Mastering NBT/BCIP Staining: A Complete Guide to Robust Chromogenic Detection for RNA In Situ Hybridization
This comprehensive guide details the NBT/BCIP staining protocol for chromogenic RNA in situ hybridization (ISH), a foundational technique for visualizing spatial gene expression in developmental biology, disease research, and drug...
Mastering NBT/BCIP Staining: A Complete Guide to Robust Chromogenic Detection for RNA In Situ Hybridization
Abstract
This comprehensive guide details the NBT/BCIP staining protocol for chromogenic RNA in situ hybridization (ISH), a foundational technique for visualizing spatial gene expression in developmental biology, disease research, and drug development. It covers core principles from probe design and tissue preparation to the final enzymatic reaction, providing a step-by-step methodological workflow adaptable to sectioned and whole-mount samples. The article delivers extensive troubleshooting for common issues like high background and weak signal, offers optimization strategies for enhanced sensitivity, and includes a comparative analysis with emerging methods to help researchers validate results and select the optimal detection platform for their specific applications.
Understanding NBT/BCIP Chemistry: The Foundation of Chromogenic ISH Detection
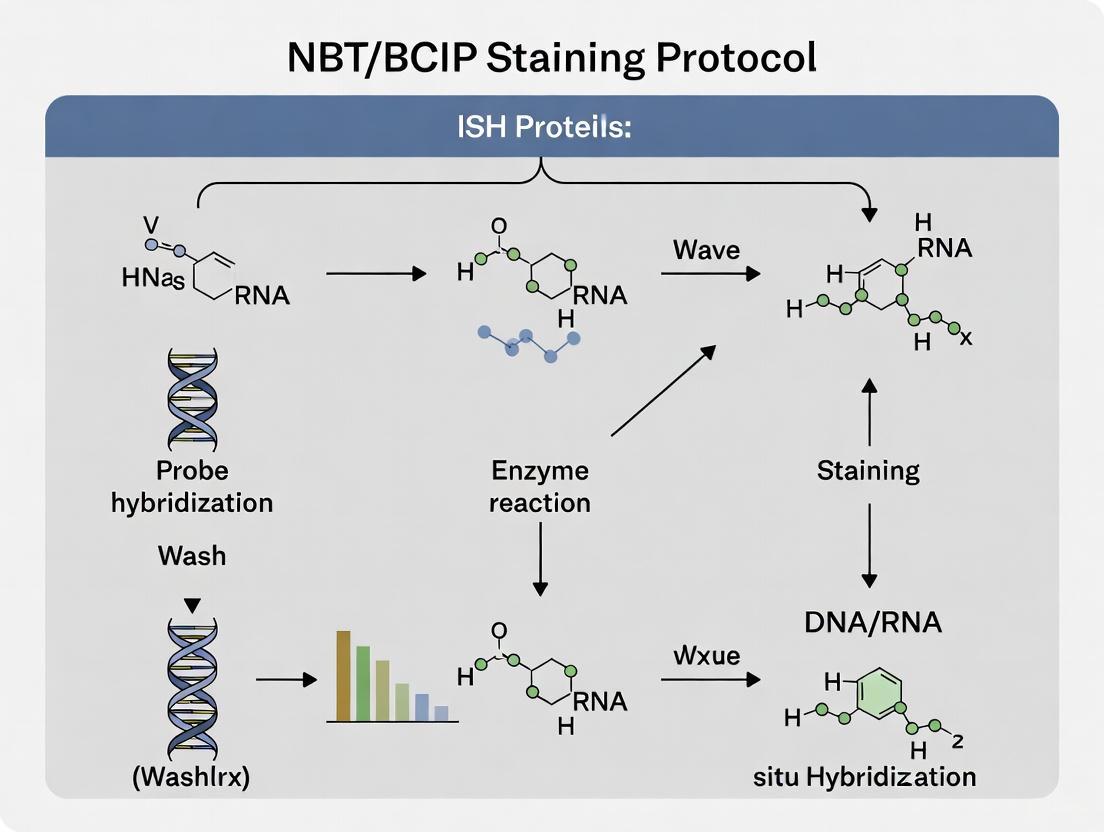
The NBT/BCIP chromogenic system is a cornerstone of non-radioactive detection for in situ hybridization (ISH) and immunohistochemical applications. This robust method utilizes the enzyme alkaline phosphatase (AP) to catalyze a reaction that yields an insoluble, dark-blue to purple precipitate at the site of target nucleic acid sequences. The precipitate forms through a coupled reaction where BCIP (5-Bromo-4-chloro-3-indolyl phosphate) is hydrolyzed, and the resulting product reduces NBT (Nitro Blue Tetrazolium) to an insoluble formazan compound [1] [2]. This system is particularly valued for its high sensitivity and the exceptional stability of its reaction product, which resists fading even when exposed to light, making it suitable for permanent record-keeping [2] [3]. When integrated into an ISH workflow, typically with anti-digoxigenin-AP conjugated antibodies, it allows for precise spatial localization of gene expression within fixed tissues and cells [4] [5].
The Enzymatic Reaction Mechanism
The generation of the characteristic dark-blue precipitate is a two-step process catalyzed by alkaline phosphatase. The reaction mechanism involves a redox reaction that culminates in the formation of an insoluble, visible product.
Reaction Steps
- Dephosphorylation of BCIP: The alkaline phosphatase enzyme hydrolyzes BCIP, cleaving its phosphate group. This reaction generates an intermediate product that is unstable and highly reactive [1].
- Reduction of NBT: The reactive reduced BCIP intermediate then acts as an electron donor, reducing the pale-yellow NBT (Nitro Blue Tetrazolium chloride). This reduction reaction converts NBT into an insoluble, dark-blue to purple compound known as NBT-formazan [1] [2].
- Precipitate Formation: The NBT-formazan precipitates at the site of enzymatic activity, providing a localized, visual signal that can be analyzed using brightfield microscopy [5] [3].
Chemical Pathway Diagram
The following diagram illustrates the logical sequence of the enzymatic reaction that leads to precipitate formation.
Application in In Situ Hybridization: A Detailed Protocol
The NBT/BCIP system is a key component in chromogenic detection for ISH. The following protocol outlines the major steps from sample preparation to imaging, with a focus on the detection phase.
Experimental Workflow for ISH with NBT/BCIP Detection
Detailed Detection and Development Protocol
The core detection procedure begins after the successful hybridization of a digoxigenin (DIG)-labeled probe and subsequent binding of an anti-DIG alkaline phosphatase conjugate [6].
Reagent Preparation: Prepare the NBT/BCIP working solution according to the manufacturer's instructions. Commercial kits are often supplied as ready-to-use tablets or liquid solutions containing separate reagents for NBT and BCIP [5] [1] [3]. A typical kit might contain:
- AP Reaction Buffer (e.g., 100 mL)
- BCIP (e.g., 1 mL)
- NBT (e.g., 1 mL) These components are combined to create the working substrate solution [1].
Color Development Reaction:
- Apply the NBT/BCIP working solution to the tissue sections, ensuring complete coverage.
- Incubate the slides in the dark at room temperature or 37°C. The development time can range from 20 minutes to several hours, and for low-abundance targets, incubation may be extended overnight for maximum sensitivity [3].
- Monitor the reaction progress microscopically at regular intervals (e.g., every 20-30 minutes) to prevent excessive background staining. The reaction should be stopped as soon as the specific signal is clearly visible and just before general background appears [6].
Stopping the Reaction: Once optimal signal development is achieved, stop the reaction by rinsing the slides thoroughly in distilled water [6].
Counterstaining and Mounting:
- Counterstaining: Apply a light counterstain to provide morphological context. Mayer's hematoxylin for 5-60 seconds is recommended, as dark counterstaining can mask the NBT/BCIP signal [6]. Other compatible counterstains include Vector Methyl Green and Nuclear Fast Red [5].
- Mounting: Mount slides with an aqueous-based mounting medium. Avoid xylene-containing mounting media (e.g., DPX), as they can cause crystal formation of the NBT/BCIP precipitate [5]. Use media like Crystalmount, Vectamount, or a custom glycerol gelatin medium for long-term preservation [5].
Troubleshooting Common Issues in NBT/BCIP-Based ISH
Even with a well-designed protocol, challenges can arise. The table below summarizes common problems and their solutions.
Table 1: Troubleshooting Guide for NBT/BCIP ISH
| Problem | Possible Cause | Recommended Solution |
|---|---|---|
| Weak or No Signal | Low target abundance, poor probe penetration, or degraded reagents. | Optimize probe concentration [4]; Ensure effective permeabilization [4]; Include a strong positive control [5]. |
| High Background | Non-specific probe binding, over-fixation, or insufficient washing. | Increase stringency of post-hybridization washes (e.g., higher temperature, lower salt) [4] [6]; Include blocking agents in hybridization buffer [4]; For lipid-rich tissues (e.g., heart), delipidize with chloroform before pre-hybridization [5]. |
| Brown-Purple vs. Blue Signal | High target abundance or suboptimal pH of detection buffer. | The color can vary with target abundance [5]. Ensure the detection reaction buffer is carefully adjusted to pH 9.5 [5]. For a deeper blue, consider alternative substrates like BM Purple [5]. |
| Uneven Staining | Inconsistent probe coverage or drying of sections during processing. | Apply probe evenly and use coverslips [4]; Use a properly sealed humidified chamber to prevent evaporation [4] [5]. |
The Scientist's Toolkit: Essential Reagents for NBT/BCIP-Based ISH
Successful execution of an ISH experiment with NBT/BCIP detection requires a suite of high-quality reagents. The following table details key solutions and their functions.
Table 2: Essential Research Reagent Solutions for NBT/BCIP ISH
| Category | Reagent | Function in the Protocol |
|---|---|---|
| Fixatives | 4% Paraformaldehyde (PBS) [4] | Preserves tissue architecture and nucleic acid integrity by cross-linking proteins. |
| Permeabilization & Wash Buffers | Phosphate Buffered Saline-Tween (PBST) [4], Proteinase K [4] | Detergents (Tween) and enzymes (Proteinase K) create pores in the tissue, allowing probe and antibody access to the target. |
| Blocking Agents | Bovine Serum Albumin (BSA), Casein, Denhardt's Solution [4] | Proteins and specialized solutions bind to non-specific sites, reducing background signal and improving signal-to-noise ratio. |
| Hybridization Buffers | Saline Sodium Citrate (SSC), Formamide [4] | Provides the ionic strength and chemical environment (e.g., with formamide) to control stringency and facilitate specific binding of the probe to its target. |
| Core Detection System | Alkaline Phosphatase Enzyme (e.g., in Anti-DIG-AP) [6] | The reporter enzyme that catalyzes the colorimetric reaction. |
| NBT/BCIP Substrate [1] [3] | The chromogenic substrate pair that is converted by AP into an insoluble, colored precipitate at the target site. | |
| Specialized Mounting Media | Glycerol Gelatin, Vectamount, Immunomount [5] | Aqueous-based media that preserve the NBT/BCIP precipitate without causing crystal formation, enabling long-term storage. |
The alkaline phosphatase-mediated reaction with NBT/BCIP substrates remains a fundamental and powerful technique for precise spatial localization of nucleic acids in biological samples. Its value in ISH research stems from the production of a highly stable, insoluble precipitate that provides excellent morphological context. Mastery of this technique involves not only a thorough understanding of the underlying enzymatic reaction but also careful optimization of the entire ISH workflow, from fixation through to final mounting. By adhering to detailed protocols and proactively troubleshooting common issues such as background staining and weak signal, researchers can reliably generate high-quality, reproducible data that advances our understanding of gene expression in its native context.
Within the framework of in situ hybridization (ISH) research, the NBT/BCIP staining protocol represents a cornerstone method for the precise cellular localization of gene expression. The robustness of this technique hinges on two core molecular components: digoxigenin (DIG)-labeled nucleic acid probes and the anti-DIG antibodies used for their detection. This application note details the roles, protocols, and reagent systems that enable highly sensitive and specific chromogenic detection of RNA or DNA targets within a histological context. The DIG/anti-DIG system provides a viable, high-resolution alternative to radioactive methods, facilitating large-scale molecular profiling at cellular resolution, as evidenced by its use in major projects like the Allen Brain Atlas [7].
Fundamental Concepts and Components
Digoxigenin (DIG): The Hapten Label
Digoxigenin is a small, plant-derived steroid hapten that is not naturally found in animal tissues, a property that minimizes non-specific background staining in biological samples [8] [9]. Its primary role is to serve as a label for nucleic acid probes (DNA, RNA, or oligonucleotides). These probes are synthesized by enzymatically incorporating DIG-conjugated nucleotides (such as DIG-11-dUTP for DNA or DIG-11-UTP for RNA) into the nucleic acid sequence [9]. A significant advantage of DIG labeling is that it does not interfere with the biological activity—specifically, the hybridization capacity—of the probe. Furthermore, probes labeled with DIG are stable for at least one year when stored at -20°C, providing flexibility for long-term research projects [9].
Anti-DIG Antibody: The Detection Conjugate
The anti-DIG antibody is the second critical component, responsible for specifically recognizing and binding to the DIG hapten attached to the hybridized probe. For chromogenic detection using the NBT/BCIP protocol, the antibody is typically conjugated to the enzyme alkaline phosphatase (AP) [10] [9]. The high specificity between the antibody and the DIG hapten results in an immunoassay system characterized by exceptionally low background noise [9]. Following the binding of the conjugate, the antibody-bound AP enzyme catalyzes the subsequent colorimetric reaction.
The NBT/BCIP Detection Mechanism
The detection of the DIG-labeled hybrids via the anti-DIG-AP conjugate is achieved through a chromogenic reaction. The alkaline phosphatase enzyme dephosphorylates the substrate 5-Bromo-4-chloro-3-indolyl-phosphate (BCIP), leading to the formation of an intermediate compound [10]. This intermediate, upon dimerization, releases hydrogen ions that reduce the second substrate, Nitroblue Tetrazolium (NBT), into an insoluble, intracellularly deposited purple compound known as NBT Diformazan [10]. This precipitate is visually detectable and stable under standard mounting media, allowing for permanent record-keeping and high-resolution digital imaging [7].
The following diagram illustrates the logical sequence and relationships in this detection system.
Experimental Protocols
Protocol 1: Synthesis of DIG-Labeled RNA Probes
The generation of high-quality riboprobes is a critical first step for a successful ISH experiment. The following table summarizes a standard protocol adapted from optimized methods [7] [11].
Table 1: Protocol for DIG-Labeled RNA Probe Synthesis
| Step | Component | Volume/Amount | Final Concentration/Note |
|---|---|---|---|
| 1. Template Prep | cDNA template (in vector) | 1 μg total | Flanked by T3/T7 RNA polymerase promoters [7]. |
| Restriction Enzyme (e.g., BssHII) | As per mfr. | Linearize plasmid downstream of insert. | |
| Purification | Post-digestion | Purify via phenol-chloroform or column [11]. | |
| 2. Transcription | DIG RNA Labeling Mix (10X) | 1.0 μL | 1X; contains DIG-11-UTP [7]. |
| Transcription Buffer (10X) | 1.0 μL | 1X | |
| Recombinant RNasin | 0.5 μL | 2 U/μL; inhibits RNases. | |
| BSA (20μg/μL) | 0.5 μL | 1 μg/μL | |
| DTT (100 mM) | 0.5 μL | 10 mM | |
| T3 or T7 RNA Polymerase | 1.0 μL | ~1.7 U/μL | |
| DEPC-treated H2O | To 10.0 μL | ||
| 3. Purification | Sephadex G-50 Column | ~250 μL | Pre-equilibrated in TE Buffer [7]. |
Procedure Summary: Combine all components from Step 2 in the order listed, incubate at 37°C for 2 hours. Purify the transcribed RNA probe from unincorporated nucleotides using a Sephadex G-50 size-exclusion column. Determine the yield and quality of the probe via spectrophotometry or gel electrophoresis before aliquoting and storing at -80°C [7].
Protocol 2: High-Throughput ISH with NBT/BCIP Detection
This optimized protocol is designed for cellular-resolution labeling on cryosections with low background and is scalable for processing large numbers of slides [7].
Table 2: High-Throughput ISH and NBT/BCIP Staining Protocol
| Step | Reagent/Solution | Conditions | Purpose |
|---|---|---|---|
| 1. Pre-hybridization | Acetylation Solution (TEA + Acetic Anhydride) | Freshly prepared, 10 min [7]. | Reduces non-specific probe binding to tissues. |
| Hybridization Buffer (50% Formamide, 2X SSPE, tRNA, BSA) | Apply to sections, 62°C [7] [10]. | Creates optimal hybridization environment. | |
| 2. Hybridization | DIG-labeled Riboprobe in Hybridization Buffer | Add to sections, incubate overnight at 62°C. | Allows probe-target mRNA hybridization. |
| 3. Post-Hybridization Washes | Wash Buffer I (50% Formamide, 2X SSPE) | 62°C, 30 min [7]. | High-stringency wash to remove unbound probe. |
| Wash Buffer II (0.1X SSPE) | Room temperature, 5 min [7]. | Lower-stringency final wash. | |
| 4. Immunological Detection | Anti-DIG-AP, Fab fragments | 1:2000 dilution in blocking buffer, 2-4 hrs [10]. | Binds specifically to DIG label on hybridized probe. |
| 5. Chromogenic Development | NBT/BCIP Substrate | In 0.1 M Tris-HCl, 0.1 M NaCl, pH 9.5. | Incubate in dark until signal develops (no background). |
| 6. Mounting | Aqueous Mounting Media (e.g., VectaMount AQ) | - | Preserves stain; some media cause fading [7]. |
The complete workflow, from tissue preparation to final imaging, is visualized below.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of the NBT/BCIP staining protocol requires a suite of specific, high-quality reagents. The following table catalogs the essential materials for this field.
Table 3: Key Research Reagent Solutions for DIG-based ISH
| Reagent / Kit | Function / Role | Example Product / Note |
|---|---|---|
| DIG RNA Labeling Mix | Provides DIG-11-UTP for efficient incorporation during in vitro transcription. | Roche Cat. No. 11277073910 [7]. |
| Anti-Digoxigenin-AP, Fab fragments | High-affinity antibody conjugate for detection; Fab fragments minimize background. | Roche Cat. No. 11093274910 [7]. |
| NBT/BCIP Stock Solution | Ready-to-use chromogenic substrate for alkaline phosphatase. | Perkin Elmer Cat. No. NEL937 [7]. |
| RNA Polymerase (T3, T7) | For synthesizing RNA probes (riboprobes) from a DNA template. | Use high-concentration versions for best yield [7]. |
| Blocking Reagent | Reduces non-specific binding of the antibody conjugate. | Provided in Roche blocking solution or use skim milk [7] [10]. |
| Aqueous Mounting Media | Preserves the NBT/BCIP precipitate for long-term storage. | VectaMount AQ recommended to prevent fading [7]. |
| DNase/RNase-Free Water | Prevents degradation of probes and target RNA throughout the protocol. | Certified nuclease-free or DEPC-treated H2O [7]. |
Advanced Applications: q2PISH for Quantitative Analysis
A significant advancement in this field is the development of qualitative and quantitative PISH (q2PISH), a method that allows for both the qualitative assessment of culture heterogeneity and the quantitative measurement of gene expression with single-cell resolution [10]. This protocol innovatively leverages the robustness of the alkaline phosphatase enzyme conjugated to the anti-DIG antibody. After hybridization and antibody binding, the AP enzyme is first used to catalyze a soluble chromogenic reaction using p-Nitrophenyl Phosphate (pNPP), the supernatant of which can be measured spectrophotometrically at 405 nm for quantitative data [10]. Following this, without inactivating the AP enzyme, the same cells are subjected to the standard NBT/BCIP reaction to generate the insoluble purple precipitate for qualitative, cellular-resolution imaging. A final nuclear stain (e.g., To-PRO-3) is used to count total cell numbers, enabling the normalization of quantitative pNPP data to a "per cell" basis [10]. This dual-substrate approach overcomes the limitations of cell-pooling methods like RT-PCR, preventing the masking of variations in heterogeneous cell populations.
Chromogenic detection, particularly using substrates like Nitro-Blue Tetrazolium/5-Bromo-4-Chloro-3-Indolyl Phosphate (NBT/BCIP), is a cornerstone technique in in situ hybridization (ISH) for locating specific nucleic acid sequences within cells and tissues. This method utilizes enzyme-labeled probes (typically alkaline phosphatase, ALP) that catalyze a colorimetric reaction, producing an insoluble, stable precipitate at the site of target binding [12]. Within the framework of a broader thesis on ISH protocols, this document details the significant advantages of chromogenic detection—robustness, permanence, and accessibility—and provides detailed application notes to ensure experimental success for researchers, scientists, and drug development professionals.
Core Advantages of Chromogenic Detection
Chromogenic detection offers a suite of benefits that make it exceptionally valuable for both research and diagnostic applications. The table below summarizes its core advantages and the underlying mechanisms.
Table 1: Core Advantages of Chromogenic Detection Systems
| Advantage | Technical Basis | Practical Implication for Research |
|---|---|---|
| Robustness | Formalin-fixed, paraffin-embedded (FFPE) tissue compatibility [12] | Enables analysis of archived clinical samples, facilitating longitudinal and retrospective studies. |
| High resistance to quenching during tissue processing [12] | Consistent results under standard histology protocols; minimal interference from complex sample preparation. | |
| Permanence | Formation of an insoluble, non-fading precipitate (e.g., from NBT/BCIP) [12] | Slides can be stored permanently and re-evaluated years later; ideal for clinical records and legal evidence. |
| Compatibility with permanent counterstains (e.g., Mayer's hematoxylin) and mounting media [12] | Creates a durable, high-contrast permanent record of the cellular context and hybridization signal. | |
| Accessibility | Standard bright-field microscopy for visualization [12] | No need for specialized, costly fluorescence microscopes; accessible to most laboratories and clinics. |
| Clear cellular localization superior to immunofluorescence in many cases [12] | Easier for pathologists to identify specific positive cell types and correlate morphology with signal. |
Quantitative Comparison with Fluorescent Detection
Choosing a detection method involves trade-offs. The following table provides a direct comparison to guide protocol selection.
Table 2: Chromogenic vs. Fluorescent Detection for ISH
| Characteristic | Chromogenic (e.g., NBT/BCIP/ALP) | Fluorescent (e.g., FITC/Fluorophore) |
|---|---|---|
| Signal Permanence | High (years) [12] | Low (prone to photobleaching) |
| Sensitivity | High, amplifiable | High |
| Multiplexing Potential | Lower (typically 1-2 targets with careful color separation) | Higher (multiple targets with different fluorophores) |
| Equipment Needs | Standard bright-field microscope | Fluorescence microscope with specific filter sets |
| Compatibility with FFPE | Excellent [12] | Excellent, but may require antigen retrieval |
| Cellular Resolution | Excellent for morphological correlation [12] | Can be difficult to correlate with complex morphology |
| Quantitative Analysis | Semi-quantitative (SQ); prone to human variability [13] | More amenable to digital quantitation (e.g., AI-driven methods) [13] |
Detailed NBT/BCIP-ISH Protocol
This protocol is designed for detecting mRNA or DNA targets in FFPE tissue sections using an ALP-conjugated probe and NBT/BCIP chromogen.
Materials and Reagent Solutions
Table 3: Research Reagent Solutions for NBT/BCIP-ISH
| Reagent/Material | Function/Description | Example/Note |
|---|---|---|
| FFPE Tissue Sections | Sample substrate for ISH. | 4-5 µm thickness mounted on charged slides [12]. |
| ALKALINE PHOSPHATASE (ALP)-Conjugated Probe | Binds specifically to the target nucleic acid sequence. | Diluted in 1% BSA-PBS; avoid repeated freeze-thaw cycles [12]. |
| NBT/BCIP Substrate | Chromogenic substrate for ALP. Produces an insoluble purple/black precipitate. | Ready-to-use solutions are commercially available. |
| Proteinase K or Pepsin | For epitope/target retrieval to expose nucleic acids. | Concentration and time are target- and fixation-dependent. |
| Hybridization Buffer | Optimal environment for specific probe-target binding. | Contains salts, Denhardt's solution, dextran sulfate, formamide. |
| Blocking Solution | Reduces non-specific background staining. | 1-2% BSA or normal serum in PBS. |
| Detection Kit (Polymer-based) | Increases sensitivity and reduces background vs. traditional methods. | e.g., HRP- or ALP-labeled polymers (Simple Stain Max, Novolink) [12]. |
| Mayer's Hematoxylin | Nuclear counterstain (blue). | Briefly apply (e.g., 10 seconds) after chromogenic reaction [12]. |
Step-by-Step Experimental Workflow
Title: NBT/BCIP ISH Experimental Workflow
Procedure:
- Dewaxing and Rehydration: Deparaffinize FFPE sections in xylene (or substitute) and rehydrate through a graded ethanol series (100%, 95%, 70%) to distilled water.
- Target Retrieval: Digest sections with Proteinase K (e.g., 10-20 µg/mL) or pepsin in a pre-warmed humidified chamber for 5-15 minutes at 37°C to expose target sequences. Note: Optimal concentration and time must be determined empirically to balance signal with tissue morphology.
- Pre-hybridization: Apply pre-warmed hybridization buffer to sections and incubate for 15-30 minutes at the probe's specific hybridization temperature in a humidified chamber to reduce non-specific binding.
- Hybridization: Apply the ALP-conjugated probe in hybridization buffer to the tissue. Cover with a parafilm or coverslip to prevent evaporation. Incubate in a humidified chamber overnight (12-16 hours) at the appropriate hybridization temperature.
- Stringency Washes: The following day, carefully remove coverslips and wash slides in pre-warmed SSC buffer (e.g., 2x SSC, 0.1x SSC) at a defined temperature to remove unbound and mismatched probe.
- Blocking: Apply blocking solution (e.g., 2% BSA in PBS) for 30-60 minutes at room temperature to block non-specific sites.
- Detection System Application: If using an indirect detection system (e.g., a primary probe followed by a secondary ALP-conjugated antibody), apply the subsequent layers with appropriate washes in between. For direct detection with an ALP-conjugated probe, proceed to the next step.
- Chromogenic Reaction (NBT/BCIP): Apply the ready-to-use NBT/BCIP substrate solution to the tissue section. Incubate in the dark at room temperature and monitor development under a microscope periodically (from 10 minutes to 2 hours). Stop the reaction by immersing the slides in distilled water when the desired signal-to-noise ratio is achieved.
- Counterstaining and Mounting: Counterstain briefly with Mayer's hematoxylin (e.g., 10-30 seconds) to visualize nuclei. Rinse in tap water, bluing if necessary. Aqueous mounting medium is recommended to preserve the chromogen. Coverslip for permanent storage [12].
Visualizing the Chromogenic Reaction Pathway
The high sensitivity and clear signal of the NBT/BCIP system are due to its robust enzymatic amplification process, visualized below.
Title: NBT/BCIP Chromogenic Reaction Mechanism
Troubleshooting and Pitfalls
Even robust protocols can encounter issues. The table below addresses common challenges in chromogenic ISH.
Table 4: Troubleshooting Guide for NBT/BCIP-ISH
| Problem | Potential Cause | Solution |
|---|---|---|
| High Background | Inadequate blocking or non-specific probe binding. | Optimize blocking solution (BSA concentration, add normal serum). Increase stringency of washes (temperature, salt concentration). |
| Over-digestion during target retrieval. | Titrate Proteinase K concentration and incubation time. | |
| Excessive development time with NBT/BCIP. | Monitor reaction microscopically and stop promptly. | |
| Weak or No Signal | Under-digestion during target retrieval. | Increase Proteinase K concentration or incubation time within limits that preserve morphology. |
| Probe degradation or inactivation. | Aliquot and store probes correctly; avoid repeated freeze-thaw cycles; use fresh dilution [12]. | |
| Low target abundance. | Consider using signal amplification systems (e.g., tyramide signal amplification) if sensitivity is critical [12]. | |
| Poor Morphology | Over- or under-fixation of original tissue. | Standardize fixation protocols. Excessive protease digestion. Optimize target retrieval step. |
A significant consideration in modern research is the semi-quantitative (SQ) nature of chromogenic analysis. Traditional SQ scoring by a pathologist, while efficient, is subject to human bias and cannot discriminate subtle changes [13]. For quantitative studies, chromogenic results can be analyzed using digital pathology strategies like positive pixel quantitation or more advanced AI-driven cellular density quantitation, which offer higher accuracy and reproducibility [13].
NBT/BCIP (Nitro-blue tetrazolium chloride/5-Bromo-4-chloro-3'-indolyphosphate p-toluidine salt) serves as a fundamental chromogenic substrate system for detecting alkaline phosphatase (AP) enzyme activity in molecular biology applications [14] [15]. This combination produces an insoluble, dark purple-blue precipitate at the site of probe hybridization, enabling precise localization of nucleic acid targets within tissue architecture [14]. The high sensitivity and resolution of NBT/BCIP make it particularly valuable for in situ hybridization (ISH) techniques, where it facilitates the visualization of gene expression patterns while maintaining excellent cellular and tissue morphology [16].
The chemical reaction mechanism involves BCIP dephosphorylation by alkaline phosphatase, followed by oxidation and dimerization of the resulting indoxyl derivative. This reduced compound subsequently reduces the yellow, water-soluble NBT to an insoluble, blue-purple formazan precipitate via an oxidation-reduction reaction [14] [17]. The robust, water-insoluble nature of this precipitate ensures minimal diffusion artifacts, providing precise spatial localization that is essential for accurate interpretation of gene expression patterns in complex tissues [14].
Ideal Applications of NBT/BCIP Staining
Application Spectrum Across Sample Types
NBT/BCIP staining demonstrates remarkable versatility across diverse experimental preparations and sample types. The table below summarizes its optimal applications in biomedical research:
Table 1: Application Spectrum of NBT/BCIP Staining Across Sample Types
| Sample Type | Ideal Applications | Key Advantages | Technical Considerations |
|---|---|---|---|
| Whole-Mount Embryos (e.g., Drosophila) | Gene expression patterning in developmental studies [18] | Preserves 3D architecture while localizing transcript expression [18] | Requires extended permeabilization; optimal for qualitative spatial analysis [18] |
| FFPE Tissue Sections | Diagnostic pathology and archival tissue analysis [6] [19] | Excellent morphological preservation; compatible with clinical archives [19] | Requires antigen retrieval and protease digestion optimization [6] [4] |
| Cryosections | RNA localization in lipid-rich tissues [17] | Superior probe accessibility to targets [17] | May require delipidization with chloroform for high lipid content tissues [17] [15] |
| Membrane Blotting | Northern, Southern, and Western blot detection [14] [15] | High sensitivity and resolution for nucleic acid or protein detection [15] | Ready-to-use formulations available; development typically within 30 minutes [14] |
Quantitative and Qualitative Analysis Applications
The application of NBT/BCIP extends beyond simple localization to include sophisticated analytical approaches:
Semi-quantitative Gene Expression Analysis: Research demonstrates that with proper standardization, NBT/BCIP signal intensity can be used for relative quantification of transcript abundance. Automated image segmentation algorithms can identify contiguous pixel groups corresponding to BCIP/NBT precipitate, enabling computational analysis of expression levels [16].
Multiplexing Approaches: While traditionally used for single-plex detection, NBT/BCIP can be combined with other detection methods in sequential protocols. The technique has been successfully integrated with immunofluorescence (IF-FISH) to simultaneously visualize both protein and mRNA expression within the same cell type, providing reliable quantification of mRNA expression levels while maintaining spatial context [18].
Genome-Scale Mapping: Large-scale projects like the Allen Brain Atlas have utilized NBT/BCIP-based ISH for genome-wide expression mapping in the mouse brain. These efforts leverage the high-throughput suitability of colorimetric ISH while developing computational methods for standardized relative quantification across multiple structures and genes [16].
Comprehensive Experimental Protocols
Core ISH Protocol with NBT/BCIP Detection
The following workflow outlines the standardized procedure for NBT/BCIP-based in situ hybridization across multiple sample types:
Diagram 1: Core ISH workflow with NBT/BCIP detection
Fixation and Permeabilization
Fixation Protocol: For most tissues, fix with 4% paraformaldehyde in PBS for 15-24 hours at 4°C [4]. Consistent fixation conditions are critical, as both under-fixation and over-fixation can dramatically impact results. Under-fixation may cause nucleic acid degradation, while over-fixation can reduce probe accessibility and increase background staining [19].
Permeabilization Optimization: Treat samples with Proteinase K (typically 1-10 µg/mL for 3-30 minutes at 37°C) [6]. The optimal concentration and duration must be determined empirically for each tissue type. Over-digestion can weaken or eliminate the ISH signal and prevent counterstaining of cell nuclei, while under-digestion may also decrease or eliminate the specific signal [6].
Hybridization and Stringency Washes
Probe Hybridization: Apply digoxigenin-labeled riboprobes diluted in hybridization buffer and incubate overnight (16-18 hours) at 37-45°C in a humidified chamber [4]. A standard hybridization buffer formulation includes 50% formamide, 1× SSC, 50 µg/mL heparin, 100 µg/mL denatured salmon sperm DNA, 1% SDS, and 0.1% Tween-20 [4].
Stringency Washes: Perform post-hybridization washes with SSC buffer at elevated temperatures (75-80°C) to remove non-specifically bound probe [6]. Increase the temperature by 1°C per slide when processing multiple slides, but do not exceed 80°C to preserve signal [6]. Inadequate stringency washing is a common cause of high background staining.
Immunodetection and Color Development
Antibody Incubation: Incubate samples with anti-digoxigenin-alkaline phosphatase conjugate (typically diluted 1:500-1:5000) for 30-60 minutes at 37°C [6] [18]. Thorough washing with PBST (PBS with 0.025% Tween-20) after antibody incubation is essential to reduce non-specific background [6].
NBT/BCIP Development: Prepare the NBT/BCIP substrate in alkaline phosphatase detection buffer (pH 9.5) and apply to tissues. Monitor development microscopically at 2-minute intervals [6]. The reaction typically produces a visible signal within 5-15 minutes at 37°C [6]. Stop the reaction by rinsing in distilled water once specific signal is optimal or when background begins to appear.
Specialized Protocols for Different Sample Types
Whole-Mount ISH for Embryonic Structures
For complex tissues like the Drosophila lymph gland, whole-mount ISH enables analysis of gene expression within a three-dimensional context [18]. This approach is particularly valuable for studying small stem cell compartments, such as the haematopoietic niche consisting of only 40-45 cells, where isolation and quantitative analysis would otherwise be difficult and prone to error [18].
The whole-mount protocol incorporates tyramide signal amplification (TSA) to enhance sensitivity for detecting low-abundance transcripts [18]. Following hybridization and antibody incubation, samples are treated with fluorescently labeled tyramide substrates, then developed with NBT/BCIP for chromogenic detection. This combination allows for both quantification of expression levels and precise spatial localization [18].
FFPE Tissue Section Processing
For formalin-fixed, paraffin-embedded (FFPE) tissues, additional steps are required to overcome protein-nucleic acid crosslinking induced by fixation:
Dewaxing and Rehydration: Thoroughly remove paraffin with xylene substitutes and hydrate through graded ethanol series to water [19]. Incomplete dewaxing can produce unstained or poorly stained areas in sections.
Heat-Induced Epitope Retrieval: Heat sections for 15 minutes starting from the time the pretreatment buffer reaches 98°C [6]. This step is crucial for breaking crosslinks and exposing target nucleic acids.
Enzyme Digestion Optimization: Between 3-10 minutes of pepsin digestion at 37°C is recommended for most tissues [6]. Prevent evaporation during this step, as drying artifacts can cause significant problems.
Troubleshooting and Optimization Guide
Common Technical Challenges and Solutions
Table 2: Troubleshooting Common NBT/BCIP Staining Issues
| Problem | Potential Causes | Recommended Solutions | Preventive Measures |
|---|---|---|---|
| Weak or Absent Signal | Poor probe penetration [4], target degradation [19], insufficient probe concentration [17] | Optimize permeabilization [4]; Check RNA integrity [4]; Test higher probe concentrations (1-8 µL per 50 µL hybridization solution) [17] | Fix tissue promptly after collection [19]; Include strong positive control [17] |
| High Background Staining | Inadequate stringency washing [6], over-fixation [15], section drying [17] | Increase stringency wash temperature (75-80°C) [6]; Add blocking agents (COT-1 DNA, salmon sperm DNA) [6]; Ensure sections remain hydrated [17] | Include Denhardt's solution in hybridization buffer [4]; Use PBST instead of PBS for washes [6] |
| Brown-Purple Instead of Blue Signal | High target abundance [17] [15], suboptimal detection pH [17] | Adjust pH of detection solution to precisely 9.5 [17] [15]; For abundant targets, consider shorter development time [17] | Optimize development time with microscopic monitoring [6]; Use BM Purple for deeper blue signals [17] |
| Uneven Staining | Incomplete coverage with reagents [4], bubbles on section surface [19], uneven dewaxing [19] | Apply probes and antibodies evenly; Ensure full coverage of sample [4]; Take particular care with dewaxing and hydration [19] | Use coverslips and properly sealed humidified chamber [4]; Avoid protein-based section adhesives [19] |
Signal Optimization and Validation
Color Development Control: The color of NBT/BCIP precipitate can vary from blue to brown or purple depending primarily on the abundance of target mRNA in the tissue [17] [15]. This color variation is also influenced by probe length and labeling intensity, with more abundant targets generally producing deeper blue color precipitates [17].
Counterstain Selection: NBT/BCIP signals are not compatible with classical counterstains that require xylene-containing mounting media [17] [15]. Recommended counterstains include Nuclear Fast Red or Methyl Green, which are compatible with aqueous mounting media [17] [15]. Avoid dark counterstaining with hematoxylin, which can mask the brown DAB product and dark blue NBT/BCIP signals [6].
Mounting Considerations: Never embed slides with NBT/BCIP signals in xylene-based mounting media because these could lead to crystal formations of the color precipitates [17] [15]. Instead, use specifically formulated mounting media such as Crystalmount, Vectamount, or Immunomount [17]. A customer-recommended glycerol gelatin mounting medium can preserve signals for several years without fading [17].
Research Reagent Solutions
Essential Materials for NBT/BCIP-Based ISH
Table 3: Key Research Reagents for NBT/BCIP ISH Applications
| Reagent Category | Specific Examples | Function & Application Notes |
|---|---|---|
| Probe Labeling Systems | DIG RNA Labeling Kit (SP6/T7) [18] | Generates digoxigenin-labeled riboprobes for high-sensitivity target detection |
| Detection Enzymes | Anti-DIGOXIGENIN-AP conjugate [18] | Antibody conjugate that binds digoxigenin-labeled probes for enzymatic detection |
| Signal Amplification | Tyramide Signal Amplification (TSA) Kit [18] | Enhances sensitivity for low-abundance targets; essential for whole-mount ISH |
| Chromogenic Substrates | NBT/BCIP ready-to-use tablets [17] or solution [15] | Produces insoluble blue-purple precipitate at sites of probe hybridization |
| Blocking Reagents | Denhardt's solution [4], salmon sperm DNA [4], COT-1 DNA [6] | Reduces non-specific probe binding to minimize background staining |
| Hybridization Buffers | Formamide-based buffer with SSC, heparin, SDS [4] | Optimizes hybridization stringency and probe specificity |
| Mounting Media | Vectashield [18], Vectamount [17], custom glycerol gelatin [17] | Preserves signal without crystal formation; compatible with NBT/BCIP precipitate |
Advanced Applications and Integration
Correlation with Genomic-Scale Data
Advanced computational methods now enable quantitative analysis of NBT/BCIP-based ISH data for cross-platform correlation studies. Research demonstrates that with proper normalization, colorimetric ISH signal can be correlated with microarray expression data, despite differences in dynamic range and probe design [16].
The methodology involves automated image segmentation algorithms that identify contiguous pixel groups corresponding to BCIP/NBT precipitate, enabling the definition of expression levels normalized to cell density in each region [16]. This approach facilitates large-scale cross-platform expression level comparisons, bridging the gap between anatomical localization and genomic-scale expression profiling.
Future Directions and Methodological Integration
The application of NBT/BCIP continues to evolve with emerging methodological integrations:
Multiplexed Detection Systems: Combining NBT/BCIP with other detection modalities, such as immunofluorescence (IF-FISH), enables simultaneous visualization of both protein and mRNA expression within the same cell type [18]. This powerful technique provides reliable quantification of mRNA expression levels while maintaining spatial context and protein co-localization information.
Quantitative Whole-Mount Analysis: In specialized applications such as Drosophila haematopoietic organ analysis, NBT/BCIP-based IF-FISH allows for densitometric quantitative measurement of transcripts within specific immunolabeled cell populations [18]. This approach is particularly valuable when dealing with small cell compartments embedded within complex organs.
Computational Image Analysis: Advanced image analysis algorithms now enable standardized relative quantification of colorimetric ISH signals, making NBT/BCIP-based detection suitable for genome-scale expression mapping projects [16]. These developments highlight the continuing relevance of this established methodology in the era of high-throughput spatial transcriptomics.
Step-by-Step NBT/BCIP Staining Protocol: From Sample to Signal
In situ hybridization (ISH) is a powerful technique for detecting specific nucleic acid sequences within histologic sections, cells, or whole-mount tissues, providing crucial spatial context for gene expression analysis. Within the broader context of a thesis on NBT/BCIP (nitro-blue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate) staining protocols for ISH research, the pre-hybridization phase emerges as the most critical determinant of experimental success. These initial steps—fixation, permeabilization, and blocking—establish the foundation upon which all subsequent procedures rely, ultimately determining the sensitivity, specificity, and interpretability of the final results. Proper execution of these stages preserves tissue architecture and nucleic acid integrity while enabling specific probe access and binding, minimizing non-specific background, and ensuring robust chromogenic development with NBT/BCIP, which produces an insoluble purple precipitate easily visualized by light microscopy [20] [21]. This protocol details the optimized methods for these essential pre-hybridization steps, framed specifically for researchers employing NBT/BCIP detection in their ISH investigations.
Theoretical Framework: The Science Behind Pre-Hybridization
The pre-hybridization cascade functions as an integrated system where each step establishes the necessary conditions for the next. Fixation serves the dual purpose of preserving morphological structure and immobilizing target nucleic acids within their native cellular environments. Inadequate fixation leads to rapid RNA degradation by endogenous nucleases, while over-fixation creates excessive cross-linking that impedes probe penetration [19]. Permeabilization strategically compromises cellular barriers without destroying structural integrity, creating portals for probe entry. The final blocking step conditions the molecular landscape to favor specific over non-specific interactions during hybridization. When optimized for NBT/BCIP detection, this trilogy of steps ensures that the alkaline phosphatase-conjugated antibodies used in later detection phases bind specifically to their target haptens (typically digoxigenin or fluorescein), resulting in precise, localized deposition of the NBT/BCIP reaction product with minimal diffusion artifact or background staining [20] [21].
Materials and Reagents
Research Reagent Solutions
The following table details essential reagents required for the pre-hybridization steps, with specific notes on their application in NBT/BCIP-based detection systems.
Table 1: Essential Reagents for Pre-Hybridization Steps
| Reagent Category | Specific Examples | Function & Application Notes |
|---|---|---|
| Fixatives | 4% Paraformaldehyde (PBS or phosphate buffer) [4], Formalin (10%) [4] | Preserves tissue morphology and nucleic acid integrity; consistent fixation conditions are critical for reproducible NBT/BCIP results [19]. |
| Permeabilization Agents | Proteinase K [4] [20], Detergents (Tween-20, Triton X-100) [4], Acetone [20], Xylenes [21] | Disrupts cellular membranes to allow probe access; concentration and time must be carefully optimized for each tissue type [21]. |
| Blocking Agents | Bovine Serum Albumin (BSA) [4], Normal Sheep Serum [20], Heparin [20], Yeast tRNA [22] [20], Salmon Sperm DNA [4] | Reduces non-specific probe and antibody binding, decreasing background in the final NBT/BCIP stain [4] [20]. |
| Buffers | Phosphate Buffered Saline (PBS) [4], Saline Sodium Citrate (SSC) [4], Tris Buffered Saline (TBS) [4], Pre-hybridization Buffer [22] | Maintains pH and ionic strength; SSC concentration affects stringency in post-hybridization washes [4]. |
Methodologies: Pre-Hybridization Protocol
Step I: Tissue Preparation and Fixation
The fixation process begins immediately following tissue collection to prevent RNA degradation.
- Tissue Collection: Handle specimens carefully and transfer promptly to fixative. Cold ischemia time should be minimized to limit RNA degradation by endogenous RNases [19].
- Fixative Solution: Prepare 4% paraformaldehyde in phosphate-buffered saline (PBS). For zebrafish embryos or Drosophila ovaries, fixation is typically performed for 2-24 hours depending on tissue size, often at 4°C [20] [21].
- Fixation Protocol: Immerse tissue in sufficient volume of fixative (approximately 10:1 fixative-to-tissue ratio). For paraffin-embedded samples, ensure complete deparaffinization using xylene substitutes and ethanol gradients before fixation [4].
- Post-Fixation Processing: Cryoprotect fixed tissues in 20% sucrose in PBS (DEPC-treated) for at least 24 hours at 4°C before embedding in optimal cutting temperature (OCT) compound for frozen sections [22]. For paraffin embedding, process through ethanol gradients and xylene using standardized protocols.
Critical Considerations for NBT/BCIP Staining: Consistent fixation conditions (time, temperature, pH) are paramount. Under-fixed tissues exhibit poor morphology and nucleic acid loss, while over-fixed tissues resist permeabilization, resulting in weak or false-negative NBT/BCIP signals [19].
Step II: Sectioning and Slide Preparation
- Sectioning: Cut thin, uniform sections (5-15 μm) using a cryostat (for frozen sections) or microtome (for paraffin sections). For frozen sections, optimal cryostat chamber temperature is typically -25°C with specimen temperature of -18°C to -20°C [22].
- Slide Selection: Use charged slides to ensure section adhesion throughout the stringent ISH procedure [19].
- Section Drying: Air-dry sections thoroughly at room temperature. Avoid using protein-based adhesives in flotation baths as they can block charged slide surfaces and cause uneven reagent pooling [19].
- Storage:
- Frozen Sections: Store at -80°C.
- Paraffin Sections: Store at room temperature, protected from dust.
Step III: Permeabilization Strategies
Permeabilization must be tailored to the tissue type and fixation method. The goal is to allow probe penetration while preserving tissue morphology and RNA integrity.
Standard Proteinase K Treatment:
- Prepare a working solution of Proteinase K (e.g., 10-50 μg/mL in PBTween) [20] [21].
- Incubate sections for 5-60 minutes at room temperature. Optimal concentration and time must be determined empirically for each tissue and fixation type [21].
- Terminate digestion by rinsing in PBS and post-fixing in 4% paraformaldehyde for 10-20 minutes.
Alternative Permeabilization Methods:
- Detergent-Based: Use RIPA buffer or solutions containing Tween-20 or Triton X-100 (0.1-1%) [4] [21].
- Solvent-Based: Treat with 80% acetone/20% water for 20 minutes at room temperature [20] or xylenes followed by ethanol gradients [21].
- Combined Approaches: For challenging tissues like Drosophila ovaries, a combination of xylenes and detergent-based permeabilization may be optimal, especially when performing subsequent protein immunofluorescence [21].
Optimization for NBT/BCIP: Excessive permeabilization damages morphology, while insufficient treatment causes weak staining. For protocols combining protein immunofluorescence with RNA FISH (IF/FISH), avoid Proteinase K as it degrades protein epitopes; instead use alternative methods like xylenes and detergents [21].
Step IV: Pre-Hybridization Blocking
Blocking minimizes non-specific binding of probes and detection antibodies, a common source of high background in NBT/BCIP staining.
Pre-hybridization Buffer Formulation: Prepare a blocking buffer containing:
- 50% Formamide (denaturing agent that lowers hybridization temperature) [22] [20]
- 5X SSC (provides appropriate ionic strength) [22]
- Blocking Agents: Include heparin (50 μg/mL), yeast tRNA (50-100 μg/mL), and/or denatured salmon sperm DNA (100 μg/mL) to compete for non-specific binding sites [4] [20].
- Detergent: 0.1% Tween-20 [20].
- Volume Exclusion Agents (Optional): Dextran sulfate (5-10%) or polyvinyl alcohol (PVA, 10%) can be added to occupy solvent space and concentrate reactants, potentially reducing stain time and non-specific background [22] [20].
Blocking Protocol:
- Apply pre-warmed pre-hybridization buffer to completely cover tissue sections.
- Incubate at 37-45°C for 30-60 minutes in a humidified chamber to prevent evaporation [4].
- Do not rinse after blocking; proceed directly to probe application.
Workflow Integration and Visualization
The following workflow diagram integrates the critical pre-hybridization steps into the broader context of an ISH protocol utilizing NBT/BCIP detection, highlighting their pivotal role in determining experimental outcomes.
Figure 1: ISH workflow highlighting critical pre-hybridization steps and their impact on NBT/BCIP results.
Troubleshooting and Optimization
Even carefully executed protocols may require optimization. The table below outlines common problems arising from suboptimal pre-hybridization steps and their solutions.
Table 2: Troubleshooting Guide for Pre-Hybridization Steps
| Problem | Potential Causes | Corrective Actions |
|---|---|---|
| High Background Staining | Inadequate blocking [4]; insufficient post-hybridization washes [4]; probe concentration too high [4]. | Increase stringency of washes (higher temperature, lower SSC) [4]; include additional blocking agents (salmon sperm DNA, tRNA) in hybridization buffer [4]; consider acetylation step to block charged amines [4]. |
| Weak or No Signal | Insufficient permeabilization [4]; over-fixation [19]; nucleic acid degradation [19]; low probe concentration or quality [4]. | Optimize Proteinase K concentration and time [4] [21]; ensure prompt fixation after tissue collection [19]; check RNA integrity [4]; verify probe quality and concentration [4]. |
| Uneven Staining | Incomplete dewaxing [19]; uneven section drying [19]; evaporation during hybridization [19]; bubbles on section [19]. | Ensure complete paraffin removal [19]; use charged slides and dry sections thoroughly [19]; use properly sealed humidified chamber [19] [4]; apply probes and reagents evenly [4]. |
| Poor Tissue Morphology | Over-permeabilization [21]; improper fixation [19]; careless tissue handling. | Reduce Proteinase K concentration/time [21]; optimize fixation conditions (time, temperature) [19]; handle tissue specimens carefully during dissection and processing [19]. |
The pre-hybridization trilogy of fixation, permeabilization, and blocking constitutes the critical foundation upon which reliable and interpretable ISH data is built, particularly when using NBT/BCIP chromogenic detection. These steps collectively determine the accessibility of the target nucleic acid, the specificity of probe binding, and the signal-to-noise ratio in the final stained section. While the protocols outlined here provide a robust starting point, researchers must remember that optimal conditions vary by tissue type, fixation method, and target abundance. Empirical optimization using appropriate controls is therefore essential. When executed with precision, these pre-hybridization steps ensure that the subsequent NBT/BCIP development accurately reveals the spatial distribution of gene expression, enabling meaningful biological insights and advancing research in gene regulation, development, and disease pathogenesis.
In situ hybridization (ISH) is an indispensable technique for visualizing the spatial and temporal distribution of specific nucleic acid sequences within cells, tissues, or entire organisms. Within the context of a broader thesis employing NBT/BCIP chromogenic detection, the choice between DNA and RNA probes represents a fundamental decision point that directly impacts experimental outcomes. This selection influences everything from hybridization efficiency and signal specificity to the preservation of tissue morphology. The NBT/BCIP staining protocol, which yields a purple-blue precipitate through alkaline phosphatase activity, provides a robust, cost-effective detection method particularly valuable for whole-mount specimens and histological sections where high contrast is essential [23]. The integrity of this final visualization is critically dependent on initial probe design and hybridization optimization, making understanding the distinctions between nucleic acid probe types essential for researchers, scientists, and drug development professionals aiming to generate reliable, interpretable data.
DNA vs. RNA Probes: A Comparative Analysis
Fundamental Characteristics and Hybridization Properties
The core difference between DNA and RNA probes lies in their chemical structure. DNA probes are composed of deoxyribonucleotides (adenine, thymine, cytosine, guanine) and can be double-stranded (dsDNA) or single-stranded (ssDNA) [24]. RNA probes (riboprobes), in contrast, are made of ribonucleotides (adenine, uracil, cytosine, guanine), which include a 2' hydroxyl group that makes the molecule more chemically labile than DNA but does not detract from its utility in controlled conditions [24]. This structural difference underlies a key functional distinction: RNA-RNA hybrids (formed when an RNA probe binds to an mRNA target) are more thermodynamically stable than RNA-DNA hybrids, which are in turn more stable than DNA-DNA hybrids [25]. This inherent stability directly influences the stringency requirements and signal strength achievable in ISH experiments.
Table 1: Core Characteristics of DNA and RNA Probes
| Feature | DNA Probes | RNA Probes (Riboprobes) |
|---|---|---|
| Chemical Structure | Deoxyribose sugar, Thymine | Ribose sugar, Uracil |
| Strandedness | Double or single-stranded | Typically single-stranded |
| Thermal Stability | DNA-DNA hybrids are least stable | RNA-RNA hybrids are most stable [25] |
| Common Synthesis Methods | Nick translation, PCR, random priming, chemical synthesis [26] [24] | In vitro transcription (IVT) from DNA templates [26] [24] |
| Primary Labeling Technique | Incorporation of labeled nucleotides during synthesis [26] | Incorporation of labeled nucleotides during IVT [26] |
| Common Applications in ISH | Chromosomal FISH (locus, centromere, whole chromosome) [24] | mRNA localization, gene expression analysis (RISH) [27] [24] |
Synthesis, Labeling, and Probe Design Specifications
The synthesis and labeling of DNA and RNA probes follow distinct pathways. DNA probes are frequently generated using enzymatic methods like nick translation or random priming, which incorporate labeled nucleotides into a DNA template [26]. For RNA probes, in vitro transcription is the predominant method, whereby a DNA template cloned downstream of a bacteriophage RNA polymerase promoter (e.g., T7, T3, SP6) is transcribed in the presence of labeled nucleotides, producing large amounts of uniformly labeled, single-stranded probe [26]. A common strategy involves flanking the DNA insert with two opposed promoters, allowing transcription of both antisense (probe) and sense (negative control) RNAs [26].
Probe design is critical for success. For RNA probes, a length of 250–1,500 bases is recommended, with probes of approximately 800 bases often exhibiting the highest sensitivity and specificity [27]. The probe sequence must possess high complementarity to the target; even >5% non-complementary base pairs can result in loose hybridization and potential signal loss during washing [27]. For DNA probes, especially those used in fluorescent ISH (FISH), sizes can range widely from 20-1000 bp for oligonucleotide probes to 1-10 Kb for genomic FISH probes [24].
Optimizing Hybridization and Stringency Conditions
Key Parameters for Hybridization
Hybridization is the critical step where the probe binds to its complementary target sequence. The specificity of this interaction, or stringency, is controlled by several factors to ensure that only perfect or near-perfect matches are stabilized.
Table 2: Optimization of Hybridization and Wash Conditions
| Parameter | Impact on Specificity | Optimization Guidelines |
|---|---|---|
| Hybridization Temperature | Primary driver of stringency [25]. | Typically 55-65°C for many protocols [27]. Start 15-25°C below Tm [23]. |
| Formamide Concentration | Reduces melting temperature, allowing lower hybridization temperatures to preserve morphology [25]. | Commonly used at 50% (v/v) in hybridization buffers [27] [4]. |
| Salt Concentration (SSC) | Higher salt stabilizes duplex formation; lower salt increases stringency [27]. | Hybridize at 1-5x SSC; wash with 0.1-2x SSC for stringency [27]. |
| Post-Hybridization Washes | Removes unbound and weakly bound probe [27]. | Adjust temperature and SSC concentration based on probe type and length [27] [25]. |
| Probe Concentration | Too high causes high background; too low causes weak signal [4]. | Requires empirical titration for each probe and tissue type. |
The optimal hybridization temperature is often determined empirically but can be estimated based on the probe's melting temperature (Tm). For RNA probes, a typical hybridization temperature ranges between 55°C and 62°C [27]. The addition of formamide to the hybridization buffer is a standard practice, as it denatures nucleic acids and allows the use of lower incubation temperatures, thereby helping to preserve tissue integrity [25].
Probe-Specific Considerations for DNA and RNA Probes
- For RNA Probes: Due to the strength of RNA-RNA hybrids, higher stringency washes can be applied to reduce background without significant signal loss. A common high-stringency wash is 50% formamide in 2x SSC at 37-45°C [27]. Background can also be reduced by digesting non-specifically bound probe with RNase A after hybridization, which cleaves single-stranded RNA but leaves the double-stranded RNA hybrid intact [25].
- For DNA Probes: Because DNA-DNA hybrids are less stable, formaldehyde should be avoided in post-hybridization washes as it can further destabilize the hybrid [27] [25]. Washes should be optimized carefully by adjusting temperature, salt, and detergent concentration to minimize non-specific interactions while retaining the specific signal [25].
Integrated Protocols for NBT/BCIP Detection
DIG-Labeled RNA Probe ISH Protocol for Paraffin Sections
This protocol is adapted for formalin-fixed, paraffin-embedded (FFPE) tissues and uses digoxigenin (DIG)-labeled riboprobes detected with an alkaline phosphatase (AP)-conjugated anti-DIG antibody, followed by development with NBT/BCIP.
Stage 1: Sample Preparation and Pre-Treatment
- Deparaffinization and Rehydration: Immerse slides in a series of washes: Xylene (2x3 min) → Xylene:100% ethanol (1:1, 3 min) → 100% ethanol (2x3 min) → 95% ethanol (3 min) → 70% ethanol (3 min) → 50% ethanol (3 min) → rinse with cold tap water. Do not allow slides to dry after this point [27].
- Proteinase K Digestion (Antigen Retrieval): Digest sections with 20 µg/mL Proteinase K in pre-warmed 50 mM Tris buffer for 10–20 min at 37°C. This step is critical and requires optimization; insufficient digestion reduces signal, while over-digestion destroys tissue morphology. A starting titration of 1-5 µg/mL for 10 minutes at room temperature is also recommended [27] [25].
- Post-fixation and Acetylation: A brief post-fixation in 4% paraformaldehyde can be performed to maintain tissue structure. An optional acetylation step (e.g., in 0.1 M triethanolamine with acetic anhydride) can be included to block positively charged amines and reduce non-specific probe binding [4].
- Dehydration: Wash slides sequentially in 70%, 95%, and 100% ethanol (∼1 min each), then air dry [27].
Stage 2: Hybridization
- Pre-hybridization: Apply 100 µL of hybridization solution (e.g., containing 50% formamide, 5x salts, blocking agents) to each slide and incubate for 1 hour in a humidified chamber at the hybridization temperature [27].
- Probe Denaturation and Application: Dilute the DIG-labeled RNA probe in hybridization solution. Denature at 95°C for 2 minutes, then immediately chill on ice. Drain the pre-hybridization buffer from the slides and apply 50-100 µL of denatured probe per section. Cover with a coverslip and incubate overnight (16-18 hours) in a humidified hybridization chamber at 65°C [27].
Stage 3: Stringency Washes and Detection
- High-Stringency Washes: Remove coverslips gently and wash slides as follows to remove unbound probe:
- Immunological Detection:
- Blocking: Wash slides twice in MABT (Maleic Acid Buffer with Tween) for 30 min at room temperature. Transfer to a humidified chamber and apply 200 µL blocking buffer (MABT + 2% BSA or serum) for 1-2 hours at room temperature [27].
- Antibody Incubation: Drain blocking buffer and apply anti-DIG antibody conjugated to Alkaline Phosphatase (AP) at the recommended dilution in blocking buffer. Incubate for 1-2 hours at room temperature [27].
- Washes: Wash slides 5x10 min with MABT to remove unbound antibody [27].
- Color Development with NBT/BCIP:
- Equilibrate slides in a pre-staining buffer (e.g., 100 mM Tris pH 9.5, 100 mM NaCl, 10 mM MgCl₂).
- Apply the NBT/BCIP chromogen substrate solution. Develop in the dark, monitoring periodically until the desired purple-blue signal intensity is achieved with minimal background.
- Stop the reaction by washing in water or an appropriate buffer [27] [23].
- Counterstain (if desired), mount with an aqueous mounting medium, and image using brightfield microscopy [4].
The Scientist's Toolkit: Essential Reagents for ISH
Table 3: Key Research Reagent Solutions for ISH with NBT/BCIP Detection
| Reagent / Solution | Function / Purpose | Example Formulation / Notes |
|---|---|---|
| Proteinase K | Digests proteins to permeabilize tissue, allowing probe access. Critical for signal strength [27] [25]. | 1-20 µg/mL in Tris buffer; requires concentration/temperature optimization [27]. |
| Hybridization Buffer | Creates the chemical environment for specific probe-target binding. | Typically contains 50% formamide, SSC (salts), Dextran sulfate (probe concentrator), and blocking agents (e.g., BSA, tRNA) [27] [4]. |
| DIG-Labeled Nucleotides | Hapten-labeled nucleotides incorporated into probes. | DIG-dUTP for DNA probes; DIG-UTP for RNA probes. A non-radioactive, highly specific label [26] [23]. |
| Anti-DIG-AP Antibody | Immunological conjugate for detecting the DIG label. | Conjugated to Alkaline Phosphatase (AP); binds to DIG on hybridized probes [27] [23]. |
| NBT/BCIP Substrate | Chromogenic substrate for AP enzyme. Produces an insoluble purple-blue precipitate at the site of hybridization [4] [23]. | Ready-to-use solutions available; development occurs in the dark and must be monitored. |
| Blocking Reagents | Reduce non-specific binding of the probe and antibody, lowering background. | BSA, serum, milk, or commercial blocking blends in a buffer like MABT or PBS-T [27] [4]. |
| Stringency Wash Buffers | Remove imperfectly matched or unbound probes after hybridization. | SSC at varying concentrations (0.1x-2x) and temperatures; formamide can be added to increase stringency [27]. |
Advanced Applications and Emerging Technologies
The field of ISH continues to evolve with new technologies enhancing multiplexing capabilities, sensitivity, and resolution. The OneSABER platform represents a recent advancement, a unified open platform that uses a single type of DNA probe which can be adapted for various signal development methods, including canonical AP-based colorimetric ISH and fluorescent ISH [28]. This "one probe fits all" approach utilizes a pool of short ssDNA oligonucleotides complementary to an RNA target, which are enzymatically extended into long concatemers via primer exchange reaction (PER) [28]. These concatemers can then be detected using secondary probes labeled with haptens like DIG, making them compatible with the standard anti-DIG/AP/NBT-BCIP detection pipeline, thereby offering a flexible and customizable alternative to traditional riboprobes [28].
Troubleshooting and Optimization Guide
Even with a well-designed protocol, challenges can arise. Here are common issues and solutions specific to probe hybridization and NBT/BCIP detection:
- High Background Staining:
- Cause: Non-specific probe binding, insufficient blocking, or inadequate post-hybridization washes.
- Solutions: Increase the stringency of washes (e.g., lower SSC concentration, higher temperature) [4]. Include blocking agents like salmon sperm DNA or tRNA in the hybridization buffer [4]. For RNA probes, a post-hybridization RNase A treatment can digest single-stranded, non-specifically bound probe [25]. Ensure the probe is not over-concentrated.
- Weak or Absent Signal:
- Cause: Poor probe penetration, degraded probe, low target abundance, or over-fixation.
- Solutions: Optimize Proteinase K concentration and incubation time for permeabilization [27] [25]. Check probe integrity by gel electrophoresis. Increase probe concentration or development time for low-abundance targets. For DNA probes, ensure formaldehyde is not used in post-hybridization washes [27].
- Non-Specific Signals:
- Cause: Off-target probe binding or endogenous enzyme activity.
- Solutions: Always run controls, including a sense probe (for RNA probes) or a no-probe control, to identify non-specific staining [4]. Use BLAST to verify probe specificity. For DIG-based systems, endogenous biotin is not a concern, which is an advantage over biotinylated probes [25].
The strategic selection and optimization of DNA or RNA probes are pivotal to the success of any ISH study, particularly those culminating in NBT/BCIP chromogenic detection. RNA probes, with their superior hybrid stability and high sensitivity, are often the preferred choice for mRNA localization studies. In contrast, DNA probes offer versatility and are fundamental for chromosomal mapping. Meticulous attention to probe design, hybridization stringency, and immunological detection steps ensures that the resulting purple-blue precipitate accurately reflects the underlying spatial gene expression pattern. By applying the principles and protocols outlined in this application note, researchers can confidently navigate the critical choices in probe design and hybridization to generate robust, publication-quality data that advances our understanding of gene function in development and disease.
In the context of the NBT/BCIP staining protocol for chromogenic in situ hybridization (ISH), the steps following the incubation of the probe are critical for the success of the experiment. Post-hybridization washes are the primary mechanism for controlling stringency, a process that selectively removes imperfectly matched or unbound probes to ensure that the final visualized signal is specific to the target nucleic acid sequence. Achieving the correct stringency is paramount for generating reliable, interpretable, and publication-quality data in gene expression studies, making this phase indispensable for researchers and drug development professionals validating therapeutic targets.
The Principles of Stringency
Stringency refers to the set of conditions that influence the stability of hydrogen bonds between the probe and its target sequence. The fundamental goal of post-hybridization washes is to discriminate between perfectly matched probe-target hybrids (desired signal) and mismatched or non-specifically bound probes (background noise). The stability of these hybrids is governed by two key physical parameters that can be controlled during the washing process:
- Temperature: Higher temperatures increase the kinetic energy of molecules, disrupting the hydrogen bonds holding the probe and target together. Mismatched hybrids, with fewer hydrogen bonds, denature at lower temperatures than perfectly matched ones.
- Ionic Strength: The concentration of salts, most commonly in the form of Saline Sodium Citrate (SSC), stabilizes nucleic acid hybrids by shielding the negative charges on the phosphate backbones. Lower salt concentrations (e.g., 0.1X SSC vs. 2X SSC) reduce this shielding effect, increasing the electrostatic repulsion between strands and thus the stringency.
By strategically manipulating these parameters, researchers can create a window of conditions where only the specific hybrids remain stable.
Quantitative Parameters for Stringency Washes
The following table summarizes the standard parameters for post-hybridization washes in a chromogenic ISH protocol. These conditions provide a starting point for optimization, which may be required based on the specific probe, tissue type, and fixation method.
Table 1: Standardized Post-Hybridization Wash Conditions for Chromogenic ISH
| Wash Step | Buffer Composition | Temperature | Duration | Purpose |
|---|---|---|---|---|
| Initial Rinse | 2X SSC [4] | Room Temperature [4] | 5-10 minutes | To remove the bulk of the hybridization buffer and coverslips gently. |
| High-Stringency Wash | 0.1X SSC to 1X SSC [4] | 37-65°C [4] | 15-30 minutes (per wash) | To denature and wash away mismatched and non-specifically bound probes. |
| Final Rinse | Tris-Buffered Saline with Tween (TBST) or similar [4] | Room Temperature | 5 minutes | To prepare the sample for the subsequent detection steps with NBT/BCIP. |
Optimization Guidance
The values in Table 1 are a generalized framework. Critical steps for optimization include:
- Probe and Target Specificity: The optimal wash stringency is intrinsically linked to the probe's GC content and length. Longer probes with higher GC content require more stringent conditions (higher temperature, lower SSC) for dissociation.
- Tissue Considerations: Different tissue types have varying levels of background adherence. Tissues with high lipid content or endogenous phosphatases may require more stringent washing to minimize background [19].
- Systematic Titration: It is recommended to perform a stringency test by varying one parameter at a time (e.g., temperature in 5°C increments or SSC concentration in two-fold dilutions) while keeping other factors constant to determine the ideal signal-to-noise ratio.
Detailed Experimental Protocol: Post-Hybridization Washes for NBT/BCIP Detection
This protocol follows the hybridization step, where slides have been incubated with the labeled probe overnight.
Materials and Reagents
- Coplin jars or automated slide staining system
- Water bath or hybridization oven (calibrated for precise temperature control) [4]
- Wash Buffers: 2X SSC, 0.2X SSC, and TBST, pre-warmed or cooled to the required temperatures [4]
- Forceps and slide racks
Step-by-Step Method
- Coverslip Removal: Carefully remove the slides from the humidified hybridization chamber. Gently immerse the slides in a Coplin jar containing pre-warmed 2X SSC until the coverslips slide off easily. Forceful removal can damage the tissue section.
- Initial Rinse: Transfer the slides to a fresh Coplin jar with a sufficient volume of 2X SSC at room temperature. Agitate gently for 10 minutes to remove residual hybridization buffer [4].
- High-Stringency Washes: This is the critical step for establishing specificity.
- Perform two washes in a pre-warmed low-salt SSC buffer (e.g., 0.2X SSC) in a water bath or hybridization oven set to 45-65°C [4]. The exact temperature must be determined empirically (see Optimization Guidance above).
- Agitate continuously for 15-30 minutes per wash [4].
- Use a sufficient volume of buffer to maintain consistent stringency throughout the wash.
- Equilibration for Detection: Rinse the slides briefly in TBST (or a similar detection buffer) at room temperature for 5 minutes. This step removes citrate from the SSC buffer, which could interfere with the subsequent enzymatic detection reaction using alkaline phosphatase (AP) and the NBT/BCIP substrate.
- Proceed to Detection: Immediately following the final wash, proceed to the blocking and application of the anti-digoxigenin-AP antibody and subsequent NBT/BCIP chromogenic development as per the broader ISH protocol.
Troubleshooting Common Issues in Post-Hybridization Washes
Even with a well-designed protocol, issues can arise that are directly traceable to the wash steps.
Table 2: Troubleshooting Wash-Related Problems in ISH
| Problem | Potential Cause | Corrective Action |
|---|---|---|
| High Background Signal [4] | Insufficient stringency (temperature too low, SSC concentration too high). | Increase wash temperature and/or decrease SSC concentration in high-stringency steps. |
| Weak or Absent Specific Signal [4] | Excessive stringency (temperature too high, SSC concentration too low). | Decrease wash temperature and/or increase SSC concentration. Validate probe and target integrity. |
| Uneven Staining [4] | Inconsistent washing, drying of slides during washes, or bubbles on sections. | Ensure adequate buffer volume and agitation. Use standardized washing steps for duration, volume, and agitation across all runs [19]. |
| Non-Specific Signals [4] | Inadequate removal of unbound probe or off-target binding. | Increase the number or duration of high-stringency washes. Include a control with RNase or DNase digestion to confirm target specificity. |
The Scientist's Toolkit: Essential Reagents for Post-Hybridization Washes
Table 3: Key Research Reagent Solutions for Stringency Control
| Reagent | Function in Post-Hybridization Washes |
|---|---|
| Saline Sodium Citrate (SSC) [4] | The primary buffer for controlling stringency via ionic strength. Dilutions from 2X to 0.1X are used to gradually increase washing stringency. |
| Tris-Buffered Saline with Tween (TBST) [4] | Used as a final rinse buffer to remove SSC and prepare slides for immunological detection steps. The detergent (Tween-20) helps reduce non-specific background. |
| Formamide [4] | Although more common in hybridization buffers, it can be included in wash buffers to lower the effective melting temperature of hybrids, allowing for high stringency at lower, more manageable temperatures. |
Workflow Visualization
The following diagram illustrates the logical sequence and decision points in the post-hybridization wash process, culminating in the chromogenic development for ISH.
Diagram 1: Post-Hybridization Wash Workflow
In the broader framework of an NBT/BCIP staining protocol, post-hybridization washes are not merely a cleaning step but a critical determinant of experimental specificity. A meticulous approach to optimizing and standardizing stringency conditions—through controlled temperature and salt concentration—is fundamental to distinguishing true signal from background noise. By adhering to the detailed protocols and troubleshooting guides outlined herein, researchers can consistently achieve high-quality, reliable ISH results that robustly support their findings in both basic research and drug development contexts.
Antibody Incubation and Developing the Signal with NBT/BCIP
Within in situ hybridization (ISH) research, the accurate localization of specific nucleic acid sequences in fixed tissues or cells is paramount. This application note details the critical phase of this process: the incubation with an enzyme-conjugated antibody and the subsequent colorimetric detection of the hybridization signal using the NBT/BCIP substrate. When optimized, this protocol yields an intense, insoluble blue/purple precipitate at the site of target gene expression, allowing for precise qualitative and quantitative analysis with single-cell resolution [10] [2].
Experimental Protocol
Antibody Incubation
Following nucleic acid probe hybridization and stringent washes to remove unbound probe, the sample is incubated with a detection antibody.
- Preparation: Dilute the anti-digoxigenin (DIG) antibody conjugated to alkaline phosphatase (AP) in an appropriate blocking buffer (e.g., 1:2000 dilution in Roche blocking buffer) [10].
- Application: Apply the antibody solution to the sample, ensuring complete coverage.
- Incubation: Incubate for the specified duration (typically 30-60 minutes) at room temperature.
- Washing: Perform extensive washing with a buffer such as PBS-Tween or TBS-Tween to eliminate any non-specifically bound antibody, which is crucial for minimizing background [10] [4].
Signal Development with NBT/BCIP
The development step utilizes the enzymatic activity of AP to convert the soluble NBT/BCIP substrate into an insoluble colored precipitate.
Substrate Preparation Table The following table summarizes the preparation of a standard NBT/BCIP working solution for membrane assays [29].
| Component | Concentration/Amount | Notes |
|---|---|---|
| Alkaline Phosphatase Buffer | 5 mL | 100 mM Tris-HCl (pH 9.5), 150 mM NaCl, 1 mM MgCl₂ |
| NBT Stock Solution | 33 µL | 50 mg/mL in 70% dimethylformamide [29] |
| BCIP Stock Solution | 16.5 µL | 50 mg/mL in 100% dimethylformamide [29] |
- Procedure: First, add the NBT to the alkaline phosphatase buffer and mix. Then, add the BCIP and mix again. The prepared working solution should be used within one hour [29].
Development Workflow:
- Apply Substrate: Add the freshly prepared NBT/BCIP working solution to the sample, ensuring it is fully covered.
- Incubate: Allow the color development to proceed in the dark at room temperature. Monitor the reaction periodically until the desired signal intensity is achieved, avoiding over-development which can increase background.
- Stop the Reaction: Once sufficient color has developed, stop the reaction by replacing the substrate solution with a stop buffer, such as Tris-EDTA buffer, or by thoroughly washing with distilled water [4].
- Mount and Image: Mount the samples with an aqueous or specific permanent mounting medium compatible with NBT/BCIP (e.g., Crystalmount or Vectamount). Avoid xylene-containing mounting media as they can cause crystal formation of the precipitate [30]. Image using a brightfield microscope.
Quantitative Application in Solution
The NBT/BCIP substrate combination can also be adapted for quantitative assays of secreted alkaline phosphatase (SEAP) activity in cell culture supernatants. The key challenge is the insolubility of the reaction products. This is overcome by adding non-ionic detergents like TWEEN 20 (0.05% v/v) to the reaction mixture, which stabilizes the products in a colloidal form, allowing for photometric quantification at the appropriate wavelength [31].
The Scientist's Toolkit: Essential Research Reagents
The table below lists key reagents and their functions for a successful ISH protocol with NBT/BCIP detection [29] [4].
| Item | Function in the Protocol |
|---|---|
| NBT/BCIP Substrate | Chromogenic substrate for Alkaline Phosphatase; produces a blue/purple precipitate at the enzyme site [29] [2]. |
| Anti-DIG-AP Antibody | Conjugated antibody that binds to DIG-labeled probes, enabling enzymatic signal detection [10]. |
| Blocking Buffer (e.g., with BSA or Casein) | Blocks nonspecific binding sites on the tissue sample to reduce background staining [4]. |
| Stringency Wash Buffers (e.g., SSC with SDS) | Removes weakly bound or mismatched probes after hybridization; critical for signal specificity [4]. |
| TWEEN 20 | Non-ionic detergent used to stabilize NBT/BCIP reaction products in colloidal form for quantitative solution assays [31]. |
| Nuclear Stain (e.g., To-PRO-3) | Fluorescent DNA-intercalating dye used for accurate total cell counting in quantitative PISH applications [10]. |
Comparative Analysis of Alkaline Phosphatase Substrates
Researchers can choose from several AP substrates depending on the required readout. The table below compares three common options.
| Substrate | Type | Readout | Key Applications |
|---|---|---|---|
| NBT/BCIP | Chromogenic (Precipitating) | Insoluble blue/purple precipitate; qualitative imaging & quantitative analysis (with cell count) [10] | ISH, Western blot, immunohistochemistry [29] [10] |
| pNPP | Chromogenic (Soluble) | Yellow soluble product; absorbance at 405 nm [10] | Quantitative ELISAs, solution-based SEAP assays [10] [31] |
| CDP-Star | Chemiluminescent | Light emission at 475 nm [10] | Highly sensitive quantitative blotting assays [10] |
Signaling Pathway and Experimental Workflow
The following diagram illustrates the core experimental workflow from sample preparation through to signal detection.
The biochemical reaction catalyzed by alkaline phosphatase to generate the detectable signal is shown below.
Troubleshooting Common Issues
Even with a well-designed protocol, challenges can arise. The table below outlines common problems and their solutions [4] [30].
| Problem | Possible Cause | Solution |
|---|---|---|
| High general blue background | Overfixation of tissue; insufficient blocking or washing; endogenous AP activity. | Optimize fixation time; ensure thorough blocking and post-antibody washes; use an endogenous AP inhibitor [30]. |
| Weak or no signal | Low probe concentration or degradation; inefficient permeabilization; low target abundance. | Optimize probe concentration and check integrity; increase permeabilization time (e.g., Proteinase K) [4]. |
| Brown-purple instead of blue signal | Very high abundance of target mRNA; pH of detection buffer is incorrect. | Adjust pH of detection buffer to 9.5; shorten development time [30]. |
| Uneven staining | Sample drying out; uneven probe or substrate coverage. | Use a properly sealed humidified chamber during incubations; ensure solutions are applied evenly [4]. |
| Precipitate formation in stock | Common for concentrated stocks in dimethylformamide. | Warm the vial gently with shaking, then centrifuge before use [30]. |
In the context of a broader thesis on the NBT/BCIP staining protocol for in situ hybridization (ISH), the steps following chromogenic development—namely mounting and imaging—are critical for data interpretation and archival. The enzymatic reaction of alkaline phosphatase with NBT/BCIP yields a blue-purple, insoluble precipitate that localizes target nucleic acids [6] [32]. Preserving this precipitate faithfully and providing cellular context through appropriate counterstaining are paramount for generating publication-quality results that are reliable and reproducible for researchers and drug development professionals. This application note details standardized methodologies to achieve these ends.
The NBT/BCIP Precipitate: Characteristics and Vulnerabilities
The formazan precipitate resulting from the NBT/BCIP reaction is notably insoluble in aqueous and alcoholic solutions, which allows for dehydration through graded ethanols during sample processing [6]. However, a critical technical consideration is its solubility in organic solvents, particularly xylene, which is a common component of many historical and conventional histological mounting protocols [33]. Using xylene-based mounting media will dissolve the developed signal, thus invalidating the experiment. Furthermore, the final color of the precipitate, which can range from blue to deep purple or even brown-purple, is influenced by the abundance of the target nucleic acid and the pH of the detection buffer, which should be carefully adjusted to pH 9.5 for optimal and consistent results [33].
Mounting Media for Signal Preservation
The choice of mounting medium is the primary determinant for the long-term preservation of the NBT/BCIP precipitate. The key selection criterion is the absence of xylene and other solubility-matched organic solvents.
Table 1: Mounting Media Compatibility for NBT/BCIP-Stained Sections
| Mounting Medium Type | Compatibility with NBT/BCIP | Key Characteristics | Example Products |
|---|---|---|---|
| Aqueous-Based | Compatible | Water-soluble, preserves precipitate integrity, may require curing time. | Immunomount, Vectamount [33] |
| Specialized Permanent | Compatible | Specifically formulated for chromogenic precipitates; provides permanent seal. | Crystalmount [33] |
| Xylene-Based | Not Compatible | Dissolves the NBT/BCIP precipitate, destroying the signal. | DPX and similar formulations [33] |
Counterstaining for Morphological Context
A well-chosen counterstain provides essential morphological context, allowing for the precise localization of the NBT/BCIP signal within specific cells or tissue structures. The ideal counterstain should offer good contrast against the blue-purple precipitate without masking it.
- Principle of Light Counterstaining: It is imperative to use a light counterstain [6]. A dark or intense counterstain, particularly with hematoxylin, can easily obscure the NBT/BCIP signal, leading to false-negative interpretations.
- Recommended Practice: For hematoxylin, a brief incubation of 5 seconds to 1 minute is typically sufficient [6]. Mayer's hematoxylin is often recommended for this purpose, as it provides a clear nuclear stain without over-staining [6].
Table 2: Compatible Counterstains for NBT/BCIP-Based ISH
| Counterstain | Compatible Mounting Media | Function | Key Consideration |
|---|---|---|---|
| Nuclear Fast Red | Aqueous-based media [33] | Provides pink/red nuclear staining. | Offers excellent contrast to blue-purple precipitate. |
| Methyl Green | Aqueous-based media [33] | Provides green nuclear staining. | A strong alternative to red counterstains. |
| Fast Green FCF | Aqueous-based media [33] | Cytoplasmic stain. | Can be used in combination with other stains. |
| Mayer's Hematoxylin | Aqueous-based media [6] | Blue nuclear stain. | Must be used lightly (5-60 seconds) to avoid signal masking [6]. |
The following workflow outlines the critical post-staining steps for preserving the NBT/BCIP signal and preparing the slide for imaging:
Imaging and Documentation
For NBT/BCIP-stained samples, brightfield microscopy is the standard imaging modality [4]. The chromogenic precipitate is opaque and easily visualized under transmitted light. To ensure accurate documentation:
- Avoid Signal Saturation: Carefully adjust the microscope's light intensity and the camera's exposure settings to capture the full dynamic range of the signal without saturation, which can obscure quantitative differences in signal intensity [4].
- Immediate Imaging: While the precipitate is stable, it is good practice to capture images soon after mounting, especially if the mounting medium is aqueous, to preserve the original quality of the stain and counterstain [4].
- Fluorescence Imaging Note: Interestingly, the NBT/BCIP precipitate has been reported to exhibit near-infrared (NIR) fluorescence [32]. This property can be exploited for high-resolution three-dimensional imaging using confocal microscopy, offering an advanced modality beyond conventional brightfield documentation.
The Scientist's Toolkit: Essential Reagents for Mounting and Imaging
Table 3: Key Research Reagent Solutions for Post-ISH Workflow
| Item | Function/Application | Technical Note |
|---|---|---|
| Aqueous Mounting Medium (e.g., Immunomount) | To preserve the NBT/BCIP signal and secure the coverslip without dissolving the precipitate. | Must be xylene-free. Provides a permanent seal [33]. |
| Mayer's Hematoxylin | A light nuclear counterstain to provide cellular context. | Counterstaining time should be optimized between 5-60 seconds to avoid masking the signal [6]. |
| Nuclear Fast Red | A pink/red nuclear counterstain that offers high contrast to the blue NBT/BCIP signal. | Compatible with aqueous mounting media [33]. |
| NBT/BCIP Stock Solution | The chromogenic substrate for alkaline phosphatase, forming the insoluble blue-purple precipitate. | The detection buffer must be at pH 9.5 for optimal color development [33]. |
| Ethanol (100%, 95%, 70%) | For dehydration of tissue sections after counterstaining and prior to mounting with non-aqueous media. | The NBT/BCIP precipitate is insoluble in alcohol, allowing for this critical dehydration step [6]. |
Troubleshooting NBT/BCIP Staining: Solving Weak Signal and High Background
Diagnosing and Fixing Weak or Absent Staining
Within the framework of a broader thesis on NBT/BCIP staining protocol for in situ hybridization (ISH) research, achieving consistent, high-quality staining is paramount for generating reliable, publication-quality data. Weak or absent staining presents a significant obstacle in gene expression studies, potentially compromising data integrity and leading to erroneous biological interpretations. This document provides detailed application notes and protocols designed to empower researchers, scientists, and drug development professionals to systematically diagnose and rectify the most common issues that undermine staining intensity and specificity in ISH workflows. The NBT/BCIP chromogenic system, which yields an insoluble purple precipitate, is a cornerstone of many ISH protocols for mRNA localization [10] [34]. Understanding the variables that influence this reaction is the first step toward robust and reproducible results.
Troubleshooting Weak or Absent Staining: A Systematic Approach
A methodical approach is essential for diagnosing the root cause of staining problems. The following table outlines common issues, their potential causes, and recommended solutions.
Table 1: Troubleshooting Guide for Weak or Absent NBT/BCIP Staining
| Problem Observed | Potential Cause | Recommended Solution |
|---|---|---|
| Weak or Faint Staining | Inadequate probe concentration or degradation [4] | Optimize probe concentration; check probe integrity via gel electrophoresis. |
| Insufficient permeability [4] | Optimize permeabilization (e.g., Proteinase K concentration and time) [19]. | |
| Sub-optimal fixation (under-fixation) [19] | Ensure consistent fixation conditions (fixative, pH, time) to preserve nucleic acids. | |
| Low sensitivity of detection system [19] | Use a fresh, high-quality AP-conjugated antibody and substrate; extend development time. | |
| Absent Staining | Target RNA degradation [19] | Use RNase-free techniques; handle specimens carefully and fix promptly. |
| Incorrect hybridization conditions [19] | Control temperature and time stringently; check probe specificity and data sheets. | |
| Inactive reagents (substrate, antibody) [4] | Use fresh substrate batches; include positive control tissues to validate the entire protocol. | |
| High Background Staining | Non-specific probe binding [4] | Increase stringency of post-hybridization washes (e.g., lower SSC concentration). |
| Inadequate blocking [4] | Include blocking agents like BSA, salmon sperm DNA, or tRNA in hybridization buffer. | |
| Probe drying on section [19] | Prevent evaporation by using a properly sealed, humidified chamber during incubations. | |
| Uneven Staining | Incomplete dewaxing or reagent coverage [19] | Ensure complete wax removal and uniform distribution of reagents across the specimen. |
| Bubbles on section surface [19] | Ensure complete wax removal and uniform distribution of reagents across the specimen. | |
| Poor section adhesion or quality [19] | Use thin, high-quality sections on charged slides; avoid protein-based adhesives. |
Visual Diagnostic Workflow
The following diagram outlines a logical, step-by-step process for diagnosing staining issues based on the observations in your experiment.
Quantitative Optimization of Critical Parameters
Successful staining is a balance of multiple interacting variables. The following quantitative data, derived from established methodologies, provides a starting point for protocol optimization [10].
Table 2: Key Reagent Formulations for NBT/BCIP-based q2PISH Protocol
| Reagent | Composition / Concentration | Function in the Protocol |
|---|---|---|
| Fixative | 4% (w/v) Paraformaldehyde (PFA) in PBS [10] | Preserves tissue architecture and immobilizes target nucleic acids. |
| Permeabilization Agent | Proteinase K Solution (e.g., 20 mg/mL) [4] | Digests proteins to allow probe access to intracellular targets. |
| Pre-hybridization Buffer | 50% Formamide, 1X SSC, 50 µg/mL Heparin, 100 µg/mL Denatured Salmon Sperm DNA, 1% SDS [4] | Blocks non-specific binding sites and prepares tissue for hybridization. |
| Hybridization Buffer | Pre-hybridization buffer with DIG-labeled RNA probe [10] | Medium for specific binding of the probe to its complementary mRNA target. |
| Wash Buffer | Saline Sodium Citrate (SSC) at varying concentrations (e.g., 2X to 0.1X) [4] | Removes unbound and weakly bound probe; concentration controls stringency. |
| Antibody Solution | Anti-DIG Antibody conjugated to Alkaline Phosphatase (AP) [10] | Binds specifically to the DIG label on the hybridized probe. |
| Substrate Solution | NBT/BCIP in 0.1 M Tris-HCl, 0.1 M NaCl, pH 9.5 [10] | AP enzyme converts substrates to insoluble purple precipitate for visualization. |
Advanced Quantitative Workflow (q2PISH)
For researchers requiring not just qualitative localization but also quantitative data, the q2PISH (qualitative and quantitative PISH) protocol can be employed. This method sequentially uses different AP substrates to first quantify and then visualize gene expression.
Table 3: Experimental Parameters for q2PISH Quantification
| Parameter | Specification | Application / Purpose |
|---|---|---|
| Cell Seeding | ~5 × 10⁵ cells/well in a 96-well plate (confluence) [10] | Ensures consistent cell density for accurate cross-well comparison. |
| pNPP Incubation | Absorbance measured at 405 nm over 192 hours [10] | Generates a soluble yellow product for spectrophotometric quantification of total transcript levels. |
| NBT/BCIP Incubation | Following pNPP step; produces insoluble purple precipitate [10] | Allows for qualitative assessment of staining heterogeneity and spatial localization. |
| Cell Nuclei Staining | To-PRO-3 fluorescent DNA intercalating dye [10] | Provides an accurate total cell count for normalizing expression data to a "per cell" basis. |
| Data Output | Normalized gene expression value (Absorbance Units/Cell) | Enables precise comparison of gene expression levels across different cell populations or treatments. |
The Scientist's Toolkit: Essential Research Reagent Solutions
A successful ISH experiment depends on the quality and appropriateness of its core reagents. The following table details key solutions and their critical functions in the NBT/BCIP staining workflow.
Table 4: Essential Research Reagents for ISH
| Reagent / Material | Function / Application | Key Considerations |
|---|---|---|
| Charged Glass Slides | Provides a strong electrostatic attraction for tissue sections [19]. | Prevents section loss during stringent washing steps. Avoid protein-based adhesives. |
| Digoxigenin (DIG) Labeling | A hapten label for RNA or DNA probes, detected by an antibody [10]. | Offers high specificity and sensitivity, reducing background compared to radioactive methods. |
| Anti-DIG-AP Conjugate | An antibody that binds DIG, conjugated to Alkaline Phosphatase enzyme [10]. | Key detection reagent; binding triggers the chromogenic reaction. |
| NBT/BCIP Substrate | Chromogenic substrate for AP; produces a purple, insoluble precipitate [10]. | Ideal for permanent mounts and brightfield microscopy. |
| Formamide | Component of hybridization buffer [4]. | Lowers the melting temperature (Tm), allowing hybridization at lower, gentler temperatures. |
| Salmon Sperm DNA | A blocking agent in pre-hybridization and hybridization buffers [4]. | Competes with the probe for non-specific binding sites, thereby reducing background noise. |
| Proteinase K | A proteolytic enzyme used for permeabilization [4]. | Must be carefully titrated; over-digestion damages tissue, under-digestion reduces signal. |
| Stringency Wash Buffers | Buffers like SSC with controlled salt concentration and temperature [4]. | Critical for removing imperfectly matched probes; higher temperature/lower salt increases stringency. |
Diagnosing and fixing weak or absent NBT/BCIP staining requires a holistic view of the ISH protocol, from tissue preparation and probe design to detection and visualization. By understanding the chemical principles behind the method, systematically troubleshooting using the provided workflow, and quantitatively optimizing key parameters as in the q2PISH protocol, researchers can transform a problematic assay into a robust and reliable source of high-quality gene expression data. The consistent application of these detailed protocols and the use of high-quality reagents, as outlined in the Scientist's Toolkit, are fundamental to achieving excellence in ISH research within the demanding contexts of scientific discovery and drug development.
Eliminating High Generalized and Non-Specific Background
In the context of NBT/BCIP staining protocols for in situ hybridization (ISH) research, achieving high signal-to-noise ratios is fundamental to data integrity. The intense blue/purple precipitate formed by the NBT/BCIP substrate provides excellent detection sensitivity for alkaline phosphatase (AP) activity, but high generalized and non-specific background staining can compromise the interpretation of spatial gene expression patterns [2]. Such background issues can obscure genuine signals, lead to false positives, and ultimately undermine the validity of experimental conclusions in both basic research and drug development applications. This application note systematically addresses the common pitfalls that lead to elevated background and provides detailed, actionable protocols for its elimination, ensuring that the resulting data is both reliable and publication-quality.
The NBT/BCIP chromogenic reaction is prized for its sensitivity and the stability of the resulting precipitate, which resists fading when exposed to light [2]. However, this same sensitivity also makes the system susceptible to non-specific interactions that manifest as a diffuse blue stain across the tissue section. A common manifestation of this problem is a general blue staining of the whole tissue, which can often be traced to overfixation [35]. Furthermore, the final color of the precipitate itself can serve as a diagnostic tool; variations from the expected deep blue to brown or purple can indicate issues with target abundance, probe characteristics, or the pH of the detection buffer, which should be carefully adjusted to pH 9.5 [35]. Understanding and controlling these variables is essential for researchers aiming to generate robust, reproducible ISH data, particularly when studying gene expression patterns in complex tissues.
Root Cause Analysis of Background Staining
Effective troubleshooting requires a systematic approach to identifying the underlying causes of background. These causes can be broadly categorized into issues related to tissue preparation, probe hybridization dynamics, and immunological detection.
Tissue-Derived Background: Incomplete removal of paraffin during deparaffinization is a primary culprit, leading to poor staining and high background [27]. Overfixation, particularly with aldehydes, can create a general blue staining across the entire tissue section by altering the tissue chemistry in a way that promotes non-specific binding [35]. Additionally, insufficient permeabilization can trap reagents, while inadequate blocking fails to prevent non-specific attachment of probes and antibodies to positively charged amines and other reactive sites in the tissue [4].
Probe and Hybridization-Derived Background: The stringency of the hybridization and post-hybridization washes is critical. If the temperature is too low or the salt concentration too high during washes, weakly bound or mismatched probes may not be effectively removed [27]. Using a probe concentration that is too high is a frequent error that satur specific binding sites and promotes binding to non-target sequences. Probe degradation or the use of probes that are too long can also increase the likelihood of non-specific hybridization events [27] [4].
Detection-Derived Background: In the detection phase, over-incubation with the NBT/BCIP substrate solution will inevitably lead to high background, as the enzyme reaction proceeds beyond the optimal point [4]. An incorrect pH of the AP detection buffer (it must be near pH 9.5) can alter reaction kinetics and precipitate color [35]. Drying of slides during any step of the procedure, particularly from the edges, is a common technical error that causes severe, uneven background staining [35]. Furthermore, endogenous alkaline phosphatase activity, if not properly inhibited, will catalyze the substrate reaction indiscriminately, generating widespread background.
Table 1: Troubleshooting Guide for High Background in NBT/BCIP Staining
| Problem Manifestation | Primary Cause | Corrective Action |
|---|---|---|
| High general blue background [35] | Overfixation of tissue | Optimize fixation time; include acetylation step to block amines [4]. |
| Brown-purple instead of blue signals [35] | Sub-optimal detection buffer pH or high target abundance | Adjust detection buffer to pH 9.5; optimize probe concentration and hybridization time [35]. |
| Uneven or patchy staining [4] | Drying of slides, uneven probe distribution | Use a humidified chamber; ensure coverslips are sealed properly; apply probe evenly. |
| Non-specific signals [4] | Off-target probe binding, insufficient stringency washes | Increase wash stringency (higher temperature, lower SSC); use control probes (sense strand); confirm probe specificity. |
Optimized Step-by-Step Protocol for Low-Background ISH
Tissue Preparation and Pre-Treatment
Proper tissue preparation lays the foundation for a clean ISH experiment. The goal is to preserve nucleic acid integrity and tissue morphology while ensuring maximum accessibility for the probe.
- Deparaffinization and Rehydration: For formalin-fixed, paraffin-embedded (FFPE) sections, complete paraffin removal is non-negotiable. Place slides in a rack and perform the following sequential washes:
- Xylene: 2 x 3 minutes
- Xylene:1:1 with 100% ethanol: 3 minutes
- 100% ethanol: 2 x 3 minutes
- 95% ethanol: 3 minutes
- 70% ethanol: 3 minutes
- 50% ethanol: 3 minutes
- Rinse with cold tap water [27]
- Critical Note: From this point onward, the slides must not be allowed to dry out, as this causes irreversible, high-level non-specific antibody binding and background staining [27].
- Permeabilization: Digest with 20 µg/mL proteinase K in pre-warmed 50 mM Tris buffer for 10–20 minutes at 37°C. The concentration and time must be optimized for each tissue type and fixation condition. Insufficient digestion reduces hybridization signal, while over-digestion damages tissue morphology [27].
- Acetylation (Optional but Recommended): Following permeabilization, an acetylation step can be introduced to chemically block positively charged amines in the tissue, which dramatically reduces non-specific binding of probes and antibodies. This is a powerful tool for combating persistent background [4].
- Pre-Hybridization Blocking: Incubate slides in a pre-hybridization buffer for 30-60 minutes at 37-45°C in a humidified chamber. A standard recipe includes 50% formamide, 1x SSC, 50 µg/mL heparin, 100 µg/mL denatured salmon sperm DNA, and 0.1% Tween-20 [4]. This conditions the sample and blocks nonspecific binding sites.
Hybridization and Stringency Washes
This phase requires precision in temperature and timing to ensure specific probe binding.
- Probe Hybridization: Denature the labeled probe at 95°C for 5 minutes and immediately chill on ice. Apply the denatured probe diluted in hybridization buffer, cover with a coverslip, and incubate overnight (16-18 hours) in a sealed, humidified chamber at the appropriate hybridization temperature (typically 55-65°C) [27] [4].
- Stringency Washes: These washes are critical for removing unbound and weakly bound probes.
- First Wash: 50% formamide in 2x SSC, 3 x 5 minutes at 37-45°C [27].
- Second Wash: 0.1-2x SSC, 3 x 5 minutes at 25-75°C [27]. The temperature and SSC concentration for this wash are key levers for controlling stringency. Use higher temperatures and lower SSC for more stringent conditions, especially for complex or repetitive probes.
Immunological Detection and NBT/BCIP Development
The final stage involves targeted detection of the hybridized probe and controlled chromogenic development.
- Antibody Blocking and Incubation: Transfer slides to a humidified chamber and block with 200 µL of blocking buffer (e.g., MABT + 2% BSA or serum) for 1-2 hours at room temperature. Drain the buffer and incubate with the anti-label antibody (e.g., anti-DIG-AP) diluted in blocking buffer for 1-2 hours at room temperature [27].
- Post-Antibody Washes: Wash slides 5 x 10 minutes with MABT (Maleic Acid Buffer with Tween) at room temperature. MABT is gentler than PBS for nucleic acid detection and helps reduce background [27].
- Color Development with NBT/BCIP: Prepare the NBT/BCIP working solution fresh. For every 5 ml of alkaline phosphatase buffer (100 mM Tris-HCl pH 9.5, 150 mM NaCl, 1 mM MgCl₂), add 33 µl of NBT (50 mg/ml) first, mix, then add 16.5 µl of BCIP (50 mg/ml), and mix again [29] [35]. Use the solution within one hour. Apply to the slides and monitor the development of the blue/purple precipitate closely. Stop the reaction by immersing the slides in distilled water when the specific signal is clear and background is minimal.
- Mounting: It is crucial to note that NBT/BCIP is not compatible with xylene-containing mounting media (e.g., DPX), as this can cause crystal formations of the color precipitate. Use specific aqueous mounting media like Vectamount or Immunomount [35].
The following workflow diagram summarizes the critical control points in the optimized ISH protocol for minimizing background.
The Scientist's Toolkit: Essential Reagents for Background Suppression
Table 2: Research Reagent Solutions for Low-Background ISH
| Reagent / Solution | Function in Background Reduction | Key Considerations |
|---|---|---|
| Proteinase K [27] [4] | Enzymatically digests proteins to permeabilize tissue, allowing probe access. | Concentration and time must be titrated; over-digestion damages morphology. |
| Pre-Hybridization Buffer (with blocking agents) [4] | Blocks non-specific binding sites in the tissue before probe application. | Typically contains Denhardt's solution, BSA, herring sperm DNA, and tRNA. |
| Formamide [27] [4] | A denaturant used in hybridization buffers and stringent washes to control the effective temperature of hybridization. | Allows for lower hybridization temperatures while maintaining stringency. |
| Saline Sodium Citrate (SSC) [27] | Determines the stringency of post-hybridization washes; lower SSC concentration increases stringency. | A 20x stock is common; use 0.1x-2x SSC for stringent washes [27]. |
| NBT/BCIP Substrate [29] [2] | Chromogenic substrate for Alkaline Phosphatase, producing an intense blue/purple precipitate. | Prepare fresh in AP buffer at pH 9.5; monitor development closely to prevent over-staining. |
| Blocking Serum/BSA [27] | Blocks non-specific binding sites for the antibody-conjugate during the detection phase. | Use from the same species as the secondary antibody host or with inert proteins like BSA. |
| Aqueous Mounting Medium [35] | Preserves the chromogenic signal without causing crystal artifacts. | Must be xylene-free to avoid dissolution or crystallization of the NBT/BCIP precipitate. |
Achieving low-background, high-quality results with NBT/BCIP staining in ISH is a attainable goal through meticulous attention to protocol details. The interplay of proper tissue pretreatment, rigorously controlled hybridization and wash stringency, and careful management of the detection steps forms the cornerstone of success. By systematically implementing the optimized protocols and troubleshooting guides provided herein—paying particular attention to critical steps such as acetylation, stringency control, and substrate pH management—researchers can consistently generate clean, reliable, and interpretable data. This level of experimental rigor is essential for advancing our understanding of gene expression in development and disease, and for supporting robust decision-making in the drug development pipeline.
In situ hybridization (ISH) stands as a pivotal technique in molecular pathology and research, enabling precise localization of specific nucleic acid sequences within intact tissues and cells. The chromogenic detection system utilizing nitro blue tetrazolium (NBT) and 5-bromo-4-chloro-3-indolyl phosphate (BCIP) represents one of the most widely employed methods for visualizing hybridization signals through enzymatic amplification. This system typically yields a deep blue-purple precipitate at the site of target nucleic acid localization, providing excellent contrast for brightfield microscopy.
However, researchers frequently encounter unexpected color variations in NBT/BCIP staining outcomes, with precipitates ranging from deep blue to brown-purple hues. These color shifts are not merely cosmetic concerns but often indicate underlying variations in experimental conditions that may affect result interpretation, quantification, and reproducibility. The NBT/BCIP chromogenic reaction involves a complex biochemical process where alkaline phosphatase dephosphorylates BCIP, generating an intermediate that reduces NBT to an insoluble purple-blue formazan precipitate. Slight alterations in this process can significantly impact the final precipitate color, potentially confounding experimental conclusions in both research and diagnostic applications.
This application note systematically addresses the factors contributing to color variations in NBT/BCIP staining, provides optimized protocols for consistent results, and offers comprehensive troubleshooting guidance to ensure reliable detection of nucleic acid targets in ISH experiments.
Factors Influencing NBT/BCIP Precipitate Color
Chemical and Environmental Determinants
The color of NBT/BCIP precipitate is influenced by multiple interconnected factors that affect the chemical reduction process. Understanding these variables is essential for controlling and reproducing staining outcomes:
pH variations: The alkaline phosphatase enzyme exhibits optimal activity within a specific pH range (typically pH 9.0-9.5). Deviations from this optimal range can alter enzyme kinetics and the chemical properties of the final formazan product, resulting in color shifts toward brownish hues [4].
Cation concentrations: Divalent cations such as magnesium (Mg²⁺) and zinc (Zn²⁺) serve as essential cofactors for alkaline phosphatase activity. Inadequate cation concentrations can reduce enzymatic efficiency, leading to incomplete reduction and atypical precipitate colors [36].
Incubation time and temperature: Excessive incubation times or elevated temperatures can cause over-development of the chromogenic reaction, increasing precipitate density and potentially shifting color perception toward darker brown-purple appearances. Conversely, insufficient development yields lighter, more diffuse blue staining [37] [38].
Fixation artifacts: Incomplete fixation or over-fixation with aldehydes can modify tissue chemistry, potentially interfering with the enzymatic reaction. Consistent fixation using 4% paraformaldehyde for optimized time periods specific to tissue thickness is critical for reproducible results [37] [19].
Endogenous phosphatase activity: Inadequately blocked endogenous phosphatases can contribute to background staining and localized color variations, particularly in tissues with high endogenous enzyme levels such as kidney and intestine [36].
Detection System Components
The specific components and quality of the detection system significantly impact precipitate formation and characteristics:
Enzyme conjugate integrity: Degraded or inactivated alkaline phosphatase conjugates yield suboptimal enzymatic activity, resulting in weak or atypical staining. Regular validation of conjugate activity is essential [38].
Substrate formulation and age: The concentration and stability of both NBT and BCIP components directly affect precipitate color. Aged or improperly stored substrates may develop oxidation products that alter staining characteristics [38].
Mounting media composition: The chemical compatibility of mounting media with the NBT/BCIP precipitate is crucial. Certain aqueous mounting media may slowly dissolve or alter the precipitate over time, potentially changing its spectral properties [38].
Quantitative Analysis of Color Variation Factors
Table 1: Chemical Parameters Affecting NBT/BCIP Precipitate Color
| Parameter | Optimal Range | Effect on Color if Suboptimal | Quantitative Impact |
|---|---|---|---|
| pH of Reaction Buffer | 9.0-9.5 | Brownish hue development | >20% color shift outside range [4] |
| Mg²⁺ Concentration | 1-5 mM | Lighter, diffuse blue staining | 50% signal reduction at <0.5 mM [36] |
| Incubation Temperature | 37°C ± 2°C | Darker brown-purple at higher temp | 15% color density change per 5°C deviation [37] |
| Incubation Time | 5-30 minutes | Increased brown tones with overtime | 2X background after 45 minutes [38] |
| NBT/BCIP Ratio | 1:1 to 1:2.5 | Altered precipitate morphology | 30% efficiency loss at 1:5 ratio [38] |
| Substrate Age | <6 months at -20°C | Increased background, color shift | 40% signal loss after 1 year [38] |
Table 2: Troubleshooting Guide for NBT/BCIP Color Variations
| Observed Color | Potential Causes | Corrective Actions | Expected Outcome |
|---|---|---|---|
| Light Blue, Diffuse | Insufficient development time | Increase incubation time to 20-45 min | More intense, localized blue-purple signal [38] |
| Brown-Purple | Over-development, high pH | Reduce time to 10-20 min; verify buffer pH | Balanced blue-purple precipitate [4] |
| Patchy/Uneven Staining | Incomplete tissue permeabilization | Optimize proteinase K concentration (1-20 µg/mL) and time | Uniform signal distribution [37] |
| High Background | Inadequate blocking or washing | Increase blocking time; use stringent washes (0.1× SSC at 60-65°C) | Clean background with specific signal [37] [38] |
| No Signal | Enzyme conjugate inactivation | Test conjugate activity with fresh substrate | Detectable specific staining [38] |
Optimized NBT/BCIP Staining Protocol
Pre-Hybridization Sample Preparation
Proper sample preparation establishes the foundation for consistent NBT/BCIP staining results:
Tissue fixation and sectioning: Fix tissues promptly in 4% paraformaldehyde (PFA) for time periods optimized according to tissue thickness: 15-30 minutes for cryosections (7-15 µm) at room temperature, or 6-48 hours for paraffin sections at 4°C. Use RNase-free conditions throughout the process to preserve nucleic acid integrity [37].
Slide preparation and adhesion: Use charged slides for optimal tissue adhesion. Avoid protein-based adhesives in flotation baths as they can block charged slide surfaces, leading to inconsistent adhesion and uneven staining due to reagent pooling beneath lifting sections [19].
Permeabilization optimization: Treat sections with proteinase K at carefully optimized concentrations (1-20 µg/mL) and incubation times (5-30 minutes at room temperature). Excessive digestion causes tissue loss and distortion, while insufficient digestion compromises probe penetration [37].
Endogenous enzyme inhibition: Incubate sections in 3% H₂O₂ at room temperature for 10-15 minutes to quench endogenous peroxidase activity, particularly important when combining ISH with immunohistochemical detection [37].
Pre-hybridization blocking: Apply pre-hybridization buffer containing denatured salmon sperm DNA (100µg/mL), heparin (50µg/mL), and SDS (1% w/v) in 50% formamide with 1× SSC. Incubate at 37-45°C for 30-60 minutes to block nonspecific binding sites [4].
Hybridization and Stringency Controls
The hybridization phase requires precise temperature and time control for specific target detection:
Probe design and application: Design probes targeting the 3' untranslated region (3' UTR) of mRNA for better sequence specificity, with optimal length of 50-150 base pairs. Apply denatured probe (0.5-2 µg/mL for mRNA targets) in appropriate hybridization buffer and incubate overnight (16-18 hours) at 37-45°C in a humidified chamber to prevent evaporation [37] [4].
Stringency washes: Perform post-hybridization washes with increasing stringency: start with 2× SSC + 0.1% SDS at room temperature for 2×5 minutes, followed by 0.1× SSC at 60-65°C for 2×15-20 minutes. Maintain precise temperature control during stringent washes as this critically affects background levels [37].
Detection system application: Apply alkaline phosphatase-conjugated antibody (e.g., anti-digoxigenin) diluted 1:500-1:2000 in appropriate buffer. Incubate for 1-2 hours at room temperature with gentle agitation [37].
Chromogenic Development with NBT/BCIP
The development phase requires careful monitoring to achieve optimal signal-to-noise ratio:
Substrate preparation: Prepare fresh NBT/BCIP substrate solution according to manufacturer specifications. Filter-sterilize if necessary to remove particulates that might create nucleation sites for non-specific precipitation [38].
Controlled development: Apply substrate to sections and incubate at room temperature or 37°C. Monitor development progress microscopically every 5-10 minutes. The optimal development time typically ranges from 5-30 minutes, depending on target abundance [37].
Reaction termination: Stop the development reaction by transferring slides to distilled water or TE buffer (pH 7.5-8.0) when specific signal is clearly visible against minimal background. Do not allow the reaction to proceed to endpoint, as this increases background and may alter precipitate color toward brown hues [38].
Counterstaining considerations: Apply a light nuclear counterstain such as Mayer's hematoxylin for 5-60 seconds only. Excessive counterstaining can mask NBT/BCIP signals, particularly when using DAB in dual detection protocols [38].
Mounting and preservation: Mount sections with aqueous mounting medium appropriate for NBT/BCIP precipitates. Avoid organic solvents that might dissolve or alter the formazan precipitate. For permanent preservation, use synthetic mounting media compatible with the precipitate characteristics [38].
Advanced Applications and Signal Enhancement
Fluorescence Detection with NBT/BCIP
While traditionally considered a chromogenic substrate for brightfield microscopy, the NBT/BCIP reaction product exhibits previously underutilized fluorescent properties that enable advanced detection modalities:
Near-infrared fluorescence: The dark purple formazan precipitate formed from NBT/BCIP demonstrates significant near-infrared (NIR) fluorescence, enabling high-resolution three-dimensional imaging using confocal microscopy. This property permits correlation of brightfield and fluorescent signals from the same reaction product [32].
Multiplexing capabilities: The fluorescent characteristics of NBT/BCIP precipitation allow combination with other fluorescent labels in multiplex detection schemes. The large Stokes shift of the precipitate facilitates spectral separation from conventional fluorophores [32].
Enhanced sensitivity: Fluorescent detection of NBT/BCIP precipitates can reveal signals that might be subvisual in brightfield microscopy, potentially increasing the sensitivity threshold for low-abundance targets [32].
Enzyme-Labeled Fluorescence (ELF) Technology as Alternative
For applications requiring high-sensitivity fluorescent detection, ELF technology provides a robust alternative to conventional chromogenic systems:
Signal amplification principle: ELF 97 phosphate serves as a substrate for alkaline phosphatase, generating a bright yellow-green fluorescent precipitate (ELF 97 alcohol) upon enzymatic cleavage. This precipitate exhibits an unusually large Stokes shift (>180 nm) and exceptional photostability [36].
Advantages over NBT/BCIP: The ELF 97 signal demonstrates superior photostability compared to fluorescein-based detection systems, with signals persisting for months to years with minimal degradation. The large Stokes shift effectively separates signal from tissue autofluorescence [36].
Application compatibility: ELF technology is compatible with standard fluorescence microscopy equipment and can be combined with other fluorophores for multiplex detection without significant spectral overlap [36].
Table 3: Research Reagent Solutions for NBT/BCIP-Based Detection
| Reagent Category | Specific Products | Function in ISH | Optimization Tips |
|---|---|---|---|
| Fixatives | 4% Paraformaldehyde (PBS or Phosphate Buffer) [4] | Preserves tissue architecture and nucleic acid integrity | Optimize fixation time based on tissue thickness: 15-30 min for cryosections, 6-48h for paraffin sections [37] |
| Permeabilization Agents | Proteinase K (20mg/mL) [4], Triton X-100, Tween-20 [37] | Enables probe access to intracellular targets | Titrate proteinase K (1-20 µg/mL); excessive digestion causes tissue damage [37] |
| Blocking Buffers | BSA, Casein (3% in TBS or PBS) [4], Denhardt's Solution [4] | Reduces non-specific probe binding | Include in pre-hybridization step; use with salmon sperm DNA for enhanced blocking [4] |
| Hybridization Buffers | Saline Sodium Citrate (20X) [4], Formamide-based buffers [4] | Provides optimal stringency for specific hybridization | Standard SSC concentration: 1-2X; formamide (50% v/v) reduces hybridization temperature [4] |
| Detection Components | Anti-DIG-alkaline phosphatase [37], NBT/BCIP substrates [37] | Enzymatic generation of detectable signal | Dilute antibody 1:500-1:2000; monitor development microscopically [37] [38] |
| Mounting Media | Aqueous mounting media [38], Antifade solutions [37] | Preserves signal and tissue morphology | Use non-organic media for NBT/BCIP; antifade for fluorescent detection [37] [38] |
The color variations observed in NBT/BCIP staining—ranging from deep blue to brown-purple precipitates—serve as important indicators of underlying biochemical conditions within the ISH experimental system. Rather than being merely aesthetic concerns, these color shifts provide valuable diagnostic information about reaction parameters that may require optimization. Through systematic control of fixation conditions, hybridization stringency, enzymatic detection parameters, and development monitoring, researchers can achieve consistent, reproducible NBT/BCIP staining results with optimal signal-to-noise characteristics.
The comprehensive protocols and troubleshooting guidance provided in this application note establish a foundation for reliable NBT/BCIP-based detection in ISH applications. Furthermore, the advanced detection modalities, including fluorescent properties of NBT/BCIP precipitates and alternative enzyme-based detection systems, offer researchers flexible options for enhancing sensitivity and enabling multiplex detection schemes. By understanding and controlling the factors that influence chromogenic development, researchers can ensure the accuracy, reproducibility, and interpretability of their ISH experiments in both basic research and diagnostic applications.
In situ hybridization (ISH) is a foundational technique in molecular biology, enabling the precise localization of nucleic acid sequences within fixed tissues and cells. The chromogenic detection of hybridized probes using Nitro-blue Tetrazolium (NBT) and 5-Bromo-4-chloro-3-indolyl-phosphate (BCIP), which yields an insoluble purple precipitate, remains a widely used method for its permanence and compatibility with brightfield microscopy. However, achieving optimal signal-to-noise ratio requires careful balancing of several critical parameters. This application note provides a detailed protocol and optimization strategies for NBT/BCIP-based detection in ISH, specifically addressing probe concentration, permeabilization methods, and development time, framed within the context of a broader thesis on robust ISH protocol development.
Optimization of Key Parameters
Probe Design and Concentration
Probe characteristics are primary determinants of hybridization efficiency and signal specificity. The table below summarizes key optimization parameters supported by empirical studies.
Table 1: Optimization Guidelines for Probe Design and Hybridization
| Parameter | Optimal Range | Experimental Support | Impact on Results |
|---|---|---|---|
| Probe Length | ~100 base pairs | Optimized for Brassica rapa mRNA ISH; provided good hybridization signal [39]. | Shorter probes (~100 bp) improve tissue penetration and access to target sequences. |
| Probe Concentration | 100 ng/μL | Effective concentration for detecting BrrCLV3 and BrrWUSa genes in turnip [39]. | Balances sufficient signal intensity with minimal non-specific background binding. |
| Hybridization Temperature | 52-62°C | 52°C was optimal for washing in turnip [39]; 62°C used for pre-hybridization/hybridization in q2PISH [10]. | Higher temperature increases stringency, reducing non-specific probe binding and background. |
Permeabilization and Fixation
Effective permeabilization is critical for probe access, while fixation preserves tissue architecture and nucleic acid integrity. The choice of method depends heavily on tissue type and fragility.
Table 2: Permeabilization and Fixation Methods for Different Tissues
| Method | Protocol Details | Application Context | Advantages |
|---|---|---|---|
| Proteinase K Digestion | 30-minute pretreatment [39]. | Standard for robust plant tissues (e.g., turnip shoot apical meristems) [39]. | Effectively digests proteins, allowing probe penetration into dense tissues. |
| Acid-Based Permeabilization (NAFA) | Nitric Acid/Formic Acid treatment; no protease digestion [40]. | Delicate tissues (e.g., planarian epidermis, regeneration blastemas, killifish fins) [40]. | Preserves delicate tissue structures and antigen epitopes for combined ISH/immunostaining. |
| Fixative Choice | FAA (Formalin-Acetic Acid-Alcohol) [39]. | Plant tissues (e.g., turnip) [39]. | Provides optimal mRNA retention for ISH in specific plant models. |
Chromogenic Development and Quantification
The NBT/BCIP development reaction is catalyzed by Alkaline Phosphatase (AP). Controlling this step is vital for preventing high background and for quantitative applications.
Table 3: Chromogenic Development and Detection Strategies
| Aspect | Recommendation | Purpose and Rationale |
|---|---|---|
| Development Time | Monitor until signal appears; avoid over-development [4]. | Prevents high background staining; optimal time is tissue and target-dependent. |
| Quantitative q2PISH | Sequential application of pNPP substrate followed by NBT/BCIP, with cell nuclei count [10]. | pNPP provides a soluble, quantifiable yellow product (405 nm absorbance); subsequent NBT/BCIP allows qualitative single-cell resolution [10]. |
| Substrate Buffer | 0.1 M Tris-HCl, pH 9.5, 0.1 M NaCl [10]. | Standard AP reaction buffer for optimal enzyme activity with NBT/BCIP. |
Experimental Protocol: mRNA ISH with NBT/BCIP Detection
Reagent Setup
- Pre-hybridization Buffer (100 mL): 50% formamide (v/v), 1X SSC, 50 μg/mL Heparin, 100 μg/mL denatured Salmon Sperm DNA, 1% (w/v) SDS, 0.1% (v/v) Tween-20, made up with RNase-free water. Filter-sterilize before use [4].
- NBT/BCIP Substrate Solution: Dilute NBT/BCIP stock solution 1:50 in 0.1 M Tris-HCl (pH 9.5), 0.1 M NaCl, 50 mM MgCl₂ [39].
- Blocking Solution: Use commercial blocking reagents or 1% (w/v) Bovine Serum Albumin (BSA) in buffer [4].
Step-by-Step Procedure
- Fixation and Permeabilization:
- Pre-hybridization:
- Incubate samples in pre-warmed pre-hybridization buffer at 37-45°C for 30-60 minutes in a humidified chamber [4].
- Hybridization:
- Denature the DIG-labeled RNA probe (100 ng/μL) at 95°C for 5 minutes, then place on ice.
- Apply the probe diluted in hybridization buffer to the sample. Ensure full coverage.
- Cover with a coverslip and incubate overnight (16-18 hours) at the appropriate hybridization temperature (e.g., 52-62°C) in a humidified chamber [10] [4] [39].
- Stringency Washes:
- Gently remove coverslips and perform post-hybridization washes. Adjust stringency using temperature and salt concentration (e.g., lower SSC concentration) to remove unbound probe [4].
- Immunological Detection:
- Incubate samples with a 1:2000 dilution of anti-DIG-AP conjugated antibody in blocking solution [10].
- Wash extensively to eliminate non-specific antibody binding.
- Chromogenic Development:
- Apply NBT/BCIP substrate solution to the samples.
- Monitor the development of the purple precipitate in a low-light environment. Avoid over-developing.
- Once the desired signal intensity is achieved, stop the reaction by washing with distilled water or a stop solution [4].
- Mounting and Imaging:
- Mount samples with an aqueous or permanent mounting medium.
- Image using brightfield microscopy [4].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Key Reagents for NBT/BCIP-based ISH
| Reagent / Solution | Critical Function | Examples & Notes |
|---|---|---|
| Fixatives | Preserves tissue morphology and nucleic acid integrity. | 4% Paraformaldehyde (PFA) [10] [4], FAA for plants [39]. |
| Permeabilization Agents | Enables probe entry into cells. | Proteinase K [4] [39], Detergents (Triton X-100, Tween-20) [4], Acid-based treatments (NAFA) [40]. |
| Blocking Reagents | Reduces non-specific binding of probes and antibodies. | BSA, Casein, Denhardt's Solution, heparin, salmon sperm DNA [4]. |
| Hybridization Buffers | Creates optimal ionic and pH conditions for specific probe binding. | Typically contains formamide, SSC, and Denhardt's solution [4]. |
| Stringency Wash Buffers | Removes weakly bound or mismatched probes post-hybridization. | Saline-sodium citrate (SSC) buffer; temperature and concentration determine stringency [4]. |
| Anti-DIG-AP Antibody | Binds to DIG-labeled probe; AP enzyme catalyzes color reaction. | Commercial conjugates (e.g., from Roche); dilution typically 1:2000 [10]. |
| NBT/BCIP Substrate | AP substrate yielding insoluble purple precipitate for detection. | Commercially available as a ready-to-use stock solution [10] [4]. |
Troubleshooting Common Issues
- High Background Signal: Increase the stringency of post-hybridization washes (e.g., higher temperature, lower SSC concentration). Ensure adequate blocking and consider adding an acetylation step after permeabilization [4].
- Weak or No Signal: Optimize probe concentration and check for RNA degradation. Increase permeabilization time or concentration (e.g., Proteinase K) and ensure effective fixation [4] [39].
- Uneven Staining: Apply probe evenly and use a properly sealed humidified chamber to prevent evaporation and drying of the sample [4].
Signaling Pathways and Workflow Visualization
<100 chars: ISH with NBT/BCIP Experimental Workflow
The reliability of in situ hybridization using NBT/BCIP detection hinges on the systematic optimization of probe concentration, permeabilization strategy, and development time. The protocols and data tables provided here serve as a detailed guide for researchers to adapt and refine these parameters for their specific experimental systems, from robust plant tissues to delicate animal models. By applying these optimized strategies, scientists can achieve high-resolution, specific localization of gene expression, thereby advancing research in developmental biology, biomarker discovery, and drug development.
Within the framework of a comprehensive thesis on the NBT/BCIP staining protocol for in situ hybridization (ISH) research, addressing sample-specific challenges is paramount for experimental reproducibility and accuracy. This application note focuses on two predominant obstacles: lipid-rich tissues and over-fixation. Lipid-rich tissues, such as liver, brain, and adipose tissue, pose a significant challenge due to the propensity for non-specific precipitation of the NBT/BCIP formazan product onto lipid droplets, complicating signal interpretation [41]. Concurrently, over-fixation can mask epitopes, reduce nucleic acid accessibility, and create high general background staining, obscuring specific signals [41] [42]. This document provides detailed, actionable protocols and data-driven solutions to empower researchers in overcoming these hurdles.
Core Challenges and Quantitative Impacts
The challenges presented by lipid-rich tissues and over-fixation manifest in distinct yet disruptive ways. The following table summarizes their primary effects on NBT/BCIP-based ISH.
Table 1: Impact of Lipid-Rich Tissues and Over-fixation on NBT/BCIP Staining
| Challenge | Primary Effect on ISH | Visual Manifestation | Underlying Cause |
|---|---|---|---|
| Lipid-Rich Tissues | Non-specific precipitate trapping [41] | Speckled or diffuse background stain mimicking true signal | NBT/BCIP formazan precipitate becomes trapped in intracellular lipid droplets [41]. |
| Over-fixation | High general blue background [41] | Uniform blue staining across the entire tissue section | Over-crosslinking from prolonged fixation increases non-specific binding of probes or detection reagents [41] [42]. |
Experimental Protocols for Challenge Mitigation
Protocol for Lipid-Rich Tissues: Delipidation
The following protocol is designed for cryosections of tissues like heart or liver, which are prone to intracellular lipid accumulation [41].
Workflow: Delipidation of Lipid-Rich Cryosections
Materials & Reagents
- Chloroform (High purity) [41]
- Ethanol Series (100%, 95%, 70%)
- Phosphate-Buffered Saline (PBS)
- Coplin jars or slide-staining dishes
Methodology
- Sectioning and Fixation: Begin with standard fixed cryosections mounted on charged slides.
- Delipidation: Immerse slides in chloroform for 10 minutes at room temperature [41]. Perform this step in a fume hood.
- Rehydration: Transfer slides through a graded ethanol series (e.g., 100%, 95%, 70%) for 2 minutes each, concluding with a brief rinse in nuclease-free PBS.
- Pre-hybridization: Immediately proceed with the proteinase K digestion and pre-hybridization steps of your standard ISH protocol.
Protocol for Over-fixed Tissues: Antigen Retrieval and Optimized Detection
This protocol mitigates the effects of over-fixation by restoring nucleic acid accessibility and optimizing detection conditions.
Workflow: Mitigating Over-fixation Effects
Materials & Reagents
- Proteinase K (e.g., 10 µg/ml for 3-10 minutes at 37°C) [20] [43]
- Heat-induced Epitope Retrieval Buffer (appropriate for your fixative)
- Stringent Wash Buffer (e.g., 1x SSC)
- NBT/BCIP Stock Solution (Product No. 11681451001, Sigma-Aldrich) [41]
- Aqueous Mounting Media (e.g., Crystalmount, Vectamount, Immunomount) [41]
Methodology
- Dewaxing and Rehydration: Ensure complete paraffin removal using fresh xylene substitutes and graded ethanols.
- Heat-Induced Epitope Retrieval: Immerse slides in pre-heated retrieval buffer and incubate for 15 minutes once the buffer reaches 98°C [43]. Allow slides to cool slowly post-retrieval.
- Optimized Enzymatic Digestion: Digest with proteinase K (e.g., 10 µg/ml) for 3-10 minutes at 37°C [43]. Critical: Over-digestion can destroy tissue architecture and signal, while under-digestion reduces probe accessibility. Optimization using positive control tissue is essential.
- Hybridization and Stringent Washes: Perform hybridization under standardized conditions. For post-hybridization washing, use a stringent wash with SSC buffer at 75-80°C for 5 minutes to reduce non-specific background [43].
- Controlled Detection: Equilibrate sections in an alkaline phosphatase reaction buffer (e.g., NTMT: 100 mM NaCl, 50 mM MgCl₂, 100 mM Tris pH 9.5, 0.1% Tween20) [20]. Add NBT/BCIP substrate and develop in the dark. Monitor the reaction microscopically every 10-15 minutes and stop the reaction by rinsing with distilled water as soon as specific signal is optimal or background begins to appear [43].
- Mounting: Use xylene-free, aqueous mounting media like Crystalmount or Immunomount. Xylene-based media can dissolve precipitates or cause crystal formation [41].
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Managing ISH Challenges
| Reagent | Function/Application | Considerations for Use |
|---|---|---|
| Chloroform | Delipidation of cryosections [41] | Use in a fume hood; 10-minute incubation at RT is sufficient for many tissues. |
| Proteinase K | Digests cross-linked proteins to unmask nucleic acid targets [43] | Concentration and time (3-10 min) are critical and must be optimized per tissue and fixation [43]. |
| NBT/BCIP Stock Solution | Alkaline phosphatase substrate yielding an insoluble purple precipitate [41] | Dissolve any precipitate in stock by warming; ensure detection buffer is at pH 9.5 [41]. |
| Aqueous Mounting Media (e.g., Vectamount) | Preserves NBT/BCIP precipitate without dissolution or crystal formation [41] | Avoid xylene-containing media (e.g., DPX) [41]. |
| Stringent Wash Buffer (e.g., SSC) | Removes imperfectly matched or non-specifically bound probes [43] | Temperature control (75-80°C) is vital for balancing background reduction and signal preservation [43]. |
Success in ISH using NBT/BCIP for challenging samples lies in a methodical and pre-emptive approach. For lipid-rich tissues, incorporating a deliberate delipidation step is non-negotiable. For over-fixed specimens, rigorous optimization of retrieval and digestion parameters, coupled with meticulously controlled detection and mounting, is required. By integrating these tailored protocols into their standard workflows, researchers and drug development professionals can significantly enhance the reliability and interpretability of their gene expression data, thereby advancing the scope of their scientific inquiries.
Validation, Controls, and Platform Comparison: Ensuring Confidence in Your Results
Within the framework of a broader thesis on optimizing NBT/BCIP staining protocols for in situ hybridization (ISH), the implementation of rigorous experimental controls is not merely a recommendation but a fundamental requirement. For researchers, scientists, and drug development professionals, data integrity is paramount. Proper controls are the bedrock of this integrity, allowing for the accurate interpretation of staining results, verification of protocol efficacy, and confirmation of target specificity. The chromogenic reaction between alkaline phosphatase and the NBT/BCIP substrate, which yields a characteristic blue-violet precipitate, is a powerful detection method. However, without the proper controls, it is impossible to distinguish true positive signals from non-specific background staining, procedural artifacts, or off-target hybridization. This application note details the three essential controls—positive, negative, and no-probe—that must be incorporated into every ISH experiment utilizing NBT/BCIP detection to ensure reliable and reproducible outcomes.
The Critical Role of Controls in NBT/BCIP-Based ISH
In situ hybridization is a complex multi-step process, and each stage—from tissue fixation and permeabilization to hybridization and chromogenic development—introduces potential variables that can affect the final result. The use of alkaline phosphatase (AP)-conjugated antibodies with NBT/BCIP substrate is particularly common for detecting hapten-labeled probes, such as those tagged with digoxigenin (DIG). The fundamental roles of experimental controls in this context are:
- Validating Technical Execution: Controls confirm that every step of the protocol, from probe hybridization to the enzymatic reaction, has functioned correctly.
- Establishing Specificity: They provide evidence that the observed blue-violet precipitate is due to the specific binding of the probe to its intended nucleic acid target and not to non-specific interactions.
- Aiding in Troubleshooting: When results are suboptimal, controls are the first and most critical diagnostic tool for identifying the source of the problem, be it weak signal, high background, or complete staining failure.
The consequences of omitting these controls are severe, potentially leading to false positive or false negative conclusions that can derail research projects or compromise diagnostic accuracy. It is therefore strongly advised that these controls be included in every experimental run [19] [6].
The table below summarizes the purpose, composition, and interpretation of the three essential controls for an ISH experiment using NBT/BCIP detection.
Table 1: Essential Experimental Controls for ISH with NBT/BCIP Detection
| Control Type | Purpose | Description | Expected Result with NBT/BCIP | Interpretation of Deviation |
|---|---|---|---|---|
| Positive Control | To verify the entire ISH protocol is functioning correctly [19] [6]. | A tissue section with a known, abundant expression of the target nucleic acid, processed identically to the test samples. | Clear, localized blue-violet precipitate in expected cellular compartments. | No stain: Indicates a fundamental failure in the procedure, such as degraded reagents, incorrect temperatures, or inactive enzyme conjugate [6]. |
| Negative Control (Non-specific Probe) | To assess the level of non-specific background staining and off-target hybridization [37]. | A section (can be the positive control tissue) hybridized with a probe that has no sequence similarity to any target in the tissue. | Absence of specific blue-violet staining; only a clean background and counterstain should be visible. | Specific staining pattern: Suggests the probe is binding non-specifically. Probe specificity should be re-checked via BLAST [37]. |
| No-Probe Control | To identify staining artifacts caused by the detection system itself (e.g., endogenous enzyme activity or non-specific antibody binding) [37]. | A section processed through the entire protocol but with the probe omitted from the hybridization buffer. | No blue-violet precipitate development. | Background staining: Points to issues with the detection system, such as insufficient blocking, non-specific antibody binding, or endogenous alkaline phosphatase activity that was not adequately blocked [6]. |
Detailed Methodologies for Control Experiments
Positive Control Protocol
The positive control is the most critical indicator of procedural success.
- Tissue Selection: Select a tissue or cell line with well-documented, high expression of the target RNA or DNA. This tissue should be fixed, embedded, and sectioned using the same methods as the experimental samples to ensure consistency [19] [6].
- Fixation: Use 4% paraformaldehyde (PFA) in RNase-free PBS (pH ≈ 7.4) for cryosections (15-30 min at room temperature) or 10% neutral buffered formalin for paraffin sections (6-48 hours) [37]. Avoid acidic fixatives or those containing heavy metals, which can degrade nucleic acids [37].
- Protocol Execution: Process the positive control slide alongside the test samples through every single step, using the same batches of reagents.
- NBT/BCIP Development: After incubation with the AP-conjugated antibody (e.g., anti-DIG-AP, typically diluted 1:500-1:2000), develop the signal with NBT/BCIP substrate. Monitor the reaction microscopically every 2-5 minutes until the signal is strong and background begins to appear, then stop the reaction by rinsing with distilled water [6] [37]. A clear, positive result confirms that the probe, hybridization conditions, and detection system are all functional.
Negative and No-Probe Control Protocol
These two controls are best run simultaneously to provide complementary information.
- Section Preparation: Two sections from the same test tissue (or the positive control tissue) are prepared on the same slide batch.
- Probe Preparation:
- For the Negative Control: The hybridization buffer contains a non-specific probe (e.g., a sense probe or a probe targeting a non-existent sequence) at the same concentration as the specific probe [37].
- For the No-Probe Control: The hybridization buffer is identical but contains no probe whatsoever [37].
- Hybridization and Washing: Both slides are carried through the hybridization and post-hybridization wash steps identically. For RNA targets, include RNase inhibitors (e.g., 0.1 U/µL RNasin) in all wash solutions prepared with DEPC-treated water to prevent RNA degradation [37].
- Detection and Analysis:
- Proceed with antibody incubation and NBT/BCIP development as with the test slides.
- Interpretation: The ideal outcome is a complete absence of specific blue-violet staining in both controls. Staining in the negative control, but not the no-probe control, indicates a probe-specificity issue. Staining in both controls indicates a problem with the detection system itself, such as inadequate blocking or excessive endogenous enzyme activity.
The Researcher's Toolkit: Essential Reagents for Controlled ISH
Table 2: Key Research Reagent Solutions for ISH Controls
| Reagent / Solution | Function in the Protocol | Key Considerations for Controls |
|---|---|---|
| Charged Slides | Provides strong adhesion for tissue sections during rigorous processing. | Prevents section loss, which is critical for ensuring controls complete the entire protocol [19]. |
| Paraformaldehyde (PFA) 4% | Primary fixative to preserve tissue morphology and nucleic acid integrity. | Consistent fixation across all control and test samples is vital for comparable results [19] [37]. |
| Proteinase K | Enzyme for tissue permeabilization, enabling probe access. | Over-digestion damages morphology; under-digestion reduces signal. Must be optimized and consistent [4] [37]. |
| Pre-hybridization Buffer | Blocks non-specific binding sites to reduce background. | Essential for achieving a clean background in the negative and no-probe controls [4]. |
| DIG-Labeled Probe | Nucleic acid probe for hybridizing to the target sequence. | For the negative control, a validated non-specific probe must be used at the same concentration [37]. |
| Anti-DIG-Alkaline Phosphatase (AP) | Enzyme-conjugated antibody for binding the DIG-labeled probe. | Confirms the detection system is working in the positive control; causes background if non-specifically bound [6]. |
| NBT/BCIP Substrate | Chromogenic substrate for AP, producing a blue-violet precipitate. | Development must be monitored closely and stopped simultaneously for all slides to allow valid comparison [37]. |
| Mayer's Hematoxylin | Nuclear counterstain. | A light counterstain (5-60 seconds) is crucial to avoid masking the specific NBT/BCIP signal [6]. |
Experimental Workflow for ISH Controls
The following diagram illustrates the logical sequence and parallel processing of test samples and the three essential controls within an ISH protocol.
The path to robust, reliable, and publication-quality in situ hybridization data is paved with rigorous experimental controls. The consistent and correct implementation of positive, negative, and no-probe controls is a non-negotiable standard in ISH research, particularly when using sensitive detection methods like NBT/BCIP. These controls provide the necessary context to confidently answer the fundamental question: "Is the staining I see real?" By embedding these controls within the broader context of an optimized NBT/BCIP staining protocol, researchers and drug developers can ensure the validity of their findings, accelerate the troubleshooting process, and ultimately advance scientific discovery with greater confidence and precision.
Correlating Signal Color and Intensity with Target RNA Abundance
In situ hybridization (ISH) research critically depends on robust detection systems to visualize and quantify target RNA abundance within tissue architecture. The chromogenic reaction utilizing Nitro Blue Tetrazolium (NBT) and 5-Bromo-4-Chloro-3-Indolyl-Phosphate (BCIP) stands as a fundamental methodology in this field, providing a permanent, high-resolution means of detecting nucleic acid localization [29]. This system operates through an enzyme-mediated precipitation process where alkaline phosphatase (AP) converts the colorless BCIP into an indoxyl derivative that subsequently reduces NBT to an insoluble, intensely colored formazan precipitate [29]. The resulting purple-blue stain deposits at the site of target RNA hybridization, enabling both morphological assessment and quantitative analysis through signal intensity correlation with target abundance.
The application of NBT/BCIP in RNA detection spans multiple research domains, from developmental biology to molecular pathology. In gametogenesis studies, researchers have employed this system to validate germline-specific gene expression patterns in oyster gonads, confirming spatial localization of markers like TDRD1 and Boll through colorimetric ISH [44]. Similarly, in cancer research, NBT/BCIP has been implemented alongside fluorescence ISH to enhance detection sensitivity for low-abundance targets [45] [46]. The system's compatibility with standard brightfield microscopy and permanent staining properties make it particularly valuable for archival tissue analysis and long-term studies where fluorescence photobleaching may present limitations.
Quantitative Correlation Between Signal Characteristics and RNA Abundance
Fundamental Principles of Signal Development
The NBT/BCIP chromogenic system generates signal through a precipitation mechanism that directly correlates with enzymatic activity. When alkaline phosphatase conjugates bind to hybridization probes via antibody recognition (typically anti-digoxigenin or anti-biotin), they catalyze the conversion of BCIP to an indoxyl derivative, which subsequently reduces NBT to an insoluble purple-blue formazan precipitate [29]. This reaction proceeds linearly with time when substrates are not limiting, resulting in cumulative precipitate deposition proportional to enzyme concentration. The color intensity and deposit density thus directly reflect the number of enzyme molecules present, which in turn correlates with the quantity of target RNA molecules accessible to hybridization probes [46].
Several factors influence the quantitative relationship between final signal intensity and target RNA abundance. The fixation duration significantly impacts miRNA preservation, with extended formalin fixation (100-144 hours) demonstrating superior signal intensity compared to shorter periods (5-24 hours) in controlled studies [47]. The precipitation kinetics follow a sigmoidal relationship where initial nucleation creates seed sites for subsequent precipitate deposition, meaning that lower abundance targets may require longer development times to reach detectable thresholds [46]. This temporal aspect necessitates standardized development periods for comparative analyses between samples.
Experimental Validation of Quantitative Relationships
Table 1: Experimental Evidence for NBT/BCIP Quantitative Detection Performance
| Study Context | Target RNA Type | Detection Limit | Quantitative Correlation Method | Reference |
|---|---|---|---|---|
| Small RNA ISH | miRNA | Single molecule sensitivity | Signal amplification systems | [46] |
| Gametogenesis marker validation | mRNA (TDRD1, Boll) | Tissue-specific expression | Chromogenic signal localization vs RNA-seq | [44] |
| miRNA ISH optimization | miR-205-5p, miR-145-5p, miR-126-3p | Low abundance miRNAs | Intensity rating scale (0-3) vs fixation parameters | [47] |
| Automated quantification | mRNA in whole-slide images | Subcellular compartments | Optical density separation algorithms | [48] |
Research has demonstrated that NBT/BCIP detection achieves sufficient sensitivity for low abundant miRNA detection when combined with appropriate signal amplification strategies [46]. The implementation of automated quantitative analysis methods based on optical density separation techniques provides objective correlation between precipitate intensity and target concentration, overcoming limitations of manual interpretation [48]. These computational approaches precisely quantify nuclear, membrane, and cytoplasmic expressions in whole-slide images by applying sophisticated algorithms that convert color intensity into quantitative metrics.
Experimental Protocols for NBT/BCIP-Based RNA Detection
Standardized NBT/BCIP Staining Protocol for RNA ISH
The following protocol provides a optimized methodology for NBT/BCIP-based RNA detection in formalin-fixed paraffin-embedded (FFPE) tissues, integrating best practices from multiple research applications:
Reagent Preparation:
- Prepare alkaline phosphatase buffer: 100mM Tris-HCl (pH 9.0), 150mM NaCl, 1mM MgCl₂
- Prepare NBT/BCIP working solution: Add 33μl NBT (50mg/ml in 70% dimethylformamide) and 16.5μl BCIP (50mg/ml in 100% dimethylformamide) to 5ml alkaline phosphatase buffer
- Add NBT first, mix thoroughly, then add BCIP and mix again
- Use prepared solution within 1 hour; discard unused portion [29]
Tissue Pretreatment and Hybridization:
- Dewaxing: Heat slides at 65°C for 2 hours, then dewax in xylene (twice, 3 min each), followed by graded ethanol series (100%, 95%, 70%, 50%) [49]
- Permeabilization: Apply proteinase K (10-20μg/ml) in citric acid solution at 37°C for 10 minutes [49]
- Acetylation: Soak slides in 20% acetic acid for 20 seconds to reduce nonspecific background [49]
- Hybridization: Apply digoxigenin-labeled probes diluted in pre-hybridization solution, incubate overnight at 37°C [49]
Post-Hybridization Washes and Detection:
- Stringency Washes: Wash twice in 2× saline sodium citrate (SSC) at 37°C for 15 minutes, then three times in 0.1× SSC at 37°C for 15 minutes [49]
- Blocking: Apply 0.5% blocking buffer for 1 hour at room temperature [49]
- Antibody Incubation: Incubate with biotinylated mouse anti-digoxigenin (1:500 dilution) for 1 hour at 37°C [49]
- Chromogenic Development: Apply NBT/BCIP detection solution at 37°C for 30 minutes (adjust time based on signal intensity monitoring) [49]
- Termination: Rinse slides thoroughly with distilled water (three times) to stop reaction [49]
Advanced Optimization Strategies
Table 2: Optimization Parameters for Enhanced NBT/BCIP Detection
| Parameter | Standard Approach | Optimized Approach | Impact on Signal |
|---|---|---|---|
| Fixation Time | 5-24 hours | 100-144 hours | Enhanced miRNA preservation and intensity [47] |
| Pretreatment | Proteinase K | HmiRR TRS pH 9 ± pepsin | Superior permeability and morphology [47] |
| Probe Design | DNA probes | LNA/DNA probes (30% LNA) | Higher specificity and affinity [46] [50] |
| Detection | Direct NBT/BCIP | Tyramide signal amplification | Enhanced signal strength for low targets [46] |
| Permeabilization | Proteinase K (20 min) | TRS pH 9 (20 min at 100°C) | Preserved morphology with intense staining [47] |
For challenging targets, particularly low abundance miRNAs, researchers should consider implementing advanced signal amplification systems. The Tyramide Signal Amplification (TSA) system provides enhanced detection sensitivity while maintaining compatibility with NBT/BCIP chromogenic development [46]. Similarly, Enzyme-Labeled Fluorescence (ELF) detection offers high cellular resolution and signal strength for single-molecule detection applications [46]. Recent optimization studies demonstrate that Heat-induced microRNA Retrieval (HmiRR) using target retrieval solution (TRS) at pH 9, with or without pepsin digestion, outperforms traditional proteinase K pretreatment across all fixation conditions [47].
Visualization of the NBT/BCIP Detection Workflow
NBT/BCIP ISH Workflow and Optimization
The visualization illustrates the complete NBT/BCIP ISH protocol with emphasis on critical optimization points that significantly impact the correlation between signal intensity and target RNA abundance. The chromogenic development phase represents the crucial quantitative step where alkaline phosphatase enzymatic activity generates the measurable signal proportional to target RNA concentration.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for NBT/BCIP ISH
| Reagent/Category | Specific Examples | Function in Protocol | Application Notes |
|---|---|---|---|
| Chromogenic Substrate | BCIP/NBT (S3771, Promega) | Alkaline phosphatase substrate generating purple-blue precipitate | Quality tested for blotting and ISH; prepare fresh [29] |
| Probe Technology | LNA/DNA probes (30% LNA) | Enhanced hybridization affinity and specificity | "Golden standard" for small RNA ISH; expensive but superior [46] [50] |
| Detection Antibodies | Anti-digoxigenin-AP conjugate | Binds digoxigenin-labeled probes for enzymatic detection | Critical for signal amplification; optimal dilution essential |
| Pretreatment Solutions | TRS pH 9, Proteinase K | Tissue permeabilization and antigen retrieval | HmiRR TRS pH 9 outperforms proteinase K for miRNA [47] |
| Hybridization Buffers | SSC-based with blockers | Controlled stringency during hybridization | Regulates probe specificity; includes blocking agents |
| Fixation Reagents | 10% neutral buffered formalin | Tissue preservation and RNA immobilization | Extended fixation (100-144h) improves miRNA detection [47] |
The selection of appropriate probe technology represents perhaps the most critical reagent choice in NBT/BCIP ISH experiments. Locked Nucleic Acid (LNA) modifications incorporated into DNA probes significantly enhance hybridization affinity and specificity, particularly valuable for challenging small RNA targets like miRNAs [47] [50]. These probes demonstrate higher melting temperatures, shorter hybridization times, and improved discriminatory power for closely related RNA family members. However, researchers should note that LNA probes require careful optimization as they can generate strong background signals at suboptimal concentrations [46].
Troubleshooting and Technical Considerations
Addressing Common Challenges
Successful implementation of NBT/BCIP staining for quantitative RNA assessment requires addressing several technical challenges:
High Background Staining:
- Over-digestion with proteinase K is a common cause; reduce concentration or incubation time
- Insufficient post-hybridization washing; increase stringency with lower SSC concentrations
- Endogenous alkaline phosphatase activity; include levamisole (2-5mM) in substrate solution
- Non-specific probe binding; optimize formamide concentration in hybridization buffer
Weak or Absent Signal:
- Inadequate permeabilization; implement HmiRR with TRS pH 9 instead of enzymatic treatment
- RNA degradation; ensure RNase-free conditions and proper tissue fixation
- Suboptimal probe design; implement LNA modifications for enhanced affinity
- Short development time; extend NBT/BCIP incubation with monitoring
Precipitate Crystallization:
- Excessive development time; monitor staining progression microscopically
- Substrate contamination; prepare fresh NBT/BCIP solution for each use
- Incorrect pH; ensure alkaline phosphatase buffer at pH 9.0
Quantitative Analysis Methodologies
For rigorous correlation between signal intensity and RNA abundance, implement computational analysis approaches. Automated quantitative methods using optical density separation algorithms can differentiate hematoxylin and NBT/BCIP staining components in whole-slide images, providing objective quantification superior to manual interpretation [48]. These methods employ sophisticated segmentation algorithms (e.g., CellViT for nuclear segmentation) and region-growing approaches to precisely quantify signal intensity within specific cellular compartments [48]. Image analysis software such as ImageJ can be utilized to calculate integrated optical density values from NBT/BCIP stained sections, enabling statistical comparison between experimental conditions [49].
When implementing quantitative analysis, include appropriate controls to account for potential non-linearities in the detection system. The relationship between target abundance and signal intensity may deviate from linearity at extreme high and low expression levels due to substrate depletion or threshold effects. Generate standard curves using samples with known expression levels when possible, and always include positive and negative controls in each experimental run to ensure quantitative reliability.
In situ hybridization (ISH) is a foundational technique in molecular biology for localizing specific nucleic acid sequences within histologic sections, cells, or entire embryos. The choice of detection system—chromogenic or fluorescent—is critical and directly impacts the sensitivity, resolution, and application scope of the experiment. This application note provides a comparative analysis of two dominant detection methodologies: the chromogenic system using Nitro Blue Tetrazolium/5-Bromo-4-Chloro-3-Indolyl Phosphate (NBT/BCIP) and fluorescent detection, particularly when enhanced by Tyramide Signal Amplification (TSA).
We frame this comparison within the context of optimizing ISH staining protocols, providing researchers, scientists, and drug development professionals with clear, actionable data to inform their experimental design. The analysis covers the fundamental principles, relative performance characteristics, and specific protocols for each method, supported by structured data and workflow visualizations.
Principles of Detection and Key Reagents
NBT/BCIP Chromogenic Detection
NBT/BCIP is a colorimetric substrate for the enzyme Alkaline Phosphatase (AP). In the presence of AP, BCIP is hydrolyzed to produce an intermediate that reduces NBT, resulting in an insoluble, blue-purple formazan precipitate at the site of target localization [51]. This method is prized for its strong signal and low background, making it a staple for chromogenic ISH and immunohistochemistry (IHC).
Fluorescent Detection and Tyramide Signal Amplification (TSA)
Fluorescent detection typically relies on fluorophore-conjugated antibodies or probes. To overcome limitations in detecting low-abundance targets, Tyramide Signal Amplification (TSA), also known as Catalyzed Reporter Deposition (CARD), is often employed. TSA is an enzyme-mediated method that utilizes Horseradish Peroxidase (HRP) to catalyze the activation and covalent deposition of labeled tyramide derivatives onto tyrosine residues proximal to the enzyme [52] [53]. This process can increase detection sensitivity by up to 100-fold compared to conventional methods [52] [53].
The Scientist's Toolkit: Essential Research Reagent Solutions
The table below details key reagents central to these detection methodologies.
Table 1: Key Research Reagent Solutions for ISH Detection
| Reagent/Solution | Function/Description | Key Features |
|---|---|---|
| NBT/BCIP Substrate | Chromogenic substrate for Alkaline Phosphatase (AP) [51]. | Produces an insoluble blue-purple precipitate; allows for reaction monitoring. |
| Vector Red Substrate | Chromogenic/fluorescent AP substrate [51]. | Yields a red precipitate that is also fluorescent, enabling dual-mode imaging. |
| Labeled Tyramides | TSA substrates (e.g., Alexa Fluor tyramides, Biotin-XX tyramide) activated by HRP [52] [53]. | Fluorophore- or hapten-labeled; enables high-density labeling of the target. |
| TSA Kits | Comprehensive kits containing tyramides, HRP conjugates, and reaction buffers [52]. | Simplifies workflow; optimized for sensitivity and specificity. |
| Zenon HRP Antibody Labeling Kits | Kits for labeling primary antibodies with HRP [52]. | Allows for direct HRP conjugation to primary antibodies, streamlining TSA protocols. |
| Anti-Digoxigenin-AP | Antibody conjugate for detecting DIG-labeled probes [51]. | High specificity; used with NBT/BCIP or other AP substrates. |
| Anti-Fluorescein-AP | Antibody conjugate for detecting FL-labeled probes [51]. | Enables sequential detection of multiple targets in multiplex ISH. |
Comparative Performance Analysis
The choice between NBT/BCIP and fluorescent TSA involves trade-offs between sensitivity, resolution, and experimental flexibility. The following table summarizes a direct comparison of their core characteristics.
Table 2: Quantitative and Qualitative Comparison of NBT/BCIP and Fluorescent TSA
| Parameter | NBT/BCIP (AP-based) | Fluorescent TSA (HRP-based) |
|---|---|---|
| Detection Modality | Chromogenic (Colorimetric) | Fluorescent |
| Sensitivity | High; suitable for detecting weak transcripts [51]. | Very high; up to 100-fold more sensitive than conventional methods [52] [53]. |
| Spatial Resolution | Good, but precipitate can diffuse slightly. | Excellent; covalent deposition ensures high-resolution localization [52]. |
| Reaction Monitoring | Yes; color development can be visually monitored and stopped [51]. | No; the reaction is rapid and fluorescent signal cannot be monitored during development [51]. |
| Enzymatic Reaction Time | Long; AP has long reactivity, beneficial for weak targets [51]. | Short; HRP activity is consumed rapidly. |
| Multiplexing Potential | Limited to 2-3 colors with distinct substrates (e.g., NBT/BCIP & Vector Red) [51]. | High; suitable for high-plex imaging via sequential TSA rounds and antibody stripping [53]. |
| Compatible Imaging | Brightfield microscopy, fluorescent microscopy (NBT/BCIP fluoresces in near-IR) [51]. | Standard, confocal, and super-resolution fluorescence microscopy. |
| Primary Applications | Single or two-color ISH where monitoring development is key [51]. | Detection of low-abundance targets, high-resolution imaging, and multiplexed experiments [52] [53]. |
Experimental Protocols
Two-Color FISH Protocol Using NBT/BCIP and Vector Red
This protocol, adapted from an improved FISH method in zebrafish embryos, leverages the advantages of AP substrates for sensitive two-color detection [51].
Day 1: Hybridization
- Fixation and Permeabilization: Fix embryos or tissue in 4% paraformaldehyde and dehydrate through an ethanol series.
- Pre-hybridization: Rehydrate samples and incubate in pre-hybridization buffer (e.g., 50% formamide, 5X SSC, 0.1% Tween 20) for 2 hours at 65°C.
- Hybridization: Incubate samples overnight at 65°C with DIG- and FL-labeled probes diluted in pre-hybridization buffer. Ideally, label the weaker probe with DIG.
Day 2: Post-Hybridization Washes and First Antibody Incubation
- Stringency Washes: Wash samples in a series of buffers (e.g., 75%, 50%, 25% pre-hybridization buffer in 2X SSC, then 0.2X SSC) at 65°C.
- Blocking: Wash samples into PBT (PBS with 0.2% Tween 20).
- Anti-DIG-AP Incubation: Incubate samples overnight at 4°C with pre-absorbed anti-DIG-AP antibody diluted in 2% lamb serum in PBT.
Day 3: First Chromogenic Development and Second Antibody Incubation
- Washing: Wash samples 6x in PBT and once in AP buffer (100 mM Tris pH 9.5, 100 mM NaCl, 50 mM MgCl₂, 0.1% Tween 20).
- NBT/BCIP Development: Develop the DIG-labeled probe in NBT/BCIP solution (e.g., 4.5 μL of 50 mg/mL NBT, 3.5 μL of 50 mg/mL BCIP per 1 mL AP buffer). Monitor the reaction and stop with PBT washes once the desired signal intensity is reached.
- Enzyme Inactivation: Fix samples in 4% PFA for 1 hour at room temperature to inactivate the AP.
- Anti-FL-AP Incubation: Process samples for incubation with anti-FL-AP antibody (in 0.2M Tris pH 8.5 with 0.1% Tween 20) overnight at 4°C.
Day 4: Second Chromogenic Development and Imaging
- Washing: Wash samples 6x in PBT and once in the appropriate buffer for the second substrate.
- Vector Red Development: Develop the FL-labeled probe using Vector Red substrate according to the manufacturer's instructions.
- Reduce Background: Dehydrate embryos in ethanol overnight to reduce background fluorescence.
- Imaging: Mount samples and acquire confocal images. NBT/BCIP fluorescence is excited with a 647 nm laser and detected with a 740 nm long-pass filter. Vector Red is excited at 561 nm and detected with a 595/50 nm emission filter [51].
The workflow for this protocol is summarized in the following diagram:
Immunofluorescence Protocol Using Tyramide Signal Amplification
This protocol outlines the basic steps for detecting a target using TSA, which can be applied to ICC, IHC, or FISH [52] [53].
- Sample Preparation: Fix and permeabilize cells or tissue sections using standard protocols. If required, block endogenous peroxidase activity.
- Primary Antibody Incubation: Incubate with a primary antibody specific to the target protein. Note: Significantly less primary antibody is typically required compared to conventional immunofluorescence due to the high amplification [53].
- HRP-Conjugated Secondary Antibody Incubation: Incubate with an HRP-conjugated secondary antibody (e.g., HRP-goat anti-rabbit IgG). Alternatively, use a Zenon HRP Labeling Kit to directly label the primary antibody with HRP [52].
- Tyramide Working Solution Preparation: Prepare the tyramide working solution by diluting the fluorophore-labeled tyramide (e.g., TyraMax dye or Alexa Fluor tyramide) in the supplied amplification buffer, which contains a low concentration of H₂O₂ [52] [53].
- Tyramide Reaction: Incubate the sample with the tyramide working solution for a specified time (e.g., 2-10 minutes). During this step, HRP catalyzes the activation of tyramide, leading to its covalent deposition near the target.
- Washing: Wash the sample thoroughly to remove unbound tyramide.
- Signal Detection: The signal can be immediately detected by fluorescence microscopy. For multiplexing, proceed with antibody elution steps (e.g., heat treatment in stripping buffer) before initiating the next round of staining with a different primary antibody and tyramide dye [53].
The core signaling pathway and workflow for TSA are illustrated below:
Discussion and Concluding Remarks
The comparative analysis reveals that NBT/BCIP and fluorescent TSA are not mutually exclusive but are complementary technologies suited for different experimental objectives.
Choose NBT/BCIP-based detection when working with easily detectable targets, when the ability to visually monitor the development of the signal is paramount to control background, or for two-color ISH experiments where the high sensitivity and distinct fluorescent properties of substrates like NBT/BCIP and Vector Red are beneficial [51]. Its compatibility with brightfield microscopy also makes it accessible for most laboratories.
Choose fluorescent TSA for the detection of low-abundance targets that are beyond the reach of conventional methods, when the highest possible spatial resolution is required, or when designing multiplex experiments requiring more than three labels [52] [53]. The need for specialized fluorescence microscopy and the inability to monitor signal development in real-time are trade-offs for the substantial gains in sensitivity.
In conclusion, a deep understanding of the principles and performance characteristics of both NBT/BCIP chromogenic and TSA-enhanced fluorescent detection empowers researchers to select the optimal tool. This choice ultimately enhances the reliability and quality of data generated in ISH research and drug development workflows.
Evaluating Emerging Unified Platforms like OneSABER
In situ hybridization (ISH) remains an indispensable technique for visualizing the spatiotemporal expression patterns of specific nucleic acid sequences within tissues and cells, providing crucial insights into gene function during development, homeostasis, and disease [28]. For decades, enzyme-catalyzed reporter deposition assays, particularly alkaline phosphatase (AP)-based colorimetric detection using substrates like NBT/BCIP, have served as the gold standard for achieving reliable signal-to-noise ratios in both sectioned and whole-mount samples [54] [28]. These traditional methods, while robust and cost-effective, present significant limitations including limited multiplexing capabilities, signal diffusion that restricts cellular resolution, and protocol inflexibility that often locks researchers into specific probe designs and detection chemistries [28].
The recent expansion of single-cell sequencing technologies has accelerated the discovery of novel cell types whose marker genes require experimental validation through in situ methods, creating pressing demand for more versatile and accessible platforms [28]. Meanwhile, researchers working with non-mammalian model organisms often face a paucity of commercial antibodies, making ISH their primary tool for gene expression analysis [28]. These challenges are compounded by the proliferation of specialized ISH approaches, each requiring custom probe types, designs, and proprietary detection chemistries, creating significant navigation barriers for beginners and increasing costs when multiple methods must be applied within a single research program [28].
The OneSABER platform emerges as a unified open platform addressing these fragmentation issues by connecting commonly used canonical and recently developed single- and multiplex, colorimetric and fluorescent ISH approaches using a single type of DNA probe [28]. This application note evaluates OneSABER within the context of traditional NBT/BCIP staining protocols, providing detailed comparative analysis, experimental protocols, and implementation guidance for researchers seeking to leverage this unified approach in their gene expression studies.
Core Principles and Mechanism
OneSABER functions as a modular "one probe fits all" approach that stems from classical ISH protocols while incorporating recent methodological advances [28]. At its core, the platform utilizes a pool of 15-30 custom user-defined short (approximately 35-45 nucleotide) single-stranded DNA (ssDNA) oligonucleotides complementary to an RNA target, with the exact number adjustable based on target RNA length, expression strength, desired resolution, and budget [28]. Each probe contains a specific 9-nucleotide 3' initiator sequence that undergoes in vitro extension through a primer exchange reaction (PER) to generate long concatemerized probes [28].
The primer exchange reaction relies on a catalytic DNA hairpin combined with a strand-displacing polymerase and competitive branch migration to repeatedly add the same sequence to the 3' end of ssDNA primers [28]. This elegant mechanism enables researchers to control signal amplification strength simply by adjusting reaction time, with longer reactions producing more extended concatemers [28]. These concatemers subsequently serve as universal landing-pad sequences for binding short (20 nucleotide) secondary oligonucleotide ssDNA probes/adapters, which are modified according to the chosen signal development method [28].
Key Advantages Over Traditional Approaches
The table below summarizes the comparative advantages of the OneSABER platform relative to traditional ISH approaches using NBT/BCIP detection:
Table 1: Comparative Analysis of OneSABER vs. Traditional NBT/BCIP ISH
| Feature | Traditional NBT/BCIP ISH | OneSABER Platform |
|---|---|---|
| Probe Type | Long hapten-labeled antisense RNA probes (typically ~800 bases) [27] | Short ssDNA oligonucleotides (35-45 nt) with PER extension [28] |
| Detection Flexibility | Limited to colorimetric AP detection [54] | Compatible with colorimetric AP, fluorescent TSA, HCR FISH [28] |
| Multiplexing Capacity | Very limited [28] | Moderate to high, depending on detection method [28] |
| Signal Amplification | Fixed; depends on substrate incubation time [54] | Tunable via concatemer length controlled by PER reaction time [28] |
| Platform Lock-in | High; probes specific to method [28] | Minimal; same probes multiple methods [28] |
| Resolution | Cellular level (signal diffusion) [28] | Potentially subcellular, depending on detection method [28] |
| Entry Cost | Lower | Moderate (requires initial oligonucleotide set) |
| Probe Design Transparency | Variable | Fully user-controlled and open [28] |
This unified approach demonstrates particular utility in challenging model organisms like the regenerative flatworm Macrostomum lignano, where ISH has previously been limited mostly to canonical AP-based colorimetric assays, significantly hindering experimental potential [28]. Similar benefits extend to other model systems including planarians and formalin-fixed, paraffin-embedded mouse tissue sections [28].
Experimental Protocols
OneSABER Workflow for Colorimetric Detection
The following workflow diagram illustrates the complete OneSABER experimental process, highlighting compatibility points with traditional colorimetric detection methods:
Diagram 1: OneSABER colorimetric ISH workflow. Yellow: sample preparation; Green: OneSABER-specific steps; Red: traditional NBT/BCIP detection; White: common ISH steps.
Detailed OneSABER Protocol with NBT/BCIP Detection
This protocol adapts the unified OneSABER approach for colorimetric detection using NBT/BCIP, enabling direct comparison with traditional ISH results while leveraging the platform's modularity.
Probe Design and Preparation
- Target Selection: Design 15-30 short ssDNA oligonucleotides (35-45 nucleotides each) complementary to the target RNA sequence. The number of probes should be adjusted based on target abundance and desired signal strength [28].
- Initiator Sequence: Append a specific 9-nucleotide 3' initiator sequence to each probe during oligonucleotide synthesis [28].
- Primer Exchange Reaction:
- Prepare PER reaction mixture containing catalytic DNA hairpin and strand-displacing polymerase.
- Extend probes for controlled duration (typically 30-120 minutes) to achieve desired concatemer length.
- Purify extended probes using standard nucleic acid purification methods [28].
- Hapten Labeling: Incubate PER-extended probes with secondary adapter oligonucleotides conjugated to digoxigenin (DIG) for compatibility with anti-DIG alkaline phosphatase antibodies and subsequent NBT/BCIP detection [28].
Sample Preparation and Hybridization
Tissue Preparation:
- For formalin-fixed paraffin-embedded (FFPE) sections: Deparaffinize through xylene series (2×3 min) and rehydrate through graded ethanol series (100%, 95%, 70%, 50%, 3 min each) to cold tap water [27].
- For frozen sections: Begin with fixation in 4% paraformaldehyde [27].
- Critical: From rehydration onward, do not allow samples to dry, as this causes non-specific antibody binding and high background [27].
Permeabilization and Pre-hybridization:
- Digest with 20 µg/mL proteinase K in pre-warmed 50 mM Tris for 10-20 minutes at 37°C. Optimize concentration and time for specific tissue type [27].
- Rinse slides 5× in distilled water.
- Immerse slides in ice-cold 20% (v/v) acetic acid for 20 seconds for additional permeabilization [27].
- Dehydrate through ethanol series (70%, 95%, 100%, 1 min each) and air dry.
- Apply pre-hybridization buffer and incubate 1 hour at hybridization temperature in humidified chamber [27].
Hybridization:
- Denature OneSABER probes at 95°C for 2 minutes in PCR block, then immediately chill on ice.
- Apply 50-100 µL diluted probe per section, covering entire sample.
- Incubate overnight at 65°C in humidified hybridization chamber with coverslip to prevent evaporation [27].
Stringency Washes and Detection
Post-hybridization Washes:
Immunological Detection:
- Transfer slides to humidified chamber and block with 200 µL blocking buffer (MABT + 2% BSA, milk, or serum) for 1-2 hours at room temperature [27].
- Drain blocking buffer and apply anti-DIG-alkaline phosphatase antibody at manufacturer's recommended dilution in blocking buffer.
- Incubate 1-2 hours at room temperature.
- Wash slides 5×10 minutes with MABT at room temperature [27].
- Equilibrate slides with pre-staining buffer (100 mM Tris pH 9.5, 100 mM NaCl, 10 mM MgCl₂) [27].
NBT/BCIP Colorimetric Development:
- Prepare NBT/BCIP stock solution according to manufacturer protocol [54].
- Develop with NBT/BCIP substrate until signal appears (typically 10 minutes to several hours).
- Critical: Ensure detection buffer is at pH 9.5 (at 20°C) and minimize exposure to air by using air-tight containers [54].
- Monitor development microscopically to prevent over-development and high background.
- Stop reaction by transferring slides to distilled water.
Mounting and Imaging:
- Mount with NBT/BCIP-compatible aqueous mounting media. Avoid xylene-containing mounting media as these can cause crystal formations of color precipitates [54].
- Image using brightfield microscopy.
- For long-term storage, avoid storing slides dry at room temperature. Store in 100% ethanol at -20°C or in plastic boxes covered with saran wrap at -20°C to -80°C [27].
Traditional NBT/BCIP ISH Protocol (Reference Method)
For comparative purposes, the core workflow of traditional NBT/BCIP ISH is summarized below:
- Probe Preparation: Generate long hapten-labeled (typically DIG) antisense RNA probes approximately 800 bases long from linearized DNA templates using in vitro transcription [27].
- Sample Preparation: Identical to Sections 3.2.2, steps 1-2.
- Hybridization: Apply denatured RNA probes in hybridization solution overnight at optimized temperature (typically 55-62°C) [27].
- Stringency Washes: Similar to Section 3.2.3, step 1, with temperatures and SSC concentrations optimized for RNA probes.
- Immunological Detection and Development: Identical to Section 3.2.3, steps 2-4.
Research Reagent Solutions
The following table details essential materials and reagents required for implementing the OneSABER platform with NBT/BCIP detection:
Table 2: Essential Research Reagents for OneSABER with NBT/BCIP Detection
| Reagent Category | Specific Product | Function and Application Notes |
|---|---|---|
| Probe Synthesis | Custom ssDNA Oligonucleotides (35-45 nt) | Target-specific binding with 3' initiator sequences for PER extension [28] |
| Primer Exchange Reaction (PER) Kit | Catalytic DNA hairpin and strand-displacing polymerase for concatemer generation [28] | |
| Sample Preparation | Proteinase K Solution (20 mg/mL) | Tissue permeabilization for probe access; requires concentration optimization [27] [4] |
| Paraformaldehyde (4%) | Tissue fixation preserving nucleic acid integrity and morphology [4] | |
| Hybridization | Formamide | Hybridization buffer component reducing stringency temperature [27] [4] |
| Saline Sodium Citrate (SSC) 20X | Buffer for hybridization and stringency washes; concentration affects specificity [27] | |
| Denhardt's Solution | Blocking agent in hybridization buffer reducing non-specific probe binding [27] | |
| Detection | Anti-DIG-Alkaline Phosphatase Antibody | Immunological detection of hapten-labeled probes [27] |
| NBT/BCIP Stock Solution | Colorimetric AP substrate producing blue-purple precipitate [54] | |
| MABT Buffer (Maleic Acid with Tween) | Gentle washing buffer for nucleic acid detection; alternative to PBS [27] | |
| Mounting & Storage | Aqueous Mounting Media | NBT/BCIP-compatible; xylene-free to prevent crystal formation [54] |
Technical Considerations and Troubleshooting
Signal Optimization and Validation
The modular nature of OneSABER requires careful optimization at critical junction points:
- Concatemer Length Titration: Systematically vary PER reaction time (30-120 minutes) to determine optimal signal-to-noise ratio for specific targets and detection methods [28].
- Probe Number Optimization: For low-abundance targets, increase the number of complementary oligonucleotides (up to 30) in the initial probe pool [28].
- Hybridization Temperature Calibration: For traditional RNA probes, typical range is 55-62°C [27]; for OneSABER DNA probes, optimal temperature is approximately 65°C [28].
- Antibody Cross-reactivity Controls: Include control samples without primary antibody to detect non-specific binding of detection reagents.
Troubleshooting Common Issues
The following workflow diagram illustrates a systematic approach to resolving common experimental challenges:
Diagram 2: Troubleshooting common NBT/BCIP issues. Red: problem identification; White: specific issues; Green: recommended solutions.
Advanced Applications and Multiplexing Potential
While traditional NBT/BCIP detection is limited to single-plex colorimetric applications, the OneSABER platform enables more advanced experimental designs:
- Sequential Multiplexing: Using the same initial probe set, researchers can perform sequential ISH experiments with different detection methods (e.g., colorimetric followed by fluorescent) on adjacent sections [28].
- Signal Amplification Tuning: The same target can be visualized with different resolution requirements by adjusting PER extension times without redesigning probe sets [28].
- Method Validation: Traditional NBT/BCIP results can be directly compared with more sensitive fluorescent detection methods using the same core probe technology, facilitating method transition and validation [28].
The OneSABER platform represents a significant advancement in ISH methodology by providing a unified framework that maintains compatibility with established techniques like NBT/BCIP colorimetric detection while offering a modular pathway to more advanced applications. This "one probe fits all" approach addresses the critical problem of methodological fragmentation in gene expression analysis, particularly benefiting research programs that require validation across multiple detection platforms or gradual transition from canonical to modern ISH methods.
For researchers entrenched in NBT/BCIP-based workflows, OneSABER offers a backward-compatible upgrade path that preserves institutional knowledge and comparative value of existing data while removing technological barriers to method diversification. The platform's demonstrated efficacy in challenging model organisms like Macrostomum lignano, planarians, and mammalian FFPE sections underscores its broad applicability across diverse research contexts [28].
By integrating the reliability of traditional NBT/BCIP detection with the flexibility of modern DNA-based probe systems, OneSABER establishes a new standard for unified experimental design in spatial transcriptomics, effectively bridging classical and contemporary approaches to gene expression analysis.
Establishing a Standardized Protocol for Reproducible Results
Within the framework of a broader thesis on in situ hybridization (ISH) research, this application note details a standardized protocol for the chromogenic detection of RNA using NBT/BCIP. Reproducibility is paramount in scientific research, particularly in drug development, where qualitative and quantitative assessments of gene expression can inform critical decisions [10]. The NBT/BCIP staining method, which yields an insoluble purple precipitate, is a cornerstone technique for localizing specific mRNA sequences within tissue sections and cells, providing invaluable spatial context lost in homogenized assays like RT-PCR [4] [10]. This protocol is designed to provide researchers and scientists with a rigorous, optimized methodology to ensure consistent and reliable results, thereby enhancing the validity and cross-comparability of data generated in both academic and industrial settings.
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table catalogues the essential materials and reagents required for a successful NBT/BCIP-based ISH experiment, along with their critical functions in the protocol.
Table 1: Key Research Reagent Solutions for NBT/BCIP ISH
| Reagent/Item | Function & Application in the Protocol |
|---|---|
| Paraformaldehyde (4%) | A key fixative that preserves tissue architecture and cellular morphology while retaining the integrity of the target nucleic acids [4] [10]. |
| Proteinase K | A permeabilization agent that digests proteins to increase tissue permeability and allow probe access to the target mRNA [4]. |
| DIG-Labeled Riboprobe | A digoxigenin-labeled nucleic acid probe that is complementary to the target mRNA; the DIG hapten is later recognized by an antibody for detection [10]. |
| Pre-hybridization Buffer | Conditions the sample and blocks nonspecific binding sites to reduce background staining and improve signal-to-noise ratio [4]. |
| Anti-DIG-AP Antibody | An alkaline phosphatase (AP)-conjugated antibody that specifically binds to the DIG epitope on the hybridized probe, enabling enzymatic detection [10]. |
| NBT/BCIP Substrate | The chromogenic substrate for Alkaline Phosphatase. AP reduces NBT to form an insoluble purple diformazan precipitate, localizing gene expression [55] [10]. |
| Salmon Sperm DNA | A blocking agent added to the pre-hybridization and hybridization buffers to bind to and sequester non-specific sites, thereby lowering background [4]. |
| Formamide | A component of hybridization buffers that lowers the effective melting temperature of DNA, allowing for stringent hybridization at manageable temperatures [4]. |
| SSC Buffer (20X) | Saline-sodium citrate buffer used for post-hybridization stringency washes; its concentration and temperature determine the stringency, removing weakly bound probes [4]. |
Quantitative Data and Cross-Platform Correlation
The quantitative potential of NBT/BCIP-based ISH, while historically underexplored, is crucial for cross-platform validation. Systematic comparisons between colorimetric ISH data and microarray expression profiles reveal both correlations and inherent technological disparities that researchers must account for.
Table 2: Key Considerations for Quantitative Analysis of NBT/BCIP Signal
| Parameter | Consideration & Impact on Quantification |
|---|---|
| Dynamic Range | Colorimetric ISH has a compressed upper dynamic range compared to microarrays, largely due to signal saturation from tyramide amplification [55]. |
| Quantification Metric | Relative signal can be measured using cellular resolution image segmentation and integrated optical density, providing an expression level analogous to microarrays [55]. |
| Cross-Platform Correlation | Studies show high concordance between ISH and microarray data when controlling for factors like probe homology and analysis methods, supporting its use for validation [55]. |
| q2PISH Method | A quantitative/qualitative method involves using pNPP substrate prior to NBT/BCIP, allowing supernatant absorbance reading followed by chromogenic localization [10]. |
Detailed Experimental Protocol for NBT/BCIP Staining
This section provides a step-by-step methodology for ISH using NBT/BCIP, from sample preparation to imaging. Adherence to this protocol is critical for achieving reproducible and high-quality results.
Fixation, Permeabilization, and Pre-hybridization
- Fixation: Fix tissue sections or cells in 4% Paraformaldehyde (PFA) for at least 20 minutes at room temperature [10]. Consistent fixation conditions (pH, time, temperature) are vital for preserving nucleic acid integrity and ensuring reproducible staining [19].
- Permeabilization: Treat samples with Proteinase K solution (e.g., 20 mg/mL) to digest proteins and allow probe penetration into the cells. Optimal concentration and time must be determined for each tissue type [4].
- Pre-hybridization Blocking: Incubate samples in a pre-warmed, filtered pre-hybridization buffer for 30-60 minutes at 37-45°C in a humidified chamber. A standard buffer formulation is:
- 50% Formamide (v/v)
- 1X SSC
- 50 µg/mL Heparin
- 100 µg/mL Denatured Salmon Sperm DNA
- 1% SDS (w/v)
- 0.1% Tween-20 (v/v)
- RNase-free Water to volume [4] This step blocks nonspecific binding sites to reduce background.
Hybridization and Stringency Washes
- Probe Denaturation: Denature the DIG-labeled riboprobe at 95°C for 5 minutes and immediately place on ice [4].
- Hybridization: Apply the denatured probe, diluted in an appropriate hybridization buffer, to the sample. Cover with a coverslip to ensure even distribution and prevent evaporation. Incubate overnight (16-18 hours) at the appropriate hybridization temperature (e.g., 37-45°C or 62°C for higher stringency) in a sealed, humidified chamber [4] [10]. Drying of reagents must be avoided, as it causes heavy non-specific staining [19].
- Stringency Washes: The following day, carefully remove the coverslip and perform a series of washes to remove unbound and weakly bound probes. Use a water bath or hybridization oven to maintain precise temperature control, as stringency is highly temperature-sensitive [4]. Standard washes include:
- 2X SSC at room temperature.
- 1X SSC or 0.5X SSC at the hybridization temperature.
- 0.1X SSC at a temperature appropriate for the desired stringency.
Immunological Detection and Chromogenic Development
- Antibody Incubation: Incubate the samples with an anti-DIG antibody conjugated to Alkaline Phosphatase (AP), typically at a dilution of 1:2000 in blocking buffer, for 1 hour at room temperature [10].
- Chromogenic Development (NBT/BCIP Reaction):
- Wash the samples thoroughly to eliminate any non-specifically bound antibody.
- Prepare the NBT/BCIP substrate solution in a buffer containing 0.1 M Tris-HCl (pH 9.5) and 0.1 M NaCl [10].
- Apply the substrate to the samples and incubate in the dark. Monitor the development of the purple/blue precipitate closely. The reaction can take from several minutes to several hours.
- Stop the reaction by washing the samples with distilled water or a prescribed stop buffer once the desired signal-to-background level is achieved.
Imaging and Analysis
- Mounting: Mount samples with an aqueous mounting medium [4].
- Imaging: Image the stained samples using a brightfield microscope [4]. For quantitative analysis, ensure images are captured under standardized lighting conditions. High-resolution images (e.g., at 10× magnification) are used to extract expression profiles, often by analyzing intensity along the embryonic antero-posterior axis or within specific tissue regions [55] [34].
Signaling Pathways and Experimental Workflows
The core chemical reaction and the overall experimental workflow are visualized below to aid in understanding the technical procedure.
NBT/BCIP Chromogenic Reaction Pathway
Experimental Workflow for ISH with NBT/BCIP
Troubleshooting and Optimization Tips
Even with a standardized protocol, issues can arise. The following table addresses common problems and provides solutions to optimize your ISH results.
Table 3: Troubleshooting Common Issues in NBT/BCIP ISH
| Problem | Potential Cause | Solution |
|---|---|---|
| High Background Signal | Non-specific probe binding, insufficient blocking, or reagent evaporation. | Increase stringency of washes (higher temperature, lower SSC). Ensure adequate blocking and use a sealed humidified chamber to prevent drying [4] [19]. |
| Weak or No Signal | Poor probe penetration, low probe concentration, probe degradation, or insufficient antibody. | Optimize permeabilization (Proteinase K concentration/time). Check probe integrity and increase concentration. Ensure detection reagents are fresh and active [4]. |
| Uneven Staining | Incomplete coverage of tissue by reagents, air bubbles, or lifting sections. | Apply probes and reagents evenly. Use coverslips correctly to avoid bubbles. Use charged slides to ensure section adhesion [4] [19]. |
| Non-Specific Signals | Off-target probe binding or tissue damage. | Always run controls: a no-probe control and a sense probe control. Validate signal specificity with RNase digestion [4]. |
Conclusion
The NBT/BCIP chromogenic staining protocol remains a cornerstone technique for spatial gene expression analysis, prized for its robustness, permanent record, and minimal equipment requirements. Mastering this method requires a thorough understanding of the underlying chemistry, meticulous attention to sample preparation and hybridization conditions, and diligent troubleshooting to balance signal intensity with background. While emerging multiplexed and highly sensitive fluorescent methods offer expanded capabilities, the NBT/BCIP ISH protocol provides an indispensable, cost-effective tool for initial gene validation and studies where cellular resolution within a complex tissue context is paramount. Its continued optimization and proper validation ensure reliable data that can powerfully inform hypotheses in developmental biology, disease mechanisms, and therapeutic development.
References
- 1. BCIP/NBT Alkaline Phosphatase Color Development Kit [https://www.apexbt.com/bcip-nbt-alkaline-phosphatase-color-development-kit.html]
- 2. BCIP/NBT | Intense blue/purple precipitate| Very Stable [https://kementec.com/products/substrates/precipitating-substrates/bcipnbt/]
- 3. BCIP/NBT Substrate Kit, Alkaline Phosphatase (AP), (5- ... [https://vectorlabs.com/products/bcip-nbt-ap-substrate-kit/?srsltid=AfmBOort6M_ZQ_hlKhlMwvlP3iD58rnkK9hm0_rTaBdUFtcNE5gtSZ5n]
- 4. ISH Protocol [https://www.bostonbioproducts.com/resources/ish-protocol/]
- 5. NBT/BCIP Ready-to-Use Tablets Protocol Troubleshooting [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/genomics/nucleic-acid-labeling-and-detection/nbt-bcip-ready-to-use-tablets?srsltid=AfmBOorYRvMwY_c4FPQ92tF__VaZpbP_tH6mK-VwD_BF3rGECr8DCE07]
- 6. In Situ Hybridization Support—Troubleshooting [https://www.thermofisher.com/us/en/home/technical-resources/technical-reference-library/gene-expression-analysis-genotyping-support-center/in-situ-hybridization-support/ish-support-troubleshooting.html]
- 7. An optimized protocol for high-throughput in situ ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4589143/]
- 8. Goat Anti-Digoxigenin/Digoxin (DIG) Antibody, DyLight 488 [https://vectorlabs.com/products/dylight-488-anti-digoxigenin-digoxin-dig/?srsltid=AfmBOoomYFaCC5y5xsvuyJ-AMPhGTSRTGrVcsBSsBs8HQ_pS2MHfWrIR]
- 9. Digoxigenin - an overview [https://www.sciencedirect.com/topics/neuroscience/digoxigenin]
- 10. A quantitative and qualitative RNA expression profiling ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5809649/]
- 11. Synthesis of RNA–Digoxigenin Probe [https://link.springer.com/protocol/10.1007/978-1-0716-4518-5_49]
- 12. Pitfalls and Caveats in Applying Chromogenic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8232789/]
- 13. Comparison of multiple quantitative strategies for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12456881/]
- 14. BCIP/NBT Membrane Alkaline Phosphatase Substrate [https://www.rockland.com/categories/supporting-reagents/bcip-nbt-membrane-alkaline-phosphatase-substrate-NBT-100/?srsltid=AfmBOoqhLNZpCOuwUDqzdjgqEfz6eoRvMD54a9NduSQ1k_BBdzcu21XZ]
- 15. NBT/BCIP Stock Solution Protocol & Troubleshooting [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/genomics/nucleic-acid-labeling-and-detection/nbt-bcip-stock-solution?srsltid=AfmBOopEEablxetOMSphD415E1jsyEpIxVhfUsvxOheNQHBb2a0RvkMD]
- 16. Quantitative methods for genome-scale analysis of in situ ... [https://genomebiology.biomedcentral.com/articles/10.1186/gb-2008-9-1-r23]
- 17. NBT/BCIP Ready-to-Use Tablets Protocol Troubleshooting [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/genomics/nucleic-acid-labeling-and-detection/nbt-bcip-ready-to-use-tablets?srsltid=AfmBOorv2gPOaJ40qNGIw3XNinjUn_Z2L_Q-0plXZdHb6EzKt3E1sdvR]
- 18. Combination of Immunofluorescence and Quantitative ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8799906/]
- 19. Steps to Better ISH Staining [https://www.leicabiosystems.com/us/knowledge-pathway/steps-to-better-ish/]
- 20. Comparative in situ hybridization protocols in zebrafish [https://pmc.ncbi.nlm.nih.gov/articles/PMC9490454/]
- 21. Optimized RNA ISH, RNA FISH and protein-RNA double ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4126239/]
- 22. Double in situ hybridisation protocols [https://www.ucl.ac.uk/~ucbzwdr/double%20in%20situ%20protocol.htm]
- 23. Detection of mRNA by Whole Mount in situ Hybridization ... [https://en.bio-protocol.org/en/bpdetail?id=3193&type=0]
- 24. Differences Between DNA and RNA Probes [https://www.creative-bioarray.com/support/differences-between-dna-and-rna-probes.htm]
- 25. 10 Tips for Optimizing In Situ Hybridization (ISH) [https://www.enzo.com/note/10-tips-for-optimizing-in-situ-hybridization-ish/]
- 26. What are the Differences Between DNA and RNA Probes? [https://www.enzo.com/note/what-are-the-differences-between-dna-and-rna-probes/]
- 27. ISH: in situ hybridization protocol [https://www.abcam.com/en-us/technical-resources/protocols/in-situ-hybridization]
- 28. One probe fits all: a highly customizable modular RNA in situ ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12148028/]
- 29. BCIP/NBT Color Development Substrate (5-bromo-4-chloro ... [https://www.promega.com/products/biochemicals-and-labware/biochemical-buffers-and-reagents/bcip_nbt-color-development-substrate-_5_bromo_4_chloro_3_indolyl_phosphate_nitro-blue-tetrazolium_/]
- 30. NBT/BCIP Stock Solution Protocol & Troubleshooting [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/genomics/nucleic-acid-labeling-and-detection/nbt-bcip-stock-solution?srsltid=AfmBOoqktkF6H22d6OLFx-8rNzKAogUfJGPLR9m6dl9aJdC9m3kDV8n3]
- 31. SEAP activity measurement in reporter cell-based assays ... [https://www.sciencedirect.com/science/article/abs/pii/S0003269719304968]
- 32. Fluorescent in situ hybridization employing the conventional ... [https://pubmed.ncbi.nlm.nih.gov/17612300/]
- 33. NBT/BCIP Stock Solution Protocol & Troubleshooting [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/genomics/nucleic-acid-labeling-and-detection/nbt-bcip-stock-solution?srsltid=AfmBOople2WOEE2zTUmYCIEfhz2Rqo9N-wlxlwHj6OWSthMzogp3MUGl]
- 34. High-resolution gene expression data from blastoderm ... [https://www.nature.com/articles/sdata20155]
- 35. NBT/BCIP Stock Solution Protocol & Troubleshooting [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/genomics/nucleic-acid-labeling-and-detection/nbt-bcip-stock-solution?srsltid=AfmBOop8U0ahYZ2DEZo2138wKjG0_iY6_WgC4ZRcWZg4VZeQAsz54PVC]
- 36. Phosphatase-Based Signal Amplification Techniques— ... [https://www.thermofisher.com/us/en/home/references/molecular-probes-the-handbook/ultrasensitive-detection-technology/enzyme-labeled-fluorescence-signal-amplification-technology.html]
- 37. 9 ISH Tips You Can't Ignore [https://www.creative-bioarray.com/9-ish-tips-you-cant-ignore.htm]
- 38. In Situ Hybridization Support—Troubleshooting [https://www.thermofisher.com/id/en/home/technical-resources/technical-reference-library/gene-expression-analysis-genotyping-support-center/in-situ-hybridization-support/ish-support-troubleshooting.html]
- 39. Establishment and optimization of mRNA in situ ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6794860/]
- 40. A powerful and versatile new fixation protocol for ... [https://bmcbiol.biomedcentral.com/articles/10.1186/s12915-024-02052-3]
- 41. NBT/BCIP Stock Solution Protocol & Troubleshooting [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/genomics/nucleic-acid-labeling-and-detection/nbt-bcip-stock-solution?srsltid=AfmBOor_bH5IAXOGylnBj_o54JnCm9IqLsrnTOYlgXDvSn0G6SLXCLyy]
- 42. Steps to Better ISH Staining [https://www.leicabiosystems.com/es-es/knowledge-pathway/steps-to-better-ish/]
- 43. In Situ Hybridization... | Thermo Fisher Scientific - CN [https://www.thermofisher.com/cn/zh/home/technical-resources/technical-reference-library/gene-expression-analysis-genotyping-support-center/in-situ-hybridization-support/ish-support-troubleshooting.html]
- 44. Integrative analysis of single-nucleus RNA-seq and bulk ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10987912/]
- 45. Fluorescence In Situ Hybridization, Immunohistochemistry, ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4350802/]
- 46. Small RNA Detection by in Situ Hybridization Methods - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4490494/]
- 47. A novel approach for microRNA in situ hybridization using ... [https://www.nature.com/articles/s41598-021-83888-5]
- 48. A fully automated quantitative analysis method based on ... [https://www.sciencedirect.com/science/article/pii/S2950261625000342]
- 49. RNA in-situ hybridization (ISH) [https://bio-protocol.org/exchange/minidetail?id=10783265&type=30]
- 50. Small RNA Detection by in Situ Hybridization Methods [https://www.mdpi.com/1422-0067/16/6/13259]
- 51. Two-color fluorescent in situ hybridization using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4336792/]
- 52. TSA and Other Peroxidase-Based Signal Amplification ... [https://www.thermofisher.com/us/en/home/references/molecular-probes-the-handbook/ultrasensitive-detection-technology/tyramide-signal-amplification-technology.html]
- 53. Tyramide Signal Amplification - Biotium [https://biotium.com/technology/primary-secondary-antibody-conjugates/tyramide-signal-amplification/]
- 54. NBT/BCIP Stock Solution Protocol & Troubleshooting [https://www.sigmaaldrich.com/US/en/technical-documents/protocol/genomics/nucleic-acid-labeling-and-detection/nbt-bcip-stock-solution?srsltid=AfmBOopzQ5DxvyAiWXs7uab-zarRuUNZGL62SiQ9KmxhzJUGHNbPc_7B]
- 55. Quantitative methods for genome-scale analysis of in situ ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2395252/]