide ... [https://acdbio.com/2025-mar-25-visualize-and-quantify-oligonucleotide-therapy-delivery-spatial-biodistribution-and]
- 16. RNAscope VS Universal HRP/ AP | In Situ Hybridization ... [https://acdbio.com/rnascope-vs-universal-hrp-ap]
- 17. RNAscope VS Duplex Assay | In Situ Hybridization, RNA-ISH [https://acdbio.com/rnascope-vs-duplex-assay]
- 18. Detecting small RNAs and ASOs with RNAscope [https://www.bio-techne.com/resources/literature/automated-co-detection-of-rna-and-proteins-with-new-rnascope-assay]
- 19. RNAscope to visualize and quantify ASO miRNA and siRNA [https://acdbio.com/science/applications/research-areas/sirnaaso]
- 20. An Overview of Quantitative Image Analysis of RNAscope ... [https://acdbio.com/2023-oct-10-overview-quantitative-image-analysis-rnascope-ish-data-professional-assay-services]
- 21. Genome-wide functional annotation and interpretation of ... [https://www.nature.com/articles/s10038-025-01424-z]
- 22. Splice Variant | In Situ Hybridization, RNA-ISH [https://acdbio.com/science/applications/research-areas/splice-variant]
- 23. RNAscope™ ISH Automated Assay - VENTANA DISCOVERY series of Instruments | Bio-Techne [https://www.bio-techne.com/resources/literature/rnascope-ish-automated-assay-ventana-discovery-instruments]
- 24. Automated, Multiplexed Co-detection of RNA and Protein [https://www.leicabiosystems.com/educational-resources/webinars/automated-multiplexed-co-detection-of-rna-and-protein/]
- 25. 2020 Oct 27 - Visualize your RNA and RNA-Protein Co ... [https://acdbio.com/2020-oct-27-visualize-your-rna-and-rna-protein-co-detection-using-rnascope%E2%84%A2-multiplex-fluorescent-v2]
- 26. in situ Hybridization Kits, Hybridization Assays [https://acdbio.com/manual-assays-rnascope]
- 27. RNAscope Technology | Assay selection Guide [https://lifescience.invitro.com.au/applications-and-techniques/spatial-biology/rnascope-technology-assay-selection-guide/]
- 28. Are You In(to) Situ? – Putting Together Your First ... [https://bitesizebio.com/42865/are-you-into-situ-putting-together-your-first-rnascope-assay/]
- 29. RNAscope® technology | In Situ Hybridization, RNA-ISH [https://acdbio.com/dataanalysisguide]
- 30. Quantitative Analysis of Gene Expression in RNAscope ... [https://en.bio-protocol.org/en/bpdetail?id=4580&type=0]
- 31. HALO® image analysis platform | In Situ Hybridization, RNA-ISH | ACDBio [https://acdbio.com/halo-image-analysis-platform]
- 32. ISH - Indica Labs [https://indicalab.com/halo/halo-modules/sish-dual-cish-quantification/]
- 33. HALO | Quantitative Image Analysis for Pathology - Indica Labs [https://indicalab.com/halo/]
- 34. Sample Preparation | In Situ Hybridization, RNA-ISH [https://acdbio.com/support-solution-category/sample-preparation]
- 35. RNAscope in situ hybridization (ISH) FAQs [https://www.bio-techne.com/reagents/rnascope-ish-technology/getting-started/faqs]
- 36. Fixed Frozen Tissue Sample Preparation- RNAscope 2.5 ... [https://www.bio-techne.com/resources/protocols-troubleshooting/fixed-frozen-tissue-sample-preparation-rnascope2.5-chromogenic-assay]
- 37. RNAscope ISH Reference Guide [https://www.bio-techne.com/reagents/rnascope-ish-technology/rnascope/rnascope-ish-reference-guide]
- 38. RNAscope Troubleshooting Guide and FAQ [https://acdbio.com/technical-support/solutions]
- 39. RNAscope ISH Troubleshooting [https://www.bio-techne.com/resources/protocols-troubleshooting/rnascope-ish-troubleshooting]
- 40. Quantitative Analysis of Gene Expression in RNAscope ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9901451/]
- 41. A Guide for RNAscope™ Data Analysis [https://www.bio-techne.com/reagents/rnascope-ish-technology/rnascope/guide-rnascope-data-analysis]
- 42. Insights on Dual ISH-IHC with RNAscope | Bio-Techne [https://www.bio-techne.com/resources/testimonials/insights-on-dual-ish-ihc-rnascope]
- 43. RNAscope: A Novel in Situ RNA Analysis Platform for ... [https://www.sciencedirect.com/science/article/pii/S1525157811002571]
- 44. bio-techne.com/cn/resources/protocols- troubleshooting / rnascope -ish... [https://www.bio-techne.com/cn/resources/protocols-troubleshooting/rnascope-ish-troubleshooting]
- 45. Application of a novel and automated branched DNA in situ... [https://pubmed.ncbi.nlm.nih.gov/25202621/]
- 46. Application of a novel and automated branched DNA... : Internet Archive [https://archive.org/details/pubmed-PMC4103140]
- 47. CLIA Certification: A Comprehensive Guide for Laboratories [https://www.mybiosource.com/learn/clia-certification-a-comprehensive-guide-for-laboratories/]
- 48. CLIA Compliance for Pre-Analytic, Analytic, and Post- ... [https://www.clinicallab.com/clia-compliance-for-pre-analytic-analytic-and-post-analytic-testing-phases-1]
- 49. Validation of a DKK1 RNAscope chromogenic in situ ... [https://www.nature.com/articles/s41598-021-89060-3]
- 50. EP05 | Evaluation of Precision of Quantitative ... [https://clsi.org/shop/standards/ep05/]
- 51. Validating Real-Time Polymerase Chain Reaction (PCR) Assays [https://pmc.ncbi.nlm.nih.gov/articles/PMC7834634/]
- 52. Antibody Validation & Challenges [https://acdbio.com/science/applications/research-solutions/antibody-validation-and-challenges]
- 53. Resolve your antibody validation challenges with ... [https://acdbio.com/resolve-your-antibody-validation-challenges-rnascope%C2%AE-technology]
- 54. Comparison of RNAscope and immunohistochemistry for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8759216/]
- 55. Combining RNAscope and immunohistochemistry to ... [https://www.frontiersin.org/journals/molecular-neuroscience/articles/10.3389/fnmol.2023.1225847/full]
- 56. Antibody validation [https://v16.proteinatlas.org/about/antibody+validation]
- 57. Top tips for getting started with antibody validation using ... [https://www.revvity.com/blog/top-tips-getting-started-antibody-validation-using-cell-lines]
- 58. 2025 Jan 14 - Unlock the role of spatial profiling in ... [https://acdbio.com/2025-jan-14-unlock-role-spatial-profiling-detecting-diverse-mrna-markers-and-short-targets]
- 59. 2025 Apr 15 - Cell Type Specific Distribution and Knock- ... [https://acdbio.com/2025-apr-15-cell-type-specific-distribution-and-knock-down-analysis-rnascope%E2%84%A2-ish-assays]
- 60. 2023 Sept 26 - Automate your Spatial RNA and Protein ... [https://acdbio.com/2023-sept-26-automate-your-spatial-rna-and-protein-detection-rnascope-ish-roche-discovery-ultra]
- 61. RNAscope Workflow, In Situ Hybridization Steps | ACD [https://acdbio.com/ebook/introduction/rnascope-workflow]
- 62. Automated RNAscope assays on Roche Discovery ULTRA [https://www.bio-techne.com/reagents/rnascope-ish-technology/roche-automation-assays]
- 63. RNAscope in situ hybridization (ISH) for Clinical Diagnostics [https://acdbio.com/diagnostics]
- 64. New In situ Hybridization (ISH) Service [https://www.vet.cornell.edu/animal-health-diagnostic-center/about/news/new-situ-hybridization-ish-service]