Membrane Protein Biogenesis: Direct Anchoring vs. Translocon-Mediated Systems in Health and Disease
This article provides a comprehensive comparison of membrane-anchored and translocation-based systems, the two principal pathways governing membrane protein biogenesis.
Membrane Protein Biogenesis: Direct Anchoring vs. Translocon-Mediated Systems in Health and Disease
Abstract
This article provides a comprehensive comparison of membrane-anchored and translocation-based systems, the two principal pathways governing membrane protein biogenesis. Tailored for researchers and drug development professionals, we explore the fundamental mechanisms of Oxa1/SecY family insertases and the Sec61/SecY translocon, detailing their distinct roles in inserting transmembrane domains with short versus long flanking segments. The scope extends to methodological advances in structural biology, strategies for troubleshooting misfolding and mislocalization, and a comparative analysis of how these systems influence drug disposition, efficacy, and the pathogenesis of genetic diseases. Understanding this dynamic interplay is crucial for developing novel therapeutic strategies targeting membrane protein homeostasis.
Core Machinery and Evolutionary Principles of Membrane Protein Integration
In cellular biology, the accurate integration of proteins into membranes is a fundamental process essential for energy generation, signal transduction, and cellular organization. Two principal macromolecular complexes have evolved to facilitate this: the Oxa1 family of insertases and the SecY translocon. The Oxa1 pathway specializes in the direct anchoring of proteins into the lipid bilayer, while the SecY translocon operates as a protein-conducting channel for translocation across membranes. Understanding their distinct mechanisms, substrates, and functional roles is critical for advancing research in membrane protein biogenesis, with implications for understanding diseases related to mitochondrial dysfunction and protein mislocalization. This guide provides a structured comparison of these two systems, supported by experimental data and methodological protocols.
Core Mechanisms and Structural Foundations
The Oxa1 family and the SecY translocon represent two evolutionarily distinct solutions to the problem of membrane protein biogenesis. Their core mechanisms stem from fundamentally different structural architectures.
Oxa1 Family: The Direct Insertase Mechanism
- Core Function: Members of the YidC/Oxa1/Alb3 family function primarily in the insertion and folding of membrane proteins [1] [2]. They are found in the bacterial cytoplasmic membrane (YidC), mitochondrial inner membrane (Oxa1), and chloroplast thylakoid membrane (Alb3) [1] [3].
- Mechanistic Principle: These insertases do not form a wide, aqueous channel. Instead, they facilitate membrane protein integration by lowering the kinetic barrier for the partitioning of hydrophobic transmembrane domains into the lipid bilayer [4]. They act as a platform that shields nascent membrane proteins from the aqueous environment and promotes their integration directly into the lipid phase.
- Co-translational Collaboration: A key feature of Oxa1 is its association with the mitochondrial ribosome, which allows it to facilitate the co-translational insertion of mitochondrially-encoded proteins [5] [3]. This direct interaction positions Oxa1 at the ribosomal exit tunnel to receive nascent chains as they are synthesized.
SecY Translocon: The Protein-Conducting Channel
- Core Function: The Sec translocon, with the SecYEG complex (in bacteria) or Sec61 complex (in the ER) at its core, mediates the translocation of unfolded polypeptides across the membrane and the co-translational insertion of nascent membrane proteins [6].
- Mechanistic Principle: SecY forms a hourglass-shaped protein-conducting channel within the membrane [6]. The channel is gated by several critical elements:
- The Pore Ring: A constriction in the middle of the channel formed by six hydrophobic residues that create a seal around the translocating polypeptide chain [6].
- The Plug: A short α-helix that blocks the channel in its idle state to prevent ion leakage [6].
- The Lateral Gate: A crevice between transmembrane segments 2 and 7 that can open to allow the integration of transmembrane domains into the lipid bilayer [6].
- Energy Coupling: Post-translational translocation through SecY is driven by the ATP-dependent motor protein SecA, which undergoes conformational changes to push the polypeptide through the channel [6]. The proton motive force (PMF) can also support translocation [6].
Fig. 1: Comparative schematic of the Oxa1 insertase and SecY translocon pathways. The Oxa1 pathway facilitates direct integration of proteins into the membrane, while the SecY translocon functions as a gated channel that requires ATP hydrolysis and utilizes a lateral gate for membrane protein insertion.
Comparative Functional Profiles
The fundamental structural differences between the Oxa1 and SecY systems underlie their distinct biological roles and substrate specificities.
Table 1: Functional Comparison of Oxa1 and SecY Systems
| Feature | Oxa1 Family Insertases | SecY Translocon |
|---|---|---|
| Primary Function | Membrane protein insertion & folding [1] [2] | Protein translocation across membrane & membrane protein insertion [6] |
| Representative Localization | Mitochondrial inner membrane (Oxa1), Bacterial plasma membrane (YidC), Chloroplast thylakoid (Alb3) [1] | Bacterial plasma membrane (SecYEG), ER membrane (Sec61) [6] |
| Key Substrates | Respiratory complex subunits (Cox1, Cox2, Cox3) [5] [3]; ATP synthase subunits [1] | Secretory proteins; multipass transmembrane proteins [6] [7] |
| Energy Source | Not well-defined; co-translational insertion coupled to translation [5] | ATP hydrolysis (via SecA); Proton Motive Force (PMF) [6] |
| Membrane Protein Topology | Primarily Nin-Cin orientation (e.g., Cox1, Cox3) [5] | Capable of complex topologies; N-tail export (Nout-Cin) [5] [6] |
| Ribosome Interaction | Direct interaction for co-translational insertion [5] [3] | Binds ribosomes for co-translational translocation/insertion [6] [7] |
| Evolutionary Conservation | Conserved from bacteria to organelles [1] [2] | Universally conserved in all domains of life [6] |
The Oxa1 pathway is specialized for the biogenesis of key respiratory chain components. Studies in yeast mitochondria demonstrated that Oxa1 is essential for the insertion of mitochondrially encoded proteins like Cox1p, Cox2p, and Cox3p [5]. Using temperature-sensitive Oxa1 mutants, researchers showed that the membrane insertion of these proteins was adversely affected, rendering them protease-inaccessible—a key indicator of failed integration into the inner membrane [5].
In contrast, the SecY translocon handles a broader range of substrates, including secretory proteins and complex multipass transmembrane proteins. Its role in quality control is exemplified by its ability to prevent the translocation of aberrant outer membrane porins (OmpC/F) with mutated constriction zones. Strikingly, this proof-reading function can be bypassed by PrlA (SecY) mutations that destabilize the closed state of the channel, demonstrating SecY's role as a gatekeeper for substrate quality [8].
Key Experimental Approaches and Data
The distinct functions of these pathways have been elucidated through well-established experimental methodologies. The following protocols are foundational to the field.
Protocol 1: Protease Accessibility Assay for Membrane Protein Insertion
Application: Determining the successful integration and topology of a membrane protein. This assay is ideal for studying Oxa1-dependent substrates [5].
Detailed Methodology:
- Isolate Organelles: Prepare intact mitochondria or bacterial membrane vesicles from wild-type and mutant (e.g.,
oxa1-ts) strains. - In vitro Synthesis: Incubate the organelles with
[³⁵S]-Methionine under conditions that support mitochondrial or bacterial protein synthesis. - Protease Treatment: Divide the sample and treat one aliquot with Proteinase K. Leave a parallel aliquot untreated as a control.
- Inhibition and Analysis: Add a protease inhibitor (e.g., PMSF) to stop the reaction. Solubilize the membranes, perform immunoprecipitation of the protein of interest (e.g., Cox2p), and analyze by SDS-PAGE and autoradiography.
Expected Outcome: A successfully inserted protein will have domains exposed to the intermembrane space (or periplasm) and will be degraded by the protease, seen as a loss of signal on the gel. A protein that fails to insert will be protected within the matrix (or cytosol) and will remain intact [5]. In oxa1-ts mutants at the non-permissive temperature, substrates like Cox2p accumulate in the uninserted, protease-protected form [5].
Protocol 2: Split GFP Complementation Assay for Translocation
Application: Monitoring the translocation of specific protein domains across a membrane. This method has been used to study SecY-mediated export [8].
Detailed Methodology:
- Genetic Engineering: Fuse the protein of interest (e.g., OmpC) to the 11th β-strand of GFP (GFP11). Express this construct in cells.
- Complementary Fragment Expression: Co-express the remaining 1-10 β-strands of GFP (GFP1-10) in the same cells, targeted to the destination compartment (e.g., the periplasm) via a signal peptide.
- Fluorescence Detection: If the protein of interest is successfully translocated and the GFP11-tagged domain reaches the destination compartment, the GFP fragments will assemble, resulting in a fluorescent signal detectable by microscopy or fluorometry.
Expected Outcome: Successful translocation of the OmpC-GFP11 fusion will result in green fluorescence in the periplasm. Mutants that abort translocation (e.g., OmpCΔH1) will fail to produce this fluorescence, confirming a translocation defect [8].
Table 2: Summary of Key Experimental Data from Literature
| Experiment | System | Key Finding | Interpretation |
|---|---|---|---|
| Protease Accessibility [5] | oxa1-ts yeast mitochondria |
~70% of newly synthesized Cox1p remained protease-inaccessible in the mutant, vs. accessible in WT. | Oxa1 is critically required for the membrane insertion of key respiratory subunits. |
| Split GFP Complementation [8] | E. coli SecYEG | OmpC variants with a deleted alpha-helical domain (ΔH1) showed no periplasmic fluorescence. | SecY-mediated translocation of the mutant porin was initiated but aborted post signal-peptide cleavage. |
| PrlA Suppression [8] | E. coli SecYEG | The PrlA4 mutation in SecY restored periplasmic export of a defective OmpC eyelet mutant. |
PrlA mutations stabilize the open SecY channel, bypassing quality control. |
| Functional Replacement [4] | Yeast mitochondria | A mitochondria-targeted Emc6–Emc3 fusion (core of ER insertase) restored respiration in Δoxa1 cells. |
The membrane insertion mechanism is structurally and functionally conserved across insertase families. |
The Scientist's Toolkit: Essential Research Reagents
The following reagents, as identified in the search results, are crucial for experimental investigation of these pathways.
Table 3: Key Research Reagents and Their Applications
| Reagent / Tool | Function / Description | Experimental Application |
|---|---|---|
Temperature-sensitive (ts) Mutants (e.g., oxa1-ts) [5] |
Allows conditional inactivation of the essential gene by shifting to a non-permissive temperature. | Studying the acute loss-of-function effects on protein biogenesis in vivo. |
PrlA Mutants of SecY (e.g., PrlA4) [6] [8] |
SecY mutants with relaxed proof-reading capability, allowing translocation of proteins with defective signal sequences. | Probing the gating and quality control mechanisms of the SecY channel. |
| BioID2 [9] | An engineered biotin ligase that biotinylates proximate proteins, enabling the mapping of protein interactions in living cells. | Identifying transient interaction partners and the molecular environment of insertases like Oxa1. |
| Mito-EMC Fusion Protein [4] | A chimeric protein consisting of the core subunits of the ER Membrane Complex (EMC) targeted to mitochondria. | Testing the functional conservation between evolutionarily related insertases from different organelles. |
Fig. 2: A decision workflow for investigating Oxa1 and SecY pathways. The experimental approach is determined by the specific biological question, with distinct genetic tools and functional assays available for each system.
The Oxa1 insertase and SecY translocon represent two fundamental, mechanistically distinct paradigms for membrane protein biogenesis. The Oxa1 pathway is a specialized machinery for the direct co-translational integration of hydrophobic proteins into the lipid bilayer, playing an indispensable role in the biogenesis of oxidative phosphorylation complexes [5] [3]. In contrast, the SecY pathway provides a versatile, gated channel for both the translocation of soluble proteins and the insertion of membrane proteins, with an inherent proof-reading function to ensure substrate quality [6] [8].
Emerging evidence, including the functional replacement of Oxa1 by engineered ER insertase components, suggests a deep evolutionary conservation among membrane insertases [4]. This comparative guide provides the foundational knowledge and experimental framework for researchers to dissect the unique and overlapping functions of these essential cellular machines, a crucial endeavor for the broader understanding of membrane biology and its implications in disease.
The biogenesis of membrane proteins is a fundamental cellular process facilitated by highly conserved molecular machinery. This guide compares the two principal systems responsible for membrane protein insertion: the SecY translocon and the Oxa1 insertase. While SecY serves as the primary channel for co-translational protein translocation and insertion across bacterial plasma membranes and the eukaryotic endoplasmic reticulum, the Oxa1/YidC family operates as a specialized machinery for the post-translational membrane insertion of specific protein subsets, particularly tail-anchored and multi-spanning membrane proteins with C-terminal transmembrane domains. Evolutionary analysis reveals these systems originated from a common ancestral mechanism, with Oxa1 homologs (including bacterial YidC and chloroplast Alb3) representing an ancient insertion pathway that has been conserved across all domains of life. This objective comparison examines their structural features, mechanistic principles, substrate specificities, and supporting experimental data, providing researchers with a framework for understanding the evolutionary progression from unassisted membrane insertion to specialized protein translocation systems.
The proper folding and assembly of membrane proteins depends critically on accurate insertion of their transmembrane domains (TMs) into the target lipid bilayer. Cellular systems have evolved two major pathways to facilitate this process: the SecY-mediated co-translational pathway that couples protein translation with membrane insertion, and the Oxa1-mediated post-translational pathway that inserts fully synthesized polypeptides. These systems work in concert to manage the diverse topogenetic challenges presented by different membrane protein architectures.
The SecY translocon constitutes the core of the general secretory (Sec) pathway, a system highly conserved across bacteria, archaea, and eukaryotes [10] [11]. This complex facilitates both the translocation of soluble proteins across membranes and the integration of membrane proteins into the lipid bilayer. In contrast, the Oxa1 superfamily represents a more specialized membrane insertase family that includes bacterial YidC, mitochondrial Oxa1, and chloroplast Alb3 [5]. While SecY primarily handles ribosome-associated nascent chains, Oxa1/YidC can operate either in conjunction with SecY or independently to mediate membrane protein insertion.
Recent research has illuminated the evolutionary relationship between these systems, suggesting that Oxa1 represents a more ancient mechanism that has been conserved alongside the more complex Sec machinery. Studies indicate that the sequences of membrane proteins themselves have co-evolved with these insertion machineries, with particular adaptations in C-terminal domains to optimize post-translational insertion via Oxa1-like mechanisms [12]. This co-evolution has resulted in specialized systems that handle distinct but overlapping subsets of the cellular membrane proteome.
The SecY Translocon System: Architecture and Mechanism
Structural Composition and Evolutionary Conservation
The SecY protein forms the central pore of the bacterial translocon complex, typically associating with SecE and SecG subunits to form the functional SecYEG translocon [11]. This core complex collaborates with additional membrane proteins (SecD, SecF) and the cytoplasmic ATPase SecA to drive protein translocation. The eukaryotic homolog, Sec61α, forms the analogous Sec61 complex in the endoplasmic reticulum membrane, demonstrating the remarkable evolutionary conservation of this translocation system [11] [13].
Structural studies reveal that SecY forms a hourglass-shaped channel with a constricted region in the middle, creating a pore ring that maintains membrane permeability barriers during protein translocation [11]. Cytoplasmic regions 2 and 3, and transmembrane domains 1, 2, 4, 5, 7, and 10 are particularly well conserved across species, with the conserved cytoplasmic regions believed to interact with cytoplasmic secretion factors, while the TM domains participate directly in protein export [11].
Mechanistic Principles of Operation
The Sec system operates through two distinct but related mechanisms: the SecB pathway for secretory proteins and the signal recognition particle (SRP) pathway for membrane proteins [10].
SecB Pathway: In many Gram-negative bacteria, proteins destined for transport to the periplasm or extracellular environment contain a removable N-terminal signal sequence recognized by the SecB chaperone, which maintains preproteins in an unfolded state and delivers them to SecA [10] [11]. SecA then hydrolyzes ATP to provide the energy for translocation through the SecYEG channel.
SRP Pathway: Membrane proteins with hydrophobic transmembrane domains are recognized by the signal recognition particle (SRP) as they emerge from the ribosome, leading to a co-translational mechanism where translation is coupled with secretion through the SecYEG channel [10]. During this process, transmembrane domains escape through the lateral gate of the SecY channel into the lipid bilayer.
Table 1: Key Components of the Bacterial Sec Translocon System
| Component | Type | Function |
|---|---|---|
| SecY | Transmembrane protein | Main transmembrane subunit; forms protein-conducting channel |
| SecE | Transmembrane protein | Essential subunit stabilizing SecY structure |
| SecG | Transmembrane protein | Enhances translocation efficiency |
| SecA | Cytoplasmic ATPase | Motor protein providing energy for translocation; binds SecB-substrate complexes |
| SecB | Cytoplasmic chaperone | Binds preproteins; maintains unfolded state; targets substrates to SecA |
| SRP | Cytoplasmic ribonucleoprotein | Recognizes hydrophobic signal sequences; mediates co-translational targeting |
| FtsY | Membrane-associated receptor | SRP receptor; facilitates delivery of ribosome-nascent chain complexes to SecYEG |
The Sec translocon mediates insertion of transmembrane helices (TMs) through a lateral gating mechanism, where TMs partition into the lipid bilayer through a seam in the SecY structure. Recent research using selective ribosome profiling has defined how and when factors for N-glycosylation and membrane insertion engage and disengage from the core Sec61 translocation channel during biogenesis of secretory and membrane proteins [14]. The fundamental principles of this cotranslational insertion mechanism are conserved across organisms, with bacteria, archaea, and eukaryotes all employing homologous systems [12].
The Oxa1 Insertase Superfamily: Specialized Membrane Insertion
Evolutionary Conservation and Family Members
The Oxa1 superfamily represents a universally conserved family of membrane insertases found in all domains of life. Family members include: Oxa1 in mitochondrial inner membranes, YidC in bacterial plasma membranes, and Alb3 in chloroplast thylakoid membranes [5]. These proteins facilitate membrane protein insertion through a mechanism distinct from the Sec translocon, specializing particularly in the post-translational membrane integration of specific substrate classes.
Evolutionary analyses suggest that the Oxa1/YidC/Alb3 protein family represents a novel evolutionarily conserved membrane protein insertion machinery that predates the Sec system [5]. The conservation of this mechanism across evolutionary boundaries highlights its fundamental importance in membrane biogenesis. Recent studies have identified SecY proteins in the mitochondrial genomes of jakobids and Mantamonas sphyraenae, representing particularly gene-rich mitogenomes that provide insights into the evolutionary transition between these systems [11].
Substrate Specificity and Mechanism of Action
Oxa1 superfamily members facilitate the membrane insertion of a diverse set of substrates with particular specialization for:
- Tail-anchored (TA) proteins with a single C-terminal transmembrane domain [12]
- C-terminal transmembrane helices (cTMs) of multi-spanning membrane proteins [12]
- Polytopic membrane proteins with N-termini retained in the matrix (mitochondria) or cytoplasm (bacteria) [5]
Recent research has revealed that evolution has refined the hydrophilicity and length of C-terminal tails following cTMs to optimize their post-translational insertion, primarily via Oxa1-like mechanisms [12]. In Escherichia coli, YidC serves as the insertase for cTMs of proteins with extracytosolic C-termini, with C-tail mutations disrupting productive cTM-YidC interaction.
Table 2: Oxa1 Superfamily Members and Their Functions
| Organism | Insertase | Localization | Key Substrates | Characteristic Features |
|---|---|---|---|---|
| Bacteria | YidC | Plasma membrane | cTMs of multi-spanning MPs, TA proteins | Cooperates with SecYEG or functions independently |
| Mitochondria | Oxa1 | Inner membrane | Cox1p, Cox2p, Cox3p, cytochrome b | General membrane insertion machinery |
| Chloroplasts | Alb3 | Thylakoid membrane | Light-harvesting chlorophyll-binding protein (LHCP) | Essential for thylakoid biogenesis |
| Eukaryotes (ER) | EMC | Endoplasmic reticulum | cTMs of multi-spanning MPs | Functional analog for post-translational insertion |
The mechanism of Oxa1-mediated insertion involves direct interaction with substrate transmembrane domains during or after their synthesis. In mitochondria, Oxa1 interacts directly with nascent polypeptide chains during their synthesis, facilitating co-translational insertion from the matrix [5]. Studies demonstrate that the interaction of Oxa1 with its substrates is particularly strong when nascent chains are inserted into the membrane, suggesting a direct function in co-translational insertion [5].
Comparative Analysis: SecY versus Oxa1 Mechanisms
Functional Overlap and Distinct Roles
While both systems facilitate membrane protein insertion, they display distinct substrate preferences, temporal coordination, and mechanistic principles. The table below summarizes key comparative features:
Table 3: Functional Comparison of SecY and Oxa1/YidC Membrane Insertion Systems
| Feature | SecY Translocon | Oxa1/YidC Insertase |
|---|---|---|
| Primary Function | Protein translocation; initial membrane insertion | Specialized membrane insertion |
| Translocation Capability | Yes (full protein chains) | Limited (transmembrane segments) |
| Insertion Mechanism | Lateral gating from central pore | Direct partitioning from protein-lipid interface |
| Temporal Coordination | Primarily co-translational | Co- and post-translational |
| Representative Substrates | Secretory proteins; most multi-spanning MPs | cTMs; tail-anchored proteins; specific multi-spanning MPs |
| Energy Source | ATP hydrolysis (SecA); proton motive force | Proton motive force; ATP-independent |
| Membrane Topology | Complex with multiple subunits | Simpler structure; can function as monomer |
| Evolutionary Conservation | Universal (SecY/Sec61) | Universal (Oxa1/YidC/Alb3/EMC) |
| Spatial Organization | Discrete translocation sites | Distributed throughout membrane |
Recent research highlights that the C-terminal tails of membrane proteins are under unique evolutionary pressure that optimizes them for insertion by specific machineries. In proteins with cytosolic C-termini (Ccyt), C-tails tend to be longer and more hydrophilic, while in proteins with extracytosolic C-termini (Cext), C-tails are significantly shorter and less hydrophilic [12]. This adaptation is particularly sharp in C-tails compared to other loop locations, suggesting specialized evolution for post-translational insertion via Oxa1-like mechanisms.
Cooperative Interactions in Membrane Protein Biogenesis
Despite their distinct functions, SecY and Oxa1/YidC often cooperate in cellular membrane biogenesis. In bacteria, YidC can associate with the SecYEG translocon to facilitate the insertion of certain transmembrane domains, particularly those in multi-spanning membrane proteins [12]. Recent structural studies have identified SND3 as a membrane insertase within a distinct SEC61 translocon complex, implying a role in co-translational insertion of multi-pass membrane proteins [14].
In mitochondria, Oxa1 represents a general membrane insertion machinery that works alongside other import components. Studies demonstrate that Oxa1 is required for efficient membrane insertion of newly synthesized Cox1p, Cox3p, and cytochrome b, with the degree of dependency varying among different mitochondrial proteins [5]. The interaction of Oxa1 with these nascent chains is stabilized by the presence of associated ribosomes, supporting its role in co-translational insertion.
Experimental Approaches and Research Methodologies
Key Experimental Models and Techniques
Research comparing SecY and Oxa1 mechanisms employs diverse experimental systems ranging from bacterial models to eukaryotic organelles. Key methodologies include:
Protease Protection Assays: Used to monitor membrane integration by assessing accessibility of protein domains to externally added proteases [5]. In mitochondrial studies, mitoplasts (mitochondria with disrupted outer membranes) are treated with proteinase K to probe for translocation of hydrophilic loops to the intermembrane space.
Cross-linking and Interaction Studies: Employed to identify proximate components during membrane insertion. Studies demonstrate that Oxa1 interacts directly with its substrates prior to completion of their synthesis, with particularly strong interactions when nascent chains are inserted into the membrane [5].
Selective Ribosome Profiling: A recently developed technique that enables genome-wide analysis of translational pausing and factor recruitment during membrane protein biogenesis [14]. This approach has defined how and when factors for N-glycosylation and membrane insertion engage with the core translocation channel.
Genetic Approaches: Temperature-sensitive mutants (e.g., oxa1-ts) enable functional dissection of essential insertion components [5]. Studies using such mutants revealed that Oxa1 function is particularly important for insertion of Cox1p and Cox2p, with varying dependency among mitochondrial proteins.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagents for Studying Membrane Protein Insertion
| Reagent/Category | Specific Examples | Research Applications | Key Features and Functions |
|---|---|---|---|
| Antibody Reagents | Anti-SecY, Anti-YidC, Anti-Oxa1 | Localization studies; immunoprecipitation; Western blotting | Validate protein expression and localization; complex isolation |
| Genetic Tools | Temperature-sensitive mutants (oxa1-ts); knockout strains; siRNA | Functional analysis; pathway requirements | Enable conditional disruption of insertion machinery |
| Chemical Inhibitors | Azide; NaN₃; specific translocation inhibitors | Energy depletion; pathway inhibition | Block ATP-dependent processes; test energy requirements |
| Protease Reagents | Proteinase K; trypsin | Protease protection assays; topology mapping | Determine membrane integration and protein orientation |
| Cross-linking Agents | Formaldehyde; DSS; BMH | Protein-protein interaction studies; complex identification | Capture transient interactions during insertion |
| Labeling Reagents | [³⁵S]-methionine; fluorescent dyes (GFP, RFP) | Pulse-chase studies; localization tracking | Visualize and quantify protein synthesis and localization |
Research Applications and Therapeutic Implications
Understanding the evolutionary origins and mechanistic distinctions between SecY and Oxa1 systems has important implications for multiple research domains:
Antibiotic Development: Bacterial YidC represents a promising target for novel antibiotics, as it is essential for viability and has no direct homolog in eukaryotic cytoplasm [10]. Its role in inserting virulence factors makes it particularly attractive for anti-virulence strategies.
Disease Mechanism Elucidation: Mutations disrupting membrane protein insertion cause human genetic diseases. Recent research has identified disease-causing mutations in human membrane proteins that increase C-tail hydrophilicity, causing misinsertion and mistrafficking consistent with misfolding [12].
Biotechnology Applications: Engineered versions of these insertion systems facilitate production of membrane proteins for structural and pharmacological studies. Modulating C-tail properties can optimize membrane protein expression for structural biology and drug screening.
Organelle Biogenesis Research: Understanding Oxa1 function provides insights into mitochondrial disorders and chloroplast development defects. The conservation between bacterial YidC and mitochondrial Oxa1 further enables comparative studies.
The evolutionary progression from unassisted membrane insertion to specialized SecY and Oxa1 machinery represents a fundamental adaptation in cellular biology. While the SecY translocon serves as the primary conduit for co-translational protein translocation and membrane insertion, the Oxa1 superfamily provides essential specialized functions for post-translational insertion of specific substrate classes, particularly proteins with C-terminal transmembrane domains.
Experimental evidence demonstrates that these systems are not mutually exclusive but rather function in an integrated network to manage the diverse membrane protein biogenesis needs of the cell. Recent findings that membrane protein sequences themselves have evolved to optimize collaboration with cellular insertion machinery highlight the sophisticated co-evolution between proteins and their biogenesis machinery [12]. This integrated view provides researchers with a comprehensive framework for investigating membrane protein biogenesis in both physiological and pathological contexts, with significant implications for therapeutic development and biotechnology applications.
The biogenesis of membrane and secretory proteins requires precise recognition and translocation of nascent polypeptides across cellular membranes. Two principal energetic factors—translocated domain hydrophobicity and length—serve as key determinants directing proteins into distinct translocation pathways such as the Sec61/SRP-dependent route or the twin-arginine translocation (TAT) system. This guide objectively compares these pathways using experimental data, highlighting how hydrophobicity and length parameters dictate mechanistic choices that impact protein folding, membrane integration, and biological function. Understanding these determinants provides critical insights for optimizing recombinant membrane protein production and targeting specific pathways for therapeutic intervention.
Integral membrane proteins and secreted proteins represent a substantial fraction of the proteome and require sophisticated machinery for correct localization and folding. The fundamental challenge lies in transporting often highly hydrophobic polypeptide segments across or into lipid bilayers—a process governed by specific physicochemical properties of the nascent chain [15] [16]. Among these properties, hydrophobicity and domain length have emerged as primary energetic factors determining which translocation pathway a protein will utilize.
The two major pathways for protein translocation—the Sec/SRP pathway and the TAT pathway—differ fundamentally in their energetic requirements and conformational preferences. The Sec61/SRP pathway typically handles unfolded polypeptides in an ATP-dependent process, threading chains through a protein-conducting channel [17] [18]. In contrast, the TAT pathway specializes in transporting fully folded proteins, often with bound cofactors, using a distinct proton-motive force mechanism [17]. This guide systematically compares these pathways, examining how hydrophobicity and length parameters dictate pathway selection through specific experimental approaches and findings.
Pathway Comparison: SRP/Sec61 vs. TAT Systems
Table 1: Key Characteristics of Major Protein Translocation Pathways
| Feature | SRP/Sec61 Pathway | TAT Pathway |
|---|---|---|
| Protein State | Unfolded polypeptide chain | Folded, cofactor-containing proteins |
| Energy Source | ATP/GTP hydrolysis | Proton motive force |
| Signal Sequence | Hydrophobic signal anchor | Twin-arginine (RR) motif |
| Hydrophobicity Requirement | High hydrophobicity essential for SRP binding | Moderate hydrophobicity; folding quality control |
| Domain Length Consideration | Extended conformation in ribosome tunnel | Pre-folded domains of varying sizes |
| Key Machinery | SRP, Sec61 translocon | TatA, TatB, TatC components |
| Typical Substrates | Membrane proteins, secreted proteins | Redox enzymes with metal cofactors |
The SRP/Sec61-Dependent Pathway: Hydrophobicity-Driven Targeting
The SRP/Sec61 pathway represents the major route for co-translational protein translocation and membrane integration. This system recognizes hydrophobic segments in nascent chains and directs them to the endoplasmic reticulum (ER) membrane or bacterial plasma membrane via the Sec61/SecYEG translocon [15] [18].
Molecular Mechanism: The process initiates when a hydrophobic signal sequence or transmembrane domain emerges from the ribosome exit tunnel. In eukaryotes, the signal recognition particle (SRP), composed of 7S RNA and six proteins including SRP54, binds to these hydrophobic segments through its M-domain, which contains a hydrophobic groove accommodating approximately 10 residues in an α-helical conformation [15]. This SRP-nascent chain interaction then recruits the ribosome-nascent chain complex to the ER membrane via interaction with the SRP receptor. The nascent chain is subsequently transferred to the Sec61 translocon complex, a heterotrimeric protein-conducting channel that facilitates polypeptide translocation across the membrane or lateral integration into the lipid bilayer [18] [7].
Recent structural insights from cryo-electron tomography studies reveal specialized translocon configurations at the ER membrane, with distinct polysomes binding to different ER translocons optimized for either signal peptide-containing proteins or multipass transmembrane proteins [7]. The translocon-associated protein complex (TRAP) appears to play a role in both configurations, facilitating signal peptide insertion.
The TAT Pathway: Folded Protein Translocation
The twin-arginine translocation (TAT) system provides an alternative route that specializes in transporting folded proteins across cytoplasmic membranes in bacteria and chloroplast thylakoid membranes [17]. This pathway is functionally distinct from the Sec pathway in both mechanism and substrate specificity.
Molecular Mechanism: TAT substrates are characterized by a specific signal peptide containing a conserved twin-arginine (RR) motif. Unlike Sec substrates, TAT substrates fold completely and often incorporate complex cofactors (such as Fe-S clusters or molybdopterin) in the cytoplasm prior to translocation [17]. The TAT machinery typically consists of TatA, TatB, and TatC components (or TatA and TatC only in minimal systems). TatBC complexes recognize the signal peptide, while TatA complexes are thought to form the actual translocation channel. The system remarkable accommodates substrate proteins of varied sizes and different physicochemical properties without compromising membrane integrity [17].
The TAT pathway includes quality control mechanisms that ensure proper folding and cofactor incorporation before export, rejecting unfolded substrates. This prevents the need for complex reassembly processes in the extracellular environment or target organelle [17].
Experimental Evidence: Hydrophobicity and Length Parameters
Hydrophobicity as the Primary Determinant for SRP Recognition
Experimental studies with G protein-coupled receptors (GPCRs) provide compelling evidence for hydrophobicity as the critical factor in SRP-mediated pathway selection.
Experimental Protocol:
- System: In vitro S-30 prokaryotic transcription/translation system
- Method: Ribosome nascent chain complexes (RNCs) of GPR35 signal anchor domain were synthesized from linear DNA templates lacking stop codons
- Measurements: Pegylation assays using PEG-MAL to assess cysteine accessibility at various nascent chain lengths (25-50 amino acids from peptidyl transferase center)
- Key Manipulation: Alteration of hydrophobicity within the first transmembrane segment while maintaining sequence length [15]
Findings: The signal anchor domain of GPR35 remained in an extended conformation while exiting the ribosome tunnel, with compaction occurring only upon interaction with SRP and Sec61 components. Critically, reducing native hydrophobicity within the first transmembrane domain destabilized SRP interaction and subsequently impaired ER membrane integration, demonstrating that hydrophobicity—not pre-formed structure—drives pathway selection [15]. This suggests that hydrophobic recognition represents the fundamental energetic basis for SRP pathway commitment.
Domain Length and Conformational Constraints
The length of translocated domains influences conformational possibilities during translocation, particularly in the constrained environment of the ribosome exit tunnel.
Experimental Approaches:
- Pegylation Accessibility Assays: Systematic measurement of PEG-MAL reactivity to engineered cysteine residues at varying distances from the peptidyl transferase center
- Cryo-EM Analysis: High-resolution structural studies of ribosome-nascent chain complexes
- Cross-linking Studies: Identification of proximate interactions between nascent chains and ribosomal components [15]
Key Observations: The ribosomal exit tunnel measures approximately 100Å in length with a diameter ranging from 10-20Å, accommodating about 30 amino acids in an extended conformation or up to 65 amino acids in a compacted helical conformation [15]. For GPR35, pegylation assays revealed that nascent chains of 25 amino acids remained completely buried within the tunnel, while chains of 30 amino acids showed partial exposure. Maximum accessibility occurred at 45-50 amino acids, indicating full exit from the tunnel. Throughout this process, the transmembrane domain remained extended rather than adopting secondary structure, emphasizing that length primarily determines exposure rather than dictating folding pathway selection [15].
Table 2: Experimental Approaches for Studying Translocation Determinants
| Method | Application | Key Findings |
|---|---|---|
| Pegylation Assays | Measure nascent chain compaction in ribosome tunnel | GPR35 TM domain remains extended until interaction with SRP/Sec61 |
| Hydrophobicity Mutagenesis | Alter hydrophobic character while maintaining length | Reduced hydrophobicity disrupts SRP binding and membrane integration |
| Cryo-ET of ER Membranes | Visualize native translocon complexes | Specialized translocons for different substrate types |
| Modular Display Systems | Test membrane anchor efficiency | Anchor-passenger interactions affect functionality independent of sequence |
Visualization of Translocation Pathways
The following diagram illustrates how hydrophobicity and domain length determine pathway selection between the SRP/Sec61 and TAT translocation systems:
Research Reagent Solutions for Translocation Studies
Table 3: Essential Research Tools for Protein Translocation Experiments
| Reagent/Category | Specific Examples | Research Application |
|---|---|---|
| In Vitro Translation Systems | S-30 Prokaryotic System, Wheat Germ Extract | Nascent chain compaction studies, co-translational folding analysis |
| Modular Display Platforms | SpyCatcher/SpyTag System with various membrane anchors (Lpp-OmpA, PgsA, INP, AIDA-I) | Testing anchor-passenger compatibility without genetic fusions |
| Chemical Probes | PEG-MAL (Polyethylene glycol-maleimide) | Pegylation assays to measure nascent chain accessibility |
| Translocon Components | Purified Sec61/SecYEG complexes, SRP components | Reconstitution experiments, mechanistic studies |
| Membrane Model Systems | ER-derived microsomes, proteoliposomes | Native environment translocation assays |
Discussion: Energetic Principles and Research Implications
The energetic basis of translocation pathway choice revolves fundamentally around the interplay between hydrophobicity and domain length. High hydrophobicity domains preferentially engage the SRP/Sec61 pathway through hydrophobic interactions with SRP54, committing the protein to co-translational membrane integration. Domain length primarily governs conformational possibilities within the ribosomal tunnel, with shorter domains remaining constrained while longer domains gain flexibility for interaction with downstream components.
These principles have profound implications for membrane protein research and biotechnology. The finding that hydrophobicity rather than secondary structure dictates SRP recognition [15] suggests that bioinformatic predictions based solely on secondary structure may be insufficient for accurately predicting membrane protein biogenesis. Similarly, the modularity demonstrated in surface display systems [19] highlights how understanding these determinants enables engineering of optimized protein localization systems.
For drug development professionals, these insights are particularly valuable. With membrane proteins constituting approximately 60% of approved drug targets [20], understanding the fundamental principles governing their biogenesis enables more strategic approaches to drug design and optimization. The ability to manipulate pathway choice through strategic alterations in hydrophobicity profiles offers potential for improving functional expression of challenging therapeutic targets.
The biogenesis of membrane proteins is a fundamental cellular process essential for energy production, metabolite exchange, and cellular signaling. This process is mediated by highly conserved molecular machines. The Sec61/SecY translocon represents the central protein-conducting channel for polypeptide translocation and membrane integration, operating universally across organisms from bacteria to humans [21]. In contrast, the YidC/Oxa1/Alb3 family of insertases, which includes bacterial YidC, mitochondrial Oxa1, chloroplast Alb3, and the endoplasmic reticulum (ER) complexes EMC and GET, facilitates membrane protein insertion through a distinct, more specialized mechanism [22] [4]. Although both systems can handle membrane proteins, their structures, precise mechanisms, and substrate preferences differ significantly. This guide provides a structured comparison of these key players, supported by experimental data and methodologies relevant to ongoing research on membrane-anchored versus translocation-based systems.
Systematic Comparison of Molecular Machines
The table below summarizes the core characteristics, mechanisms, and substrates for the central translocon and the Oxa1 family insertases.
Table 1: Comparative Overview of the Sec61/SecY Translocon and Oxa1 Family Insertases
| Feature | Sec61/SecY Translocon | YidC/Oxa1/Alb3 Family | EMC Complex | GET Complex |
|---|---|---|---|---|
| Core Identity & Structure | Heterotrimeric complex (α, β, γ subunits); Sec61α forms an hourglass-shaped aqueous channel [21]. | Monomeric proteins with 5 conserved transmembrane domains (TMDs) [22]. | Multisubunit ER complex; core formed by Emc3-Emc6, structurally related to YidC [4]. | Heterodimeric ER complex (Get1-Get2/WRB-CAML); core structurally related to YidC [4]. |
| Primary Localization | Endoplasmic Reticulum (ER) in eukaryotes; plasma membrane in bacteria (SecY) [21]. | Bacterial cytoplasmic membrane (YidC), mitochondrial inner membrane (Oxa1), chloroplast thylakoid membrane (Alb3) [22]. | Endoplasmic Reticulum (ER) [23]. | Endoplasmic Reticulum (ER) [4]. |
| Core Mechanism | Forms a gated, water-filled pore; facilitates lateral release of TMDs into the lipid bilayer [21]. | Functions as an insertase, likely catalyzing lipid partitioning and compression without a large aqueous pore [4] [24]. | Functions as an insertase; rectifies topology for multipass proteins and handles post-translational insertion [23]. | Specialized insertase for the post-translational insertion of tail-anchored (TA) proteins [4]. |
| Key Substrates & Functions | Co-translational insertion of most multi-pass membrane proteins; translocation of secretory proteins [21] [24]. | Insertion of respiratory chain subunits (bacteria/mitochondria) and photosynthetic complexes (chloroplasts) [22]. | Post-translational insertion of the final TMD of multipass proteins (e.g., Cys-loop receptors); co-translational insertion of first TMDs [23]. | Post-translational insertion of tail-anchored proteins, which have a single C-terminal TMD [4]. |
| Representative Experimental Substrates | Rhodopsin [24], ASGR1 [23]. | Cytochrome oxidase subunits (Cox1, Cox2, Cox3), F0F1-ATP synthase subunits [22]. | GABAA receptor subunit (GABRA1), Squalene Synthase (SQS) [23]. | Not specified in search results. |
Table 2: Key Functional and Genetic Evidence from Cross-Complementation Studies
| Experimental Approach | Key Finding | Implication |
|---|---|---|
| EMC Replacement of Oxa1 | A mitochondria-targeted fusion of EMC core proteins (Emc6-Emc3) partially restored respiratory competence and supported the insertion of proteins like Cox2 in yeast lacking Oxa1 (Δoxa1) [4]. | The core insertase function is functionally conserved and transferable between the ER and mitochondria, despite different evolutionary origins. |
| Functional Conservation | Chloroplast Alb3 and Alb4 from Arabidopsis thaliana can replace E. coli YidC function [22]. | The YidC/Oxa1/Alb3 family members are functionally interchangeable across kingdoms of life. |
| Substrate-Specific Limitations | The mito-EMC fusion could not facilitate the assembly of the Atp9 ring in mitochondria [4]. | While core insertion mechanisms are conserved, specific assembly functions may require specialized factors not present in the heterologous system. |
Essential Research Tools and Methodologies
To investigate these systems in a laboratory setting, researchers employ a suite of well-established protocols and reagents. The table below outlines key components of the experimental toolkit.
Table 3: The Scientist's Toolkit: Key Reagents and Methodologies
| Tool / Reagent | Function in Research | Example Application |
|---|---|---|
| Semi-Permeabilized Cells (SPCs) | In vitro system that provides native ER membranes with functional translocons and insertases for protein biogenesis assays [23]. | Used to reconstitute the membrane insertion of radiolabeled proteins (e.g., GABRA1) to test the dependence on specific factors like EMC [23]. |
| Protease Protection Assay | Determines whether a protein domain has been translocated across or integrated into a membrane, as membrane-protected fragments are resistant to digestion [23]. | Validated the insertion of TMDs; the C-terminal fragment of GABRA1 was protected in wild-type but not ΔEMC SPCs [23]. |
| Glycosylation Tagging | Using the endogenous oligosaccharyltransferase (OST) complex, a engineered glycosylation site reports on the translocation of a specific protein segment into the ER lumen [23]. | A glycosylated C-terminal tail on GABRA1-glyc reporter confirmed EMC-dependent post-translational translocation [23]. |
| Genetic Complementation | Tests whether a gene from one system can rescue the function of a deleted gene in another system, indicating functional homology [4]. | Mitochondrial expression of EMC core components (mito-EMC) to rescue the viability of a yeast Δoxa1 mutant [4]. |
| Cryo-Electron Microscopy (Cryo-EM) | High-resolution structural technique for visualizing macromolecular complexes in near-native states [24]. | Revealed structures of ribosome-translocon complexes and dynamic interactions of TMDs with the Sec61 lateral gate and other factors [24]. |
Core Experimental Protocol: In Vitro Reconstitution of Membrane Protein Insertion
This foundational protocol is used to dissect the molecular requirements for inserting a protein into the ER membrane [23].
- Template Design: Clone the cDNA of the target membrane protein (e.g., GABRA1) under a suitable promoter. To monitor translocation, engineer a glycosylation consensus sequence (N-X-S/T) into the region of interest (e.g., the C-terminal tail).
- In Vitro Transcription/Translation: Synthesize the mRNA in vitro and then translate it in a cell-free system like rabbit reticulocyte lysate in the presence of ³⁵S-labeled methionine to produce radiolabeled nascent proteins.
- Incubation with Semi-Permeabilized Cells (SPCs): Mix the newly synthesized, radiolabeled protein with SPCs derived from either wild-type or mutant (e.g., ΔEMC) mammalian cells. These SPCs provide the necessary membrane machinery.
- Membrane Insertion Assay:
- Glycosylation Readout: After incubation, solubilize the membranes and perform immunoprecipitation for the target protein. Analyze by SDS-PAGE and autoradiography. A shift to a higher molecular weight indicates successful glycosylation and thus translocation of the tagged segment.
- Protease Protection Readout: Treat the membrane mixture with proteinase K. Subsequently, solubilize the membranes, immunoprecipitate the protein with antibodies against different domains (e.g., N-terminal and C-terminal), and analyze by SDS-PAGE. The appearance of protected fragments indicates which domains have been integrated into or translocated across the membrane.
Mechanisms and Pathways Visualized
The diagrams below illustrate the core mechanisms and functional relationships of these molecular players.
The Sec61 Translocon and its Accessory Systems
Diagram Title: Functional Specialization at the ER Translocon
The Oxa1 Superfamily Across Organelles
Diagram Title: Evolutionary and Functional Relationships of the Oxa1 Superfamily
The comparative analysis underscores a fundamental division of labor in membrane protein biogenesis. The Sec61/SecY translocon serves as the central, versatile channel for polypeptide translocation and the co-translational integration of the majority of transmembrane domains. In contrast, the diverse members of the Oxa1 superfamily, including the EMC and GET complexes, have evolved as specialized insertases that handle specific, challenging substrates—such as tail-anchored proteins or the final TMDs of multipass proteins—often through post-translational mechanisms [4] [23].
The functional replacement of mitochondrial Oxa1 by the core of the ER's EMC complex provides compelling evidence that these distantly related systems adhere to similar topological principles and possess a functionally conserved core [4]. This functional conservation, despite limited sequence similarity, suggests that the basic mechanism of insertase-mediated membrane protein integration is an ancient, evolutionarily optimized process. Future research will likely focus on obtaining high-resolution structures of these complexes in action with their native substrates, further elucidating the precise molecular choreography of TMD integration and the regulatory mechanisms that govern the choice between the Sec61 pathway and its partner insertases.
The biogenesis of integral membrane proteins is a fundamental cellular process governed by a sophisticated set of topological rules. This review systematically compares the mechanisms determining transmembrane domain (TMD) orientation and integration efficiency, focusing on the interplay between sequence-encoded topogenic signals and the cellular machinery that interprets them. We synthesize recent structural and biochemical findings that elucidate how the translocon decodes sequence features such as hydrophobicity and charge distribution, and how membrane lipid composition further refines topological outcomes. Experimental data across multiple systems reveal how competing topological determinants are balanced during membrane protein biogenesis, providing a framework for predicting and engineering membrane protein topology for basic research and therapeutic development.
Integral membrane proteins constitute approximately 25% of all protein-coding genes and perform crucial functions in transport, signaling, and catalysis [25]. Their biogenesis represents one of the most ancient biological processes, with core machinery conserved across all domains of life. A fundamental aspect of membrane protein biogenesis is topogenesis—the establishment of correct transmembrane topology, defined by the number, orientation, and integration efficiency of TMDs within the lipid bilayer [26] [27].
The process of membrane protein topogenesis must accommodate tremendous diversity—the human genome alone encodes approximately 5,000 integral membrane proteins containing roughly 20,000 TMDs that vary widely in sequence, biophysical properties, and topological arrangement [25]. This diversity necessitates multiple targeting and insertion pathways tailored for different substrate classes. This review systematically compares the rules governing TMD orientation and integration, synthesizing recent structural insights with biochemical evidence to provide a comprehensive framework for understanding how sequence features dictate membrane protein topology.
Core Topogenesis Rules and Determinants
Primary Sequence Determinants
Membrane protein topology is governed by topogenic signals encoded within the amino acid sequence that are interpreted by the cellular machinery during membrane insertion.
Table 1: Primary Sequence Determinants of TMD Topology
| Determinant | Mechanistic Basis | Experimental Support |
|---|---|---|
| Hydrophobicity | Primary driving force for membrane integration; total hydrophobicity and position-dependent effects influence insertion efficiency | Biological free-energy scales predict insertion probability; tryptophan/tyrosine position effects [26] |
| Positive-Inside Rule | Enrichment of positively charged residues (Lys, Arg) in cytoplasmic flanking regions; positive charges are 4x more abundant on cytoplasmic side | Statistical analysis of membrane proteomes; mutagenesis studies [27] |
| Charge Difference Rule | Net electrical charge difference between flanking regions (positive charges minus negative charges) determines orientation | Engineering of inverted topologies in yeast and E. coli by charge manipulation [27] |
| Hydrophobicity Gradient | The most hydrophobic terminus of a TMD is preferentially translocated across the membrane | N-terminus translocation favored by long hydrophobic sequences; C-terminus by short sequences [26] |
The Role of Membrane Lipid Composition
Beyond sequence features, membrane lipid composition critically influences topological outcomes through the Charge Balance Rule, an extension of the Positive-Inside Rule. The net zero-charged phospholipid phosphatidylethanolamine dampens the translocation potential of negatively charged residues while enhancing the cytoplasmic retention potential of positively charged residues [28] [27]. This explains the dominance of positive residues as topological signals under physiological conditions and enables post-insertional topological reversibility in response to changes in lipid composition, a phenomenon demonstrated both in vivo and in synthetic liposomes [28] [27].
Comparative Analysis of Translocon Systems
Translocon Machinery and Accessory Complexes
The Sec61/SecY translocon serves as the central protein-conducting channel for membrane protein insertion, with accessory complexes dynamically recruited based on substrate requirements.
Recent transcriptome-wide selective ribosome profiling reveals that translocon composition changes repeatedly and reversibly during synthesis of topologically complex multipass membrane proteins [29]. The OST-A complex (catalyzing N-glycosylation) is preferentially recruited to open Sec61 channels engaged in polypeptide translocation, while the multipass translocon (MPT) complexes (GEL, PAT, and BOS) are recruited synchronously to closed Sec61 channels stabilized by newly inserted TMDs [29].
Quantitative Analysis of Translocon Client Interactions
Table 2: Translocon Accessory Complex Client Specificity and Recruitment Dynamics
| Complex | Primary Function | Enriched Client Classes | Enrichment Frequency | Recruitment Trigger |
|---|---|---|---|---|
| OST-A | N-glycosylation of luminal domains | SP-only proteins, Type I/II single-pass | 97%, 96%, 92% respectively | Translocation of long segments (>90 residues post-SP) |
| MPT (GEL/PAT/BOS) | Multipass membrane protein biogenesis | Type I/II/III multipass proteins | 83%, 87%, 98% respectively | Emergence of first TMD or TMD pair from ribosome |
| Sec61 Core | Protein-conducting channel | All membrane and secretory proteins | Universal | Signal sequence emergence from ribosome |
The recruitment dynamics of these complexes are precisely coordinated. OST-A engagement begins when approximately 90 residues have been synthesized beyond the signal peptide, corresponding to ~35 residues entering the ER lumen—sufficient length to sample the STT3A active site for N-glycosylation [29]. For multipass proteins, MPT recruitment is progressively stabilized following emergence of the first TMD or closely spaced TMD pair from the ribosome [29].
Experimental Approaches for Topogenesis Analysis
Methodologies for Determining Membrane Protein Topology
Several experimental strategies have been developed to determine TMD orientation and integration efficiency:
4.1.1 Fusion Partner Systems for N-terminal Orientation A modular system utilizing bacteriorhodopsin mutants (D94N) enables determination of N-terminal orientation in E. coli. D94N with cytoplasmic C-terminus (Cin) enhances expression of Nin targets, while fusion with a designed 3-TM linker (D94N-3TM) creates a Cout orientation suitable for Nout targets [30]. The compatibility between fusion partner and target protein orientation determines expression efficiency, allowing topological assessment without mutagenesis or chemical labeling.
4.1.2 Reporter Enzyme Assays C-terminal fusions with β-lactamase (BlaM) or green fluorescent protein (GFP) enable determination of C-terminal orientation. BlaM confers ampicillin resistance only when translocated to the periplasm, providing selective pressure for topological assessment [30]. These assays confirmed the membrane integration and orientation of computationally designed TM bundles, demonstrating their utility for validating engineered membrane proteins [30].
4.1.3 Modular Surface Display Systems The SpyCatcher/SpyTag bioconjugation system enables modular combination of passengers with different membrane anchors (Lpp-OmpA, PgsA, INP, AIDA-I) without constructing direct genetic fusions [19]. This approach allows rapid comparison of anchor performance, revealing significant impacts on cell growth, membrane integrity, and display capacity—critical factors for biotechnology applications [19].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for Topogenesis Studies
| Reagent/System | Function/Application | Key Features |
|---|---|---|
| D94N-3TM Fusion System | Determining N-terminal orientation of TM proteins | Compatible with both Nin and Nout targets; requires no mutagenesis [30] |
| β-lactamase (BlaM) Reporter | C-terminal topology determination | Confers ampicillin resistance when periplasmic; enables selective screening [30] |
| SpyCatcher/SpyTag System | Modular membrane protein display | Covalent isopeptide bond; enables rapid anchor comparison [19] |
| SecYEG Proteoliposomes | In vitro translocation assays | Reconstituted minimal system for studying insertion mechanisms [31] |
| Selective Ribosome Profiling | Global analysis of translocon interactions | Identifies native clients and binding sites for translocon components [29] |
Comparative Analysis of Targeting Pathways
Membrane protein targeting occurs through distinct pathways specialized for different substrate classes:
The signal recognition particle (SRP) pathway mediates co-translational targeting for proteins with TMDs located at least 65 amino acids from the C-terminus [25]. Tail-anchored (TA) proteins with TMDs closer than 65 residues to the C-terminus utilize post-translational pathways: the GET pathway for high-hydrophobicity TMDs and the ER membrane complex (EMC) for low-hydrophobicity TMDs [25]. An additional SRP-independent (SND) pathway handles substrates whose first TMD is far from the N-terminus but not tail-anchored [25].
Structural Insights into Translocon Mechanism
Recent cryo-EM structures of SecY translocon complexes with TM substrates reveal detailed mechanisms of membrane protein insertion. Structures of SecY with FtsQ-LacY substrates illustrate type II TM insertion, where the N-terminal hydrophobic residues are exposed to lipids outside the lateral gate while the C-terminal segment is clamped by TM3 and TM7 of SecY [31]. These structural insights demonstrate how the translocon acts as a chaperone during membrane protein insertion, facilitating the transition of TMDs from the aqueous channel environment into the lipid bilayer [31].
The structural data support a model where TMD insertion occurs through a hydrophobic gate consisting of SecY transmembrane segments 2b, 3, and 7, which rearrange to facilitate TMD movement into the membrane. This gate mechanism accommodates the diverse biophysical properties of natural TMDs while maintaining membrane integrity during protein insertion [31].
Computational and Engineering Approaches
Deep Learning for Membrane Protein Design
Recent advances in deep learning have enabled the computational design of soluble analogues of integral membrane protein folds. A pipeline combining AlphaFold2 inversion with ProteinMPNN sequence optimization successfully designed soluble versions of complex membrane protein topologies including claudin, rhomboid protease, and GPCR folds [32]. This approach demonstrates that membrane protein topologies can be recapitulated in soluble form, potentially enabling new approaches in drug discovery by bringing membrane protein functions to the soluble proteome [32].
Experimental Validation of Designed Membrane Proteins
Computationally designed membrane proteins can be experimentally validated using the methodologies described in Section 4. For example, a designed 4-helical bundle membrane protein (4TM) derived from cytochrome b562 was confirmed to integrate into E. coli membranes with the predicted topology using BlaM and GFP fusion assays [30]. The successful expression and membrane integration of such designed proteins demonstrates our growing understanding of the rules governing membrane protein topogenesis.
The establishment of membrane protein topology emerges from a complex interplay of sequence-encoded determinants and their interpretation by cellular machinery. The hydrophobicity of TMDs provides the primary driving force for membrane integration, while charged residues in flanking regions determine orientation according to the Positive-Inside Rule and Charge Difference Rule. These sequence features are interpreted initially by the translocon machinery, with final topological outcomes refined by the membrane lipid composition through the Charge Balance Rule.
Recent structural and biochemical advances reveal an unexpected dynamicity in topogenesis, with TMDs capable of reorientation during and after insertion, and translocon composition remodeling repeatedly during synthesis of complex membrane proteins. This integrated understanding enables more accurate prediction of membrane protein topology and provides novel approaches for engineering membrane proteins with customized topological features for basic research and therapeutic applications. As deep learning methods advance our ability to design membrane protein analogues, the fundamental rules governing topogenesis will continue to provide essential guidance for membrane protein engineering and design.
Techniques for Studying and Exploiting Membrane Protein Biogenesis Pathways
The structural elucidation of membrane protein insertion intermediates is pivotal for understanding fundamental biological processes, including protein biogenesis, trafficking, and function. For decades, visualizing these transient states at high resolution posed a significant challenge due to their dynamic nature and the complexities of membrane environments. The resolution revolution in cryo-electron microscopy (cryo-EM) has transformed this landscape, providing structural biologists with an expanding toolkit to capture these elusive complexes. This guide objectively compares two powerful cryo-EM modalities—single-particle analysis (SPA) and microcrystal electron diffraction (MicroED)—for studying membrane insertion intermediates, providing researchers with a framework for selecting the optimal technique based on their scientific questions and sample characteristics.
Technical Comparison: Cryo-EM SPA vs. MicroED
Cryo-EM encompasses several distinct techniques. Single-particle analysis (SPA) images individual macromolecules frozen in vitreous ice, followed by computational reconstruction into 3D structures [33] [34]. In contrast, microcrystal electron diffraction (MicroED) is a crystallographic method that collects diffraction patterns from nanometer-sized 3D crystals to determine atomic structures [33] [35] [36]. While both are performed using a transmission electron microscope (TEM) under cryogenic conditions, their underlying principles, sample requirements, and data processing workflows differ substantially.
Table 1: Core Technical Specifications and Applications
| Feature | Cryo-EM SPA | MicroED |
|---|---|---|
| Core Principle | Imaging of individual particles [33] | Diffraction from 3D crystals [33] [37] |
| Typical Sample State | Purified complexes in solution [34] | Sub-micron 3D crystals [33] [36] |
| Ideal Crystal Size | Not applicable | ~200 nm - 1 μm thick [37] [36] |
| Membrane Mimetics Used | Nanodiscs, Amphipols, Detergents [34] [37] | Lipidic Cubic Phase (LCP), Bicelles [37] [38] |
| Key Advantage for Membrane Proteins | Studies non-crystalline samples and large complexes [34] [39] | High-resolution from nanocrystals; details protonation states [35] [40] |
Table 2: Performance and Data Output Metrics
| Metric | Cryo-EM SPA | MicroED |
|---|---|---|
| Best Resolution (General) | Atomic (≤ 1.2 Å) [34] | Sub-atomic (≤ 1.0 Å) [41] [40] |
| Typical Resolution (Membrane Proteins) | ~3 Å [34] [39] | ~2 - 3 Å [37] [38] |
| Data Collection Temperature | Cryogenic (≈ 100 K) [33] [34] | Cryogenic (≈ 100 K) [33] [36] |
| Sample Consumption | Low (μL of μM concentration) [34] | Very low (nanogram quantities) [35] [36] |
| Data Collection Time | Minutes to hours per dataset [34] | ~5-10 minutes per crystal [36] |
Experimental Protocols for Membrane Protein Studies
Sample Preparation and Membrane Mimetics
Successful structural biology of membrane proteins hinges on extracting them from native membranes and stabilizing them in a functional state using membrane mimetics.
- For Cryo-EM SPA: Detergents like digitonin are commonly used for extraction, with a trend toward gentler alternatives to preserve complex integrity [34]. Subsequently, samples are often reconstituted into nanodiscs (membrane scaffold protein belts encircling a lipid bilayer) or amphipols (polymers that wrap around transmembrane domains) to create a more native-like environment ideal for SPA [34] [37]. The sample is then applied to a grid, blotted, and vitrified.
- For MicroED: Crystallization is required. The Lipidic Cubic Phase (LCP) method has been extremely successful for membrane proteins like GPCRs [37] [38]. This method involves creating a 3D lipid matrix that mimics the native membrane environment, promoting crystal growth with protein surrounded by lipid [37]. Bicelles (aqueous lipid-detergent assemblies) are another successful mimetic for crystallization [37].
Specialized Workflow for MicroED of LCP-Embedded Crystals
A robust protocol for challenging membrane protein targets, such as GPCRs, involves correlated light and ion-beam microscopy [38].
- Crystallization and Fluorescent Labeling: Crystallize the target membrane protein in LCP. The protein can be fluorescently tagged for identification.
- Grid Preparation and Vitrification: Apply the LCP crystal mixture to an EM grid and vitrify by plunging into liquid ethane.
- Crystal Identification via iFLM: Transfer the grid to a plasma Focused Ion Beam-Scanning Electron Microscope (pFIB-SEM) with an integrated fluorescence light microscope (iFLM). Use fluorescence to locate crystals embedded within the opaque LCP [38].
- Plasma FIB Milling: Use a plasma FIB (with Xenon or Argon ions) to mill the LCP surrounding the targeted crystal, creating a thin, electron-transparent lamella (optimal thickness ~300 nm) for MicroED data collection [38].
- Data Collection: Collect continuous rotation MicroED data on the lamella using an ultra-low dose electron beam (< 1 e⁻/Ų/s) and a direct electron detector in counting mode [37] [41] [38].
Data Processing and Structure Determination
- Cryo-EM SPA: Processing involves motion correction, contrast transfer function (CTF) estimation, particle picking, 2D classification, 3D initial model generation, and high-resolution refinement to produce a final electron density map for model building [34].
- MicroED: The continuous rotation data (collected as a movie) is processed using established X-ray crystallographic software such as XDS or DIALS [33] [35] [36]. This includes indexing, integration, and scaling of diffraction intensities. Phasing can be achieved by molecular replacement or, as recently demonstrated, by ab initio methods using direct electron detectors [40].
Workflow Visualization
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials and Reagents for Structural Studies of Membrane Proteins
| Item | Function / Application | Technique |
|---|---|---|
| Membrane Scaffold Protein (MSP) | Forms protein-lipid nanodiscs to stabilize membrane proteins in a native-like lipid environment [34] [37]. | Cryo-EM SPA |
| Lipidic Cubic Phase (LCP) Lipids | Creates a membrane-mimetic matrix for crystallizing membrane proteins [37] [38]. | MicroED |
| Amphipols | Synthetic polymers that stabilize detergent-solubilized membrane proteins in solution, replacing detergent molecules [34] [37]. | Cryo-EM SPA |
| Digitonin | A gentle detergent used for extracting and solubilizing membrane proteins while preserving complex integrity [34]. | Both |
| Cryo-EM Grids | Supports (e.g., holy carbon grids) for applying and vitrifying the sample for cryo-TEM analysis. | Both |
| Plasma FIB (Xe/Ar) | Uses xenon or argon plasma ions for precise, low-damage milling of vitrified samples to optimal thickness [38]. | MicroED |
| Direct Electron Detector | High-sensitivity camera for recording high-resolution images or diffraction patterns with minimal noise [41] [40]. | Both |
Both Cryo-EM SPA and MicroED are powerful techniques for visualizing membrane protein insertion intermediates, each with distinct strengths. SPA excels in studying large, heterogeneous complexes in a near-native lipid environment without the need for crystallization. MicroED complements this by achieving exceptionally high resolution from vanishingly small crystals, often revealing fine chemical details like ion binding and hydrogen networks [35]. The choice between them hinges on project goals: SPA is ideal for capturing conformational states of large complexes, while MicroED is superior for obtaining atomic-level detail from nanocrystals, with emerging methods like pFIB milling making it increasingly applicable to challenging membrane targets [38].
The biogenesis of membrane proteins—the process by which these vital proteins are targeted to, inserted into, and folded within lipid bilayers—is a fundamental biological process. Roughly one-fourth of all genes in an organism code for integral membrane proteins, which perform crucial functions including ion and nutrient transport, signal transduction, and cellular adhesion [25]. In vitro reconstitution of these processes is essential for deepening our understanding of membrane biology and for facilitating drug discovery, given that membrane proteins constitute approximately 60% of all drug targets [42]. Traditional methods using detergent micelles often fail to provide a native-like lipid environment, which can lead to protein denaturation and loss of function [43] [42].
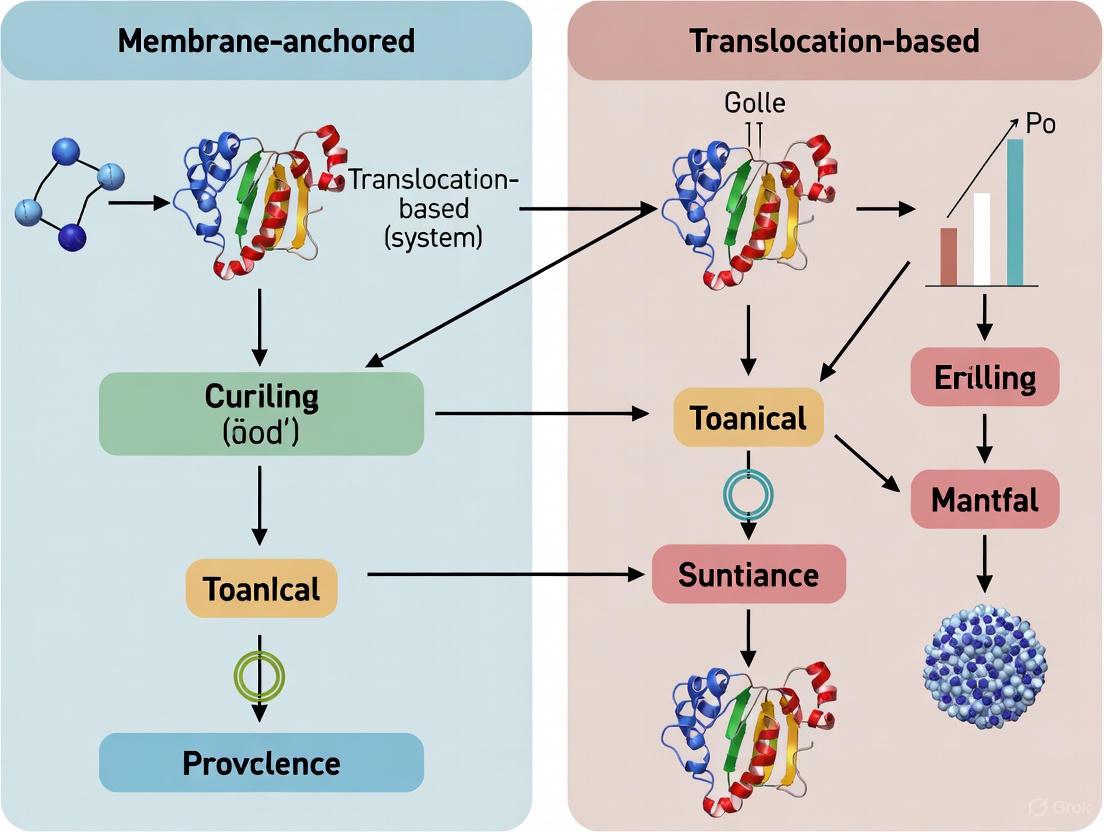
This guide compares two leading approaches for creating in vitro insertion assays: membrane-anchored systems, which use nanodiscs to provide a stable, native-like lipid bilayer patch, and translocation-based systems, which employ engineered, spontaneously inserting proteins within synthetic membranes. The emergence of novel nanodisc technologies, particularly detergent-free methods, and the creative repurposing of bacterial toxins for synthetic biology, are pushing the boundaries of what is possible in membrane protein research.
Technology Platform Comparison
The core of modern in vitro insertion assays lies in the membrane mimetic used. The table below provides a high-level comparison of the primary platforms.
Table 1: Comparison of Membrane Mimetic Platforms for In Vitro Insertion Assays
| Platform | Key Constituents | Key Advantage | Ideal for Assay Type | Typical Size Range |
|---|---|---|---|---|
| MSP Nanodiscs [43] | Phospholipids, Membrane Scaffold Protein (MSP) | Controlled size and monodispersity; well-established | Ligand-binding studies, oligomeric state control, functional characterization of stabilized proteins [44] | 7 - 17 nm [43] |
| Polymer Nanodiscs (e.g., SMALPs) [42] | Phospholipids, Styrene Maleic Acid (SMA) copolymer | Direct extraction from native membranes; no detergent needed [42] | Studying proteins in near-native lipid environment; detergent-sensitive complexes [42] | Poly-disperse, but tunable [42] |
| Peptide-Based Nanodiscs (DeFrND) [45] | Phospholipids, Engineered Amphipathic Peptides (DeFrMSPs) | Detergent-free reconstitution; preserves function of sensitive complexes [45] | Functional studies of transporters and receptors that are unstable in detergents [45] | 10 - 20 nm [45] |
| Synthetic Membranes with Engineered Inserters [46] | Lipid Vesicles, Engineered Pore-Forming Toxins (e.g., α-hemolysin) | Genetically encodable; enables external display from within artificial cells [46] | Building communicative artificial cells; synthetic tissue engineering [46] | N/A (Vesicle size variable) |
The Nanodisc Toolkit: Membrane-Anchored Systems
Nanodiscs are self-assembled, nanosized, disc-shaped phospholipid bilayer structures stabilized by a surrounding scaffold [43]. This scaffold defines the type of nanodisc and its application.
- MSP Nanodiscs: The classic system uses Membrane Scaffold Proteins (MSPs), derived from apolipoprotein A-I, which wrap around the lipid bilayer to form a soluble disc. The assembly is triggered by the removal of detergent from a mixture of lipids, MSP, and the target membrane protein [43] [44]. A key advantage is the ability to control the oligomeric state of the membrane protein by selecting the nanodisc size [43].
- Polymer-Based Nanodiscs: Using amphipathic polymers like Styrene Maleic Acid (SMA), these nanodiscs can directly solubilize membrane proteins and their surrounding lipids from native cell membranes without the use of detergents, forming SMALPs [42]. This preserves a more native lipid environment.
- Peptide-Based Nanodiscs (DeFrND): A cutting-edge 2025 development, the DeFrND system uses engineered membrane-scaffolding peptides (DeFrMSPs) for detergent-free reconstitution [45]. These peptides, such as the fatty acid-modified "18A" peptide, can directly transform proteoliposomes into nanodiscs, maintaining the functional integrity of complexes like the MalFGK2 transporter, which is often inactivated by detergents or polymers [45].
Synthetic Membranes & Engineered Inserters: Translocation-Based Systems
For building from the ground up, synthetic membranes like Giant Unilamellar Vesicles (GUVs) serve as minimal artificial cells. A major challenge has been functionalizing these membranes with proteins from the inside. A novel solution involves engineered self-translocating proteins.
- Engineered α-hemolysin (αHL): The bacterial toxin α-hemolysin naturally forms a β-barrel pore that spontaneously inserts into membranes. Researchers have engineered its extracellular loop (amino acids 128-131) to translocate functional peptides of up to 52 amino acids across the lipid bilayer [46]. This system allows proteins expressed inside an artificial cell to be displayed on the exterior, enabling programmed interactions like the formation of tissue-like structures [46].
Experimental Data and Performance Comparison
Functional Performance in Biophysical Assays
Different mimetics can profoundly impact the activity and stability of a membrane protein. The following table summarizes experimental data from recent studies.
Table 2: Experimental Performance Data of Membrane Mimetics in Functional Studies
| Mimetic System | Membrane Protein Studied | Key Experimental Readout | Performance Outcome |
|---|---|---|---|
| Peptide-Based (DeFrND) [45] | Bacterial ABC transporter MalFGK2 | ATPase activity coupled to maltose and MBP | Preserved functional coupling, unlike the uncoupled, hyper-active state in detergents or inactive state in polymer nanodiscs [45] |
| MSP Nanodiscs [44] | GPCR Neurotensin Receptor 1 (NTS1) | Ligand binding & signaling properties via biophysical assays | Enabled study of receptor in a defined monomeric or oligomeric state; lipid charge was shown to influence signaling [44] |
| Synthetic Membranes with Engineered αHL [46] | Various peptide-tagged αHL mutants | Membrane binding, pore formation (dye leakage), and external peptide display | Mutants with inserts up to 52 amino acids retained full pore-forming functionality and enabled specific external interactions [46] |
| Polymer Nanodiscs (SMALPs) [42] | Various (e.g., Bacteriorhodopsin, GPCRs) | Stability and monodispersity via Fluorescence Correlation Spectroscopy (FCS) | Provided excellent thermal and temporal stability, keeping proteins monodisperse and functional for over a week at 4°C [42] |
Workflow and Experimental Protocols
A critical difference between these systems lies in their experimental workflows. The diagrams below outline the core processes for detergent-free nanodisc reconstitution and synthetic membrane functionalization.
Detergent-Free Native Nanodisc Reconstitution (DeFrND)
Diagram 1: Workflow for detergent-free nanodisc reconstitution using DeFrMSP peptides. This method bypasses the need for initial detergent solubilization, preserving the functional state of delicate membrane protein complexes [45].
Synthetic Membrane Functionalization via Engineered Translocon
Diagram 2: Workflow for functionalizing synthetic membranes from within using engineered α-hemolysin. This genetically encodable system allows artificial cells to self-display interaction motifs, enabling the formation of higher-order structures [46].
The Scientist's Toolkit: Essential Research Reagents
Success in reconstituting biogenesis relies on a specific set of reagents and tools. The following table details key solutions for researchers in this field.
Table 3: Key Research Reagent Solutions for In Vitro Insertion Assays
| Reagent / Material | Function in Assay | Examples & Notes |
|---|---|---|
| Membrane Scaffold Pro/Peptides | Forms the stabilizing belt around the lipid bilayer in nanodiscs. | MSP1D1: Classic ApoA-I derived protein [42]. DeFrMSPs: Engineered peptides (e.g., 18A) for detergent-free reconstitution [45]. |
| Amphipathic Polymers | Directly solubilizes native membrane patches to form polymer nanodiscs. | SMA (Styrene Maleic Acid): Original polymer, sensitive to divalent cations. DIBMA: Di-isobutylene Maleic Acid, milder alternative [42]. |
| Engineered Pore-Forming Toxins | Enables genetically encoded protein translocation across synthetic membranes. | α-hemolysin (αHL) mutants: Engineered in loop 128-131 to display functional peptides (e.g., 6XHis, GLP1) externally [46]. |
| Cell-Free Protein Expression System | Produces membrane proteins or scaffold components directly for in vitro assembly. | Used for co-translational incorporation of membrane proteins into nanodiscs, bypassing detergent purification [42]. |
| Lipid Mixtures | Forms the foundational bilayer of nanodiscs or synthetic vesicles. | Can be tailored with specific phospholipids, cholesterol, or labeled lipids to mimic native membranes or enable detection [42]. |
The choice between membrane-anchored nanodisc systems and translocation-based synthetic membranes is not a matter of which is universally better, but which is most appropriate for the specific biological question.
For fundamental biophysical and biochemical characterization of membrane proteins—such as determining structural details, studying lipid-protein interactions, or conducting high-throughput ligand screening—nanodiscs offer a superior, controlled environment. The recent advent of detergent-free peptide nanodiscs (DeFrND) is a significant leap forward, finally allowing the functional study of membrane protein complexes that are too fragile for traditional detergent-based methods [45].
Conversely, for synthetic biology and bottom-up construction of cellular mimicry—such as engineering communicative artificial cells or programming self-assembling tissue scaffolds—the translocation-based system using engineered proteins like αHL is unparalleled. Its genetic encodability and ability to direct external display from within a compartment align with the goals of creating life-like systems from simple components [46].
In conclusion, the ongoing development of both membrane-anchored and translocation-based systems is driving a more nuanced and powerful paradigm in membrane research. By leveraging the strengths of each platform, researchers can now design more physiologically relevant in vitro insertion assays, thereby accelerating our understanding of membrane protein biogenesis and its applications in therapeutic development.
The Sec61 complex, known as SecYEG in prokaryotes, is the central protein-conducting channel in the endoplasmic reticulum (ER) membrane of eukaryotic cells and the plasma membrane of prokaryotes [47]. This heterotrimeric complex serves as the essential gateway for the biogenesis of approximately 25-30% of the proteome, including secreted proteins and integral membrane proteins with complex topologies [25] [47]. The translocon facilitates both co-translational translocation (where proteins enter the channel during their synthesis) and post-translational translocation (where fully synthesized proteins are threaded through the channel) [47]. Given its fundamental role in protein secretion and membrane integration, the Sec61 translocon has emerged as a compelling target for therapeutic intervention in diseases ranging from cancer to autoimmune disorders and viral pathogenesis [48].
Several structurally diverse small molecules have been identified that specifically inhibit Sec61 function through distinct yet overlapping mechanisms. Among these, mycolactone, cotransin (and related compounds), and eeyarestatin I (ESI) represent powerful pharmacological tools for probing translocon function and developing novel therapeutics. These inhibitors exhibit varying degrees of selectivity, potency, and mechanism of action, making them valuable for both basic research and clinical applications. This review provides a comprehensive comparison of these inhibitors, focusing on their molecular mechanisms, experimental applications, and potential therapeutic utility within the broader context of membrane protein biogenesis research.
Comparative Analysis of Sec61 Inhibitors
The following table summarizes the key characteristics of mycolactone, cotransin, and eeyarestatin I, highlighting their origins, mechanisms, and research applications.
Table 1: Comparative Profile of Sec61 Translocon Inhibitors
| Inhibitor | Source/Origin | Primary Molecular Target | Inhibitory Mechanism | Cellular IC₅₀ | Key Research Applications |
|---|---|---|---|---|---|
| Mycolactone | Mycobacterium ulcerans | Sec61 complex [49] | Binds lateral gate, stabilizes closed plug, inhibits co-translational translocation [48] [50] | Nanomolar range [49] | Studying ISR activation, immune suppression, Buruli ulcer pathogenesis [49] |
| Cotransins | Fungal cyclic heptadepsipeptides [48] | Sec61 complex [48] | Client-selective inhibition via lateral gate binding [48] [50] | Varies by client protein [48] | Substrate-specific translocation studies, selective protein downregulation [48] |
| Eeyarestatin I (ESI) | Synthetic compound [48] | Sec61 complex [48] | Binds lateral gate, stabilizes closed plug [48] [50] | Micromolar range [48] | Probing ER-associated degradation (ERAD), substrate-agnostic translocation inhibition [48] |
Table 2: Functional Consequences of Sec61 Inhibition
| Cellular Process | Mycolactone Effect | Cotransin Effect | ESI Effect |
|---|---|---|---|
| Secretory Protein Production | Broad inhibition [49] | Selective inhibition based on signal sequence [48] | Broad inhibition [48] |
| Membrane Protein Integration | Inhibits most Type I and Type II TMPs [49] | Client-dependent inhibition | Broad inhibition |
| ER Stress Sensor Activation | Activates PERK only (uncoupled ISR) [49] | Not well characterized | Not well characterized |
| Integrated Stress Response | Strong induction of p-eIF2α, ATF4, CHOP [49] | Minimal data available | Minimal data available |
| Therapeutic Potential | Immunosuppression, anti-inflammatory [49] | Cancer, autoimmune diseases [48] | Cancer, viral infection [48] |
Structural Mechanisms of Translocon Inhibition
Recent structural biology advances have revolutionized our understanding of how small molecules inhibit the Sec61 translocon. Cryo-electron microscopy studies from 2023 reveal that despite their chemical diversity, mycolactone, cotransin, ESI, and other inhibitors share a common binding site on the Sec61 complex [48] [50].
Common Binding Pocket and Molecular Interactions
All analyzed Sec61 inhibitors bind to a lipid-exposed pocket formed by the partially open lateral gate and the plug domain of Sec61α [50]. This pocket is situated approximately halfway across the membrane and is composed of transmembrane helices TM2b, TM3, and TM7 of the Sec61α subunit [50]. The inhibitors stabilize the plug domain in a closed state, thereby preventing the conformational changes necessary for protein translocation pore opening [50].
The binding of these inhibitors to the lateral gate region physically obstructs the lateral exit of transmembrane domains (TMDs) into the lipid bilayer, explaining their potent inhibition of membrane protein biogenesis [48]. Mutations that confer resistance to these inhibitors are clustered precisely at this binding pocket, providing genetic evidence for its functional importance [48].
Distinct Structural Footprints and Mechanism Variations
While sharing the same general binding region, each inhibitor exhibits distinct interactions within the pocket:
- Mycolactone and cotransin compete for overlapping binding sites, suggesting similar but non-identical binding modes [49] [48]
- Cotransin exhibits client-selective inhibition, likely through subtle differences in how it stabilizes the closed conformation that preferentially affects substrates with specific signal sequence properties [48]
- Despite common mechanisms, each inhibitor induces slightly different degrees of lateral gate opening, explaining their varying potency and client selectivity [50]
The following diagram illustrates the common structural mechanism of Sec61 inhibition by these small molecules:
Figure 1: Common structural mechanism of Sec61 inhibition. Small molecules bind the lateral gate and plug domain, stabilizing the channel in a closed state and preventing protein translocation.
Mycolactone: A Unique Probe for Uncoupling Integrated Stress Response from ER Stress
Mycolactone, the primary virulence factor of Mycobacterium ulcerans, has emerged as a particularly valuable tool for understanding the complex relationship between Sec61 inhibition and cellular stress responses.
Experimental Evidence and Key Findings
Research using translational profiling in macrophages exposed to mycolactone revealed that this inhibitor activates an integrated stress response (ISR) without concurrent ER stress [49]. This uncoupled response features:
- Phosphorylation of eIF2α via multiple kinases (PERK, GCN, PKR) without activation of the ER stress sensors IRE1 or ATF6 [49]
- Transcriptional induction of ATF4 and CHOP, key regulators of stress-induced apoptosis [49]
- Translation attenuation followed by chronic activation of autophagy and apoptosis pathways [49]
The specificity of this response to Sec61 inhibition was confirmed through experiments showing that cells expressing mycolactone-resistant Sec61α variants failed to activate this pathway [49].
Methodological Approach for Studying Mycolactone Effects
Researchers employing mycolactone as an experimental tool should consider these established protocols:
- Cell treatment: Typically 100-500 nM mycolactone for 2-8 hours, depending on cell type and sensitivity [49]
- Translational profiling: Polysome fractionation followed by microarray or RNA-seq analysis of polysomal vs. sub-polysomal transcripts [49]
- Validation assays: Western blotting for p-eIF2α, ATF4, CHOP; qPCR for stress response genes; XBP-1 splicing assays to exclude IRE1 activation [49]
The following diagram illustrates the unique signaling pathway activated by mycolactone-mediated Sec61 inhibition:
Figure 2: Mycolactone activates an integrated stress response uncoupled from ER stress. This unique signaling pathway makes it a valuable tool for studying stress response mechanisms.
Experimental Toolkit for Sec61 Research
Essential Research Reagents and Materials
Table 3: Key Reagents for Studying Sec61 Inhibition
| Reagent/Material | Function/Application | Example Sources |
|---|---|---|
| Mycolactone | Broad-spectrum Sec61 inhibition, ISR studies | Purified from M. ulcerans or synthetic |
| Cotransin/CAM741 | Client-selective translocation inhibition | Commercial suppliers, custom synthesis |
| Eeyarestatin I (ESI) | Substrate-agnostic Sec61 inhibition | Commercial chemical suppliers |
| Sec61α Mutants | Resistance validation, mechanism studies | CRISPR-edited cell lines |
| Antibodies for p-eIF2α, ATF4, CHOP | ISR pathway validation | Multiple commercial providers |
| XBP-1 Splicing Assay Reagents | IRE1 activation monitoring | Commercial kits available |
| Polysome Profiling Components | Translational efficiency analysis | Sucrose gradients, fractionation systems |
Methodological Considerations for Sec61 Studies
When designing experiments with Sec61 inhibitors, several methodological approaches yield robust results:
- Translocation assays: Monitor processing of model substrates like preprolactin or VSV-G protein [49] [48]
- Membrane protein integration assays: Use glycosylation reporters or protease protection assays for topology determination [25]
- Structural approaches: Cryo-EM of inhibitor-bound Sec61 complexes for mechanistic insights [48] [50]
- Functional genomics: CRISPR screens to identify resistance mutations and synthetic lethal interactions [48]
Research Applications and Therapeutic Implications
Basic Research Applications
These Sec61 inhibitors serve as powerful tools for addressing fundamental questions in cell biology:
- Protein trafficking mechanisms: Determining signal sequence requirements and translocation kinetics [48]
- Membrane protein biogenesis: Elucidating pathways for different classes of integral membrane proteins [25]
- Cellular stress responses: Decoupling ER stress from integrated stress response pathways [49]
- Organelle communication: Understanding how ER priming events trigger responses in other cellular compartments [49]
Therapeutic Development and Clinical Potential
The therapeutic applications of Sec61 inhibitors stem from their ability to selectively modulate protein secretion and membrane expression:
- Oncology: Downregulation of oncogenic receptors, growth factors, and immunomodulatory proteins [48]
- Autoimmune diseases: Suppression of cytokine and adhesion molecule production [49] [48]
- Viral infections: Inhibition of viral envelope protein biogenesis and host factor dependency [48]
- Inflammatory disorders: Selective modulation of inflammatory mediator secretion [48]
Notably, one Sec61 inhibitor has entered phase I clinical trials for solid tumor malignancies, demonstrating the translational potential of this target class [48].
Mycolactone, cotransins, and ESI represent complementary tools for probing Sec61 translocon function and developing novel therapeutics. While they share a common binding pocket and fundamental inhibitory mechanism, their distinct properties make each uniquely valuable for specific research and clinical applications. Mycolactone's unique ability to uncouple integrated stress response from ER stress provides particular insight into cellular stress signaling, while cotransins offer remarkable substrate selectivity for precision interventions.
Future research directions should include developing next-generation inhibitors with improved selectivity profiles, exploring combination therapies that leverage Sec61 inhibition with other treatment modalities, and further elucidating the structural basis for client-specific inhibition. As our understanding of Sec61 biology deepens, these inhibitors will continue to serve as essential tools for both basic research and therapeutic development in the rapidly advancing field of membrane protein biogenesis.
The study of membrane protein structures is fundamental to understanding cellular communication, signal transduction, and metabolic transport, with membrane proteins constituting approximately 60% of all pharmaceutical drug targets [20]. Despite their biological and therapeutic significance, membrane proteins represent less than 1% of structures in the Protein Data Bank, primarily due to challenges in achieving high-yield functional expression [51] [52]. The inherent hydrophobicity of membrane proteins often causes misfolding, aggregation, and cellular toxicity when overexpressed in heterologous systems, creating a critical bottleneck for structural biology [53]. This guide provides a comprehensive comparison of expression strategies, focusing on the optimization of membrane-anchored versus translocation-based systems to overcome these barriers and enable successful high-resolution structural studies.
Membrane Protein Expression Challenges and System Selection
Fundamental Obstacles in Membrane Protein Production
Membrane protein production presents unique challenges distinct from those of soluble proteins. The primary difficulties include:
- Cellular Toxicity and Stress: Overexpression of membrane proteins can disrupt the host cell's membrane integrity, inhibit growth, and trigger stress responses, ultimately leading to cell death [53].
- Targeting and Insertion Complexity: Membrane proteins require sophisticated cellular machinery for correct targeting, translocation, and insertion into the appropriate membrane, processes that can be overwhelmed during overexpression [20] [53].
- Solubilization and Stability: Extracting proteins from their native lipid environment requires detergents that can destabilize their native conformation, leading to aggregation or loss of function [51] [52]. Maintaining stability throughout purification often necessitates extensive screening of detergents and lipids.
Selecting an Appropriate Expression System
Choosing the right expression host is the first critical step toward success. The decision should be guided by the protein's origin, complexity, and post-translational modification requirements.
Table 1: Comparison of Major Expression Systems for Membrane Proteins
| Expression System | Typical Yield | Folding Capacity | Post-Translational Modifications | Cost & Speed | Best Suited For |
|---|---|---|---|---|---|
| E. coli | Variable (High for some) | Moderate | None (prokaryotic) | Low / Fast | Prokaryotic proteins; Simple eukaryotic proteins without PTMs [54] |
| Yeast | Moderate to High | Good | Core glycosylation (high-mannose) | Low-Moderate / Moderate | Eukaryotic proteins requiring basic glycosylation [54] |
| Insect Cells (Baculovirus) | Moderate | Very Good | Paucimannose-type N-glycans | Moderate / Slow | Complex eukaryotic proteins, including many GPCRs and ion channels [54] [52] |
| Mammalian Cells | Low to Moderate | Excellent | Complex, human-like glycosylation | High / Very Slow | Highly complex eukaryotic proteins requiring precise human-like PTMs [54] |
The following decision pathway can help researchers select the optimal expression system:
Comparative Analysis of Membrane Anchoring and Translocation Systems
Membrane-Anchored Fusion Systems
Membrane-anchored systems utilize fusion partners to direct and integrate recombinant proteins into cellular membranes, often enhancing expression and stability.
Table 2: Comparison of Membrane Anchor Systems for Surface Display in E. coli
| Membrane Anchor | Origin | Mechanism of Surface Display | Impact on Cell Growth | Demonstrated Passenger (This Study) | Key Advantage |
|---|---|---|---|---|---|
| Lpp-OmpA | E. coli | Hybrid: Lpp signal + OmpA transmembrane domain | Significant impact on viability [19] | Cytochrome P450 BM3 (119 kDa) [19] | Highly efficient for many functional enzymes [19] |
| PgsA | Bacillus subtilis | Membrane-integrated protein for poly-γ-glutamate synthesis | Minimal impact on viability [19] | Cytochrome P450 BM3 (119 kDa) [19] | Robust display without reduced cell viability [19] |
| INP (INPNC) | Pseudomonas syringae | GPI-anchored N-terminal domain + C-terminal domain | Minimal impact on viability [19] | Cytochrome P450 BM3 (119 kDa) [19] | Spacer domain controls distance from cell surface [19] |
| AIDA-I | E. coli | Autotransporter: β-barrel channel facilitates passenger transport | Data not specified | Cytochrome P450 BM3 (119 kDa) [19] | Effective for large, cofactor-containing enzymes [19] |
The pOmpF fusion system represents another successful anchoring strategy. This E. coli-based system utilizes an engineered fragment of outer membrane protein F (OmpF) to direct target membrane proteins into inclusion bodies, minimizing toxicity associated with membrane disruption [51]. When used to express full-length human RAMP1, a type I integral membrane protein, pOmpF achieved high-yield expression where other common fusions (trpLE, BclXL, GST) failed. The fusion was successfully solubilized in zwitterionic fos-choline detergents and purified to homogeneity, maintaining a predominantly α-helical structure [51].
Translocation-Based Secretion Systems
Translocation systems direct the secretion of recombinant proteins into specific cellular compartments or the extracellular space, which can aid proper folding and reduce proteolytic degradation.
In E. coli, proteins can be directed to the periplasm by adding a signal sequence (e.g., phoA, pelB, ompA) to the N-terminus. The periplasmic space provides an oxidizing environment conducive to disulfide bond formation, which is critical for the stability of many eukaryotic proteins [54]. However, a major limitation is that not all expressed protein may successfully translocate, often resulting in lower overall yields compared to cytoplasmic production [54].
Advanced engineering of translocation pathways has shown promise for improving yields. Recent work with the yeast Pichia pastoris has focused on engineering the Sec complex, a central component of the general secretory pathway, to enhance the efficiency of recombinant protein secretion [55].
High-Yield Expression and Stability Optimization
Experimental Protocol for Screening Membrane Anchors
The modular SpyCatcher/SpyTag (SC/ST) system provides an efficient method for rapidly comparing different membrane anchors without constructing full genetic fusions for each combination [19].
Methodology:
- Genetic Construct Design: Fuse the SC domain (113 aa) to the C- or N-terminus of the selected membrane anchor (e.g., Lpp-OmpA, PgsA, INP, AIDA-I) using a flexible (GGGGS) linker [19].
- Separate Passenger Expression: Express the passenger protein (e.g., cytochrome P450 BM3) with the complementary ST domain (13 aa) [19].
- In Situ Assembly: Co-express or mix the membrane anchor-SC and passenger-ST components. The SC and ST domains spontaneously form a covalent isopeptide bond, attaching the passenger to the anchor [19].
- Evaluation: Assess system performance by measuring:
- Cell Growth: Optical density (OD600) over time to monitor toxicity.
- Membrane Integrity: Using assays based on fluorescent dyes (e.g., propidium iodide).
- Display Efficiency: Flow cytometry or fluorescence microscopy with a labeled antibody against the passenger.
- Biocatalytic Activity: For enzymes, measure the conversion of a substrate to product by whole cells [19].
Optimizing Expression and Stabilization Conditions
Once a promising anchor or fusion system is identified, optimizing expression conditions is crucial for maximizing yields of stable, functional protein.
Key Optimization Parameters:
- Promoter and Induction: Use tightly regulated promoters (e.g., T7, araBAD) and optimize inducer concentration (e.g., IPTG) and induction time to balance yield and cell viability [53].
- Temperature Shift: Lowering the incubation temperature from 37°C to 32°C or even lower after induction can slow cell growth, reduce stress, and improve folding by allowing the cellular machinery more time to process the recombinant protein [56] [53].
- Strain Selection: Use specialized E. coli strains like Lemo21(DE3) or C41(DE3)/C43(DE3), which are engineered to tolerate membrane protein overexpression better than standard BL21(DE3) [53].
- Detergent Screening: Systematically screen detergents for solubilization and purification. Zwitterionic detergents like Fos-Choline-14 (FC-14) and Lauryl Maltose Neopentyl Glycol (LMNG) are often successful at solubilizing membrane proteins while maintaining structural integrity [51] [56].
The following workflow illustrates a high-throughput pipeline for membrane protein production:
The Scientist's Toolkit: Essential Research Reagents
Successful membrane protein production relies on a suite of specialized reagents and tools. The following table details key solutions for critical steps in the workflow.
Table 3: Essential Research Reagent Solutions for Membrane Protein Production
| Reagent / Tool | Function / Application | Example Products / Types |
|---|---|---|
| Codon Optimization Tools | Enhances transcription and translation efficiency in the heterologous host by adapting codon usage. | GeneArt GeneOptimizer Algorithm [56] |
| Specialized E. coli Strains | Tolerates toxicity from membrane protein overexpression; improves folding and integration. | Lemo21(DE3), C41(DE3), C43(DE3) [53] |
| Detergent Screening Kits | Solubilizes membrane proteins while preserving native structure and function. | LMNG/CHS, DDM/CHS, Fos-Choline series [56] [51] |
| Affinity Purification Resins | Captures and purifies recombinant proteins based on a fused affinity tag. | Ni-NTA resins (for His-tag), Anti-DYKDDDDK Resin (for FLAG-tag) [56] |
| Membrane Protein Extraction Kits | Efficiently extracts membrane proteins from cultured cells or tissues with minimal denaturation. | Mem-PER Plus Kit, GPCR Extraction & Stabilization Reagent [56] |
| Cell-Free Expression Systems | Produces membrane proteins in vitro, bypassing cellular toxicity. Ideal for toxic proteins or labeling. | E. coli-based CFE systems, Wheat Germ systems [54] [52] |
Achieving high-yield expression of membrane proteins for structural studies requires a strategic and systematic approach. The choice between membrane-anchored and translocation-based systems is not universal; it must be guided by the specific characteristics of the target protein. As comparative data shows, modular systems like SpyCatcher/SpyTag enable rapid screening of anchors such as Lpp-OmpA and PgsA to identify the optimal configuration for a given passenger protein with minimal workload. Furthermore, fusion systems like pOmpF demonstrate that strategic fusion partners can overcome expression bottlenecks for challenging targets like full-length human RAMP1. Success ultimately depends on the integrated optimization of the expression host, genetic design, fusion partner, and subsequent solubilization and purification protocols. By leveraging the comparative data and experimental frameworks outlined in this guide, researchers can rationally navigate this complex landscape to increase the probability of producing high-quality membrane protein samples suitable for high-resolution structural analysis.
Integral membrane proteins represent approximately 25% of all protein-coding genes in organisms and perform crucial functions in ion transport, signaling, and cellular communication [25]. The biogenesis of these proteins—encompassing their targeting, insertion, folding, and assembly—occurs through distinct pathways tailored to the protein's topological and biophysical characteristics [25]. In recent years, these fundamental biological processes have emerged as promising targets for therapeutic intervention in drug development. By strategically modulating specific membrane protein insertion pathways, researchers can potentially influence protein function, trafficking, and ultimate localization—thereby altering disease-relevant cellular processes. This guide provides a comparative analysis of membrane-anchored versus translocation-based systems, examining how their distinct biogenesis mechanisms present unique opportunities for pharmaceutical innovation. We focus particularly on the therapeutic potential of targeting tail-anchored protein insertion pathways and the Sec translocon-mediated insertion systems, highlighting experimental approaches for investigating these pathways in drug development contexts.
Comparative Analysis of Membrane Protein Insertion Pathways
Pathway Classification and Key Characteristics
Membrane protein insertion mechanisms are primarily classified based on their temporal relationship with protein synthesis (co-translational vs. post-translational) and their specific molecular machinery. The table below compares the major insertion pathways relevant to drug development.
Table 1: Comparison of Major Membrane Protein Insertion Pathways
| Pathway | Insertion Timing | Key Machinery | Substrate Characteristics | Therapeutic Relevance |
|---|---|---|---|---|
| SRP-Sec61 | Co-translational | Signal Recognition Particle (SRP), Sec61 translocon [25] | Proteins with N-terminal signal sequences or TMDs located ≥65 amino acids from C-terminus [25] | Primary pathway for most membrane proteins; target for secretory protein production |
| GET Pathway | Post-translational | GET complex, EMC insertase [25] [12] | Tail-anchored proteins with high-hydrophobicity TMDs near C-terminus [25] | Emerging target for modulating protein localization; engineering surface display systems |
| EMC Pathway | Post-translational | ER Membrane Complex (EMC) [25] [12] | Tail-anchored proteins with low-hydrophobicity TMDs [25] [12] | Alternative insertion route; potential compensatory pathway when GET is impaired |
| YidC/Oxa1 Family | Post-translational | YidC (bacteria), Oxa1 homologs [12] | Multispanning membrane proteins with C-terminal TMs requiring post-translational insertion [12] | Conservation from bacteria to humans highlights fundamental importance |
Structural Determinants of Pathway Selection
The specific insertion pathway employed by a membrane protein is largely determined by sequence features and structural characteristics:
Tail-anchored proteins are defined by a single transmembrane domain (TMD) located close to the C-terminus (within ~65 amino acids) and utilize post-translational insertion pathways [25]. Their TMD hydrophobicity determines whether they preferentially utilize the GET pathway (high hydrophobicity) or EMC pathway (low hydrophobicity) [25].
C-terminal transmembrane domain (cTM) properties significantly impact insertion efficiency, particularly for multispanning membrane proteins. Recent research reveals that the hydrophilicity and length of C-terminal tails flanking these TMs are evolutionarily optimized to ensure proper membrane insertion [12].
Hydrophobicity thresholds serve as critical determinants for pathway selection, with subtle differences in transmembrane domain characteristics directing proteins to distinct insertion mechanisms [25] [12].
Experimental Approaches for Investigating Insertion Pathways
Quantitative Analysis of Insertion Efficiency
Researchers have developed multiple experimental frameworks to quantify membrane protein insertion efficiency across different pathways. The following table summarizes key quantitative findings from recent studies:
Table 2: Experimental Data on Insertion Pathway Efficiency and Regulation
| Experimental System | Key Manipulation | Measured Outcome | Implications for Drug Development |
|---|---|---|---|
| Emerin-based display system [57] | Sequential deletion of N-terminal domains (Δ190, Δ200, Δ210) | Increased plasma membrane localization with longer deletions | Optimization of tail-anchored protein scaffolds for surface display |
| C-tail hydrophilicity modulation [12] | Systematic variation of C-terminal tail hydrophilicity in cTMs | Decreased insertion efficiency with increased C-tail hydrophilicity | Sequence optimization for proper membrane protein folding |
| Lipid composition studies [58] | Addition of short-chain DL lipids to POPE/POPG membranes | Inhibition of CLC-ec1 dimerization even at <1% DL ratios | Lipid microenvironment as regulator of membrane protein oligomerization |
| Pathway competition assays [25] | Genetic disruption of GET vs. EMC pathways | Substrate-specific dependence on alternative pathways | Identification of compensatory mechanisms for backup targeting |
Core Methodologies for Pathway Analysis
Cell Surface Display Using Tail-Anchored Proteins
Experimental Protocol:
- Construct Design: Fuse reporter proteins (e.g., mRuby) and target proteins to N- and C-termini of emerin TMD, respectively [57].
- Expression System: Transfect constructs into mammalian cells (HeLa or HEK293) using transfection reagents like X-tremeGENE [57].
- Localization Assessment: Monitor reporter fluorescence (mRuby) to confirm cytoplasmic expression and membrane integration [57].
- Functional Validation: For enzyme display (e.g., biotin protein ligase), add fluorescently labeled substrates (biotin carboxyl carrier protein) and detect surface capture [57].
Key Applications in Drug Development:
- Target protein screening through surface display
- Biosensor development using membrane-anchored receptors
- Engineering of therapeutic cell surfaces for enhanced trafficking
Genetic and Biochemical Dissection of Insertion Pathways
Experimental Protocol:
- Genetic Manipulation: Use CRISPR/Cas9 or RNAi to disrupt specific insertion components (GET complex, EMC, YidC) [12].
- Substrate Tracking: Monitor localization and stability of candidate membrane proteins following pathway disruption.
- Interaction Mapping: Employ crosslinking and co-immunoprecipitation to define physical interactions between substrates and insertion machinery.
- Functional Assays: Measure activity of membrane proteins (transport, signaling) following pathway-specific perturbations.
Pathway Visualization and Molecular Relationships
Membrane Protein Insertion Pathways
Research Reagent Solutions Toolkit
Table 3: Essential Research Reagents for Studying Insertion Pathways
| Reagent/Category | Specific Examples | Research Application | Key Function |
|---|---|---|---|
| Expression Plasmids | Emerin pEGFP-N2, mRuby2-C1 [57] | Protein localization studies | Fluorescent tagging and tracking |
| Cell Lines | HeLa, HEK293 [57] | Heterologous expression systems | Consistent protein production and visualization |
| Transfection Reagents | X-tremeGENE [57] | Plasmid delivery | Efficient introduction of DNA constructs |
| Pathway-Specific Reporters | VAMP2 (GET substrate), SQS (EMC substrate) [25] | Pathway activity assessment | Selective monitoring of insertion routes |
| Hydrophobicity Analysis Tools | ΔGapp prediction algorithms [12] | Bioinformatics screening | Quantitative assessment of insertion potential |
Therapeutic Applications and Future Directions
Current Therapeutic Opportunities
The strategic manipulation of membrane protein insertion pathways offers multiple avenues for therapeutic intervention:
Chimeric Antigen Receptor (CAR)-T Cell Engineering: Synthetic biology approaches now enable engineering of mammalian cells for therapeutic applications. CAR-T cells represent a prominent example where membrane protein expression is strategically controlled for cancer immunotherapy [59]. These engineered receptors combine antigen-binding domains with T-cell activating domains, requiring proper membrane insertion for function [59].
Biosensor and Display System Development: Tail-anchored proteins provide advantageous scaffolds for surface display systems due to their distinct topology. Unlike type I transmembrane proteins that display N-termini externally, tail-anchored proteins naturally display C-termini on the cell surface [57]. This enables attachment of target proteins via their N-termini, preserving C-terminal functionality that might be essential for activity.
Correcting Pathogenic Misfolding: Disease-causing mutations that increase C-tail hydrophilicity can be addressed through compensatory mutations or pharmacological chaperones that restore proper membrane insertion [12].
Emerging Research Frontiers
Several promising research directions are emerging in the field of membrane protein insertion targeting:
Lipid Microenvironment Engineering: Recent studies demonstrate that lipid composition influences membrane protein oligomerization through preferential solvation effects rather than specific binding sites [58]. This reveals opportunities for modulating protein function through membrane lipid composition rather than direct protein targeting.
Dual-Pathway Inhibition Strategies: Given the redundancy in some insertion pathways, combined targeting of primary and backup insertion mechanisms may yield enhanced therapeutic effects for specific membrane protein classes.
Synthetic Biology Approaches: Engineered cells equipped with synthetic DNA circuits can be programmed to respond to specific biomarkers, enabling controllable release of therapeutic agents [59]. These approaches increasingly rely on precise manipulation of membrane protein insertion to achieve proper localization and function.
The continuing elucidation of membrane protein biogenesis mechanisms—including recent insights into post-translational insertion by YidC in E. coli and EMC in human cells [12]—promises to reveal new therapeutic opportunities for targeting these fundamental cellular processes in drug development.
Diagnosing and Correcting Biogenesis Failures in Membrane Proteins
The faithful insertion of proteins into membranes is a fundamental biological process with far-reaching implications for cellular health and biotechnology. In both native cellular environments and engineered systems, proteins destined for membranes or secretion must navigate complex biogenesis pathways. Faulty insertion—the failure of a protein to correctly integrate into a membrane or translocate across it—can trigger a cascade of detrimental outcomes, including misfolding, mislocalization, and aggregation [60] [61]. These failure points are not merely inefficiencies; they represent critical junctures where cellular function breaks down, leading to lost productivity in engineered systems and contributing to severe human diseases, notably neurodegenerative disorders [60] [62] [63].
This guide provides a comparative analysis of two primary strategies for managing membrane protein biogenesis: membrane-anchored systems and translocation-based systems. Membrane-anchored systems typically involve the direct fusion of a protein of interest (the "passenger") to a membrane anchor protein, tethering it to the cell surface [19]. In contrast, translocation-based systems, such as the Sec or Tat pathways, actively transport proteins across lipid bilayers, often employing specialized machinery like the Sec61 complex in the endoplasmic reticulum (ER) or the TatABC complexes in bacteria [64] [65] [7]. By objectively comparing the performance, failure modes, and experimental methodologies of these systems, this guide aims to equip researchers with the knowledge to diagnose and mitigate insertion-related pathologies.
Comparative Analysis of Protein Display and Insertion Systems
The choice between membrane-anchored and translocation-based systems involves a fundamental trade-off between control and complexity, each with distinct vulnerabilities.
Membrane-Anchored Display Systems function by genetically fusing a passenger protein to a native membrane protein anchor. This complex is then expressed and routed to the membrane. A prominent example is the Lpp-OmpA anchor, which fuses the signal peptide and first 9 N-terminal amino acids of the E. coli lipoprotein (Lpp) to the transmembrane domain of outer membrane protein A (OmpA) [19]. However, this direct genetic fusion approach is prone to several failure points. The genetic linkage itself can lead to unfavorable domain interactions, causing the hybrid protein to misfold or be degraded by cellular quality control systems [19]. Furthermore, certain anchors, like Lpp-OmpA, can exert significant toxicity and impact membrane integrity when overexpressed, compromising cell viability and overall system performance [19].
Translocation-Based Systems are essential for transporting proteins across membranes. The Sec pathway, found in the ER of eukaryotes and the plasma membrane of bacteria, is the primary route for the co-translational translocation of unfolded polypeptides [64] [7]. Its core component, the Sec61/SecYEG complex, forms a aqueous channel that opens in response to a signal peptide on the nascent chain. Key failure points here include the inefficient recognition of signal peptides and the inability to transport pre-folded proteins, which can lead to jamming and abortive translocation [65] [7]. In contrast, the Tat pathway specializes in the remarkable feat of transporting fully folded proteins across energized membranes [65]. This system is critical for inserting cofactor-containing enzymes into membranes. Its failure modes often involve membrane stress and destabilization due to the physical act of pushing a rigid structure across the lipid bilayer, which can trigger stress responses like the phage shock protein (Psp) system [65].
Performance Comparison of Membrane Anchors in a Modular Display System
Conventional genetic fusion approaches to surface display are laborious, requiring the construction of a new plasmid for every passenger-anchor combination. A innovative solution to this problem is the use of modular systems that leverage post-translational coupling, such as the SpyCatcher/SpyTag (SC/ST) technology [19]. In this system, the membrane anchor is fused to the SpyCatcher peptide, while the passenger protein is fused to the small SpyTag. When these separate components are expressed, they spontaneously form a covalent bond, assembling the display complex on the cell surface. This modularity allows researchers to rapidly screen multiple anchor-passenger combinations without constructing direct genetic fusions.
A study utilizing this S3D (SpyCatcher/SpyTag Surface Display) system compared the performance of four different membrane anchors for displaying a challenging passenger protein, cytochrome P450 BM3 (BM3, 119 kDa) on E. coli [19]. The quantitative results of this comparison are summarized in the table below.
Table 1: Performance Comparison of Membrane Anchors in a Modular Display System for Cytochrome P450 BM3 [19]
| Membrane Anchor | Origin / Type | Key Structural Features | Impact on Cell Growth & Membrane Integrity | Surface Display Capacity | Whole-Cell Biocatalytic Activity |
|---|---|---|---|---|---|
| Lpp-OmpA | E. coli / Hybrid | N-terminal Lpp signal peptide, OmpA transmembrane domain, C-terminal SC fusion. | Strong negative impact on cell viability upon overexpression. | High display capacity. | Efficient for BM3 display. |
| PgsA | B. subtilis / Membrane-integrated | Full-length protein involved in poly-γ-glutamate synthesis, C-terminal SC fusion. | No major negative impact reported. | Efficient display capacity. | Efficient for BM3 display. |
| INP (INPNC) | P. syringae / Truncated ice nucleation protein | N- and C-terminal domains of InaK (central repeats largely deleted), C-terminal SC fusion. | Used successfully without affecting cell viability. | Successful display of BM3 in direct fusion systems. | Not specified in results. |
| AIDA-I | E. coli / Autotransporter | N-terminal SC fusion, native linker, C-terminal β-barrel pore. | Used successfully for large enzymes. | Successful display of BM3 in direct fusion systems. | Not specified in results. |
The data demonstrates that the membrane anchor is not a passive tether but actively influences the entire system's performance. While Lpp-OmpA and PgsA were both efficient for BM3 display in the S3D system, their impact on cellular health differed significantly [19]. This highlights a critical failure point: anchor-induced toxicity. The study also proved the modular system's power by overcoming a known size limitation of the Lpp-OmpA anchor, which was previously thought to be restricted to passengers under 70 kDa [19]. The success in displaying the 119 kDa BM3 suggests that the post-translational, modular approach can mitigate steric and folding issues inherent in direct genetic fusions.
Experimental Protocols for Investigating Failure Points
Protocol 1: Assessing Insertion and Localization via Cryo-Electron Tomography
To visualize the fundamental process of protein insertion and identify points of failure at molecular resolution, cryo-Electron Tomography (cryo-ET) has emerged as a premier technique. A recent groundbreaking study used cryo-ET to visualize the translation and translocation machinery at the ER membrane in near-native conditions [7].
Workflow Overview:
- Sample Preparation: ER-derived vesicles (microsomes) are rapidly isolated from human cells (e.g., HEK 293F cells) within about one hour to preserve native complexes.
- Vitrification: The sample is plunge-frozen in liquid ethane to create a frozen-hydrated state, preserving cellular structures in amorphous ice.
- Data Acquisition: A transmission electron microscope is used to acquire a series of 2D images (a "tilt series") of the sample as it is tilted along an axis.
- Tomogram Reconstruction: The 2D tilt series are computationally merged to reconstruct a 3D volume (tomogram) of the cellular environment.
- Subtomogram Averaging and Classification: Thousands of individual particles (e.g., ribosome-translocon complexes) are extracted from the tomograms, aligned, and classified to obtain high-resolution structures of different functional states.
Key Insights and Identified Failure Points [7]:
- The study visualized distinct polysomes binding to different ER translocons specialized for secreting soluble proteins or inserting multipass transmembrane proteins.
- It resolved the structure of the most abundant translocon variant, comprising the SEC61 channel, the translocon-associated protein complex (TRAP), and the oligosaccharyltransferase complex A (OSTA).
- By classifying various ribosomal intermediates, the method can pinpoint stalling in the translocation or elongation cycle, which are potential precursors to misfolding and aggregation. For instance, failure to properly engage the translocon with the ribosome (SEC61 gating failure) would lead to cytosolic mislocalization of a secretory protein.
The following diagram illustrates the key steps and components in this protocol.
Protocol 2: Evaluating Membrane Integrity and Stress During Tat-Dependent Translocation
The translocation of folded proteins via the Tat system imposes unique physical stresses on the membrane. The following protocol is based on research investigating the interaction between the Tat translocon and the membrane-stress response [65].
Objective: To assess the membrane stress and integrity challenges associated with the translocation of folded proteins via the Tat system, and to identify the recruitment of stress-response proteins as a failure mitigation mechanism.
Methodology:
- Strain and Plasmid Design: Use E. coli strains with chromosomal deletions of Tat components (e.g., ΔtatAE) or phage shock protein (Psp) components (e.g., ΔpspA, ΔpspB, ΔpspC, ΔpspF). Express Strep-tagged TatA or its mutants from rhamnose-inducible plasmids in these backgrounds.
- Affinity Purification and Interaction Analysis: Isolate TatA complexes from membranes without using detergents where possible. Use affinity chromatography (e.g., Strep-Tactin) to pull down TatA and co-purifying proteins. Identify interacting partners like PspA via mass spectrometry or immunoblotting.
- Electron Microscopy: Visualize purified TatA and PspA complexes using negative stain electron microscopy to confirm direct physical contact.
- Membrane Integrity Assays: Prepare inverted membrane vesicles (INV) from strains with elevated TatA expression. Use fluorescence quenching assays (e.g., with ACMA) to monitor the dissipation of the proton motive force, a direct indicator of membrane leakiness and destabilization.
- Physiological Growth Assays: Test the physiological consequence of membrane stress by comparing anaerobic growth of wild-type and ΔpspA strains on minimal medium with Trimethylamine N-oxide (TMAO) as a terminal electron acceptor. TMAO respiration requires the Tat-dependent translocation of the large, folded TMAO reductase (TorA). Impaired growth in the ΔpspA mutant indicates a failure to cope with Tat-induced stress.
Key Findings and Identified Failure Points [65]:
- The N-terminal transmembrane domain of TatA directly interacts with the membrane-stabilizing protein PspA.
- Overexpression of TatA complexes induces membrane stress, increasing membrane permeability and triggering a Psp stress response.
- Under native expression levels, the absence of PspA impaired TMAO respiratory growth, indicating that the Psp system is crucial for managing the membrane stress inherent to the translocation of large, folded Tat substrates. A failure in this stress response system constitutes a critical failure point that limits the functional efficiency of the Tat pathway.
The Scientist's Toolkit: Essential Research Reagents and Solutions
The following table lists key reagents, constructs, and tools used in the experimental studies cited in this guide, providing a resource for designing related investigations.
Table 2: Key Research Reagents and Solutions for Studying Membrane Protein Insertion
| Reagent / Tool | Function / Description | Example Application / Context |
|---|---|---|
| SpyCatcher/SpyTag (SC/ST) | A protein ligation system that forms a spontaneous, covalent isopeptide bond. | Enables modular surface display (S3D system); allows separate expression of passenger and anchor [19]. |
| Lpp-OmpA, PgsA, INP, AIDA-I | Model membrane anchors from different biological origins. | Used for comparative performance testing of surface display systems in E. coli [19]. |
| Cytochrome P450 BM3 (BM3) | A large (119 kDa) model passenger protein containing heme and diflavin cofactors. | A challenging substrate used to test the limits and efficiency of surface display systems [19]. |
| SEC61 Complex | The core heterotrimeric protein-conducting channel for Sec-based translocation in the ER. | Central subject of structural studies on co-translational translocation and translocon gating [64] [7]. |
| TatABC Complexes | Membrane protein complexes that facilitate the translocation of folded proteins. | Studied to understand the mechanisms of folded protein transport and associated membrane stress [65]. |
| Phage Shock Protein A (PspA) | A membrane-stabilizing protein induced by various stresses, including Tat activity. | Used as a marker and effector to study membrane integrity in response to Tat-mediated translocation [65]. |
| Cryo-Electron Tomography (cryo-ET) | An imaging technique for obtaining 3D structures of cellular components in a near-native state. | Used to visualize ribosome-translocon complexes and associated factors in ER-derived microsomes [7]. |
The comparative analysis of membrane-anchored and translocation-based systems reveals a landscape of shared and distinct failure points. Membrane-anchored systems are highly susceptible to issues of misfolding and cytotoxicity stemming from direct genetic fusions and anchor overexpression [19]. The adoption of modular technologies like SpyCatcher/SpyTag presents a powerful strategy to decouple the passenger from the anchor, mitigating these risks and accelerating combinatorial optimization [19]. In contrast, translocation-based systems face fundamental biophysical challenges: the Sec pathway is limited to unfolded proteins and can jam, while the robust Tat pathway, capable of moving folded proteins, inherently destabilizes membrane integrity and requires dedicated cellular systems to manage the resultant stress [65] [7].
For researchers and drug development professionals, these insights are critical for system selection and optimization. When working with complex, cofactor-dependent proteins, the Tat pathway or modular membrane-anchored systems may be preferable despite their associated stress and complexity. For high-throughput display of simpler peptides, traditional Sec-based translocation or direct fusion anchors may suffice. Ultimately, diagnosing failure points—whether it's aggregation in the cytosol, mislocalization to the wrong compartment, or stress-induced membrane collapse—requires a toolkit of sophisticated methods, from molecular phenotyping like cryo-ET [7] to physiological growth assays [65]. By understanding these core vulnerabilities, scientists can better engineer robust cellular systems for biocatalysis, therapeutic protein production, and the fundamental study of membrane biology.
The biogenesis of multispanning membrane proteins is a complex process wherein the accurate insertion of transmembrane helices (TMs) is critical for proper folding and function. While the Sec61/SecYEG translocon mediates the co-translational insertion of the majority of TMs, a significant biological challenge arises for C-terminal TMs (cTMs) located close to the end of the protein sequence. Recent research reveals that cTMs with cytosolic tails shorter than approximately 45-60 amino acids cannot be accommodated by the canonical co-translational insertion mechanism due to geometric constraints of the ribosome exit tunnel, necessitating an alternative post-translational insertion pathway [66] [12]. This guide comprehensively compares the sequence features and cellular machinery required for efficient post-translational cTM insertion across biological systems, providing a foundational resource for researchers engineering membrane proteins for therapeutic and biotechnological applications.
Comparative Analysis of Sequence Determinants for cTM Insertion
Key Sequence Parameters Governing Insertion Efficiency
Systematic studies across organisms have identified two paramount sequence features that direct successful post-translational cTM insertion: the length and the hydrophilicity of the C-terminal tail (C-tail) following the final transmembrane helix.
Table 1: Comparative Role of C-Tail Features in Post-Translational Insertion
| Sequence Feature | Role in Insertion | Experimental Evidence |
|---|---|---|
| C-Tail Length | Dictates insertion route; tails <45-60 residues necessitate post-translational insertion [12]. | Bioinformatic proteome analyses in E. coli, yeast, and human [12]; Molecular length requirement for Sec61 translocon access [66]. |
| C-Tail Hydrophilicity | Determines efficiency of post-translational insertion, especially for proteins with extracytosolic C-termini (Cext) [12]. | Evolutionary analysis showing constrained hydrophilicity in Cext proteins; Mutagenesis increasing hydrophilicity causes insertion defects [12]. |
| cTM Hydrophobicity | Less correlated with post-translational insertion efficiency compared to C-tail properties [12]. | Comparison of cTM apparent free energy (ΔGapp) with other TMs shows inconsistent organism-specific trends [66] [12]. |
Organism-Specific Prevalence and Sequence Adaptation
The requirement for post-translational insertion is pervasive across membrane proteomes. Genomic analyses indicate that a majority of multispanning membrane proteins in E. coli (567 of 673 proteins) and approximately half in humans (1385 of 2483 proteins) possess C-tails shorter than 45 residues, compelling them to utilize this pathway [12]. Evolution has fine-tuned the C-tail composition to optimize this process. Specifically, proteins with an extracytosolic C-terminus (Cext) exhibit significantly shorter and less hydrophilic C-tails compared to those with cytosolic C-termini (Ccyt), facilitating the translocation of the tail across the membrane during post-translational insertion [12].
Table 2: Comparative Analysis of cTM Insertion Across Model Systems
| Organism / System | Primary Insertase | Key C-Tail Feature | Experimental Validation |
|---|---|---|---|
| E. coli | YidC (Oxa1 family) [12]. | Low hydrophilicity for Cext proteins [12]. | C-tail mutations disrupting YidC interaction cause misfolding [12]. |
| Human Cells | ER Membrane Protein Complex (EMC) [12]. | Low hydrophilicity for Cext proteins [12]. | Disease-causing mutations increase C-tail hydrophilicity, leading to misinsertion [12]. |
| General Sec61 Translocon | Sec61 (Post-translational retention) [66]. | Short tails (<60 aa) with low hydrophobicity [66]. | Poor ER insertion of cTMs with short tails; Sec61 retention provides assembly window [66]. |
Experimental Workflows and Methodologies
Key Workflow: Analyzing Insertion Efficiency and Assembly
A common experimental pipeline for investigating cTM insertion involves perturbing sequence features and quantitatively assessing the consequences on membrane integration, protein complex assembly, and cellular trafficking.
Diagram 1: Experimental analysis workflow for cTM insertion and assembly.
Detailed Experimental Protocols
Glycosylation Mapping for Insertion Topology
This protocol tests whether a cTM is properly integrated into the membrane or translocated into the ER lumen.
- Construct Design: Append a consensus N-glycosylation site (e.g., N-X-T/S) to the immediate C-terminus of the protein of interest [66].
- Transfection & Expression: Express the construct in a suitable cell line (e.g., HEK293) or in vitro translation system supplemented with microsomes.
- Glycosylation Detection: Resolve the protein via SDS-PAGE. A shift to a higher molecular weight indicates glycosylation, implying full C-tail translocation into the ER lumen and failed cTM membrane insertion. Treat samples with Endoglycosidase H (Endo H) to confirm the shift is due to N-linked glycosylation [66].
- Quantification: The ratio of glycosylated to non-glycosylated protein provides a quantitative measure of insertion efficiency.
Cycloheximide Chase for Protein Stability Assay
This assay determines the half-life of a protein and assesses its degradation upon failed insertion or assembly.
- Treatment: Treat cells expressing the target protein with cycloheximide to inhibit new protein synthesis.
- Time-Course Sampling: Collect cell lysates at predetermined time points (e.g., 0, 1, 2, 4 hours) post-treatment.
- Immunoblotting: Detect the target protein and a loading control at each time point via Western blotting.
- Inhibitor Confirmation: To confirm degradation is mediated by the proteasome via ERAD, repeat the assay in the presence of a proteasome inhibitor such as MG132. Stabilization of the protein confirms ERAD involvement [66].
Cell-Free Expression for High-Throughput Screening
Recent advances enable high-throughput screening of PTM installation and protein assembly using cell-free systems.
- System Setup: Utilize a cell-free gene expression (CFE) system, such as PUREfrex, supplemented with fluorescently labeled lysine (e.g., FluoroTect GreenLys) to monitor expression [67].
- Parallelized Expression: Express hundreds of protein variants with different C-tail sequences in parallel in a 384-well plate format.
- Interaction Screening: Use a bead-based assay like AlphaLISA to detect interactions between the synthesized cTM-containing protein and its partner protein or insertase, facilitating rapid binding landscape characterization without purification steps [67].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Studying Post-Translational Membrane Insertion
| Reagent / Tool | Function / Application | Example Use Case |
|---|---|---|
| MG132 (Proteasome Inhibitor) | Inhibits proteasomal degradation, stabilizing ERAD substrates for detection [66]. | Confirms ERAD-mediated degradation of misinserted cTM proteins in chase assays [66]. |
| Cycloheximide | Halts cytoplasmic protein synthesis, enabling measurement of protein half-life [66]. | Used in pulse-chase experiments to monitor the stability and turnover of membrane proteins [66]. |
| Endoglycosidase H (Endo H) | Removes N-linked glycans of the high-mannose type in the ER, confirming glycosylation status [66]. | Verifies that a gel shift is due to ER lumenal translocation of the C-tail [66]. |
| PUREfrex CFE System | Reconstituted cell-free translation system for high-throughput protein expression [67]. | Rapid expression and screening of C-tail variant libraries for insertion/assembly efficiency [67]. |
| AlphaLISA Beads | Bead-based proximity assay for detecting molecular interactions in solution in a high-throughput format [67]. | Measures binding between cTM proteins and their partner subunits or insertases without protein purification [67]. |
The strategic optimization of C-terminal tail length and hydrophilicity is a fundamental principle for ensuring the efficient biogenesis of multispanning membrane proteins. The experimental data and comparative analysis presented herein establish that the cellular machinery for post-translational cTM insertion—namely YidC in bacteria and EMC in humans—exerts distinct evolutionary pressure on C-tail sequences. For researchers, this underscores that membrane protein sequences cannot be viewed in isolation; they are fine-tuned for productive collaboration with the specific biogenesis machinery of the host system. As the field progresses, leveraging high-throughput cell-free expression and AI-driven structural prediction [67] [68] will accelerate the engineering of membrane proteins with optimized insertion signatures, paving the way for advances in the production of therapeutic membrane proteins and the targeting of membrane-associated diseases.
The biogenesis of integral membrane proteins represents a fundamental cellular process essential for numerous physiological functions, including signal transduction, nutrient transport, and cell adhesion. Despite evolutionary optimization, an estimated 20-30% of inherited diseases result from mutations that disrupt protein folding and membrane insertion [25] [12]. These "biogenesis hurdles" manifest as misfolding, mislocalization, or premature degradation of functionally important membrane proteins. In response, researchers have developed two primary intervention strategies: chaperone co-expression to guide proper folding and trafficking, and genetic suppressors to bypass fundamental assembly bottlenecks. This review objectively compares these approaches within the context of membrane-anchored versus translocation-based systems, providing experimental data and methodologies to inform therapeutic development.
Comparative Analysis of Rescue Strategies
Table 1: Strategic Comparison of Protein Rescue Approaches
| Parameter | Chaperone Co-expression | Genetic Suppressors |
|---|---|---|
| Primary Mechanism | Physical interaction with client proteins to facilitate folding/insertion [69] [70] | Mutational alteration of machinery components to overcome biogenesis blocks [71] [12] |
| Typical Experimental Timeframe | Days to weeks (transient/stable expression) | Weeks to months (selection pressure applied) |
| Key Readouts | Protein localization, complex assembly, functional activity [69] [72] | Growth assays, OMP levels, suppressor mutation identification [71] |
| Therapeutic Applicability | Protein-based therapeutics; challenging delivery | Small molecules mimicking suppressor effects; more druggable |
| Notable Example | PcrH-PopB interaction in P. aeruginosa T3SS [69] | BamA mutants (S715R, E470K) bypassing BamD essentiality [71] |
Table 2: Quantitative Assessment of Rescue Efficacy in Model Systems
| Experimental System | Rescue Strategy | Efficiency Metric | Result | Reference |
|---|---|---|---|---|
| P. aeruginosa T3SS | Chaperone (PcrH) binding to PopB | Translocation competence | Stable insertion into bilayers | [69] |
| E. coli BAM complex | BamA(S715R) mutation | Viability in ΔbamD background | Normal growth | [71] |
| E. coli BAM complex | rcsF(G117R) mutation | Crosslinking to BamA | >70% reduction in jamming | [71] |
| Chickpea (Plant) | AtBAG4 expression | Nodulation under drought | 306 vs 109 nodules (transgenic vs control) | [72] |
| Chickpea (Plant) | AtBAG4 expression | Seed nitrogen content | 4.03% vs 3.3% (transgenic vs control) | [72] |
Chaperone Co-Expression: Direct Folding Assistance
Core Mechanisms and Experimental Evidence
Chaperone co-expression leverages the cell's natural quality control systems to rescue defective membrane proteins. The foundational principle involves overexpressing specific chaperones that recognize and bind to misfolded clients, providing them an opportunity to attain proper conformation. This approach has demonstrated efficacy across diverse biological systems, from bacterial secretion apparatus to plant symbiotic signaling.
In Pseudomonas aeruginosa, the type III secretion system (T3SS) requires precise assembly of translocator proteins PopB and PopD within host membranes. Research has demonstrated that the chaperone PcrH stabilizes these translocators in the bacterial cytoplasm and employs a conserved binding interface to prevent premature aggregation [69]. Structural analyses revealed that PopB residues 51-59 lie within the concave face of PcrH, utilizing primarily backbone atoms for binding interactions that exclude ternary complex formation [69].
In plant systems, the cytoprotective co-chaperone AtBAG4 has shown remarkable ability to enhance stress tolerance without compromising normal cellular functions. Transgenic chickpea lines expressing AtBAG4 maintained robust nodulation capacity under drought conditions (306 nodules per plant in transgenics versus 109 in controls) while simultaneously increasing seed nitrogen content (4.03% vs 3.3%) [72]. This demonstrates how chaperone co-expression can potentially rescue defective proteins without metabolic tradeoffs.
Essential Research Reagents and Methodologies
Table 3: Key Research Reagents for Chaperone Studies
| Reagent/Tool | Function/Application | Example from Literature |
|---|---|---|
| Affinity Tags | Protein purification and detection | His-tag for PopB/PcrH complex purification [69] |
| Proteoliposomes | In vitro membrane insertion assays | PS:PC liposomes for PopB insertion studies [69] |
| β-lactamase Reporter Assays | Monitoring translocation efficiency | Eukaryotic cell translocation assays [69] |
| Limited Proteolysis | Mapping solvent-accessible regions | Trypsin digestion of membrane-bound PopB [69] |
| Sucrose Gradient Centrifugation | Separating membrane-bound and soluble proteins | Characterization of lipid-bound translocators [69] |
Experimental Protocol: Chaperone-Translocator Interaction Analysis
Objective: Characterize chaperone-translocator interactions and membrane insertion competence.
Methodology Details:
Complex Purification: Co-express chaperone (PcrH) and translocator (PopB) using pETDuet vector system in E. coli. Purify complex via nickel-affinity chromatography exploiting His-tagged components [69].
Liposome Preparation: Create lipid films from 2:3 phosphatidylserine:phosphatidylcholine mixtures in chloroform. Dissolve in PBS with 0.7% octyl-β-D-glucopyranoside, followed by extrusion through 100μm membranes [69].
Membrane Insertion Assay: Incubate purified proteins with pre-formed liposomes overnight at room temperature. Assess insertion stability by challenging with high-salt (1M KCl), urea (6M), or detergent treatments [69].
Sucrose Gradient Analysis: Layer proteoliposome mixtures onto 0-45% sucrose gradients. Ultracentrifuge at 230,000 × g for 16 hours in SW41.Ti rotor. Collect 1ml fractions and analyze by Western blotting [69].
Trypsin Digestion: Treat proteoliposomes with trypsin at 1:500 (trypsin:protein ratio) at room temperature. Quench reactions at timed intervals and analyze cleavage patterns by SDS-PAGE and Western blotting [69].
Genetic Suppressors: Bypassing Biogenesis Essentiality
Fundamental Principles and Key Findings
Genetic suppressor strategies exploit the cell's capacity to overcome protein biogenesis defects through compensatory mutations that restore function without directly correcting the original defect. This approach has revealed fundamental insights into the flexibility of essential biological systems and identified potential therapeutic targets.
Landmark research on the β-barrel assembly machinery (BAM) complex in E. coli has challenged long-standing assumptions about essentiality. While BamD was considered essential for outer membrane protein (OMP) biogenesis, recent work demonstrates that its essentiality is both conditional and substrate-specific [71]. Specific point mutations in BamA (S715R, E470K) or RcsF (S70R, G117R) can completely bypass BamD requirement, revealing that BamD's essential function primarily involves preventing RcsF-mediated jamming of the BamA barrel [71].
The insertion of C-terminal transmembrane helices (cTMs) presents another biogenesis challenge that organisms have evolved sequence-based solutions for. Research demonstrates that the hydrophilicity and length of C-terminal tails are evolutionarily optimized to ensure proper membrane insertion [12]. In E. coli, proteins with extracytosolic C-termini possess short, hydrophobic C-tails that facilitate post-translational insertion via YidC, while in humans, the ER membrane complex (EMC) performs an analogous function [12]. Mutations that increase C-tail hydrophilicity disrupt this process, causing protein misfolding and disease.
Experimental Protocol: Genetic Suppressor Identification
Objective: Identify and characterize genetic suppressors that bypass essential gene requirements.
Methodology Details:
Conditional Depletion Strain Construction: Create chromosomal BamD depletion strain using PBAD promoter system. Clone wild-type and mutant bamA alleles on low-copy pZS21 plasmid [71].
Growth Assays Under Depletion Conditions: Grow overnight cultures in arabinose-containing media for BamD expression. Wash and serially dilute for spot cultures on LB and minimal media with glucose (repression conditions). Monitor growth over 24-48 hours [71].
OMP Assembly Assessment During Depletion: In minimal media with glycerol, initiate BamD depletion by switching to glucose-containing media. Dilute cultures periodically to maintain exponential growth. Collect samples at 4h and 8h time points for immunoblot analysis of OMP levels [71].
Crosslinking Analysis: Perform biochemical crosslinking in ΔbamE background to quantify RcsF/BamA complex formation. Assess restoration of RcsF/OmpA assembly as indicator of suppressed jamming phenotype [71].
Whole-Genome Sequencing Validation: Confirm suppressor mutations and exclude secondary mutations by whole-genome sequencing of viable depletion strains [71].
Integrated Analysis: Strategic Application Guidance
Context-Dependent Strategy Selection
The comparative data reveal that chaperone co-expression and genetic suppressors represent complementary rather than competing approaches, each with distinct application domains based on the nature of the biogenesis defect:
For folding efficiency bottlenecks where proteins possess marginal stability but correct topological information, chaperone co-expression typically provides superior rescue. The PcrH-PopB system exemplifies this scenario, where dedicated chaperone binding maintains translocation competence until membrane engagement [69].
For essentiality constraints where core biogenesis machinery becomes limiting, genetic suppressors offer more fundamental solutions. The BamD bypass suppressors demonstrate how identifying alternative conformational states in machineries like the BAM complex can overcome previously absolute requirements [71].
Technical Implementation Considerations
Temporal factors significantly influence strategy selection. Chaperone co-expression typically yields functional data within days to weeks, making it suitable for rapid intervention testing. Genetic suppressor identification often requires longer timeframes (weeks to months) for selection and validation but can reveal more fundamental biological insights.
Therapeutic development paths differ substantially between approaches. Chaperone-based strategies face delivery challenges but offer protein-based intervention possibilities. Suppressor-informed drug development aims to identify small molecules that mimic suppressor effects on target proteins, potentially offering more druggable approaches.
The systematic comparison of chaperone co-expression and genetic suppressor strategies reveals a sophisticated toolkit for addressing membrane protein biogenesis defects. Chaperone co-expression provides direct folding assistance with relatively rapid implementation, while genetic suppressors identify fundamental restructuring opportunities within biogenesis pathways. The emerging paradigm recognizes that these approaches are context-dependent rather than universally superior, with optimal selection requiring careful analysis of the specific biogenesis hurdle. As structural biology advances illuminate increasingly detailed mechanisms [73] [74], the integration of these complementary strategies promises enhanced capacity to rescue defective proteins underlying human disease.
Membrane proteins (MPs) are pivotal players in a vast array of physiological processes, including signal transduction, molecular transport, and cell recognition. Their significance is underscored by the fact that they constitute approximately 60% of all approved drug targets [20]. However, a major bottleneck impedes their study: their inherent hydrophobicity and complex interactions with lipid bilayers make them notoriously difficult to solubilize and stabilize outside their native membrane environment [75] [76]. Overcoming this challenge is a critical prerequisite for functional and structural studies, most notably for single-particle cryo-electron microscopy (cryo-EM) which has emerged as a leading technique for resolving MP structures [75]. This guide provides a comparative analysis of the principal strategies developed to tackle these issues, focusing on the context of membrane-anchored versus translocation-based systems, and provides a toolkit of experimental protocols for researchers.
Comparative Analysis of Solubilization and Stabilization Systems
The core challenge in MP research lies in replacing the native lipid bilayer with a synthetic environment that maintains the protein's native conformation and function. The following section objectively compares the performance of conventional and emerging technologies.
Table 1: Comparison of Major Membrane Protein Stabilization Systems
| System Category | Key Examples | Mechanism of Action | Advantages | Disadvantages / Stability Data |
|---|---|---|---|---|
| Conventional Detergents | DDM, OG, LDAO | Form micelles around hydrophobic MP domains [75]. | Well-established protocols; wide commercial availability [77]. | Often lead to protein denaturation and instability over time; low success rate for complex MPs [75] [77]. |
| Pendant-Bearing Detergents | GNG, Mal-X, P-GNGs | Alkyl chains at micelle interface reduce water penetration and enhance protein-detergent interactions [77]. | Improved stability over conventional detergents; tunable properties [77]. | A study showed P-GNGs increased stability for 4 out of 6 test MPs, whereas DDM failed for 5 of them [77]. |
| Amphipols / Nanodiscs | A8-35, MSP Nanodiscs | Amphipols trap MPs in a soluble state; Nanodiscs use a lipid bilayer encircled by membrane scaffold proteins [75]. | Nanodiscs provide a near-native lipid environment; both can enhance stability [75]. | Complex preparation; Amphipols can have lower solubility and are difficult to remove [75]. |
| Polymer-Based Lipid Particles | SMA, DIBMA | Amphiphilic copolymers that directly solubilize membranes, forming nanodiscs with native lipids [75]. | Preserves native lipid environment; high stability for cryo-EM; detergent-free [75]. | SMA is sensitive to low Mg2+ and low pH; DIBMA is more gentle but may form larger particles [75]. |
Table 2: Performance Comparison of Different Membrane Anchor Systems for Surface Display
| Membrane Anchor | Origin / Type | Display Capacity (Model Passenger: P450 BM3, 119 kDa) | Impact on Cell Growth & Membrane Integrity |
|---|---|---|---|
| Lpp-OmpA | E. coli hybrid anchor | Effective display achieved via modular system, overcoming a previous ~70 kDa size limit [19]. | Strong negative impact on cell viability after overexpression [19]. |
| PgsA | Bacillus subtilis | Efficient display capacity demonstrated [19]. | No significant negative impact on viability reported [19]. |
| INP (INPNC) | Pseudomonas syringae | Previously used for successful BM3 display via direct genetic fusion [19]. | No significant negative impact on viability reported [19]. |
| AIDA-I | Autotransporter | Previously used for successful BM3 display via direct genetic fusion [19]. | Performance comparable to Lpp-OmpA and PgsA in modular system [19]. |
Detailed Experimental Protocols for Key Methodologies
Modular Cell Surface Display Using SpyCatcher/SpyTag
This protocol enables the rapid, modular comparison of different membrane anchors without constructing direct genetic fusions for each combination [19].
- Step 1: Construct Design. Genetically fuse the SpyCatcher (SC) domain to the C- or N-terminus of the selected membrane anchor (e.g., Lpp-OmpA, PgsA, INP, AIDA-I) using a flexible (GGGGS) linker [19].
- Step 2: Separate Expression. Express the following two components separately in E. coli:
- The membrane anchor-SpyCatcher fusion protein.
- The passenger protein of interest (e.g., cytochrome P450 BM3) fused to the SpyTag (ST) peptide.
- Step 3: Post-Translational Assembly. Combine the expressed components in situ. The SpyTag and SpyCatcher domains spontaneously form a covalent isopeptide bond, anchoring the passenger protein to the cell surface via the membrane anchor [19].
- Step 4: Analysis. Assess performance by measuring cell growth, membrane integrity, surface display capacity (e.g., via flow cytometry), and whole-cell biocatalytic activity [19].
High-Throughput Screening of Artificial Synthesis Environments with MEMPLEX
The MEMPLEX platform uses machine learning to design optimal cell-free synthesis conditions for MPs [78].
- Step 1: Fluorescent Reporter Construction. Fuse the membrane protein gene to the C-terminus of a split GFP fragment (the 11th beta-strand) via a 12-amino-acid flexible linker [78].
- Step 2: Combinatorial Printing. Use a custom nanoliter droplet printer to assemble cell-free reactions in a 384-well plate format. Each reaction contains the plasmid, E. coli cell-free extract, and variable components such as different lipid types (e.g., liposomes), small molecule concentrations (e.g., Mg2+, K+), and chaperone proteins [78].
- Step 3: Synthesis and Reconstitution. Incubate reactions to allow for co-translational insertion of the synthesized MP into the supplied liposomes.
- Step 4: Fluorescence Detection. Add the complementary, bulk-purified large GFP fragment to the reactions. Fluorescence develops only if the MP is correctly integrated into the liposome and the GFP tag is accessible for complementation. Aggregate MPs produce minimal background [78].
- Step 5: Machine Learning Optimization. Use the initial fluorescence data to train an ensemble of deep neural networks. The model then predicts more effective artificial synthesis environments for subsequent iterative rounds of synthesis and screening [78].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagent Solutions for Membrane Protein Research
| Reagent / Material | Function / Application | Specific Examples / Notes |
|---|---|---|
| Styrene Maleic Acid (SMA) Copolymer | Direct solubilization of membranes to form SMA Lipid Particles (SMALPs), preserving native lipids [75]. | A detergent-free alternative that is particularly valuable for cryo-EM studies [75]. |
| Pendant-Bearing Detergents | Enhanced stabilization of MPs by filling voids in micelles and reducing water penetration [77]. | GNG, Mal-101/111/12_1; P-GNGs with optimized pendant chains [77]. |
| SpyCatcher/SpyTag System | Modular, post-translational conjugation for cell surface display and other protein assembly applications [19]. | Enables rapid comparison of different membrane anchors without genetic re-engineering [19]. |
| MEMPLEX Platform & Split GFP Reporter | High-throughput, AI-guided design of cell-free synthesis conditions and rapid assessment of MP solubilization [78]. | Reports successful liposome insertion; correlates with function (e.g., demonstrated with Bacteriorhodopsin) [78]. |
| Cryo-EM Grids & Optimized Detergents | High-resolution structural determination of membrane proteins. | The choice of solubilizing agent (e.g., SMA, DIBMA, novel detergents) is critical for achieving high-quality, near-native structures [75]. |
Workflow and Pathway Visualizations
MP Research Workflow
This diagram outlines the critical path for membrane protein research, from expression to structure determination, highlighting key decision points.
Modular Anchor System Comparison
This diagram illustrates the modular SpyCatcher/SpyTag system used to compare the performance of different membrane anchors for surface display, as detailed in [19].
The field of membrane protein research is rapidly evolving beyond traditional detergents. The comparative data presented in this guide demonstrates that no single system is universally superior; the choice depends on the specific MP and research goals. Detergent-free alternatives like SMA are revolutionizing structural biology by preserving the native lipid environment, while modular systems like SpyCatcher/SpyTag accelerate the optimization of membrane-anchored platforms. Looking forward, the integration of machine learning platforms like MEMPLEX and the computational design of soluble MP analogues [32] promise to systematically dismantle the long-standing bottlenecks in MP research. These advanced strategies, which leverage high-throughput data and AI-driven design, are poised to significantly expand our understanding of the "dark" membrane proteome, ultimately accelerating drug discovery and therapeutic development.
The faithful insertion and folding of membrane proteins are fundamental cellular processes critical for signal transduction, transport, and metabolism. Central to these processes are two evolutionarily conserved membrane protein insertases: YidC in bacteria and its eukaryotic counterpart, the ER Membrane Protein Complex (EMC). These complexes facilitate the biogenesis of a vast range of membrane proteins, and mutations disrupting their function are increasingly linked to human diseases. YidC operates in bacterial membranes, functioning both independently and in conjunction with the SecYEG translocon to insert proteins into the lipid bilayer [79] [80]. In eukaryotes, the EMC performs analogous functions within the endoplasmic reticulum (ER), acting as an insertase for tail-anchored proteins and facilitating the proper biogenesis of multi-pass transmembrane proteins [81] [82]. This guide provides a comparative analysis of the mechanisms through which genetic mutations disrupt these essential complexes, framing the discussion within broader research on membrane-anchored versus translocation-based systems. Understanding these disruptive mechanisms provides a foundation for therapeutic interventions aimed at rescuing protein function or bypassing defective biogenesis pathways.
The YidC and EMC systems, though evolutionarily related, exhibit distinct architectural complexities and functional scopes. The table below summarizes their core characteristics.
Table 1: Comparative Overview of YidC and EMC
| Feature | Bacterial YidC | Eukaryotic EMC |
|---|---|---|
| Organism | Bacteria | Eukaryotes |
| Subunit Composition | Single protein (6 transmembrane segments) | 8-9 subunit complex (e.g., EMC1-8, EMC10) |
| Primary Localization | Cytoplasmic membrane | Endoplasmic Reticulum (ER) membrane |
| Core Insertase Function | Sec-dependent & Sec-independent insertion | Insertase for TA proteins & multi-pass membrane proteins |
| Key Functional Domains | Hydrophobic slide (TM3, TM5), Charged groove | EMC3 (YidC homolog), Lumenal domain (EMC1), Cytosolic TPR (EMC2) |
| Associated Machinery | SecYEG translocon, SecDF | Sec61 translocon, Get complex |
YidC is a single-component insertase with a relatively simple architecture. Its core function relies on a hydrophobic slide formed by transmembrane segments 3 and 5 (TM3 and TM5), which guides nascent transmembrane domains into the lipid bilayer [80]. It can function alone for small proteins like the M13 procoat protein or in concert with the SecYEG translocon for more complex substrates [79]. The interaction with SecYEG is critical; residues like G355 in E. coli YidC are essential for this communication [79].
In contrast, the EMC is a multi-subunit complex with a more elaborate architecture. Its insertase activity is primarily attributed to the EMC3 subunit, which shares evolutionary homology with YidC [81] [82]. However, the complex possesses additional domains that expand its functional repertoire: a large ER lumenal domain (primarily in EMC1) thought to act as a chaperone, and cytosolic tetratricopeptide repeats (TPRs) in EMC2 that may facilitate client protein recognition [81]. This structural complexity allows the EMC to handle a diverse clientele and engage in functions beyond mere insertion, such as the stabilization of folding intermediates [81].
Mechanisms of Mutation and Disruption
Genetic mutations disrupt the function of YidC and EMC through distinct yet analogous mechanisms. The impact of these mutations can be quantified through systematic interaction profiling, as illustrated in the diagram below.
Disruption of YidC Function
Mutations in YidC primarily compromise its ability to interact with partner proteins or to form a functional substrate-binding site.
- Disruption of SecYEG Interaction: A quintuple serine mutant of E. coli YidC (YidC-5S, with mutations in TM3 and TM5) is specifically defective in Sec-dependent insertion, as shown for the subunit a of the F₀F₁ ATP synthase, while maintaining Sec-independent insertion capability. This functional defect stems from an inability to interact molecularly with SecYEG, demonstrated by a lack of FRET signal between purified and labeled YidC-5S and SecYEG in proteoliposomes [80].
- Critical Residues for Translocon Interaction: Synthetic lethal screens have identified specific, highly conserved residues in YidC, such as G355 and M471, that are critical for viability in the absence of the accessory subunit SecDF. Overexpression of SecY can rescue the viability of strains with these YidC mutations, indicating these residues participate in direct or indirect interactions with SecY [79].
Disruption of EMC Function
The multi-subunit nature of the EMC provides several targets for disruptive mutations, with effects on its insertase and chaperone activities.
- Perturbation of the Insertase Path: Structural and functional analyses of the EMC reveal a path for transmembrane helix insertion from the cytoplasm into the membrane. Mutations in key membrane-embedded cavities, including a gated cavity and a lipid-filled cavity, disrupt the insertion of clients like squalene synthase and the beta-adrenergic receptor (β1AR) [81].
- Loss of Lumenal Chaperone Activity: The large EMC1 lumenal domain provides a chaperone function distinct from insertase activity. Mutations in this domain can specifically affect the stability of clients like sigma intracellular receptor 2 without impairing the insertion of other proteins, indicating a role in post-insertion folding or quality control [81].
- Impaired Biogenesis of Multi-pass Proteins: EMC disruption affects proteins beyond its direct insertase substrates. For example, the EMC is required for the biogenesis and stability of complex multi-pass membrane proteins, including ionotropic acetylcholine receptors and the beta-adrenergic receptor, likely by assisting in the co-translational insertion of specific challenging transmembrane domains [82].
A Broader Perspective on Protein-Protein Interaction Disruption
Large-scale studies reveal that missense mutations disrupting protein-protein interactions (PPIs) are a widespread phenomenon. One systematic analysis of 2,009 missense variants found that ~10.5% of missense variants per individual are disruptive, with disruptive mutations enriched at conserved residues and lower allele frequencies, though still present even in common variants [83]. This highlights that PPI disruption is a major mechanism of mutational effect, directly relevant to understanding how variants impact complexes like YidC and EMC.
Experimental Data and Comparative Analysis
Quantitative data from key experiments provides direct evidence for the disruptive mechanisms outlined above.
Table 2: Experimental Data on Mutation Disruption in YidC and EMC
| Experimental System | Mutation/Variant | Quantitative Effect | Functional Consequence |
|---|---|---|---|
| E. coli YidC FRET Binding [80] | YidC Wild-Type | Kd = 27 (± 10) nM with SecYEG | Normal SecYEG interaction and Sec-dependent insertion |
| YidC-5S (430,435,468,505,509S) | No FRET signal with SecYEG | Loss of SecYEG interaction; defective insertion of subunit a | |
| E. coli YidC Complementation [79] | YidC G355, M471 mutants | Synthetic lethality without SecDF; rescued by SecY OE | Impaired interaction with SecY |
| Human EMC Client Stability [81] | EMC lumenal domain mutants | Decreased stability of Sigma Intracellular Receptor 2 | Loss of chaperone function |
| EMC membrane cavity mutants | Disrupted insertion of Squalene Synthase & β1AR | Loss of insertase function | |
| Human PPI Disruption Survey [83] | 298 disruptive ExAC variants | 10.5% of missense variants/individual disrupt PPIs | Widespread functional impact of interaction disruption |
The experimental workflow for characterizing such mutations typically involves a combination of genetic, biochemical, and structural techniques, as visualized below.
Methodologies for Experimental Investigation
A detailed understanding of disruption mechanisms relies on robust experimental protocols. Key methodologies are outlined below.
Yeast Two-Hybrid (Y2H) for Protein-Protein Interaction Disruption
- Principle: This high-throughput method tests the impact of hundreds to thousands of missense single nucleotide variants (SNVs) on binary protein-protein interactions.
- Protocol: The gene encoding a protein of interest (the "bait") is cloned into a plasmid containing a DNA-binding domain. Genes for its interaction partners ("preys") are cloned into a plasmid containing a transcription activation domain. SNVs are introduced into the bait. Bait and prey plasmids are co-transformed into yeast. An interaction that activates reporter genes indicates the SNV does not disrupt the interaction, while loss of reporter signal indicates disruption. This method was used to profile 2,009 human variants across 2,185 interactions [83].
- Validation: Positive and negative interactions are typically validated using an orthogonal method like Protein Complementation Assay (PCA) in human cells [83].
Fluorescence Resonance Energy Transfer (FRET) for Complex Binding
- Principle: FRET measures the proximity between two proteins (e.g., YidC and SecYEG) in a purified system or proteoliposomes.
- Protocol:
- Protein Purification and Labeling: Engineer single cysteine residues into periplasmic loops of YidC (e.g., W23C) and SecY (e.g., M142C). Purify the proteins and label them with donor (e.g., Atto520) and acceptor (e.g., Atto647N) fluorophores, respectively [80].
- In Vitro Binding Assay: Titrate increasing amounts of labeled SecYEG into a solution containing a fixed concentration of labeled YidC in detergent. Excite the donor fluorophore and measure emission of both donor and acceptor.
- Reconstitution into Proteoliposomes: Co-reconstitute labeled YidC and SecYEG into lipid vesicles (e.g., DOPC) at a defined protein-to-lipid ratio.
- FRET Measurement: Measure fluorescence emissions. A decrease in donor signal and a concomitant increase in acceptor signal indicates energy transfer and thus protein interaction. A lack of FRET, as seen with the YidC-5S mutant, indicates a disrupted interaction [80].
- Data Analysis: The FRET efficiency is calculated, and binding affinity (Kd) can be determined from titration curves.
In Situ Structural Analysis by Cryo-Electron Tomography (Cryo-ET)
- Principle: This technique visualizes protein structures directly inside cells (in situ), capturing intermediate states in biological processes.
- Protocol:
- Sample Preparation: Use bacterial minicells or thin sections to achieve suitable thickness for imaging. Induce overexpression of the target protein (e.g., GspD secretin) [84].
- Data Collection: Vitrify the sample and acquire tilt series images using a cryo-electron microscope.
- Tomogram Reconstruction and Subtomogram Averaging: Manually or automatically pick particle subtomograms from the reconstructed 3D volume. Align and average these particles to achieve a higher resolution structure. Incorporate membrane geometry as a constraint during alignment to correctly determine particle orientation [84].
- Application: This method has been used to visualize different intermediate states of secretin translocation from the inner to the outer membrane, providing mechanistic insights into membrane protein biogenesis [84].
The Scientist's Toolkit: Essential Research Reagents
The following table lists key reagents and tools used in the experiments cited in this guide.
Table 3: Research Reagent Solutions for Studying Insertase Function
| Reagent / Tool | Function / Application | Example Use Case |
|---|---|---|
| Cysteine-less YidC (C0) [80] | Background for introducing single cysteine residues for site-specific labeling | FRET binding studies with SecYEG |
| Unstable miniF Plasmid (pRC7) [79] | Maintenance of genes in a low-copy, unstable manner for complementation assays | Studying synthetic lethal interactions (e.g., YidC mutants + ΔsecDF) |
| Proteoliposomes (DOPC) [80] | Reconstitution of membrane proteins into a near-native lipid environment | In vitro FRET analysis of YidC-SecYEG interaction |
| PIONEER AI Tool [85] | Publicly available web database and software to predict the impact of mutations on PPIs | Identifying key disruptive mutations in human diseases for drug targeting |
| DisruPPI Computational Method [86] | Structure-based computational redesign to disrupt specific PPIs while preserving stability | Deimmunization of therapeutic proteins by removing antigenic sites |
The comparative analysis of YidC and EMC reveals both shared and unique vulnerabilities to genetic mutation. While both are central to membrane protein biogenesis, YidC's function is more readily disrupted by mutations that impair its partnership with the Sec translocon. In contrast, the multi-domain architecture of the EMC means its functions—ranging from insertase to chaperone—can be independently targeted by specific mutations. The experimental approaches outlined, from high-throughput interaction screens to in situ structural biology, provide a powerful toolkit for deciphering these mechanisms.
Future research will increasingly leverage computational tools like PIONEER and DisruPPI to predict the disruptive impact of mutations on these complexes, guiding experimental validation and drug discovery [86] [85]. As the structural understanding of these complexes deepens, the design of small molecules or biologics that can either inhibit these complexes (in the case of bacterial YidC for antibiotics) or modulate their function to rescue defective client proteins (in the case of EMC for genetic diseases) becomes a tangible goal. The continued objective comparison of these membrane-anchored systems is fundamental to advancing both basic science and therapeutic development.
Functional Crossroads: A Head-to-Head Evaluation of Anchoring vs. Translocon Systems
In eukaryotic cells, a fundamental challenge exists in ensuring that thousands of integral membrane proteins are correctly sorted to their destination organelles. This process requires exquisite precision, as approximately 30% of genes in most genomes encode integral membrane proteins that perform numerous biological processes from signaling to transport [87]. The concept of substrate specificity and triage provides a unifying framework for understanding how transmembrane domains (TMDs) are assigned to different cellular pathways. Rather than a single deterministic system, cellular sorting represents a sophisticated quality control and decision-making process where TMD sequences are recognized, evaluated, and directed based on their physicochemical properties [87] [88].
This guide compares two principal systems governing membrane protein fate: membrane-anchored systems that primarily recognize and retain sorted proteins, and translocation-based systems that actively participate in membrane integration and sorting decisions. We objectively evaluate their performance through experimental data, detailing how TMD properties including length, hydrophobicity, and amino acid asymmetry determine pathway assignment in the secretory pathway.
Comparative Analysis of TMD Sorting Systems
System Definitions and Functional Roles
Table 1: Core Functions of Membrane Protein Sorting Systems
| System Type | Primary Function | Key Recognition Signals | Cellular Location | Representative Machinery |
|---|---|---|---|---|
| Translocation-Based | Co-translational membrane integration; Client triage and chaperoning | TMD hydrophobicity; topological signals | Endoplasmic Reticulum | Sec61 translocon; ER Membrane Protein Complex (EMC) |
| Membrane-Anchored | Post-insertion recognition; Retention and retrieval | Organelle-specific TMD length; residue composition | Golgi, Plasma Membrane, Early Endosomes | Unknown recognition machinery |
Performance Comparison by Sorting Accuracy
Table 2: System Performance Metrics in TMD Recognition and Sorting
| Performance Metric | Translocation-Based Systems | Membrane-Anchored Systems |
|---|---|---|
| TMD Length Discrimination | Limited discrimination during initial insertion [87] | High sensitivity to organelle-specific length variations (ER: ~19 aa; PM: ~23 aa) [87] |
| Hydrophobicity Sensitivity | High sensitivity for membrane integration [87] [88] | Moderate sensitivity for spatial organization [87] |
| Amino Acid Asymmetry Detection | Limited detection during insertion [87] | High precision in recognizing residue volume asymmetries [87] |
| Client Specificity | Broad range with preference for suboptimal TMDs [88] | Narrow specificity for organelle-matched TMD features [87] |
| Topological Control | Direct determination during membrane integration [88] | Indirect influence through spatial organization [87] |
Experimental Analysis of TMD-Environment Interactions
TMD Property Analysis Across Organelles
Systematic computational analyses of fungal and vertebrate TMDs reveal that transmembrane domains are not generic but possess organelle-specific properties that direct their cellular localization [87].
Table 3: Quantitative TMD Properties by Cellular Compartment
| Organelle | Mean TMD Length (amino acids) | Key Residue Preferences | Characteristic Structural Features |
|---|---|---|---|
| Endoplasmic Reticulum (ER) | ~19 | Shorter hydrophobic cores; minimal asymmetry | Reduced amino acid asymmetry across bilayer |
| Golgi Apparatus | ~20-21 | Intermediate length; moderate asymmetry | Developing compositional asymmetry |
| Plasma Membrane | ~23 | Longer hydrophobic cores; valine enrichment exoplasmically | Strong asymmetry linked to residue size |
| Early Secretory Pathway | 19-21 | Less pronounced asymmetry | Dichotomy in TMD length pre-/post-Golgi |
Experimental Approaches for TMD Sorting Analysis
Computational TMD Property Mapping
Objective: To systematically compare TMD length and residue composition across different organelles [87].
Methodology:
- Dataset Curation: Compile proteins with experimentally determined topology and single TMD (bitopic proteins)
- Ortholog Expansion: Use BLAST searches to collect orthologous proteins from complete fungal and vertebrate genomes
- Redundancy Reduction: Apply BLASTClust to cluster sequences based on TMD and flanking region similarity, removing sequences with >30% identity
- TMD Alignment: Implement scanning algorithm with Goldman-Engelman-Steitz (GES) hydrophobicity scale to define hydrophobic spans
- Orientation Standardization: Align TMDs from cytosolic to exoplasmic sides rather than N- to C-terminus
- Frequency Calculation: Compute amino acid frequency at each position through hydrophobic regions
Key Controls: Parallel analyses with Wimley-White and Biological hydrophobicity scales to verify scale-independent results; alignment based on "positive-inside rule" for consistent orientation [87].
Chaperone-Client Interaction Mapping
Objective: To define EMC chaperone binding preferences and client features [88].
Methodology:
- Site-Specific Crosslinking: Incorporate unnatural amino acid 4-Benzoyl-phenylalanine (Bpa) at 17 positions within EMC1 lining lipid-filled cavity
- Crosslinking Activation: Photoactivate Bpa crosslinker to capture endogenous interaction partners
- Immunoprecipitation: Isolate crosslinked adducts under stringent washing conditions
- Mass Spectrometry: Identify interacting proteins through quantitative proteomics
- Validation: Confirm interactions through co-immunoprecipitation and western blotting of selected hits
- Minimal System Verification: Test isolated TMD-EMC interactions using TMDs fused to superfolder GFP with ER-targeting signal
Key Controls: Verification of correct membrane integration of TMD-GFP fusions; comparison to non-crosslinked controls for specificity determination [88].
Visualization of TMD Sorting Pathways
Cellular TMD Sorting and Triage Pathway: This pathway illustrates how transmembrane domains are synthesized, evaluated by the ER membrane protein complex (EMC), and sorted to specific organelles based on their physicochemical properties, with organelle-specific systems providing quality control and retention.
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagents for TMD Sorting Studies
| Reagent/Cell Line | Primary Function | Experimental Application |
|---|---|---|
| HEK 293T Cells | Protein expression platform | EMC interactome studies through site-specific crosslinking [88] |
| Bpa (4-Benzoyl-phenylalanine) | Photoactivatable crosslinker | Capturing transient EMC-client interactions for MS identification [88] |
| Saccharomyces cerevisiae Strains | Yeast model system | Genetic analysis of membrane protein biogenesis and complementation assays [87] [89] |
| SGY1528 Yeast Strain (Mat a ade 2–1 can 1–100 his 3–11,15 leu 2–3,112 trp 1–1 ura 3–1 trk 1::HIS3 trk 2::TRP1) | Potassium uptake-deficient yeast | Functional assessment of potassium channel TMD mutants through complementation [89] |
| pYES2 Plasmid System | Heterologous expression vector | Expression of K+ channel mutants in yeast complementation assays [89] |
| TMHMM Prediction Algorithm | Computational TMD identification | Initial assignment of transmembrane domains in protein sequences [87] |
| GES (Goldman-Engelman-Steitz) Scale | Hydrophobicity measurement | Precisely defining hydrophobic spans in TMD alignment studies [87] |
| Minimal Consensus Membrane Protein (ConMem) | Model transmembrane protein system | Defining EMC binding preferences without confounding chaperone sites [88] |
Mechanism of TMD Recognition and Triage
The ER membrane protein complex (EMC) functions as a central triage manager that recognizes membrane proteins with signatures of incomplete structure formation [88]. The EMC possesses two distinct substrate engagement sites: a canonical insertase function located on one side of the complex, and a lipid-filled cavity on the opposite side that serves as a chaperone site [88]. This lipid-filled cavity, primarily formed by EMC1 subunits, can engage TMDs and modulate their orientation within the lipid bilayer. Productive TMD assembly reduces binding to the EMC chaperone site, providing a release mechanism for properly folded membrane proteins [88].
Organelle-specific discrimination occurs through recognition of distinct TMD features. The plasma membrane exhibits a striking preference for longer TMDs (~23 amino acids) compared to ER-resident proteins (~19 amino acids), reflecting adaptation to the thicker bilayers of post-ER compartments [87]. Additionally, TMDs from post-ER organelles show striking asymmetries in amino acid composition across the bilayer, with valine enrichment on the exoplasmic side, linked to residue size and varying between organelles [87]. This pervasive presence of organelle-specific features enables collective contribution to membrane properties and provides a physical basis for spatial segregation.
The substrate specificity and triage model provides a framework for understanding how cells manage membrane protein biogenesis—not as a simple deterministic pathway, but as a sophisticated quality control system where TMD sequences are continuously evaluated and directed. The comparison between translocation-based and membrane-anchored systems reveals complementary functions: while translocation systems like the EMC perform initial triage based on fundamental TMD properties, membrane-anchored systems provide organelle-specific recognition that reinforces spatial organization.
This understanding has significant implications for drug development, particularly for diseases associated with membrane protein misfolding and mislocalization. The EMC's role in safeguarding the biogenesis of diverse clients including ABC transporters, connexins, and rhodopsin [88] suggests potential therapeutic targets for modulating membrane protein expression and function. Furthermore, the organelle-specific TMD properties identified through comparative analyses [87] provide design principles for engineering membrane proteins with tailored subcellular localization, opening new avenues for targeted therapeutic delivery and precision membrane protein engineering.
The biogenesis of membrane proteins is a fundamental cellular process orchestrated by distinct targeting and insertion pathways. These routes can be broadly categorized into co-translational and post-translational mechanisms, primarily defined by whether the protein is inserted into the membrane during or after its synthesis on the ribosome [25]. The choice of pathway has profound implications for the subsequent folding, assembly, and ultimate function of the protein. In eukaryotic cells, most membrane proteins are inserted into the endoplasmic reticulum (ER) membrane, while in prokaryotes, insertion occurs directly into the plasma membrane [25]. This guide provides a comparative analysis of these routes, focusing on their mechanistic bases, impact on protein folding and function, and the experimental data used to distinguish between them, framed within the context of membrane-anchored versus translocation-based systems.
Comparative Analysis of Processing Routes
Mechanism and Substrate Specificity
The primary distinction between the two major processing routes lies in the timing of membrane insertion relative to protein synthesis.
Co-translational Insertion (SRP Pathway): This pathway is mediated by the Signal Recognition Particle (SRP) and its receptor (SR) [25]. The SRP recognizes hydrophobic targeting sequences—which can be a cleavable signal peptide or the first transmembrane domain (TMD)—as they emerge from the ribosome exit tunnel. It then delivers the entire ribosome-nascent chain complex to the Sec translocon (SecYEG in prokaryotes, Sec61 in eukaryotes) at the membrane [25]. A critical constraint for this pathway is that the targeting sequence must be located at least ~65 amino acids from the protein's C-terminus to ensure translation does not terminate before SRP-mediated delivery is complete [25]. This route is the default for the majority of multi-pass membrane proteins.
Post-translational Insertion: This mechanism is essential for proteins that cannot engage the SRP pathway. The most characterized class is Tail-Anchored (TA) proteins, which possess a single TMD very close to their C-terminus [25] [12]. These proteins complete synthesis in the cytosol and are then chaperoned to the membrane. The specific pathway used depends on the hydrophobicity of the TMD. High-hydrophobicity TA proteins (e.g., VAMP2) use the GET pathway, while those with low-hydrophobicity TMDs (e.g., SQS) rely on the ER membrane complex (EMC) [25]. Recent research has revealed that C-terminal TMs (cTMs) of multi-spanning membrane proteins also often require post-translational insertion, frequently mediated by insertases like YidC in E. coli and its homolog EMC in human cells [12].
Table 1: Core Characteristics of Co-translational and Post-translational Insertion Pathways
| Feature | Co-translational (SRP-Sec) Pathway | Post-translational Pathways (GET/EMC/YidC) |
|---|---|---|
| Key Machinery | SRP, SR, Sec Translocon (SecYEG/Sec61) | GET Complex, EMC, YidC Insertase |
| Timing | During translation | After translation is complete |
| Classic Substrates | Multi-pass MPs, secreted proteins | Tail-anchored (TA) proteins, C-terminal TMs (cTMs) of multi-span MPs |
| Targeting Signal | N-terminal signal peptide or first TMD | C-terminal TMD (for TA proteins) |
| Spatial Constraint | Targeting signal >~65 residues from C-terminus [25] | Targeting signal very close to C-terminus |
| Energetics | GTP hydrolysis by SRP and SR | ATP hydrolysis (chaperones), pmf (YidC) |
Impact on Protein Folding and Function
The route of membrane insertion directly influences the folding trajectory and functional integrity of a protein by shaping its biogenesis environment.
Folding Efficiency and Topology: The co-translational pathway allows TMs to exit the ribosome exit tunnel and integrate directly into the lipid bilayer via the Sec translocon's lateral gate, minimizing the exposure of hydrophobic segments to the aqueous cytosol [25]. This step-wise insertion as the chain is synthesized helps establish the correct topology early. In contrast, post-translational insertion faces the challenge of integrating a fully synthesized hydrophobic domain. The success of this process is highly dependent on the sequence features of the C-terminal tail (C-tail). For proteins with an extracytosolic C-terminus (Cext), evolution has selected for short and hydrophobic C-tails that are compatible with translocation by insertases like YidC and EMC, which struggle with long, hydrophilic segments [12]. Increasing C-tail hydrophilicity disrupts productive interaction with YidC, leading to misinsertion, mistrafficking, and loss of function [12].
Assembly and Complex Formation: For multi-subunit membrane protein complexes, the co-translational pathway provides a controlled environment at the translocon where early folding intermediates can be shielded by auxiliary factors and assembly can begin co-translationally [25]. The post-translational folding of a fully synthesized subunit before insertion might present a higher risk of aggregation or non-productive interactions with other cytosolic proteins.
Functional Consequences and Disease Links: The critical importance of the post-translational pathway is highlighted by its direct link to human genetic diseases. Mutations that increase the hydrophilicity of the C-tail in human membrane proteins have been identified as disease-causing, as they impair cTM insertion by EMC, leading to protein misfolding and degradation [12]. This demonstrates that membrane protein sequences are finely tuned to collaborate effectively with a specific biogenesis machinery, and deviations from this optimization can be pathological.
Experimental Data and Methodologies
Key Experimental Findings
Quantitative data and targeted experiments are crucial for comparing the efficiency and outcomes of different insertion pathways.
Table 2: Comparative Experimental Data on Processing Routes
| Experimental Approach | Key Finding | Implication |
|---|---|---|
| Proteome-wide topology analysis [12] | In E. coli, 567 of 673 multi-span MPs have C-tails <45 residues; in humans, 1385 of 2483 multi-span MPs have C-tails <45 residues. | Post-translational insertion of the cTM is a pervasive necessity across organisms. |
| C-tail hydrophilicity manipulation [12] | Increasing hydrophilicity of the C-tail in Cext proteins disrupts cTM insertion in both E. coli and human cells, causing misfolding. | C-tail hydrophilicity is an evolutionarily tuned parameter critical for successful post-translational insertion. |
| Genetic deletion of insertases [12] | Deletion of yidC in E. coli or perturbation of EMC in human cells specifically impairs the biogenesis of proteins dependent on post-translational insertion. | YidC/EMC are dedicated insertases for cTMs in their respective organisms. |
| Modular display system (SpyCatcher/SpyTag) [19] | Different membrane anchors (Lpp-OmpA, PgsA, INP, AIDA-I) impact passenger protein display capacity, whole-cell biocatalytic activity, and host cell health. | The choice of membrane anchor, which determines the insertion pathway, directly affects functional protein yield and host viability. |
Detailed Experimental Protocols
To generate comparative data like that summarized in Table 2, researchers employ several key protocols.
3.2.1 Assessing Membrane Protein Insertion and Topology
A common method to determine whether a protein has been correctly inserted and folded is through accessibility assays.
- Procedure:
- Cell Permeabilization: Cells expressing the target membrane protein are divided into samples. One sample is treated with a detergent (e.g., Triton X-100) to permeabilize all membranes, making all epitopes accessible. Another sample is left untreated or is treated with a non-ionic detergent that only permeabilizes the plasma membrane.
- Protease Protection Assay: An exogenous protease (e.g., Proteinase K) is added to both samples. For a correctly inserted protein, domains facing the cytosol will be degraded in both samples. Domains facing the lumen or extracellular space will be protected from protease digestion in the non-fully permeabilized sample but will be degraded in the fully permeabilized sample.
- Detection: The protein is detected by SDS-PAGE and immunoblotting. The pattern of proteolytic fragments reveals the membrane topology and, by extension, the success of insertion [12].
3.2.2 The S3D System for Comparing Membrane Anchors
The SpyCatcher/SpyTag Display (S3D) system is a powerful modular tool for rapidly comparing how different membrane anchors affect passenger protein function without constructing direct genetic fusions for each combination [19].
- Procedure:
- Modular Construct Design: The gene for the passenger protein (e.g., cytochrome P450 BM3) is fused to the 13-amino acid SpyTag. Separately, genes for different membrane anchors (e.g., Lpp-OmpA, PgsA, INP, AIDA-I) are fused to the 113-amino acid SpyCatcher.
- Separate Expression: The SpyTag-passenger and SpyCatcher-anchor constructs are expressed separately in E. coli.
- In vivo Assembly: The two cell populations are mixed, or the proteins are combined in vitro. The SpyTag and SpyCatcher domains spontaneously form a covalent isopeptide bond, anchoring the passenger to the membrane.
- Functional Assay: The whole-cell biocatalytic activity of the displayed enzyme is measured (e.g., by monitoring substrate conversion). Flow cytometry can also be used to quantify surface display efficiency [19].
Visualization of Key Pathways and Systems
Co-translational vs. Post-translational Membrane Protein Insertion
This diagram illustrates the fundamental mechanistic differences between the two primary insertion pathways.
The Modular S3D System for Testing Membrane Anchors
This workflow details the experimental setup for the SpyCatcher/SpyTag system used to compare different membrane anchors.
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Studying Membrane Protein Biogenesis
| Reagent / Tool | Function / Description | Application Example |
|---|---|---|
| Protease K | A broad-spectrum serine protease that digests accessible protein domains. | Protease protection assays to confirm membrane topology and insertion fidelity [12]. |
| Digitonin | A mild detergent that selectively permeabilizes cholesterol-containing membranes (e.g., plasma membrane) but not organellar membranes. | Differential permeabilization in topology assays to distinguish cytosolic from luminal domains. |
| SpyCatcher/SpyTag System | A protein ligation system that forms a spontaneous, covalent isopeptide bond between the two partners. | Modular surface display (S3D system) to rapidly test passenger-anchor combinations without genetic fusions [19]. |
| YidC/EMC Knockout Cells | Genetically engineered bacterial or human cell lines lacking the key insertase for post-translational insertion. | Validating the dependence of specific substrate proteins on the post-translational YidC/EMC pathway [12]. |
| cDNA Display Proteolysis Library | A high-throughput method combining cell-free display and deep sequencing to measure protein folding stability. | Assessing the thermodynamic stability of thousands of membrane protein domains or variants under identical conditions [90]. |
The precise insertion of proteins into membranes is a fundamental biological process critical for cellular function. Disruptions in the machinery facilitating membrane insertion and translocation are increasingly linked to severe pathologies, including genetic diseases and cancer. This guide compares two primary strategies for membrane protein localization—membrane-anchored systems and translocation-based systems—by examining their molecular mechanisms, roles in disease pathogenesis, and experimental approaches for their study. We synthesize current research that quantitatively evaluates the performance of different systems, provides detailed methodologies, and identifies key reagents, offering a resource for researchers and drug development professionals working to target these processes therapeutically.
The biogenesis of membrane proteins is a complex process essential for cellular and organismal health. Proteins destined for membranes typically follow one of two major pathways: they are inserted co-translationally via the Sec translocon as the polypeptide chain emerges from the ribosome, or they are inserted post-translationally after synthesis is complete, often requiring specialized machinery distinct from the Sec system [12]. The proper folding and insertion of multispanning membrane proteins, which contain multiple transmembrane helices (TMs), hinge on the accurate placement of each TM into the lipid bilayer. Inaccurate insertion can lead to protein misfolding, loss of function, and disease [12].
This guide focuses on linking deficiencies in these insertion pathways to human disease. We compare two broad classes of systems:
- Translocation-based systems primarily handle the membrane integration of proteins, with deficiencies directly linked to genetic disorders.
- Membrane-anchored systems often play roles in displaying proteins on cell surfaces, with dysregulation implicated in processes like cancer metastasis.
Understanding the distinct mechanisms, performance, and experimental analysis of these systems provides a foundation for developing targeted therapeutic interventions.
Experimental Comparison of Membrane Protein Display Systems
A critical application of membrane protein engineering is the surface display of proteins for biocatalysis, vaccine development, and biosensor design. Research has systematically compared different membrane anchors to optimize the display of a model passenger protein, cytochrome P450 BM3 (BM3), in E. coli [19].
Experimental Protocol: Modular Surface Display Screening
The following methodology enables the rapid, modular comparison of different membrane anchors without constructing individual genetic fusions for each combination [19].
- Modular Construct Design: The membrane anchor and the passenger protein are expressed as separate genetic fusions. The anchor is fused to a SpyCatcher (SC) domain, while the passenger is fused to a SpyTag (ST) domain. These two domains spontaneously form a covalent isopeptide bond upon contact.
- Anchor Selection: Four established membrane anchors were compared:
- Lpp-OmpA: A hybrid anchor comprising the N-terminus of the E. coli lipoprotein (Lpp) and the transmembrane domain of outer membrane protein A (OmpA).
- PgsA: A Bacillus subtilis protein involved in poly-γ-glutamate synthesis.
- INP (INPNC): The N- and C-terminal domains of the ice nucleation protein from Pseudomonas syringae.
- AIDA-I: An autotransporter protein from the E. coli adhesin involved in diffuse adherence.
- Expression and Assembly: The SC-anchor and ST-passenger constructs are co-expressed in E. coli. In situ post-translational coupling occurs via SpyCatcher/SpyTag interaction, assembling the complete surface-displayed protein.
- Performance Evaluation: Systems are assessed for:
- Cell Growth: Optical density (OD) measurements over time.
- Membrane Integrity: Fluorescence-based assays using dyes that penetrate compromised membranes.
- Display Capacity: Flow cytometry or fluorescence microscopy using labeled antibodies against the passenger protein.
- Biocatalytic Activity: Whole-cell assays measuring the conversion of a substrate specific to the displayed enzyme (e.g., BM3 monooxygenase activity).
Quantitative Performance Data
The performance of the four membrane anchors in the modular display system varied significantly, as summarized in the table below.
Table 1: Comparative performance of membrane anchors for surface display of cytochrome P450 BM3 in E. coli [19].
| Membrane Anchor | Impact on Cell Growth | Effect on Membrane Integrity | Surface Display Capacity | Whole-Cell Biocatalytic Activity |
|---|---|---|---|---|
| Lpp-OmpA | High negative impact | Significant disruption | High | High |
| PgsA | Low negative impact | Minimal disruption | High | High |
| INP (INPNC) | Moderate negative impact | Moderate disruption | Moderate | Moderate |
| AIDA-I | Moderate negative impact | Moderate disruption | Low | Low |
Key Findings and Interpretation
The data reveal a critical trade-off between display efficiency and cellular health. While Lpp-OmpA and PgsA were most efficient for displaying the large BM3 passenger, Lpp-OmpA's strong negative impact on cell viability is a major drawback for applications requiring healthy, reusable whole-cell biocatalysts [19]. PgsA emerges as a superior anchor in this context, offering high display capacity and activity with minimal membrane disruption. The modular SpyCatcher/SpyTag approach dramatically accelerated this comparative screening, highlighting its utility for optimizing surface display systems.
Insertion Deficiencies in Genetic Diseases and Cancer
Defects in the cellular machinery that inserts proteins into membranes are a direct cause of genetic disease. Similarly, the overexpression of certain translocation-associated proteins can drive cancer progression.
cTM Insertion Deficiencies and Genetic Disease
The post-translational insertion of C-terminal transmembrane helices (cTMs) is a vulnerable point in membrane protein biogenesis. Research shows that the sequences flanking cTMs, particularly the C-terminal tail (C-tail), are evolutionarily fine-tuned to collaborate with the insertase machinery [12].
- Molecular Mechanism: In proteins with an extracytosolic C-terminus (Cext), the C-tail must be translocated across the membrane. A short and hydrophobic C-tail is essential for efficient post-translational insertion by insertases like YidC in bacteria and the ER membrane complex (EMC) in humans [12].
- Disease Link: Mutations that increase the hydrophilicity of the C-tail disrupt the productive interaction with the insertase, leading to cTM misinsertion, protein misfolding, loss of function, and disease. Specific disease-causing mutations in human membrane proteins that operate through this mechanism have been identified [12].
This pathway can be visualized as follows:
TRAM2 Overexpression in Cancer Metastasis
In cancer, the dysregulation of translocation-associated machinery can promote aggressive disease. The translocon-associated membrane protein 2 (TRAM2) is overexpressed in oral squamous cell carcinoma (OSCC) [91].
- Experimental Evidence: In OSCC cell lines and clinical samples, TRAM2 mRNA and protein levels are significantly accelerated. TRAM2-positive OSCC tissues correlate closely with metastasis to regional lymph nodes and vascular invasiveness [91].
- Mechanistic Insight: Functional experiments show that knockdown of TRAM2 inhibits metastatic phenotypes, including cellular migration, invasiveness, and transendothelial migration. This is associated with a significant decrease in protein kinase RNA-like ER kinase (PERK) and key matrix metalloproteinases (MT1-MMP, MMP2, and MMP9), which are enzymes crucial for degrading the extracellular matrix to enable cancer cell invasion [91].
The signaling pathway driven by TRAM2 overexpression can be summarized as:
The Scientist's Toolkit: Key Research Reagents and Methodologies
Research into membrane insertion and its disease links relies on a suite of specialized reagents and experimental protocols.
Table 2: Essential research reagents and methods for studying membrane insertion and display systems.
| Reagent / Method | Function / Purpose | Example Application |
|---|---|---|
| SpyCatcher/SpyTag System | Post-translational, covalent coupling of protein modules; enables modular surface display. | Rapidly comparing different membrane anchors without generating genetic fusions [19]. |
| YidC / EMC Insertase | Facilitates post-translational insertion of transmembrane helices, particularly cTMs. | Studying the biogenesis of multispanning membrane proteins; modeling genetic diseases caused by misinsertion [12]. |
| siRNA / shRNA Knockdown | Silences gene expression to determine protein function. | Validating the role of specific genes (e.g., TRAM2) in cancer metastasis [91]. |
| Cellular Migration & Invasion Assays | Measures the metastatic potential of cancer cells in vitro. | Quantifying changes in invasiveness after knockdown of a target gene like TRAM2 [91]. |
| Western Blotting / qRT-PCR | Detects and quantifies protein and mRNA expression levels. | Confirming overexpression of a target (e.g., TRAM2, MMPs) in disease models [91]. |
The comparative analysis of membrane-anchored and translocation-based systems reveals a common theme: the fidelity of membrane protein biogenesis is paramount to cellular health. Deficiencies in the post-translational insertion of cTMs, driven by miscommunication between protein sequence and insertase machinery, are a direct pathway to genetic disease. Conversely, the hijacking of translocation-associated components, such as TRAM2, can empower cancer metastasis. The experimental frameworks and reagents summarized here provide a roadmap for further dissecting these mechanisms. Future research and drug discovery efforts aimed at correcting insertion deficiencies or targeting overactive translocation systems hold significant promise for treating a range of human diseases.
Membrane transporters are critical gatekeepers that profoundly influence the pharmacokinetics of pharmaceutical compounds. These proteins, which represent approximately a third of all proteins in living organisms and about 60% of approved drug targets, mediate the movement of drugs across biological barriers throughout the body [20]. The process by which these proteins are synthesized, folded, and correctly assembled into membranes—their biogenesis—directly determines their functional capacity and ultimately shapes drug absorption, distribution, and elimination [73] [92]. Within the context of membrane protein research, a fundamental distinction exists between membrane-anchored systems (where proteins are permanently integrated into lipid bilayers) and translocation-based systems (which facilitate the movement of substrates across membranes). Understanding the differences between these systems is essential for predicting drug disposition and designing effective therapeutics.
Transporters are primarily categorized into two superfamilies: the ATP-binding cassette (ABC) transporters, which are efflux pumps that utilize ATP hydrolysis to move substrates against concentration gradients, and the solute carrier (SLC) transporters, which facilitate the cellular uptake of drugs through various mechanisms including facilitated diffusion and secondary active transport [93] [94] [95]. The proper biogenesis of these transporters in key epithelial barriers—such as the intestine, liver, kidney, and blood-brain barrier—ensures they can perform their physiological roles in drug transport. Disruptions in their assembly or trafficking can lead to altered drug exposure, potentially resulting in therapeutic failure or adverse drug reactions [93] [96].
Transporter Biogenesis: From Ribosome to Functional Membrane Protein
Fundamental Biogenesis Pathways
The biogenesis of membrane transporters begins with their synthesis on ribosomes and proceeds through a complex sequence of events that culminates in their integration into target membranes. For integral membrane proteins, this process involves permanent anchorage within the lipid bilayer, with transmembrane proteins spanning the membrane at least once [20]. These proteins are classified as either integral polytopic proteins (spanning the membrane multiple times) or integral monotopic proteins (attached to only one side of the membrane) [20].
Research comparing different biogenesis systems reveals that translocation-based mechanisms must be precisely interrupted to achieve proper membrane integration rather than complete protein secretion [92]. As each transmembrane segment emerges from the ribosome, it sequentially translocates across the membrane, yet integration into the bilayer is often delayed until after protein synthesis is complete [92]. During this critical period, transmembrane segments appear to be stabilized by salt-sensitive electrostatic bonds within an aqueous-accessible compartment before final insertion [92].
Comparative Biogenesis Systems Across Organisms
Recent structural biology advances have revealed striking differences in transporter biogenesis machinery across evolutionary domains, particularly in the β-barrel-assembly machinery (BAM) complex responsible for folding and inserting outer membrane proteins in Gram-negative bacteria.
Table 1: Comparison of BAM Complex Components Across Species
| Component | E. coli (Canonical BAM) | F. johnsoniae (Bacteroidota BAM) | Functional Role |
|---|---|---|---|
| Core Subunit | BamA | BamA | Central folding portal |
| Essential Lipoprotein | BamD | BamD | Complex stability |
| Novel Components | BamB, BamC, BamE | BamG, BamH, BamM, BamP | Species-specific adaptations |
| Extracellular Structures | Minimal | Extensive canopy | Handling complex substrates |
| Membrane Integration | Periplasmic accessory factors | Transmembrane BamG subunit | Enhanced membrane association |
The canonical BAM complex in Escherichia coli consists of BamA plus four periplasmic lipoprotein subunits, with only BamA and BamD being individually essential [73]. In contrast, the BAM complex from Flavobacterium johnsoniae (a member of the Bacteroidota phylum) features a dramatically different organization with five novel proteins in addition to BamA, including BamG (a transmembrane OMP) and BamH/BamM (surface lipoproteins that form an extensive extracellular canopy) [73]. This enhanced complexity in Bacteroidota reflects the need to assemble unusually complex membrane proteins with large extracellular domains that are characteristic of this phylum [73].
The Bacteroidota BAM complex represents a membrane-anchored system with enhanced stabilization, where the interaction between BamA and BamG is reinforced by lipid binding on either side of the protein interface [73]. This creates a more robust structural framework compared to the more dynamic translocation-based systems observed in canonical BAM complexes.
Structural Dynamics and Functional Implications
Once correctly assembled, membrane transporters exhibit specific dynamic behaviors within the membrane environment that directly influence their function. The fluid mosaic model initially proposed that membrane proteins would undergo free translational diffusion, but current understanding reveals that various restriction mechanisms profoundly limit this mobility [97].
Table 2: Modes of Membrane Transporter Dynamics and Functional Consequences
| Mobility Mode | Characteristics | Functional Impact | Example Transporters |
|---|---|---|---|
| Random Diffusion | Brownian motion in homogeneous membrane | Increases interaction opportunities | Some unassociated transporters |
| Confinement | Restricted to specific subdomains | Corralling in functional microdomains | Lipid raft-associated proteins |
| Hop Diffusion | Transient confinement with rare relocation | Balanced localization and mobility | Various SLC transporters |
| Directed Motion | Active transport along scaffolds | Targeted delivery to specific sites | Signaling complex components |
These mobility patterns are governed by multiple factors including: interactions with membrane lipids (particularly cholesterol-enriched membrane microdomains), steric interactions and complex formation with other membrane proteins, binding to intracellular and extracellular structures, and self-organization tendencies that drive hierarchical ordering of membrane proteins [97]. The cytoskeleton frequently governs modes of membrane protein motion by stably anchoring proteins through direct-binding interactions, restricting diffusion through steric interactions, or facilitating directed motion [97].
Methodological Approaches for Studying Transporter Biogenesis and Function
Experimental Systems for Transporter Characterization
Researchers employ multiple complementary approaches to investigate transporter biogenesis and function, each with distinct advantages and limitations for studying membrane-anchored versus translocation-based systems.
Table 3: In Vitro Systems for Transporter Studies
| System Type | Best Applications | Key Advantages | Notable Limitations |
|---|---|---|---|
| Recombinant Cells (HEK293) | Specific SLC transporter interactions | Low background expression; clear mechanistic data | Reduced physiological complexity |
| Inside-Out Membrane Vesicles | ABC efflux transporter studies | Direct transporter access; ATP-dependent transport measurable | Limited for low permeability compounds |
| Polarized Cell Monolayers (MDCK) | ABC transporter substrate studies for medium/high permeability drugs | Models cellular barriers; measures vectorial transport | Unsuitable for low permeability drugs |
| Primary Hepatocytes | Hepatic uptake and biliary excretion | Contains full complement of native transporters | Donor variability; limited lifespan |
| Caco-2 Cells | Intestinal absorption prediction | Enterocyte-like characteristics; models absorption | Long culture time; variable expression |
For the assessment of specific SLC transporter interactions, recombinant cells overexpressing a single transporter (such as HEK293) are preferred due to their human origin and low background endogenous transporter expression [95]. Substrate studies in these systems compare the uptake of investigational drugs into transporter-overexpressing cells relative to control cells, while inhibition studies monitor the uptake of known probe substrates in the presence of the investigational drug [95].
ABC efflux transporters are commonly studied in inside-out membrane vesicles isolated from transporter-expressing recombinant cells, where drugs have direct access to the transporter in the membrane [95]. For medium/high permeability compounds, monitoring flux through polarized cell monolayers transfected to overexpress a single ABC transporter often provides more relevant data [95].
Visualization of Transporter Biogenesis Pathways
The following diagram illustrates the complex pathway of membrane transporter biogenesis, highlighting key steps from synthesis to functional membrane integration:
Membrane Transporter Biogenesis Pathway
Essential Research Reagents and Solutions
The following toolkit represents critical reagents required for investigating membrane transporter biogenesis and function:
Table 4: Research Reagent Solutions for Transporter Studies
| Reagent/Category | Specific Examples | Primary Research Function |
|---|---|---|
| Recombinant Cell Lines | HEK293-OATP1B1, MDCK-II-P-gp | Isolate specific transporter functions; study substrate/inhibitor interactions |
| Transporter-Proteoliposomes | P-gp reconstituted vesicles | Study ATP-dependent transport in defined membrane environment |
| Selective Chemical Inhibitors | Ko143 (BCRP), Rifampicin (OATPs) | Confirm transporter-specific contributions in complex systems |
| Membrane Protein Antibodies | Anti-P-gp monoclonal antibodies | Detect protein expression, localization, and quantification |
| Primary Human Cells | Cryopreserved hepatocytes, renal proximal tubule cells | Assess transporter function in physiologically relevant contexts |
| LC-MS/MS Systems | Quantitative proteomics platforms | Measure transporter abundance and post-translational modifications |
Specialized antibodies against membrane proteins are particularly valuable reagents, with discovery methods ranging from immune antibody library construction by phage display to native antibody discovery by antigen-specific B lymphocytes cytometry technology [20]. These tools enable researchers to detect protein expression, determine cellular localization, and quantify transporter levels in various tissue samples.
Pharmacokinetic Implications of Transporter Biogenesis
Clinical Relevance in Drug Disposition
The proper biogenesis and function of membrane transporters directly impacts all phases of drug pharmacokinetics, with particular importance for drugs that are substrates of specific transporters expressed in pharmacological barrier tissues. The International Transporter Consortium has identified key transporters with strong evidence for roles in drug absorption and disposition, including P-glycoprotein (P-gp), breast cancer resistance protein (BCRP), organic anion-transporting polypeptides (OATP1B1, OATP1B3), and organic cation transporters (OCT2) [93].
In the intestine, ABC transporters such as P-gp and BCRP limit oral drug absorption by actively effluxing substrates back into the intestinal lumen, thereby reducing systemic exposure [94] [95]. In the liver, uptake transporters like OATP1B1 and OATP1B3 mediate the clearance of many drugs from circulation, while efflux transporters such as BCRP and MRP2 facilitate biliary excretion [94]. In the kidney, transporters including OAT1, OAT3, and OCT2 on the basolateral membrane work in concert with apical MATE and P-gp transporters to mediate active renal secretion [94]. The blood-brain barrier expresses high levels of P-gp and BCRP, which restrict central nervous system penetration of many substrates [95].
Experimental Data on Transporter-Drug Interactions
The critical role of transporters in drug disposition is demonstrated by quantitative studies of drug-drug interactions and genetic polymorphisms. The following table summarizes clinically relevant interactions for major transporters:
Table 5: Quantitative Drug-Transporter Interactions and Clinical Impact
| Transporter | Probe Substrate | Inhibitor | Magnitude of Interaction | Clinical Consequence |
|---|---|---|---|---|
| OATP1B1 | Statins (rosuvastatin) | Cyclosporine | 2-7 fold AUC increase | Increased myopathy risk |
| P-gp | Digoxin, dabigatran | Quinidine, verapamil | 1.5-3 fold AUC increase | Potential toxicity |
| BCRP | Sulfasalazine | Curcumin | ~2 fold AUC increase | Altered efficacy/toxicity |
| OAT1/3 | Furosemide, cephradine | Probenecid | ~3 fold AUC increase | Enhanced diuresis |
| OCT2/MATEs | Metformin | Cimetidine | <2 fold AUC increase | Reduced glucose control |
The extended clearance classification system (ECCS) provides a framework for predicting clearance mechanisms using physicochemical properties and in vitro data, helping researchers anticipate which transporters will be most relevant for specific compounds [94]. This classification considers factors such as molecular weight, ionization, and membrane permeability to categorize compounds and predict their predominant clearance pathways [94].
Visualization of Transporter Effects on Drug Disposition
The integrated role of transporters in organ-specific drug disposition can be visualized through the following diagram:
Transporter-Mediated Drug Disposition Pathways
The biogenesis of membrane transporters represents a critical determinant of drug pharmacokinetics, with distinct implications for membrane-anchored systems that provide stable structural frameworks versus translocation-based systems that enable dynamic substrate movement. The proper assembly, folding, and membrane integration of these transport proteins directly influences their functional capacity to mediate drug absorption, distribution, and elimination across key pharmacological barriers.
Current research challenges include the difficulty in expressing functionally folded membrane proteins in sufficient amounts for structural and functional studies, limitations in crystallizing membrane proteins compared to soluble proteins, and the still-incomplete structural information for many clinically relevant transporters [20] [98]. Future research directions should focus on elucidating the complete biogenesis pathways for pharmaceutically important transporters, developing more physiologically relevant in vitro systems that recapitulate native membrane environments, and leveraging structural information to predict and mitigate problematic drug-transporter interactions early in drug development.
As structural biology techniques continue to advance, particularly with developments in cryo-electron microscopy and computational modeling approaches, our understanding of how transporter biogenesis influences drug disposition will continue to grow, enabling more rational drug design and personalized medicine approaches based on individual variations in transporter expression and function [73] [98].
The mechanistic target of rapamycin (mTOR) functions as a central processing unit that integrates nutrient availability, energy status, and growth signals to command cellular biogenesis and membrane trafficking. This master regulatory kinase, operating through two distinct complexes (mTORC1 and mTORC2), directly controls the synthesis, translocation, and organization of cellular membranes and their associated machinery. This review provides a systematic comparison of mTOR-mediated regulatory mechanisms governing membrane dynamics, drawing on experimental data that delineate how mTOR signaling coordinates anabolic processes with nutrient sensing. We examine cutting-edge methodologies for investigating mTOR-dependent pathways and present key reagent solutions for probing the intricate relationship between nutrient availability and membrane biogenesis.
The mammalian target of rapamycin (mTOR) is an evolutionarily conserved serine-threonine kinase that serves as a central integrator of environmental and cellular signals to regulate growth, metabolism, and proliferation [99]. mTOR nucleates at least two structurally and functionally distinct multi-protein complexes: mTOR complex 1 (mTORC1) and mTOR complex 2 (mTORC2) [100]. These complexes process information from growth factors, cellular energy status, oxygen availability, and amino acids to coordinate anabolic and catabolic processes throughout the cell [100] [99].
The regulation of membrane traffic and biogenesis represents a fundamental aspect of mTOR's governance over cellular homeostasis. As cells grow and divide, they must expand their membrane systems, including the endoplasmic reticulum (ER), Golgi apparatus, and plasma membrane. mTOR signaling directly controls the synthesis of lipids, membrane proteins, and the biogenesis of organelles, thereby dictating the scope and composition of cellular membrane networks [101] [102]. This review systematically examines how mTOR and associated nutrient-sensing pathways regulate these processes, with particular emphasis on comparative mechanistic insights and experimental approaches.
mTOR Complexes: Architecture and Regulatory Networks
Structural Organization of mTOR Complexes
The mTOR protein contains several functional domains, including HEAT repeats at the N-terminus, FAT and FRB domains, a catalytic kinase domain, and a FATC domain at the C-terminus [99]. Cryo-electron microscopic analyses reveal that mTOR complexes form hollow rhomboid structures with dimensions of approximately 280 × 210 × 130 ų [99].
mTORC1 comprises five core components: mTOR (catalytic subunit), Raptor (scaffolding protein), mLST8/GβL, PRAS40, and DEPTOR [100] [99]. Raptor facilitates substrate recruitment and complex assembly, while PRAS40 and DEPTOR serve as endogenous inhibitors. The complex displays a symmetric organization with two mTOR and two Raptor subunits forming dimeric interfaces [99].
mTORC2 contains six core components: mTOR, Rictor, mLST8/GβL, mSIN1, Protor-1/2, and DEPTOR [100] [99]. Rictor and mSIN1 establish the structural foundation of mTORC2 and determine its substrate specificity. This complex forms a hollow rhombohedral structure (220 × 200 × 130 Å) with two copies of each component binding symmetrically to the mTOR dimer [99].
Table 1: Core Components of mTOR Complexes and Their Functions
| Component | mTORC1 | mTORC2 | Primary Functions |
|---|---|---|---|
| mTOR | ✓ | ✓ | Catalytic kinase subunit |
| Raptor | ✓ | Substrate recruitment, complex stability | |
| Rictor | ✓ | Structural component, substrate recognition | |
| mLST8/GβL | ✓ | ✓ | Kinase domain stabilization |
| DEPTOR | ✓ | ✓ | Endogenous inhibitor of both complexes |
| PRAS40 | ✓ | mTORC1-specific inhibitor | |
| mSIN1 | ✓ | mTORC2 structural integrity, substrate specificity | |
| Protor-1/2 | ✓ | Regulation of SGK1 phosphorylation |
Upstream Regulation of mTOR Signaling
mTORC1 integrates signals from multiple upstream pathways:
- Growth factors activate mTORC1 through the PI3K-AKT pathway, which phosphorylates and inhibits TSC2, leading to RHEB-GTP accumulation and mTORC1 activation [99].
- Amino acids activate mTORC1 via Rag GTPases and Ragulator complex, recruiting mTORC1 to lysosomal membranes where RHEB resides [103].
- Cellular energy status (AMP/ATP ratio) regulates mTORC1 through AMPK, which phosphorylates both TSC2 and Raptor under low energy conditions [104].
- Oxygen availability affects mTORC1 through regulation of HIF-1α and REDD1 expression during hypoxia [99].
mTORC2 is primarily activated by growth factors through PI3K signaling but appears less responsive to nutrient status [99]. Insulin and IGF-1 stimulate mTORC2-mediated phosphorylation of AKT at Ser473, facilitating full AKT activation.
The following diagram illustrates the core mTOR signaling pathway and its upstream regulation:
mTOR Regulation of Membrane Traffic and Organelle Biogenesis
Endoplasmic Reticulum and Protein Biogenesis Machinery
The endoplasmic reticulum serves as the primary site for synthesis of secretory and membrane proteins. Recent cryo-electron tomography studies have revealed the intricate organization of the ribosome-translocon complex at the ER membrane, where mTOR signaling exerts profound influence [7]. The dynamic ribosome-translocon complex produces a major fraction of the human proteome, governing synthesis, translocation, membrane insertion, N-glycosylation, folding, and disulfide-bond formation of nascent proteins [7].
mTORC1 controls the translation of nucleus-encoded mitochondrial proteins and ER-related biosynthetic factors through its downstream targets 4E-BP1 and S6K1 [102]. Phosphorylation of 4E-BP1 by mTORC1 prevents its binding to eIF4E, enabling cap-dependent translation of mRNAs encoding secretory pathway components [100] [99]. This mechanism directly links nutrient sensing to the production of ER machinery and membrane proteins.
Table 2: mTOR-Dependent Processes in ER and Membrane Biogenesis
| Process | Regulatory Mechanism | Functional Outcome |
|---|---|---|
| Protein Translation | mTORC1 phosphorylates 4E-BP1, releasing eIF4E for cap-dependent translation | Increased synthesis of ER cargo proteins and membrane receptors |
| Translocon Organization | mTOR-dependent translation controls abundance of SEC61, TRAP, and OST complex components | Regulation of protein translocation efficiency and membrane insertion |
| ER Chaperone Expression | mTORC1 modulates translation of BiP, calnexin, and protein disulfide isomerases | Enhanced folding capacity and quality control in ER lumen |
| Membrane Lipid Synthesis | mTORC1 activates SREBPs, increasing expression of lipid biosynthetic enzymes | Expansion of ER membrane surface area and lipid bilayer production |
Mitochondrial Biogenesis and Function
mTORC1 serves as a critical regulator of mitochondrial activity and biogenesis through 4E-BP-mediated control of nucleus-encoded mitochondrial gene translation [102]. This mechanism establishes a feed-forward loop that links mRNA translation to oxidative phosphorylation, providing essential energy for ongoing biosynthetic processes [102].
Key mitochondrial processes under mTOR control include:
- Oxidative phosphorylation: mTORC1 stimulates mitochondrial oxygen consumption and ATP production capacity [104] [102].
- Mitochondrial gene expression: mTORC1 promotes expression of electron transport chain components and mitochondrial ribosomal proteins [104].
- Mitochondrial dynamics: mTORC1 regulates mitochondrial fusion and fission processes, influencing network morphology and function [104].
- Mitophagy: mTORC1 inhibits autophagy, including selective removal of damaged mitochondria, through phosphorylation of ULK1 and regulation of the ATG13-FIP200-ULK1 complex [100].
The coordination between mitochondrial biogenesis and membrane trafficking is particularly evident in the provision of ATP for vesicle formation, fusion, and protein processing within the secretory pathway.
Lipid Synthesis and Membrane Architecture
mTORC1 directly regulates cellular lipid homeostasis by controlling the activity of sterol regulatory element-binding proteins (SREBPs), key transcription factors governing fatty acid, phospholipid, and cholesterol synthesis [101] [99]. Through SREBP activation, mTORC1 promotes the expression of enzymes involved in lipid biosynthesis, thereby influencing membrane composition and physical properties.
Recent research reveals that mTOR inhibition reprograms cellular lipid homeostasis through unexpected mechanisms. When mTOR activity is suppressed, cells activate alternative lipid uptake pathways and promote cholesterol transport systems to maintain membrane integrity [101]. Specifically, mTOR inhibition increases low-density lipoprotein receptor-related protein 6 (LRP6), enhancing lipid uptake and activating survival signaling despite reduced lipid biosynthesis [101].
The following diagram illustrates mTOR regulation of organelle biogenesis and membrane trafficking:
Experimental Comparison: Methodologies for Investigating mTOR in Membrane Dynamics
Modular Display Systems for Membrane Protein Studies
A modular surface display system employing SpyCatcher/SpyTag bioconjugation technology enables rapid comparison of different membrane anchors without generating direct genetic fusions for each combination [19]. This approach significantly reduces the workload for identifying optimal membrane anchors for specific applications.
Experimental Protocol: Modular Surface Display System
- Construct Design: Generate separate expression vectors for:
- Passenger protein fused to SpyTag
- Membrane anchors (Lpp-OmpA, PgsA, INP, AIDA-I) fused to SpyCatcher
- Co-expression: Transform E. coli with both constructs and induce protein expression
- Surface Assembly: SpyCatcher and SpyTag spontaneously form covalent isopeptide bonds, displaying passenger on surface
- Functional Assessment: Measure display efficiency, cell growth, membrane integrity, and passenger functionality
Table 3: Comparison of Membrane Anchors in Modular Display Systems
| Membrane Anchor | Structure Features | Display Efficiency | Impact on Cell Viability | Passenger Size Limit |
|---|---|---|---|---|
| Lpp-OmpA | Hybrid anchor: Lpp signal peptide + OmpA transmembrane domain | High | Significant impact on viability | >119 kDa (BM3 cytochrome) |
| PgsA | Bacillus subtilis membrane protein for poly-γ-glutamate synthesis | High | Minimal impact on viability | Previously <77 kDa, now >119 kDa |
| INP (INPNC) | Ice nucleation protein with N/C-terminal domains, minimal central repeats | Moderate | Minimal impact on viability | >119 kDa (BM3 cytochrome) |
| AIDA-I | Autotransporter with β-barrel channel for passenger transport | Moderate | Moderate impact on viability | >119 kDa (BM3 cytochrome) |
mTOR Inhibition Studies and Membrane Effects
Experimental approaches using mTOR inhibitors have revealed crucial insights into how nutrient sensing regulates membrane dynamics:
Protocol: mTOR Inhibition and Lipid Homeostasis Assessment
- Treatment Conditions:
- First-generation inhibitors: Rapamycin (mTORC1-specific)
- Second-generation: Torin1 (mTORC1/2 inhibitor)
- Third-generation: RapaLink-1 (mTORC1/2 inhibitor)
- Duration Considerations: Acute (2-6 hours) vs. chronic (24-72 hours) treatment
- Lipidomic Analysis:
- Extract lipids using methyl-tert-butyl ether/methanol/water system
- Perform LC-MS/MS with multiple reaction monitoring
- Quantify phospholipids, cholesterol, and neutral lipids
- Protein Analysis:
- Western blotting for p-S6K1 (T389), p-4E-BP1 (S65), p-AKT (S473)
- Immunofluorescence for receptor localization
- Functional Assays:
- LDL uptake measurements using fluorescent conjugates
- Membrane fluidity assessment with pyrene-decanoic acid
- Lipid raft isolation via detergent-resistant membrane fractionation
Experimental data demonstrates that mTOR inhibition decreases proteins involved in lipid biosynthesis but activates compensatory lipid uptake through LRP6 and promotes cholesterol transport, enabling cells to maintain membrane integrity despite metabolic stress [101].
Research Reagent Solutions for mTOR and Membrane Traffic Studies
Table 4: Essential Research Reagents for mTOR-Membrane Studies
| Reagent Category | Specific Examples | Research Applications | Key Features |
|---|---|---|---|
| mTOR Inhibitors | Rapamycin (Sirolimus), Temsirolimus, Everolimus, Torin1, INK128, RapaLink-1 | Pharmacological modulation of mTOR signaling | Varying specificity for mTORC1 vs. dual mTORC1/2 inhibition |
| Antibodies for Western Blot | p-S6K1 (T389), p-4E-BP1 (S65), p-AKT (S473), total AKT, SREBP1, LDLR | Detection of mTOR activity and downstream effectors | Validate mTOR pathway inhibition/activation |
| Modular Display System Components | SpyCatcher (113 aa), SpyTag (13 aa), Lpp-OmpA, PgsA, INP, AIDA-I anchors | Surface display of membrane-associated proteins | Covalent isopeptide bonding without genetic fusion |
| Lipid Metabolism Assays | Filipin III (cholesterol staining), Bodipy-labeled lipids, LDL-Dylight conjugates | Visualization and quantification of lipid uptake and distribution | Monitor lipid trafficking and membrane composition |
| Cell Lines and Models | HEK293T (high transfection efficiency), FaDu (HNSCC model), MEFs with mTOR mutations | In vitro assessment of mTOR signaling | Genetic tractability and pathway characterization |
The system-wide interdependence between mTOR signaling and membrane biogenesis represents a fundamental regulatory axis coordinating cellular growth with nutrient availability. Through its governance of protein synthesis, lipid metabolism, and organelle biogenesis, mTOR serves as a master conductor of membrane traffic and composition. The experimental approaches and reagent solutions outlined here provide researchers with robust methodologies for investigating these complex relationships.
Future research directions should focus on:
- Developing more precise temporal control over mTOR activity to dissect acute versus chronic effects on membrane dynamics
- Elucidating mTOR's role in specialized membrane domains such as lipid rafts and contact sites between organelles
- Exploring cell-type-specific differences in mTOR regulation of membrane homeostasis
- Investigating the therapeutic potential of targeting mTOR-membrane interactions in disease contexts
As our understanding of mTOR-mediated regulation of membrane traffic deepens, new opportunities will emerge for modulating these processes in metabolic diseases, cancer, and aging-related conditions where mTOR signaling is frequently dysregulated.
Conclusion
The comparison between membrane-anchored and translocation-based systems reveals a deeply integrated and evolutionarily conserved framework for membrane protein biogenesis. The choice between the Oxa1 pathway for short-tail substrates and the SecY pathway for long-tail substrates is not merely sequential but represents a fundamental triage mechanism that expands the functional diversity of the membrane proteome. Key takeaways include the critical role of sequence features in directing this choice, the direct link between insertion defects and human disease, and the emerging potential of these pathways as therapeutic targets. Future research must focus on obtaining high-resolution structures of insertion intermediates, developing more specific modulators of these pathways, and elucidating how cellular metabolic status, via pathways like mTOR, regulates the entire membrane protein biogenesis network. These advancements will profoundly impact the development of treatments for protein misfolding diseases, cancer, and infectious diseases, and refine drug development pipelines by accounting for transporter biogenesis.
References
- 1. YidC/Alb3/Oxa1 Family of Insertases [https://www.sciencedirect.com/science/article/pii/S0021925820443583]
- 2. YidC/Alb3/Oxa1 Family of Insertases [https://pubmed.ncbi.nlm.nih.gov/25947384/]
- 3. Review The multifaceted mitochondrial OXA insertase [https://www.sciencedirect.com/science/article/pii/S096289242300020X]
- 4. The ER membrane complex (EMC) can functionally replace ... [https://journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.3001380]
- 5. Oxa1p acts as a general membrane insertion machinery for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC145526/]
- 6. SecA-Mediated Protein Translocation through the SecYEG ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10957188/]
- 7. Visualization of translation and protein biogenesis at the ... [https://www.nature.com/articles/s41586-022-05638-5]
- 8. SecY-mediated quality control prevents the translocation of ... [https://www.nature.com/articles/s41598-020-73185-y]
- 9. ZapE/Afg1 interacts with Oxa1 and its depletion causes a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7314023/]
- 10. Bacterial Secretion Systems – An overview - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4804464/]
- 11. SecY protein [https://en.wikipedia.org/wiki/SecY_protein]
- 12. Features of membrane protein sequence direct post-translational insertion | Nature Communications [https://www.nature.com/articles/s41467-024-54575-6]
- 13. Review The protein-conducting channel SecYEG [https://www.sciencedirect.com/science/article/pii/S0167488904000916]
- 14. Protein translocation - Latest research and news | Nature [https://www.nature.com/subjects/protein-translocation]
- 15. Hydrophobicity, rather than secondary structure, is essential ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6439181/]
- 16. Protein translocation across membranes [https://pubmed.ncbi.nlm.nih.gov/2842866/]
- 17. Membrane translocation of folded proteins - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9251779/]
- 18. The Sec translocase [https://www.sciencedirect.com/science/article/pii/S000527361000297X]
- 19. A Modular System for the Rapid Comparison of Different ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9298812/]
- 20. Membrane Protein Overview [https://www.creative-biolabs.com/blog/index.php/membrane-protein-overview/]
- 21. Mechanism of Protein Translocation by the Sec61 Translocon ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9808579/]
- 22. YidC/Alb3/Oxa1 Family of Insertases - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC4463434/]
- 23. EMC rectifies the topology of multipass membrane proteins [https://www.nature.com/articles/s41594-023-01120-6]
- 24. Structural analysis of the dynamic ribosome-translocon ... [https://elifesciences.org/reviewed-preprints/95814]
- 25. The mechanisms of integral membrane protein biogenesis [https://pmc.ncbi.nlm.nih.gov/articles/PMC12108547/]
- 26. Lipid-Dependent Membrane Protein Topogenesis - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3033430/]
- 27. Lipids and topological rules governing membrane protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4057987/]
- 28. Lipids and topological rules governing membrane protein ... [https://www.sciencedirect.com/science/article/pii/S0167488913004266]
- 29. Global analysis of translocon remodeling during protein ... [https://www.nature.com/articles/s41594-025-01691-6]
- 30. Determining the N-terminal orientations of recombinant ... [https://www.nature.com/articles/srep15086]
- 31. SecY translocon chaperones protein folding during ... [https://www.sciencedirect.com/science/article/abs/pii/S0092867425001060]
- 32. Computational design of soluble and functional membrane ... [https://www.nature.com/articles/s41586-024-07601-y]
- 33. The cryo-EM method microcrystal electron diffraction (MicroED) [https://pmc.ncbi.nlm.nih.gov/articles/PMC6568260/]
- 34. Better, Faster, Cheaper: Recent Advances in Cryo–Electron ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10393189/]
- 35. Beyond protein structure determination with MicroED - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC7321661/]
- 36. MicroED: a versatile cryoEM method for structure ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6112783/]
- 37. Studying membrane proteins with MicroED - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9022970/]
- 38. A robust approach for MicroED sample preparation of ... [https://www.nature.com/articles/s41467-023-36733-4]
- 39. MicroED vs Cryo-EM SPA: Key Differences and Applications [https://www.creative-biostructure.com/resource-microed-vs-cryo-em-spa.htm?srsltid=AfmBOoqmXDEInuO1sTolxR1TB-zTY8YmBl1K3c-ejKLa2tzGxo7KLneL]
- 40. MicroED - Gonen Lab - UCLA [https://cryoem.ucla.edu/research/microed]
- 41. Energy filtering enables macromolecular MicroED data at ... [https://www.nature.com/articles/s41467-025-57425-1]
- 42. Biophysical Characterization of Membrane Proteins ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9028781/]
- 43. The application of nanodiscs in membrane protein drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11445163/]
- 44. Biophysical characterization of membrane proteins in ... [https://www.sciencedirect.com/science/article/abs/pii/S1046202312003015]
- 45. DeFrND: detergent-free reconstitution into native ... [https://www.nature.com/articles/s41467-025-63275-8]
- 46. Encoding extracellular modification of artificial cell ... [https://www.nature.com/articles/s41467-024-53783-4]
- 47. Translocon [https://en.wikipedia.org/wiki/Translocon]
- 48. A common mechanism of Sec61 translocon inhibition by ... [https://www.nature.com/articles/s41589-023-01337-y]
- 49. Inhibition of Sec61-dependent translocation by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5852046/]
- 50. A common mechanism of Sec61 translocon inhibition by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11458068/]
- 51. High-yield membrane protein expression from E. coli using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3610049/]
- 52. High-throughput expression and purification of membrane ... [https://www.sciencedirect.com/science/article/abs/pii/S1047847710001176]
- 53. Engineering expression and function of membrane proteins [https://www.sciencedirect.com/science/article/abs/pii/S1046202317304449]
- 54. A concise guide to choosing suitable gene expression ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10643540/]
- 55. Engineering protein translocation pathway to improve ... [https://www.sciencedirect.com/science/article/pii/S259026282400008X]
- 56. Membrane protein expression and purification [https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-expression/membrane-protein-expression.html]
- 57. Cell surface display of a protein based on a tail-anchored ... [https://www.sciencedirect.com/science/article/abs/pii/S0006291X25004528]
- 58. Molecular basis for the regulation of membrane proteins ... [https://www.nature.com/articles/s41589-025-02032-w]
- 59. Applications of synthetic biology in medical and ... [https://www.nature.com/articles/s41392-023-01440-5]
- 60. Misfolded Protein Aggregates: Mechanisms, Structures and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3175247/]
- 61. Misfolding and aggregation in neurodegenerative diseases [https://pmc.ncbi.nlm.nih.gov/articles/PMC11365204/]
- 62. Molecular Mechanisms of TDP-43 Misfolding and ... [https://www.frontiersin.org/journals/molecular-neuroscience/articles/10.3389/fnmol.2019.00025/full]
- 63. Protein Misfolding and Aggregation as a Mechanistic Link ... [https://www.mdpi.com/1467-3045/47/4/259]
- 64. Protein translocation across the ER membrane [https://www.sciencedirect.com/science/article/pii/S0005273610002130]
- 65. The Tat System for Membrane Translocation of Folded ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3431631/]
- 66. C-terminal tail length guides insertion and assembly of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7667985/]
- 67. Characterizing and engineering post-translational ... [https://www.nature.com/articles/s41467-025-60526-6]
- 68. Leveraging AI to explore structural contexts of post ... [https://jcheminf.biomedcentral.com/articles/10.1186/s13321-025-01019-y]
- 69. Membrane and Chaperone Recognition by the Major ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3916559/]
- 70. The landscape of molecular chaperones across human ... [https://www.nature.com/articles/s41467-021-22369-9]
- 71. Bypassing BamD essentiality by mutations in a non-essential ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12421809/]
- 72. The cytoprotective co-chaperone, AtBAG4, supports ... [https://www.nature.com/articles/s41598-023-45771-3]
- 73. A new paradigm for outer membrane protein biogenesis in ... [https://www.nature.com/articles/s41586-025-09532-8]
- 74. Hurdles and advancements in experimental membrane ... [https://pubmed.ncbi.nlm.nih.gov/41027549/]
- 75. Advances in solubilization and stabilization techniques for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11974516/]
- 76. Smoothing membrane protein structure determination by ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7079970/]
- 77. Pendant-bearing detergents: strategies and membrane ... [https://www.sciencedirect.com/science/article/abs/pii/S2589597425001807]
- 78. Designer artificial environments for membrane protein ... [https://www.nature.com/articles/s41467-025-59471-1]
- 79. Identification of YidC Residues That Define Interactions with ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3911256/]
- 80. Molecular communication of the membrane insertase YidC ... [https://www.nature.com/articles/s41598-021-83224-x]
- 81. Structural and mechanistic basis of the EMC-dependent ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7785296/]
- 82. The Role of EMC during Membrane Protein Biogenesis [https://www.sciencedirect.com/science/article/abs/pii/S0962892419300182]
- 83. Extensive disruption of protein interactions by genetic ... [https://www.nature.com/articles/s41467-019-11959-3]
- 84. Membrane translocation process revealed by in situ ... [https://www.nature.com/articles/s41467-023-39583-2]
- 85. New AI tool predicts protein-protein interaction mutations in ... [https://www.lerner.ccf.org/news/article/?title=New+AI+tool+predicts+protein-protein+interaction+mutations+in+hundreds+of+diseases&id=8f8ae79a0e14e0f074307238dd2423b74f3ee925]
- 86. DisruPPI: structure-based computational redesign ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6022686/]
- 87. A Comprehensive Comparison of Transmembrane Domains ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2928124/]
- 88. The EMC acts as a chaperone for membrane proteins [https://www.nature.com/articles/s41467-025-62109-x]
- 89. Membrane Anchoring and Interaction between ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3064186/]
- 90. Mega-scale experimental analysis of protein folding ... [https://www.nature.com/articles/s41586-023-06328-6]
- 91. Overexpression of Translocation Associated Membrane ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6160669/]
- 92. Article Biogenesis of Polytopic Membrane Proteins ... [https://www.sciencedirect.com/science/article/pii/S0092867400811162]
- 93. Membrane transporters in drug development - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC3326076/]
- 94. Navigating Transporter Sciences in Pharmacokinetics ... [https://www.sciencedirect.com/science/article/abs/pii/S0090955624055417]
- 95. Drug transporter studies in vitro – understanding ... [https://admescope.com/drug-transporter-studies-in-vitro-understanding-drug-disposition-and-interactions/]
- 96. Development of Human Membrane Transporters: Drug ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4823323/]
- 97. Membrane Protein Dynamics and Functional Implications in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4193470/]
- 98. The Important Role of Transporter Structures in Drug ... [https://www.sciencedirect.com/science/article/abs/pii/S0090955624010638]
- 99. Multifaceted role of mTOR (mammalian target of rapamycin ... [https://www.nature.com/articles/s41392-023-01608-z]
- 100. mTOR signaling at a glance - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC2758797/]
- 101. mTOR inhibition reprograms cellular lipid homeostasis by ... [https://www.sciencedirect.com/science/article/abs/pii/S1097276525007063]
- 102. mTORC1 Controls Mitochondrial Activity and Biogenesis ... [https://www.sciencedirect.com/science/article/pii/S1550413113004130]
- 103. New perspectives on mTOR inhibitors (rapamycin, rapalogs ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5061789/]
- 104. mTORC1 as a Regulator of Mitochondrial Functions and a ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6923780/]