Optimized Embryo Tissue Dissociation: A 2025 Guide to High-Viability Single-Cell Suspensions
This article provides a comprehensive guide for researchers and drug development professionals on dissociating embryonic tissue into high-quality single-cell suspensions.
Optimized Embryo Tissue Dissociation: A 2025 Guide to High-Viability Single-Cell Suspensions
Abstract
This article provides a comprehensive guide for researchers and drug development professionals on dissociating embryonic tissue into high-quality single-cell suspensions. Covering foundational principles to advanced applications, it details established enzymatic and mechanical methods alongside groundbreaking non-enzymatic and contactless technologies like Hypersonic Levitation. The content offers direct protocols, troubleshooting for common issues such as low viability and aggregation, and a comparative analysis of modern platforms. Finally, it outlines rigorous validation techniques to ensure cell integrity for downstream applications including single-cell RNA sequencing, organoid culture, and drug screening, positioning this technique as a cornerstone for advancements in regenerative medicine and developmental biology.
Understanding Embryonic Tissue Architecture and Dissociation Fundamentals
The Critical Role of Single-Cell Suspensions in Developmental Biology and Drug Discovery
The isolation of high-quality single-cell suspensions represents a foundational prerequisite for modern developmental biology and drug discovery pipelines. Tissues are complex systems of cells that display a high degree of heterogeneity, comprising multiple cell types that behave very differently with a high degree of heterogeneity even within populations of the same cell type [1]. The process of preparing single-cell suspensions enables researchers to characterize this cellular heterogeneity, which has led to the field of single-cell analysis [1]. For developmental biology, where understanding lineage commitment and cellular differentiation is paramount, and for drug discovery, where identifying rare cell populations and their responses to therapeutic intervention is crucial, the ability to effectively dissociate tissues into viable single cells is indispensable. This technical capability serves as the critical gateway to powerful single-cell technologies including flow cytometry, single-cell RNA sequencing, and high-content screening platforms that are revolutionizing both basic and translational research.
The current bottleneck in manufacturing of tissue-engineered and cell-based regenerative medicine therapies or single-cell isolation for downstream applications is the lack of rigorous, standardized, and validated systems that enable the reproducible dissociation of tissues into highly purified cell population(s) [1]. Conventional methods face significant challenges regarding viability, yield, long processing times, as well as the potential for the processing to create artifacts that can distort downstream analyses [1]. This application note examines the latest advancements in tissue dissociation technologies, provides detailed protocols optimized for diverse tissue types, and presents a framework for implementing these methods within developmental biology and drug discovery contexts, with special consideration for the unique challenges of embryonic tissues.
Technological Advances in Tissue Dissociation
Current State of Dissociation Technologies
Traditional tissue dissociation methods rely primarily on enzymatic and mechanical approaches. Enzymatic methods utilize cocktails containing collagenase, trypsin, dispase, papain, hyaluronidase, or other proteases to digest extracellular matrix components and intercellular junctions [1]. These are typically combined with mechanical mincing of the tissue and agitation of the resultant tissue fragments [1]. While widely used, these conventional approaches present several significant drawbacks: enzymatic digestion can require hours or even overnight processing, limiting analytical speed and increasing contamination risks; enzymes can damage cell surface proteins and reduce viability; and the heterogeneous nature of tissues has led to independently developed protocols with little standardization [1].
Recent technological innovations have focused on addressing these limitations through three primary avenues: optimization of traditional enzymatic methods, adaptation of these methods to microfluidic platforms, and development of non-enzymatic alternatives [1]. The table below summarizes the performance characteristics of various dissociation technologies across different tissue types:
Table 1: Performance Comparison of Tissue Dissociation Technologies
| Technology | Tissue Type | Dissociation Efficacy | Cell Viability | Processing Time | Key Advantages |
|---|---|---|---|---|---|
| Enzymatic + Mechanical | Bovine Liver Tissue | 92% ± 8% | >90% | 15 min | Established protocol, cost-effective [1] |
| Optimized Enzymatic | Human Breast Cancer | 2.4 × 10⁶ viable cells | 83.5% ± 4.4% | >1 h | High cell yield [1] |
| Mixed Modal Microfluidic | Mouse Kidney | ~20,000 cells/mg (epithelial) | ~95% (epithelial) | 1-60 min | Cell type-specific efficiency [1] |
| Electric Field Dissociation | Human Glioblastoma | >5× higher than traditional methods | ~80% | 5 min | Rapid, enzyme-free [1] |
| Ultrasound Dissociation | Bovine Liver | 72% ± 10% (with enzyme) | 91%-98% | 30 min | Enhanced enzyme penetration [1] |
| Hypersonic Levitation & Spinning (HLS) | Human Renal Cancer | 90% tissue utilization | 92.3% | 15 min | Non-contact, preserves rare cells [2] |
Emerging and Innovative Dissociation Platforms
Several cutting-edge technologies show particular promise for addressing the challenges of embryonic tissue dissociation:
Hypersonic Levitation and Spinning (HLS) represents a revolutionary contact-free tissue dissociation approach that capitalizes on a uniquely designed triple-acoustic resonator probe. This technology enables target tissue samples to levitate and execute a 'press-and-rotate' operation within a confined flow field, generating microscale 'liquid jets' that exert precise hydrodynamic forces in a non-contact manner [2]. Through this mechanism, shear forces on the tissue are enhanced, facilitating rapid and efficient dissociation while safeguarding cell integrity. Comprehensive experiments on human renal cancer tissue dissociation demonstrate that HLS greatly outperforms traditional techniques in tissue utilization (90% in 15 minutes vs. 70% in 60 minutes) and excels in maintaining high cell viability (92.3%) while preserving rare cell populations [2].
Microfluidic Adaptations have transformed tissue dissociation by enabling precise control over mechanical forces, reducing reagent usage, and automating workflows. These systems can integrate multiple dissociation mechanisms including enzymatic, mechanical, and electrical methods in a single platform [1]. Modern microfluidic systems have evolved beyond simple channel-based designs to incorporate sophisticated droplet generation and piezoelectric sorting, often with real-time AI-guided selection capabilities [3]. The mixed modal microfluidic platform demonstrates exceptional performance across multiple tissue types, achieving 60%-90% viability for different cell populations with processing times of 20-60 minutes [1].
AI-Enhanced Cell Sorting & Isolation has introduced adaptive, intelligent approaches to single-cell processing. Morphology-based intelligent sorting can now identify cells using subtle morphological features without fluorescent labels, preserving cellular integrity while revealing new biological states [3]. Predictive cell state analysis uses machine learning algorithms to analyze high-dimensional data in real-time, predicting cellular states beyond what current markers can detect—particularly valuable for isolating rare subpopulations with developmental or therapeutic significance [3].
Detailed Experimental Protocols
Enzymatic-Mechanical Dissociation for Complex Tissues
This optimized protocol combines enzymatic digestion with mechanical disruption to achieve high yields of viable single cells from complex tissues, adaptable for embryonic tissues:
Table 2: Research Reagent Solutions for Tissue Dissociation
| Reagent | Function | Example Application | Considerations |
|---|---|---|---|
| Collagenase IV | Digests collagen in extracellular matrix | Lung tissue, tumor dissociation [4] | Concentration typically 0.2-1 mg/mL; tissue-specific optimization required |
| DNase I | Degrades DNA released by damaged cells | Prevents cell clumping [4] | Use at 0.05 mg/mL; especially important for necrotic tissues |
| Papain | Cysteine protease for gentle dissociation | Retinal tissue [5] | Superior for preserving cell surface epitopes |
| EDTA | Chelating agent for cell-cell junctions | Often combined with trypsin [1] | Disrupts calcium-dependent cell adhesion |
| Ficoll-Paque | Density gradient medium | Mononuclear cell isolation [4] | Enriches for specific cell populations |
| RBC Lysis Buffer | Lyses red blood cells | Hematopoietic tissues [6] | Critical for blood-rich tissues |
Step-by-Step Protocol:
- Tissue Collection and Preservation: Place freshly isolated tissue in a flat-bottom 6-well cell culture plate with 2 mL of cold flow media (RPMI 1640 + 10% FBS + 1% Penicillin/Streptomycin + 1% L-Glutamine) until processing [6].
- Preparation of Enzymatic Digestion Buffer: Prepare digestion buffer consisting of RPMI 1640 containing 10% FBS supplemented with 0.2 mg/mL Collagenase IV and 0.05 mg/mL DNase I [4].
- Tissue Mincing: Transfer tissue to a petri dish and mince thoroughly with sterile scissors into approximately 1-2 mm fragments [4].
- Enzymatic Digestion: Transfer minced tissue to a 15 mL conical tube containing 6 mL of working enzyme concentration. Incubate in a shaking water bath at 37°C for 20-45 minutes (tissue-dependent) [6].
- Mechanical Disruption: At 10-minute intervals, vortex the tube and pipette up and down with a Pasteur pipette to further break up tissue [6].
- Filtration and Cell Collection: Transfer the suspension through a 70 μm cell strainer into a 50 mL tube, rinsing with DPBS or flow media to maximize cell recovery [6].
- Cell Washing and Counting: Centrifuge at 1800 RPM for 8 minutes at 4°C, decant supernatant, and resuspend pellet in 7 mL of flow media. Count cells using Trypan Blue or AO/PI staining [5] [6].
Assessment of Single-Cell Suspension Quality
Proper assessment of single-cell suspension quality is critical for downstream applications. Research indicates that Acridine Orange/Propidium Iodide (AO/PI) staining enables more rapid and precise evaluation of retinal single-cell suspensions compared to Trypan Blue (TPB) staining [5]. Flow cytometric analysis has shown that single-cell suspensions dispersed with papain and trypsin exhibit reduced cell adhesion, though trypsin digestion may affect antibody binding in some applications [5].
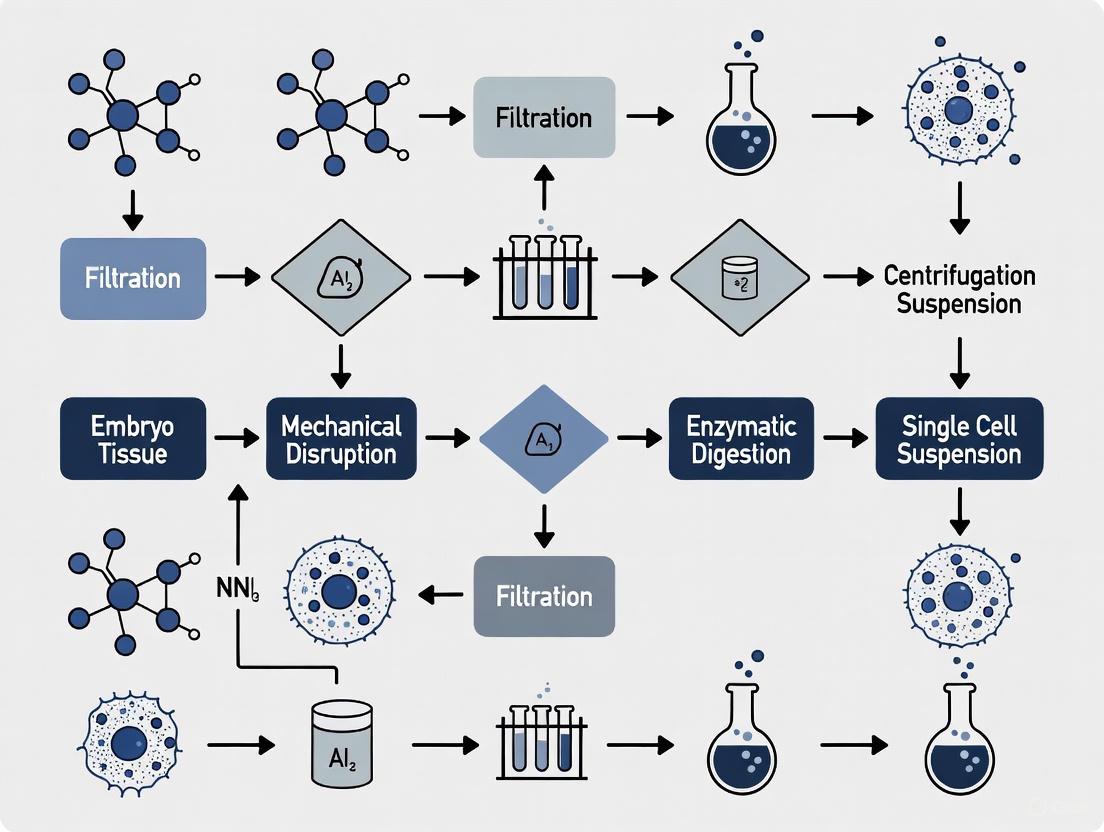
Diagram 1: Tissue Dissociation and Quality Control Workflow. This workflow outlines the critical steps for obtaining high-quality single-cell suspensions, with emphasis on quality assessment parameters essential for downstream applications.
Specialized Protocol for Sensitive Tissues
For particularly sensitive tissues such as embryonic structures or neural tissues, a modified approach using papain digestion has demonstrated superior results:
- Gentle Dissociation with Papain: Prepare papain solution according to manufacturer recommendations. Incubate minimally minced tissue for 20-30 minutes at 37°C with gentle agitation [5].
- Inhibition of Enzymatic Activity: After digestion, carefully remove papain solution and replace with inhibitor solution containing albumin and protease inhibitors.
- Trituration: Gently triturate tissue using fire-polished Pasteur pipettes with progressively smaller openings.
- Optional Density Gradient: For removal of debris and dead cells, layer cell suspension over 70% Percoll and centrifuge at 1500 RPM for 30 minutes at 4°C with brake disabled [6].
- Cell Collection: Collect cells from the interface, wash with PBS, and resuspend in appropriate buffer for downstream applications.
Applications in Developmental Biology and Drug Discovery
Single-Cell Analysis in Developmental Biology
The application of single-cell suspension technologies to embryonic tissues has revolutionized our understanding of developmental processes. By enabling comprehensive profiling of individual cells throughout embryogenesis, researchers can now reconstruct developmental trajectories, identify novel progenitor populations, and decipher the molecular mechanisms governing lineage specification. For embryonic research specifically, the preservation of rare progenitor populations and maintenance of native transcriptional states is paramount, necessitating particularly gentle dissociation approaches.
Recent advances in spatial transcriptomics-integrated isolation allow researchers to maintain architectural context while still achieving single-cell resolution [3]. Enhanced Laser Capture Microdissection now offers subcellular precision with integrated RNA preservation, enabling investigation of subcellular transcript localization within specific embryonic regions [3]. These approaches are particularly valuable for understanding patterning and morphogenetic events where spatial context is functionally significant.
Applications in Drug Discovery and Development
In pharmaceutical research, quality single-cell suspensions enable high-content screening, mechanism of action studies, and biomarker discovery. The ability to profile cell-to-cell heterogeneity in response to compound treatment reveals subpopulations of responsive and non-responsive cells, potentially identifying resistance mechanisms early in development. For immune-oncology applications, monitoring changes in immune cell composition and activation states within tumors following treatment provides critical insights into therapeutic efficacy and potential combination strategies.
Single-cell RNA sequencing has become particularly powerful for clinical biomarker studies, with recent comparisons showing that technologies from 10× Genomics, PARSE Biosciences, and HIVE successfully capture transcriptomes of sensitive cell populations like neutrophils, which were previously challenging to profile [7]. The implementation of fixed RNA profiling panels now enables stabilization of cells at clinical sites with subsequent analysis at central testing facilities, facilitating multi-site clinical trials [7].
Diagram 2: Application Pipeline for Single-Cell Suspensions. High-quality single-cell suspensions enable diverse applications across basic research, drug discovery, and clinical development, with specific methodologies optimized for each context.
Implementation Guidelines and Future Perspectives
Selection Guidelines for Technology Implementation
Choosing the appropriate dissociation technology requires careful consideration of experimental goals and tissue characteristics:
- For high-content single-cell analysis of embryonic tissues, microfluidic droplet platforms offer the best balance of throughput and information depth at reasonable cost [3].
- When maximum cell viability is crucial for functional assays, acoustic sorting systems provide exceptional gentle processing due to absence of labels, electrical fields, or high pressures [3].
- For applications preserving spatial context, spatial LCM and barcoding approaches serve different needs—LCM provides better precision for specific regions while spatial barcoding offers higher throughput [3].
- When working with limited starting material (common in embryonic research), AI-enhanced FACS systems provide intelligent gating and high recovery rates through real-time adaptive gating [3].
Emerging Technologies and Future Directions
The field of tissue dissociation and single-cell analysis continues to evolve rapidly, with several promising technologies approaching maturity:
CRISPR-Activated Cell Sorting represents a paradigm shift from surface marker-based isolation to functional characterization. This approach uses CRISPR activation of reporter genes linked to cellular functions, enabling isolation of cells based on functional states rather than static markers [3]. Current applications under investigation include isolating neurons based on immediate early gene activation and identifying cancer stem cells using stemness pathways [3].
Quantum Dot Barcoding pursues higher multiplexing capabilities through semiconductor particles with narrow, tunable emission spectra and exceptional brightness. Current systems can theoretically distinguish over 100 different barcodes, enabling unprecedented multiplexing for high-parameter studies [3].
Organoid-Based Isolation Systems represent a fundamentally different approach that selects cells based on organizational potential rather than immediate markers. This technology identifies cells capable of forming specific organoid structures, typically involving single-cell suspension followed by limited culture and isolation based on contribution to developing structures [3].
As these technologies mature, they will further enhance our ability to probe developmental processes and accelerate drug discovery by providing deeper insights into cellular heterogeneity and function. The continuing refinement of tissue dissociation methodologies will remain foundational to these advances, enabling researchers to unlock the profound biological insights contained within complex tissues at single-cell resolution.
Deconstructing the Embryonic Extracellular Matrix and Cell Junctions
Within the developing embryo, the extracellular matrix (ECM) and cell-cell junctions form a dynamic scaffold that is fundamental to structural integrity, mechanotransduction, and cellular communication. Deconstructing this intricate architecture to generate single-cell suspensions presents a significant research challenge, as the process must be carefully balanced to achieve high cell yield and viability while preserving native cellular states [1]. The ECM is not a static structure; it is a complex, tissue-specific network of proteins and glycans that confers mechanical properties and provides biochemical signals essential for development, homeostasis, and differentiation [8] [9]. Simultaneously, junctional complexes, including adherens junctions and desmosomes, maintain tissue cohesion and respond to mechanical forces [10] [11]. This Application Note details the latest protocols and mechanistic insights for the dissociation of embryonic tissues, framed within a thesis on fundamental developmental biology and its applications in drug development.
Quantitative Characterization of the Embryonic ECM and Junctions
The embryonic ECM and junctional complexes exhibit distinct compositional and mechanical properties that must be considered for effective dissociation. The following tables summarize key quantitative data and components.
Table 1: Core Matrisome Components of the Embryonic Microenvironment [9]
| Matrisome Category | Key Components | Primary Structural Function |
|---|---|---|
| Collagens | Fibrillar (I, II, III, V, XI); Network-forming (IV, VIII); FACITs (IX, XII); Transmembrane (XIII, XVII) | Confers resistance to tensile force and stretching. |
| Proteoglycans | Decorated with Chondroitin Sulfate, Dermatan Sulfate, Heparan Sulfate, Keratan Sulfate | Resists compressive forces, forms hydrated gels, and regulates growth factor signaling. |
| Glycoproteins | Fibronectin, Laminin, Nidogen, Elastin | Provides elasticity, connects ECM components, and facilitates cell adhesion. |
Table 2: Performance Metrics of Tissue Dissociation Techniques [1] [2]
| Technology / Protocol | Dissociation Type | Reported Viability | Processing Time | Key Applications & Notes |
|---|---|---|---|---|
| Optimized for Breast Cancer [1] | Mechanical & Enzymatic | 83.5% ± 4.4% | >1 hour | Single-cell RNA sequencing of human tissue. |
| Optimized for Skin Biopsy [1] | Mechanical & Enzymatic | ~93% | ~3 hours | Yields ~24,000 cells per 4mm punch. |
| Hypersonic Levitation (HLS) [2] | Non-contact, Hydrodynamic | 92.3% | 15 minutes | High rare-cell population preservation; 90% tissue utilization. |
| Electric Field Dissociation [1] | Electrical | ~80% (Glioblastoma) | 5 minutes | Rapid processing; >5x higher yield than traditional methods for some tissues. |
Experimental Protocols for Embryonic Tissue Dissociation
The following protocols provide detailed methodologies for deconstructing embryonic tissues, ranging from established enzymatic workflows to novel non-contact technologies.
Standardized Enzymatic-Mechanical Dissociation Workflow
This protocol is adapted for robust single-cell suspension preparation from complex tissues for downstream applications like single-cell RNA sequencing [1].
- Key Reagents: Collagenase, Dispase, Hyaluronidase, Trypsin, EDTA, DNase I, Fetal Bovine Serum (FBS), Phosphate Buffered Saline (PBS).
- Equipment: Sterile surgical instruments, tissue culture hood, 37°C shaking incubator or water bath, 40 µm and 100 µm cell strainers, centrifuge.
Step-by-Step Procedure:
- Tissue Mincing: In a sterile Petri dish, finely mince the fresh embryonic tissue (1-5 mm³) into ~1 mm³ pieces using sterile scalpels or razor blades. Keep the tissue moist with cold PBS containing antibiotics.
- Enzymatic Digestion: Transfer the minced tissue into a digestion buffer (e.g., PBS or DMEM) containing a customized enzyme cocktail. A typical cocktail may include:
- Collagenase (1-2 mg/mL)
- Dispase (1-2 mg/mL)
- Hyaluronidase (0.1-0.5 mg/mL)
- DNase I (10-50 µg/mL) to prevent cell clumping from released DNA.
- Incubation with Agitation: Incubate the tissue-enzyme mixture at 37°C for 30-90 minutes with constant agitation (e.g., on a shaking platform). The duration must be optimized for the specific embryonic stage and tissue type.
- Mechanical Disruption: Periodically triturate the digesting tissue every 15-20 minutes using a serological pipette (e.g., 10 mL) to aid in physical dissociation. For more robust tissues, gentle pipetting with a fire-polished Pasteur pipette may be required.
- Reaction Termination: Add a stop solution containing FBS (to a final concentration of 10%) or a specific enzyme inhibitor to halt the digestion process.
- Filtration and Washing: Pass the cell suspension through a 100 µm cell strainer followed by a 40 µm cell strainer to remove undigested tissue fragments and large aggregates. Centrifuge the filtrate at 300-400 x g for 5 minutes.
- Red Blood Cell Lysis (if needed): Resuspend the cell pellet in an appropriate red blood cell lysis buffer (e.g., ACK buffer) for 2-5 minutes at room temperature. Stop the reaction with excess PBS.
- Final Resuspension and Counting: Resuspend the final cell pellet in a suitable buffer (e.g., PBS with 0.04% BSA) for downstream applications. Determine cell count and viability using a hemocytometer or automated cell counter with Trypan Blue or similar dye.
Advanced Non-Contact Dissociation via Hypersonic Levitation and Spinning (HLS)
This protocol describes a contact-free method using hydrodynamic forces for rapid and high-viability single-cell isolation, ideal for fragile embryonic cells or preserving rare populations [2].
- Key Reagents: HLS Automated Dissociation Apparatus, standard cell culture medium or enzyme solution.
- Equipment: HLS system with triple-acoustic resonator probe, conical confinement structure, integrated fluid handling modules.
Step-by-Step Procedure:
- System Priming: Power on the HLS apparatus and prime the fluidic pathways with the desired dissociation medium (which can be a simple buffer or contain low-concentration enzymes).
- Sample Loading: Place the small piece of embryonic tissue (up to ~1-2 mm³) into the sample chamber filled with medium.
- Acoustic Activation: Activate the GHz-frequency acoustic resonator probe. The generated hypersonic streaming field will cause the tissue sample to levitate and execute a rapid "press-and-rotate" self-spinning motion within the conical confinement.
- Dissociation Process: The microscale "liquid jets" and enhanced shear forces generated by the spinning action will disrupt cell-cell and cell-ECM connections. Allow the process to run for a defined period, typically 15-30 minutes. The hypersonic streaming also enhances enzyme penetration if used.
- Automated Fluid Replacement and Filtration: The integrated system will automatically flush the dissociated cells from the dissociation chamber, replace the fluid, and filter the output to remove any remaining small aggregates.
- Cell Collection: Collect the single-cell suspension from the output chamber. The cells are now ready for counting and downstream applications like primary culture, flow cytometry, or single-cell RNA sequencing.
Visualization of Key Signaling and Workflow Pathways
The following diagrams illustrate the core mechanisms of junctional remodeling and the operational principles of advanced dissociation technologies.
Mechanosensitive Junctional Remodeling in Development
Principle of Hypersonic Levitation and Spinning (HLS)
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Materials for Embryonic Tissue Dissociation
| Reagent / Material | Function / Application | Specific Examples / Notes |
|---|---|---|
| Collagenase | Digests collagen, a major structural component of the ECM. | Crude collagenase blends are often used for complex tissues; purified types (e.g., Collagenase I-V) offer specificity. |
| Trypsin / EDTA | Proteolytic enzyme that cleaves adhesion proteins; EDTA chelates Ca²⁺, disrupting cadherin-dependent junctions. | Commonly used for epithelial cells and established cell lines. Can be harsh; exposure time must be carefully controlled. |
| Dispase | Neutral protease that cleaves fibronectin and collagen IV, often gentler on cell surface receptors. | Ideal for liberating intact epithelial sheets and organoids. |
| Hyaluronidase | Degrades hyaluronic acid, a major glycosaminoglycan in the ECM. | Used in combination with other enzymes to disrupt the ground substance. |
| DNase I | Degrades DNA released from damaged cells, preventing cell clumping and reducing solution viscosity. | Critical for tissues prone to high levels of cell death during dissociation. |
| ROCK Inhibitor (Y-27632) | Inhibits Rho-associated kinase, reducing apoptosis in single cells (anoikis) and improving viability post-dissociation. | Add to cell culture medium after dissociation, especially for primary and stem cells. |
| Integrin-Blocking Antibodies / RGD Peptides | Blocks integrin-ECM interactions, preventing re-aggregation of cells and potentially aiding in dissociation. | Useful for studies where integrin signaling must be controlled. |
| FRET-Based Tension Sensors | Genetically encoded sensors to measure molecular-scale tension across proteins like E-cadherin or Dsg3. | Used in basic research to understand mechanobiology of junctions pre- and post-dissociation [10]. |
Dissociating embryo tissue into a single-cell suspension is a critical first step in developmental biology research, single-cell analysis, and cell-based therapeutic development. The fundamental challenge lies in disrupting complex tissue architecture and cell-cell junctions while preserving the viability, function, and native transcriptional state of individual cells. Achieving this balance is particularly crucial for embryonic tissues, which often contain rare progenitor populations and are highly sensitive to micro-environmental cues. This document outlines the core principles, compares advanced dissociation technologies, and provides detailed protocols to guide researchers in optimizing this essential process.
The Dissociation Trilemma: Efficiency, Viability, and Function
The process of tissue dissociation inherently involves navigating a trilemma between three competing factors:
- Dissociation Efficiency: The completeness of tissue breakdown and the total yield of single cells.
- Cell Viability: The proportion of isolated cells that remain alive and intact post-dissociation.
- Cell Function/State Preservation: The maintenance of native transcriptional profiles, surface markers, and functional capacities, free from dissociation-induced stress artifacts.
Traditional methods often force trade-offs. For instance, prolonged enzymatic digestion can increase cell yield but may compromise viability and alter cell surface proteins [1]. Mechanical force can rapidly disaggregate tissue but risks physical damage to delicate cells [12]. The goal of modern protocols is to leverage novel techniques and principles to maximize all three factors simultaneously, especially for sensitive embryo tissues.
Current Technologies and Methodological Comparisons
Recent advancements have expanded the toolkit for tissue dissociation beyond conventional enzymatic and mechanical approaches. The table below summarizes the key characteristics of these methods, providing a quantitative comparison to inform protocol selection.
Table 1: Quantitative Comparison of Tissue Dissociation Technologies
| Technology/Method | Dissociation Type | Reported Cell Viability | Processing Time | Key Advantages | Key Limitations |
|---|---|---|---|---|---|
| Cold-Active Protease [13] | Enzymatic (Low-Temp) | >90% (Transcriptome preservation) | 30-60 min | Preserves native transcriptome; minimizes stress artifacts. | Limited protocol history; requires optimization for new tissues. |
| Hypersonic Levitation & Spinning (HLS) [2] | Non-contact Physical | 92.3% | 15 min | High speed; contactless; preserves rare cells. | Specialized equipment required; not yet widely adopted. |
| Optimized Enzymatic/Mechanical [1] | Combined Enzymatic/Mechanical | 83.5% - 92.75% | 1 - 3 hours | Well-characterized; accessible. | Can induce transcriptional stress; longer processing. |
| Mixed Modal Microfluidic [1] | Microfluidic Enzymatic/Mechanical | 50% - 95% (cell type dependent) | 1 - 60 min | Automated; controlled shear forces. | Risk of channel clogging; lower throughput. |
| Electric Field Dissociation [1] | Non-enzymatic/Electrical | ~80% - 90% | 5 min | Extremely rapid; enzyme-free. | Potential for membrane damage; optimization complexity. |
Detailed Experimental Protocols
Protocol: Cold-Active Protease Dissociation for Transcriptome Preservation
This protocol is optimized for preserving the native transcriptional state of cells from embryo tissues, such as zebrafish cranial tendons, and is ideal for downstream single-cell RNA sequencing [13].
Research Reagent Solutions
- Cold Protease Stock Solution: 100 mg/mL protease from B. licheniformis in PBS. Aliquot and store at -80°C.
- DNase Stock Solution: 20 U/μL DNase I in PBS. Aliquot and store at -20°C.
- Cold Protease Working Solution: Combine 100 μL cold protease stock, 5 μL 1M CaCl₂, 1 μL 0.5M EDTA, and 5 μL DNase stock. Bring to 1 mL with ice-cold DPBS. Keep on ice.
- DPBS-BSA Solution: 0.01% Bovine Serum Albumin (BSA) in DPBS. Keep on ice.
- Ringer's Solution: 116 mM NaCl, 2.9 mM KCl, 10 mM CaCl₂·2H₂O, 5 mM HEPES; pH to 7.2.
Procedure
- Tissue Harvesting: Dissect embryo tissues (e.g., zebrafish cranial tendons at 72 hpf) in ice-cold Ringer's solution.
- Tissue Transfer: Transfer the pooled tissue to a 1.5 mL microcentrifuge tube pre-filled with 1 mL of ice-cold DPBS-BSA solution.
- Centrifugation: Centrifuge at 300 x g for 3 minutes at 4°C. Carefully aspirate the supernatant.
- Protease Digestion: Resuspend the tissue pellet in 1 mL of cold protease working solution.
- Incubation: Incubate the tube on an ice bath (4°C) for 30 minutes. Gently agitate the tube by inverting it every 10 minutes.
- Reaction Termination: Add 2 mL of ice-cold DPBS-BSA solution to terminate the digestion.
- Trituration: Gently triturate the tissue 10-15 times using a fire-polished glass Pasteur pipette with a narrowed opening.
- Filtration: Filter the cell suspension through a 40 μm cell strainer into a new tube.
- Washing: Centrifuge the filtrate at 300 x g for 5 minutes at 4°C. Aspirate the supernatant and resuspend the cell pellet in an appropriate buffer for downstream applications (e.g., FACS, scRNA-seq).
Protocol: Hypersonic Levitation and Spinning (HLS) for Rapid, Contactless Dissociation
This protocol leverages a non-contact, acoustic-based technology to achieve rapid dissociation with high viability, suitable for tissues where mechanical stress must be minimized [2].
Procedure
- Apparatus Setup: Initialize the automated HLS apparatus, ensuring the triple-acoustic resonator probe and conical confinement structure are clean and sterile.
- Sample Loading: Place the minced embryo tissue sample (≈1 mm³ pieces) into the digestion chamber of the apparatus.
- Buffer Addition: Add an appropriate enzyme solution (e.g., a tailored collagenase/dispase blend) to the chamber, fully submerging the tissue.
- HLS Activation: Activate the hypersonic probe. The generated GHz-frequency acoustic field will levitate the tissue and induce a rapid 'press-and-rotate' spinning motion.
- Dissociation: Run the system for 10-15 minutes. The combination of hydrodynamic shear forces from microscale "liquid jets" and enhanced enzymatic penetration will dissociate the tissue.
- Automated Processing: The integrated apparatus automatically performs fluid replacement and filtration, transferring the single-cell suspension to the collection chamber.
- Collection: Collect the single-cell suspension from the outlet for immediate use or analysis.
Workflow Visualization and Logical Pathways
The following diagrams outline the logical decision-making process for method selection and the specific workflow for the cold-active protease protocol.
Diagram 1: Decision Workflow for Embryo Tissue Dissociation Method Selection
Diagram 2: Cold-Active Protease Experimental Workflow
The dissociation of complex embryo tissues into high-quality single-cell suspensions is a critical prerequisite for advanced analytical techniques such as single-cell RNA sequencing (scRNA-seq). This process represents a significant bottleneck in single-cell research, with the quality of the initial cell suspension directly determining the reliability and resolution of all downstream data. Success in this endeavor is measured by three interdependent metrics: cell yield (the number of cells recovered), cell viability (the percentage of living cells), and transcriptomic preservation (the integrity of RNA content). This application note details optimized protocols and quantitative benchmarks for embryo tissue dissociation, providing researchers with a standardized framework to maximize data quality while minimizing technical artifacts.
Quantitative Metrics for Tissue Dissociation
The following tables synthesize performance data across various dissociation technologies and tissue types, providing reference benchmarks for evaluating experimental outcomes.
Table 1: Performance Comparison of Tissue Dissociation Technologies
| Technology | Tissue Type | Cell Yield | Cell Viability | Processing Time | Source |
|---|---|---|---|---|---|
| Chemical-Mechanical Workflow | Bovine Liver Tissue | 92% ± 8% (of total cells) | >90% | 15 min | [1] |
| Optimized Enzymatic Protocol | Human Skin Biopsy | ~24,000 cells/4 mm punch | 92.75% | ~3 hours | [1] |
| Hypersonic Levitation (HLS) | Human Renal Cancer | 90% tissue utilization | 92.3% | 15 min | [2] |
| Mixed Modal Microfluidic Platform | Mouse Kidney | ~20,000 epithelial cells/mg | ~95% (epithelial) | 20-60 min | [1] |
| Electric Field Dissociation | Human Glioblastoma | >5x higher than traditional methods | ~80% | 5 min | [1] |
Table 2: scRNA-seq Method Performance on Immune Cells (from Cell Line Mix Studies)
| scRNA-seq Method | Median Genes Detected per Cell (EL4 Cell Line) | Median UMIs Detected per Cell (EL4 Cell Line) | Cell Recovery Rate |
|---|---|---|---|
| 10x Genomics 3' v3 | 4,776 | 28,006 | ~30-80% |
| 10x Genomics 5' v1 | 4,470 | 25,988 | ~30-80% |
| 10x Genomics 3' v2 | 3,882 | 21,570 | ~30-80% |
| ddSEQ | 3,644 | 10,466 | <2% |
| Drop-seq | 3,255 | 8,791 | <2% |
Detailed Experimental Protocols
Protocol 1: Enzymatic-Mechanical Dissociation for Complex Tissues
This protocol, adapted from optimized workflows for human skin and other challenging tissues, prioritizes high viability and RNA integrity [1] [14].
Reagents and Materials:
- Collagenase (Type IV or suitable for target tissue)
- Dispase
- Hyaluronidase
- DNase I
- EDTA Solution (for chelating calcium)
- Wash Buffer: PBS with 0.04% BSA or 1% FBS
Procedure:
- Tissue Preparation: Mince the embryo tissue thoroughly into fragments of approximately 1-2 mm³ using sterile surgical blades or a scalpel on a chilled surface.
- Enzymatic Digestion: Prepare an enzyme cocktail (e.g., 2 mg/mL Collagenase, 1 mg/mL Dispase, 0.5 mg/mL Hyaluronidase) in a suitable buffer. Use 5-10 mL of cocktail per 100 mg of tissue.
- Incubation: Incubate the tissue fragments in the enzyme solution with gentle agitation (e.g., on a rotator in a 37°C incubator). The incubation time must be empirically determined (typically 30-90 minutes). Avoid exceeding 2 hours to preserve RNA integrity.
- Mechanical Agitation: Every 15-20 minutes, triturate the digesting tissue gently 10-15 times using a wide-bore pipette tip or a sterile Pasteur pipette with a fire-polished end.
- Reaction Termination: Add an equal volume of cold Wash Buffer containing 1-5% FBS to neutralize the enzymes.
- Filtration and Washing: Filter the cell suspension through a 40-70 µm cell strainer. Centrifuge the filtrate at 300-400 x g for 5 minutes at 4°C and resuspend the pellet in cold Wash Buffer. A DNase I treatment (10-20 µg/mL for 5 min) can be included at this stage if clumping is observed.
- Assessment: Count cells and assess viability using a hemocytometer or fluorescence-based automated counter.
Protocol 2: Methanol Fixation for Transcriptomic Preservation of Rare Populations
For studies focusing on rare cell populations (e.g., specific embryonic progenitors), fixation allows for sample pooling and sorting without significant RNA degradation [15].
Reagents and Materials:
- Methanol (pre-cooled to -80°C)
- Rehydration Buffer: PBS with 1% BSA and RNasin Ribonuclease Inhibitor (1:80 dilution)
- Viability Stain (e.g., DRAQ7 or similar)
- Intracellular Staining Antibodies (if applicable)
Procedure:
- Single-Cell Suspension: Begin with a freshly dissociated, high-viability single-cell suspension.
- Fixation: Gently resuspend the cell pellet in a small volume of cold PBS. While vortexing at a low speed, slowly add dropwise 10 volumes of pre-cooled (-80°C) methanol to achieve a final concentration of ~90% methanol. Incubate for 15 minutes on ice.
- Storage: Cells can be pelleted and stored in 90% methanol at -80°C for at least one month (and up to 3 years) without significant RNA degradation [15].
- Rehydration: Centrifuge fixed cells and carefully remove the methanol. Resuspend the pellet gently in Rehydration Buffer and incubate for 10-15 minutes on ice.
- Staining and Sorting: Proceed with viability staining and intracellular staining for FACS. The RIN (RNA Integrity Number) of rehydrated cells is typically ~8.7, which is suitable for scRNA-seq [15].
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents for Tissue Dissociation and Single-Cell Analysis
| Reagent / Material | Function | Example Use Case |
|---|---|---|
| Collagenase | Digests collagen in the extracellular matrix | General tissue dissociation cocktail [1] |
| Dispase | Proteolytic enzyme that cleaves fibronectin and collagen IV | Preferable for epithelial cell isolation [1] |
| DNase I | Degrades extracellular DNA released by damaged cells | Reduces cell clumping during dissociation [1] |
| EDTA | Chelates Ca²⁺ ions required for cell adhesion | Used in combination with enzymes for improved dissociation [1] |
| RNasin/RNase Inhibitor | Inhibits RNase activity | Preserves RNA integrity in buffers during and after dissociation [15] |
| Wide-Bore Pipette Tips | Minimizes shear stress on cells during pipetting | Essential for gentle mechanical trituration [16] |
Critical Validation and Quality Control Workflows
A robust quality control pipeline is non-negotiable for generating reliable single-cell data.
Cell Counting and Viability Assessment:
- Best Practice: Use manual hemocytometer counting or fluorescence-based automated counters. Studies show that trypan blue-based automated counters can consistently overestimate viability [16].
- Thresholds: Aim for a final cell viability of >80-90% prior to loading on a scRNA-seq platform. Lower viability increases background noise from ambient RNA.
scRNA-seq Data Quality Assessment:
- Key Metrics: Upon receiving sequencing data, assess the following for each library:
- Fraction of Reads in Cells: Should be high (e.g., >85% for fresh cells) [15].
- Median Genes per Cell: A low count suggests poor mRNA capture.
- Mitochondrial Gene Percentage: A high percentage (>10-20%) often indicates cell stress or apoptosis during dissociation. Methanol-fixed cells typically show lower mitochondrial read percentages [15].
- Knee Plot: Visualizes the separation between cell-containing barcodes and background, indicating good encapsulation efficiency [15].
Workflow for Single-Cell Preparation and QC
The choice of dissociation strategy represents a balance between yield, viability, and transcriptional fidelity. While optimized enzymatic protocols remain the workhorse for many applications, novel non-enzymatic methods like Hypersonic Levitation and Spinning (HLS) and Electrical Dissociation offer compelling advantages, including dramatically reduced processing times (minutes versus hours) and the elimination of enzyme-induced transcriptional artifacts [1] [2].
For embryonic tissues, which often contain fragile and rare progenitor populations, the dissociation protocol must be tailored to preserve these critical cells. The methanol fixation protocol provides a powerful tool for the logistical handling of such samples, enabling the pooling, sorting, and in-depth analysis of rare embryonic cell types without compromising RNA quality [15].
In conclusion, achieving excellence in single-cell embryo research requires rigorous attention to the initial dissociation process. By adhering to the quantitative metrics, detailed protocols, and quality control frameworks outlined in this application note, researchers can ensure that their single-cell suspensions are of the highest quality, thereby laying a solid foundation for groundbreaking discoveries in developmental biology.
Step-by-Step Dissociation Protocols and Cutting-Edge Techniques for Embryonic Tissue
Within the framework of a broader thesis on dissociating embryonic tissue into single-cell suspensions, the optimization of enzymatic cocktails is a critical foundational step. This process is vital for advancing research in developmental biology, drug discovery, and regenerative medicine, where the quality of the initial single-cell suspension profoundly impacts downstream applications like single-cell RNA sequencing, flow cytometry, and cell line establishment [1]. Traditional enzymatic methods, primarily utilizing trypsin, collagenase, and dispase, face significant challenges regarding cell viability, yield, and the potential introduction of transcriptional artifacts that can distort analytical results [1] [13]. This application note provides a detailed, optimized framework for employing these enzymes specifically for embryonic tissue, presenting structured quantitative data, step-by-step protocols, and essential visual guides to ensure the production of high-quality single-cell suspensions for research and drug development.
Enzyme Mechanisms and Selection Guide
The effectiveness of an enzymatic dissociation cocktail hinges on the complementary actions of its components, which target different proteins within the extracellular matrix (ECM) and cell-cell junctions. Embryonic tissues, rich in diverse ECM components, require a balanced approach to achieve efficient dissociation while preserving cell integrity.
The following diagram illustrates the strategic workflow for selecting and optimizing an enzymatic dissociation protocol, from primary objective to final validation:
Core Enzyme Mechanisms
- Trypsin: A serine protease that cleaves peptide bonds on the C-terminal side of lysine and arginine amino acids. It is highly effective at digesting proteins that mediate cell-cell adhesion but can damage cell surface proteins if overused, making it a concern for downstream surface protein analysis (SPEX) [17] [18].
- Collagenase: Targets collagen, a major structural component of the ECM. Collagenase is crucial for breaking down the tissue scaffold, particularly in fibrous tissues [1] [18].
- Dispase: A neutral protease that cleaves fibronectin, collagen IV, and other proteins in the basement membrane. It is known for generating intact cell sheets or clusters with less damage to cell surface markers compared to trypsin [19] [18].
Quantitative Cocktail Performance
The table below summarizes key performance metrics of common enzymes and cocktails, as reported in recent literature. These values serve as a critical benchmark for protocol optimization.
Table 1: Enzymatic Dissociation Efficacy Across Tissue Types
| Technology / Cocktail | Dissociation Type | Tissue Type | Dissociation Efficacy / Yield | Cell Viability | Time |
|---|---|---|---|---|---|
| Dispase I + Collagenase IV + Trypsin (D/C/T) | Enzymatic | Human Skin | 2–6 fold more cells vs. other protocols [17] | Reported as high | ~3 hours [19] |
| Dispase I + Cold-Active Protease (D/CP) | Enzymatic (Cold) | Human Skin | Lower than D/C/T protocol [17] | Preserved | Not Specified |
| Collagenase IV Alone | Enzymatic | Zebrafish Tendon | Effective | High stress gene expression [13] | 4-18 hours [18] |
| Cold-Active Protease (Subtilisin A) | Enzymatic (Cold) | Zebrafish Tendon | Effective | >90%; Low stress genes [13] | 6-18 hours (at 4°C) [13] |
| Trypsin-EDTA (0.25%) | Enzymatic | General Primary Tissue | Effective | >90% (if optimized) [18] | 20-30 min (at 37°C) [18] |
The selection of an enzyme cocktail must balance dissociation efficiency with the preservation of cell integrity and surface markers. The optimal combination depends heavily on the specific embryonic tissue and the requirements of the downstream application.
Table 2: Enzyme Profiles and Applications
| Enzyme | Primary Target | Typical Concentration | Key Advantage | Major Consideration | Recommended for Embryonic Tissue? |
|---|---|---|---|---|---|
| Trypsin | Peptide Bonds (Cell-cell adhesion) | 0.25% [18] | Rapid, highly effective | Damages cell surface proteins [1] [17] | Use with caution; limit exposure time |
| Collagenase | Collagen (ECM) | 50-200 U/mL [18] | Breaks down tissue scaffold | Less damaging to surface markers than trypsin | Yes, often essential |
| Dispase | Fibronectin, Collagen IV | 0.6-2.4 U/mL [18] | Gentle; preserves surface markers [17] | May be slower for tough tissues | Yes, ideal for sensitive applications |
| Cold-Active Protease | Broad spectrum (at low temp) | 10 mg/mL [13] | Minimizes transcriptional stress [13] | Requires long incubation at 4°C | Highly recommended for transcriptomics |
Experimental Protocols
Optimized Triple-Enzyme Cocktail Workflow (for robust dissociation)
This protocol is adapted from a highly effective method used for human skin, which can be a robust starting point for dense embryonic tissues [17].
The following diagram outlines the key steps in the tissue dissociation and validation workflow:
Step-by-Step Method:
- Tissue Preparation: Place the embryonic tissue in a sterile Petri dish with a cold balanced salt solution (e.g., DPBS). Mince the tissue into 3-4 mm fragments using a sterile scalpel or scissors. Wash the fragments 2-3 times with the salt solution to remove debris [18].
- Enzymatic Digestion:
- Dispase Incubation: Submerge the tissue fragments in Dispase I solution (0.6-2.4 U/mL) in a sterile tube. Incubate at 37°C for 30-60 minutes with slow agitation [18].
- Collagenase/ Trypsin Incubation: After the Dispase step, you can proceed in one of two ways:
- Option A (Sequential): Decant the Dispase solution. Add a pre-warmed mixture of Collagenase IV (60-100 U/mL) and Trypsin-EDTA (0.25%) [17] [18]. Incubate at 37°C for 30-90 minutes with agitation.
- Option B (Combined): A combination of Dispase and Collagenase can be used together from the start for more efficient dissociation [18].
- Reaction Termination: Add a volume of cold, serum-containing complete media (e.g., with 10% Fetal Bovine Serum) that is at least equal to the volume of the enzyme solution. Serum inhibits trypsin and other proteases.
- Mechanical Dissociation and Filtration:
- Cell Washing and Resuspension: Centrifuge the filtered suspension at 100-300 × g for 5 minutes. Discard the supernatant and resuspend the cell pellet in an appropriate buffer or culture medium.
- Cell Counting and Viability Assessment: Determine viable cell density and percent viability using an automated cell counter or hemocytometer with a dye like Trypan Blue. Viability should be routinely greater than 90% for high-quality downstream applications [18].
Cold-Active Protease Protocol (for transcriptome preservation)
For applications like single-cell RNA sequencing where preserving the native transcriptome is paramount, a cold-active protease protocol is highly recommended to minimize dissociation-induced stress [13].
Step-by-Step Method:
- Solution Preparation: Prepare a cold protease working solution containing protease from B. licheniformis (e.g., 10 mg/mL), DNase I (100 U/mL), 5 mM CaCl₂, and 0.5 mM EDTA in DPBS. Keep the solution on ice [13].
- Tissue Preparation: Mince the embryonic tissue into small fragments as described in section 4.1, performing all steps on ice.
- Cold Digestion: Submerge the tissue fragments in the ice-cold protease working solution. Incubate the mixture at 4°C for 6-18 hours on a rocker or nutator to allow for gentle, continuous digestion [13].
- Mechanical Dissociation and Filtration: After incubation, gently pipet the mixture. Filter the cell suspension through a cell strainer (40-70 µm).
- Cell Washing and Resuspension: Centrifuge and wash the cells with an ice-cold DPBS solution containing 0.01% BSA [13]. Resuspend the final pellet in your desired cold buffer.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagent Solutions
| Item | Function / Application | Example Source / Identifier |
|---|---|---|
| Dispase II | Digests basement membrane proteins; gentle dissociation. | Roche, Cat#04942078001 [19] |
| Collagenase IV | Digests native collagen in the extracellular matrix. | Worthington, Cat#LS004189 [19] |
| Trypsin-EDTA (0.25%) | Cleaves peptide bonds for rapid cell detachment. | Thermo Fisher Scientific, Cat#25200056 [19] |
| Protease from B. licheniformis | Cold-active protease for low-stress dissociation. | Sigma-Aldrich, Cat#P5380 [13] |
| DNase I | Degrades DNA released from damaged cells to prevent clumping. | Roche, Cat#11284932001 [19] [13] |
| Dulbecco's PBS (DPBS) | Balanced salt solution for washing and reagent preparation. | Thermo Fisher Scientific, Cat#14190144 [19] |
| Cell Strainer (40 µm and 70 µm) | Filtering single-cell suspensions to remove debris and clusters. | Corning, Cat#352340 & 352350 [19] |
| Fetal Bovine Serum (FBS) | Enzyme inactivation and supplementation of culture media. | Corning, Cat#35-079-CV [19] |
The dissociation of embryo tissue into a single-cell suspension is a critical first step in many developmental biology, drug discovery, and regenerative medicine applications. The choice of dissociation method directly impacts cell viability, yield, and the preservation of rare cell populations, all of which are crucial for downstream analyses like single-cell RNA sequencing and cell culture. Gentle mechanical dissociation techniques offer distinct advantages, particularly the avoidance of enzymatic artifacts that can alter cell surface markers and transcriptional profiles. This application note provides a detailed comparison of three gentle mechanical dissociation methods—pipetting, mincing, and automated grinding—and presents optimized protocols for their implementation in embryonic tissue research.
Quantitative Comparison of Gentle Mechanical Dissociation Methods
The following table summarizes the key performance metrics of different gentle mechanical dissociation techniques, helping researchers select the most appropriate method for their specific embryonic tissue type and downstream application.
Table 1: Performance Metrics of Gentle Mechanical Dissociation Methods
| Method | Typical Processing Time | Relative Cell Viability | Key Advantages | Primary Limitations | Suitable for Embryonic Tissues? |
|---|---|---|---|---|---|
| Manual Pipetting | Varies with protocol | High (when optimized) | Low cost, high control, minimal equipment [20] | Operator-dependent, low throughput, potential shear stress on cells [20] | Yes, for delicate pre-digested tissue |
| Fine Mincing | 15-30 minutes (pre-enzymatic step) | High (preserves fragile cells) | Simple, preserves tissue architecture for further processing [20] | Incomplete on its own, always requires subsequent enzymatic/other steps [20] | Yes, as a universal first step |
| Automated Grinders (e.g., TIGR) | < 2 minutes [21] | High [21] | Rapid, enzyme-free, standardized, high throughput [21] | Initial equipment cost, requires protocol optimization per tissue [21] | Yes, protocol-dependent |
Detailed Experimental Protocols
Protocol for Manual Pipetting and Mincing
This combined protocol is foundational for processing delicate embryonic tissues.
Key Research Reagent Solutions:
- Cold Isotonic Buffer: e.g., PBS or Hanks' Balanced Salt Solution (HBSS). Function: Maintains osmotic balance and cell viability during processing [20].
- Bovine Serum Albumin (BSA) or Fetal Bovine Serum (FBS): Function: When added to buffers, protects cells from shear stress and stabilizes the cell membrane [20].
- RNase Inhibitors: Function: Critical for preserving RNA integrity if downstream application is single-cell RNA sequencing [20].
Workflow:
- Tissue Preparation: Place the harvested embryonic tissue in a petri dish containing cold, sterile isotonic buffer. Keep the tissue cold to minimize metabolic stress and preserve viability [20].
- Fine Mincing: Using sterile scalpels or scissors, meticulously mince the tissue into fine fragments of approximately 1–2 mm³. This increases the surface area for subsequent dissociation steps [20].
- Mechanical Dissociation by Pipetting:
- Transfer the minced tissue fragments into a tube using a serological pipette.
- Using a smaller-bore pipette (e.g., P1000 set to 500-700 µL), gently but repeatedly pipette the tissue suspension up and down. The number of repetitions and force required must be empirically determined for each embryonic tissue type.
- Monitor the dissociation progress visually. The goal is to break down the tissue fragments into a cloudy cell suspension.
- Filtration and Washing: Pass the resulting suspension through a sterile cell strainer (e.g., 40 µm or 70 µm) to remove any remaining undigested clumps or tissue debris [20]. Centrifuge the filtrate at low speed (e.g., 300-400 x g for 5 minutes) to pellet the cells. Carefully aspirate the supernatant and resuspend the cell pellet in fresh buffer or culture medium.
- Validation and Counting: Assess cell viability using Trypan Blue exclusion or an automated cell counter. Determine cell concentration and adjust as needed for the downstream application [20].
Protocol for Automated Tissue Grinding (e.g., TIGR System)
This protocol leverages technology for rapid, enzyme-free dissociation, ideal for standardizing workflows across multiple samples.
Workflow:
- System Setup: Place the sterile, disposable 50 mL grinding tube, which contains a rotor-stator unit with counter-rotating teeth, into the TIGR benchtop instrument [21].
- Sample Loading: Transfer the freshly collected embryonic tissue into the grinding tube along with a suitable volume of cold buffer.
- Program Selection and Execution: Select a pre-programmed protocol optimized for your specific embryonic tissue type (e.g., protocols may exist for liver, heart, etc.). If no established protocol exists, begin with a default gentle program and optimize parameters such as duration, rotation speed, and bidirectional movement [21].
- Grinding Process: Initiate the program. The grinding typically completes in under two minutes. The rotor-stator teeth generate controlled shearing and milling forces, gently dissociating the tissue into single cells without enzymatic cleavage of membrane proteins [21].
- Collection and Filtration: After the cycle is complete, retrieve the single-cell suspension from the grinding tube. Filter the suspension through a compatible cell strainer to remove any rare large aggregates.
- Validation: Proceed with cell counting and viability assessment as described in section 2.1.
Workflow Integration and Decision Pathway
The following diagram illustrates the logical process for selecting and integrating these gentle mechanical dissociation methods into a research workflow for embryonic tissues.
Essential Research Reagent Solutions
The following table lists key materials and reagents essential for successful implementation of gentle mechanical dissociation protocols.
Table 2: Key Research Reagent Solutions for Mechanical Dissociation
| Item | Function/Application | Example/Notes |
|---|---|---|
| Dounce Homogenizer | Gentle shearing of minced tissue through manual grinding [20]. | Available in different pestle clearances; loose for initial breakdown, tight for final dissociation. |
| Cell Strainers | Removal of undigested clumps and tissue debris from the single-cell suspension [20]. | Typically 40 µm or 70 µm nylon mesh. Use pre-wet strainers to improve cell yield. |
| BSA or FBS | Added to dissociation buffers to protect cells from mechanical stress and stabilize the cell membrane [20]. | Use at 0.1-2% concentration. |
| Cold-Active Proteases | Optional for combined protocols; can be used at lower temperatures to minimize cellular stress [1]. | An alternative for researchers seeking to reduce enzymatic artifacts while aiding dissociation. |
| Trypan Blue | Dye exclusion-based assessment of cell viability post-dissociation [20]. | A standard for quick viability checks. Automated cell counters can also be used. |
| Disposable Grinding Tubes | For use with automated grinders; ensure sterility and prevent cross-contamination [21]. | Often contain integrated rotor-stator units designed for specific instruments. |
Gentle mechanical dissociation methods are indispensable tools for creating high-quality single-cell suspensions from embryonic tissues. While manual techniques like mincing and pipetting offer simplicity and fine control, automated grinders provide unparalleled speed, reproducibility, and throughput for enzyme-free dissociation. The optimal method depends on the specific embryonic tissue, the required cell yield and viability, and the constraints of the research pipeline. By following the detailed protocols and utilizing the decision framework provided, researchers can effectively integrate these techniques to advance their studies in developmental biology and drug development.
The generation of high-quality single-cell suspensions from embryonic organoids is a critical foundational technique in developmental biology and regenerative medicine. This protocol provides a detailed guide for the enzymatic-mechanical dissociation of embryonic organoids, framing the process within the broader research objective of creating viable single-cell suspensions for downstream applications such as single-cell RNA sequencing, flow cytometry, and subsequent recellularization or reprogramming experiments. The choice of dissociation strategy significantly impacts cell viability, yield, and the preservation of rare cell populations, all of which are crucial for the integrity of subsequent research data [1]. This document compares two primary methods—mechanical dissociation and enzymatic digestion—to equip researchers with the knowledge to select the optimal approach based on their specific experimental requirements [22].
Materials and Reagents
Research Reagent Solutions
Table 1: Essential Reagents for Organoid Dissociation
| Reagent Category | Specific Examples | Function |
|---|---|---|
| Enzymes | Collagenase, Trypsin, Dispase, Hyaluronidase [1] | Digests extracellular matrix (ECM) components and cell-cell junctions. |
| Chelating Agents | Ethylene Diamine Tetra-acetic Acid (EDTA) [1] [23] | Chelates calcium, disrupting cell-cell adhesion. |
| Enzyme Inhibitors | DNase [23] | Degrades excess DNA released from necrotic cells to reduce viscosity. |
| Basement Membrane Matrix | Matrigel [23] | Provides a 3D support structure for organoid culture pre-dissociation. |
| Buffers & Media | Phosphate Buffered Saline (PBS), Cell Culture Media | Washes and maintains cells in a physiological environment. |
Laboratory Equipment
- Centrifuge: For pelleting cells and organoid fragments.
- Water Bath or Incubator: Maintained at 37°C for enzymatic digestion.
- Biosafety Cabinet: For sterile tissue culture procedures.
- Pipettes and Tips: For handling liquids.
- Cell Strainers (e.g., 40µm, 70µm): To remove large aggregates and obtain a single-cell suspension.
- Hemocytometer or Automated Cell Counter: For assessing cell count and viability.
Method Comparison and Selection
The decision between mechanical and enzymatic dissociation involves a trade-off between preserving the native cellular microenvironment and achieving a homogeneous single-cell suspension. The following diagram outlines the decision-making workflow.
Quantitative Method Comparison
Table 2: Comparison of Mechanical and Enzymatic Dissociation Methods
| Parameter | Mechanical Dissociation | Enzymatic Digestion |
|---|---|---|
| Core Principle | Physical disruption via mincing, pipetting, or devices [22] | Chemical breakdown of ECM and adhesions using enzymes [1] |
| Processing Time | Faster (e.g., minutes) [1] | Slower (e.g., 30 mins to several hours) [1] |
| Cell Viability | Can be lower due to shear stress [1] | Typically higher when optimized [1] |
| Key Advantage | Preserves tumor microenvironment and cellular heterogeneity [22] | Generates a more homogenous cell population; better for reproducibility [22] |
| Key Disadvantage | Inconsistent yield, operator-dependent [1] | Can damage cell surface markers; requires optimization [1] |
| Ideal Application | Personalized medicine, TME studies [22] | Large-scale drug screening, controlled experiments [22] |
Step-by-Step Protocol
Combined Enzymatic-Mechanical Dissociation Workflow
This integrated protocol leverages the consistency of enzymatic digestion with the speed of mechanical force to achieve high yields of viable single cells. The overall workflow is summarized below.
Step 1: Harvesting and Washing Embryonic Organoids
- Gently transfer the organoids embedded in Matrigel or other 3D matrix to a conical tube.
- Dissolve the basement membrane matrix using a chelating agent like EDTA or a cell recovery solution [23].
- Wash the organoids twice with a cold, sterile buffer (e.g., PBS) to remove residual matrix and culture media.
Step 2: Initial Mechanical Mincing
- Using sterile surgical scalpel or scissors, mince the washed organoid fragments into the finest possible pieces in a small volume of dissociation buffer. This increases the surface area for enzymatic action.
Step 3: Enzymatic Digestion
- Prepare an enzymatic cocktail appropriate for the embryonic tissue type. A common combination includes Collagenase (e.g., 1-2 mg/mL) and Dispase (e.g., 1-2 U/mL) in a suitable buffer, potentially with added DNase [23].
- Incubate the minced organoid fragments in the enzymatic solution. The incubation time (typically 15-60 minutes) and temperature (usually 37°C) must be optimized for each organoid line [1]. Gently agitate the tube every 10-15 minutes.
Step 4: Termination of Digestion
- Once the tissue fragments appear dispersed (check under a microscope), neutralize the enzymatic reaction by adding a large volume (e.g., 2-3x the digestion volume) of cold complete cell culture media containing serum, which inhibits trypsin and other proteases.
Step 5: Gentle Mechanical Trituration
- Pipette the cell suspension up and down gently (10-20 times) using a serological pipette or a fire-polished Pasteur pipette. This mechanical action helps break down remaining small clumps into single cells. Avoid creating excessive foam.
Step 6: Filtration and Cell Pellet Collection
- Pass the cell suspension through a sterile cell strainer (e.g., 40µm) into a new conical tube to remove any remaining aggregates or debris.
- Centrifuge the filtrate at a low speed (e.g., 300-400 x g for 5 minutes) to pellet the cells. Carefully decant the supernatant.
Step 7: Resuspension and Assessment
- Resuspend the cell pellet in an appropriate buffer or culture medium.
- Determine cell viability using Trypan Blue exclusion and count the cells with a hemocytometer or automated cell counter. Target viability should be >85% for most downstream applications [1].
Quality Control and Troubleshooting
Table 3: Troubleshooting Common Issues
| Problem | Potential Cause | Solution |
|---|---|---|
| Low Cell Viability | Over-digestion with enzymes; harsh mechanical force. | Optimize enzyme concentration and incubation time; use gentler trituration. |
| Low Cell Yield | Incomplete digestion; inefficient dissociation. | Increase mincing fineness; optimize enzyme cocktail; extend incubation slightly. |
| Excessive Clumping | Incomplete neutralization of enzymes; DNA release. | Ensure sufficient serum in neutralization media; add DNase to the digestion mix [23]. |
Advanced and Emerging Techniques
While traditional methods are widely used, novel technologies are addressing their limitations. The Hypersonic Levitation and Spinning (HLS) method uses a non-contact, acoustic-based approach to generate precise hydrodynamic forces for tissue dissociation [2]. This technology has demonstrated high performance in dissociating human renal cancer tissue, achieving 92.3% cell viability and a 90% tissue utilization rate in just 15 minutes, thereby outperforming traditional methods in speed, yield, and preservation of rare cell populations [2]. Furthermore, microfluidic dissociation platforms offer automated, integrated systems that combine mechanical and enzymatic processes with filtration, enabling rapid processing (1-60 minutes) and consistent yields for various tissues, including kidney, heart, and liver [1].
The dissociation of delicate tissues, such as those from embryos, into viable single-cell suspensions represents a critical bottleneck in developmental biology research. Traditional enzymatic methods, which operate at 28-37°C, often induce significant cellular stress responses that compromise transcriptomic data integrity and cell viability [24] [25]. These limitations are particularly problematic for embryonic tissues and cell types hypersensitive to their microenvironment. Emerging cold-active protease technologies address these challenges by enabling effective tissue dissociation at low temperatures (4-6°C), dramatically reducing stress-induced artifacts and better preserving native transcriptional states [24] [1]. This application note details the implementation, optimization, and advantages of cold-active protease methodologies within the specific context of embryonic tissue research for single-cell applications.
Cold-active proteases, primarily subtilisin A derived from the psychrophilic bacterium Bacillus licheniformis, maintain high catalytic activity at low temperatures (4-6°C) where standard mammalian enzymatic activity is minimal [24] [25]. This fundamental property enables a paradigm shift in tissue dissociation strategy.
The core advantage lies in the profound reduction of transcriptional stress artifacts. During standard warm dissociation, cellular transcriptional machinery remains active, leading to rapid induction of stress-response genes that can obscure native biological signatures. In contrast, cold-active proteases operate at temperatures that largely preserve the transcriptional profile of the cell at the moment of harvest [24]. Research across diverse tissues demonstrates that collagenase-based dissociation at 37°C consistently upregulates a conserved core set of over 500 stress response genes, including immediate early genes like FOS and JUN, and heat shock proteins. This response is minimized when using cold-active proteases [25].
Table 1: Key Characteristics of Cold-Active Protease Versus Traditional Dissociation Methods
| Characteristic | Cold-Active Protease | Traditional Enzymatic (37°C) |
|---|---|---|
| Typical Temperature | 4-6°C | 28-37°C |
| Transcriptional Stress | Minimized | Significant (512+ core stress genes) [25] |
| Key Enzyme | Subtilisin A | Collagenase, Trypsin, Pronase |
| Cell Viability | High (>90% reported) [24] | Variable, often lower |
| Ideal for Sensitive Tissues | Embryonic, connective, tumor | Robust, less sensitive tissues |
| Impact on ECM Genes | Better preserved | Often downregulated [24] |
This technology is particularly vital for embryonic and other delicate tissues where cell-cell interactions and extracellular matrix (ECM) signaling are critical for understanding developmental processes. Traditional methods can specifically downregulate hallmark genes involved in cell specification and ECM production, skewing biological interpretation. Cold-active protease dissociation mitigates this, ensuring that downstream single-cell RNA sequencing (scRNA-seq) data more accurately reflects the in vivo state [24].
Quantitative Data and Performance Comparison
Empirical studies directly comparing dissociation methods provide compelling evidence for the superiority of the cold-active approach in preserving cellular integrity and transcriptomic fidelity.
In a study focusing on zebrafish embryonic tendons, dissociation with subtilisin A at 4°C resulted in reduced stress signatures and enhanced preservation of key tenocyte marker genes and ECM genes compared to 37°C collagenase treatment [24]. This is critical for embryonic research where understanding precise gene expression patterns is paramount.
A comprehensive analysis of 155,165 cells from various tissues, including patient-derived xenografts and cell lines, quantified the stark contrast between methods. The 37°C collagenase digestion induced a consistent and robust stress response across all cell types, which was significantly minimized by dissociation with a cold-active protease at 6°C [25]. This conserved stress response can confound biological interpretation, particularly in studies of cellular responses to external stimuli or in disease modeling.
Table 2: Performance Metrics of Dissociation Methods in scRNA-seq Studies
| Metric | Cold-Active Protease (6°C) | Traditional Enzymatic (37°C) | Context |
|---|---|---|---|
| Stress Gene Signature | Minimal induction | Strong induction (512-gene core set) [25] | Multiple tissues & cell lines |
| Mitochondrial Gene % | Lower, more stable | Higher, variable (indicates stress) [25] | Lymphoblastoid cell line |
| Cell Viability | >90% [24] | Variable (often lower) | Protocol optimization |
| Preservation of Native Markers | High (e.g., tenocyte genes) | Low (specific marker downregulation) [24] | Zebrafish embryo tendons |
| MHC Class I Expression | Basal levels | Upregulated (stress response) [25] | Tumor tissues |
Detailed Experimental Protocol for Embryonic Tissues
The following protocol is adapted from established methodologies for cold protease dissociation of zebrafish embryo tissues and solid tumors, optimized for general use with delicate embryonic tissues [24] [25].
Reagents and Solutions
- Cold Protease Stock Solution (100 mg/mL): Dissolve 100 mg of protease from B. licheniformis (Sigma-Aldrich, P5380) in 1 mL of 1x PBS (without calcium or magnesium). Aliquot and store at -80°C [24].
- Cold Protease Working Solution (10 mg/mL): Combine 100 µL of cold protease stock, 5 µL of 1M CaCl₂ (5 mM final), 1 µL of 0.5M EDTA (0.5 mM final), and 5 µL of 20 U/µL DNase I stock (100 U/mL final). Bring to 1 mL with ice-cold DPBS. Keep on ice until use [24].
- DPBS-BSA Solution (0.01%): Dissolve 0.5 mg of Bovine Serum Albumin (BSA) in 5 mL of ice-cold DPBS. Keep on ice [24].
- Ringer's Solution: 116 mM NaCl, 2.9 mM KCl, 10 mM CaCl₂·2H₂O, 5 mM HEPES pH 7.2. Adjust pH to 7.2 with NaOH [24].
Step-by-Step Dissociation Procedure
Tissue Harvest and Preparation:
- Sacrifice embryos according to institutional ethical guidelines.
- Rapidly dissect target tissues in ice-cold Ringer's solution or DPBS to minimize pre-digestion stress.
- Transfer tissues to a silicone polymer-coated or low-attachment plate on ice. Critical: Maintain low temperature throughout the preparatory phase.
Mechanical Mincing:
- Using fine surgical tools or needles (e.g., 21G), meticulously mince the tissue into the finest possible fragments in a small volume of cold DPBS. This increases the surface area for enzymatic action without heat generation.
Cold Protease Digestion:
- Remove the DPBS and add the ice-cold Cold Protease Working Solution (e.g., 500 µL for a small tissue volume).
- Incubate the tissue fragments on ice or in a 4°C cold room for a determined period (e.g., 30-90 minutes) with gentle agitation. Note: Digestion time must be empirically optimized for each embryonic tissue type and stage.
- Gently triturate the tissue fragments using a low-retention pipette tip every 15-20 minutes to aid dissociation.
Reaction Termination and Cell Collection:
- Once a single-cell suspension is achieved (monitor under a microscope), add an equal volume of ice-cold DPBS-BSA solution to halt the protease activity.
- Filter the cell suspension through a pre-wet 40 µm cell strainer to remove any remaining aggregates or debris.
- Centrifuge the flow-through at 300-400 x g for 5 minutes at 4°C to pellet the cells.
Cell Washing and Resuspension:
- Carefully aspirate the supernatant and gently resuspend the cell pellet in an appropriate ice-cold buffer (e.g., DPBS-BSA or cell culture medium).
- Perform a cell count and viability assessment using Trypan Blue exclusion or an automated cell counter.
- The single-cell suspension is now ready for downstream applications like scRNA-seq, flow cytometry, or cell culture.
Diagram 1: Cold-active protease dissociation workflow for embryonic tissues.
The Scientist's Toolkit: Essential Research Reagents
Successful implementation of this protocol relies on specific, high-quality reagents. The following table details the essential components.
Table 3: Key Research Reagent Solutions for Cold-Active Protease Dissociation
| Reagent / Material | Function / Purpose | Example Specification / Source |
|---|---|---|
| Protease from B. licheniformis | Cold-active enzyme that digests extracellular matrix proteins at low temperatures. | Sigma-Aldrich, P5380 [24] |
| DNase I | Degrades extracellular DNA released by damaged cells, preventing cell clumping. | Roche, 11284932001 [24] |
| Calcium Chloride (CaCl₂) | Cofactor for optimal protease activity and stability. | Component of working solution [24] |
| EDTA (Ethylenediaminetetraacetic acid) | Chelating agent that helps disrupt cell-cell adhesions by sequestering divalent cations. | Component of working solution [24] |
| DPBS (Dulbecco's PBS), no Ca²⁺/Mg²⁺ | Isotonic buffer for tissue washing and reagent preparation. | Gibco, 14190-144 [24] |
| Bovine Serum Albumin (BSA) | Acts as a protein stabilizer and helps quench protease activity after digestion. | Component of DPBS-BSA solution [24] |
| Low-Binding Tips & Tubes | Minimizes cell loss due to adhesion to plastic surfaces. | Critical for maintaining high cell yield |
| 40 µm Cell Strainer | Removes undissociated tissue chunks and large debris from the single-cell suspension. | Pluriselect, 43-10040-40 [24] |
Mechanism of Action: Preserving Native Biology
The fundamental superiority of cold-active proteases is rooted in their biochemical mechanism and its interaction with cellular processes. The following diagram and explanation outline this process.
Diagram 2: Mechanism of native transcriptome preservation with cold-active proteases.
At a mechanistic level, the cold-active protease subtilisin A efficiently hydrolyzes peptide bonds within the extracellular matrix (ECM) and proteins mediating cell-cell adhesions. It performs this function effectively at 4-6°C, a temperature at which the endogenous transcriptional machinery of the mammalian cell is largely dormant [24] [25]. Consequently, as the tissue is dissociated, the cells are unable to mount a significant new transcriptional response to the changing microenvironment.
In stark contrast, traditional enzymes like collagenase require 37°C to function optimally. At this physiological temperature, cells remain fully transcriptionally active. The process of digesting the ECM and detaching cells is interpreted by the cell as a severe stressor, triggering rapid stress-induced signaling pathways (e.g., MAPK/ERK) that lead to the upregulation of immediate early genes (FOS, JUN), heat shock proteins (HSPs), and genes involved in apoptosis and immune recognition (e.g., MHC class I) [25]. This results in a transcriptome that is a mixture of the native state and a dissociation artifact state, complicating data interpretation.
The adoption of cold-active protease technology for dissociating delicate embryonic tissues represents a significant advancement in single-cell research methodology. By operating at low temperatures, this method effectively minimizes technical artifacts, providing a cellular transcriptome that is a more faithful representation of the in vivo state. The detailed protocol and reagent toolkit provided herein offer a robust foundation for researchers to implement this technique, thereby enhancing the quality and biological relevance of downstream single-cell analyses in developmental biology and drug discovery.
The study of early embryonic development at a single-cell resolution requires the preparation of high-quality single-cell suspensions. Traditional tissue dissociation methods often compromise cell viability, alter transcriptional states, and preferentially lose rare but biologically critical cell populations. The Hypersonic Levitation and Spinning (HLS) technology represents a transformative approach that enables contactless tissue dissociation through precise hydrodynamic forces [2]. This technology is particularly valuable for embryonic research, where preserving the native cellular state is paramount for understanding developmental trajectories, lineage specification, and the molecular foundations of pluripotency [26] [27].
HLS technology addresses critical limitations of both mechanical and enzymatic dissociation methods. Conventional approaches typically involve long processing times (often 60 minutes), achieve limited tissue utilization (approximately 70%), and frequently result in reduced cell viability and loss of fragile cell types [2]. In contrast, HLS achieves superior performance with 90% tissue utilization in just 15 minutes while maintaining 92.3% cell viability, making it uniquely suited for processing precious embryonic samples where cell integrity and representation must be preserved for downstream single-cell analyses [2] [28].
Fundamental Mechanism
The HLS system employs a triple-acoustic resonator probe that generates GHz-frequency acoustic waves to create a confined flow field around tissue samples [2]. This configuration enables two simultaneous phenomena: stable levitation of the target tissue and a 'press-and-rotate' operation that induces rapid self-rotation of the sample. The hypersonic streaming effect transfers acoustic energy into the kinetic energy of the surrounding fluid, producing microscale 'liquid jets' that exert precise hydrodynamic shear forces in a completely non-contact manner [2] [28].
The dissociation process occurs through multiple synergistic mechanisms. The eccentric torque generated by asymmetric fluid pressure creates continuous spinning of the tissue block, enhancing shear stress distribution across the tissue surface. Simultaneously, the hypersonic streaming effect improves the penetration of enzyme solutions into deeper tissue layers by disrupting cell-to-cell connections and facilitating the binding of enzymes to collagen and matrix proteins. This dual mechanical and chemical action enables thorough tissue dissociation while protecting cellular integrity [2].
Automated System Configuration
For standardized operation in research settings, an automated tissue dissociation apparatus integrating the HLS technology has been developed. This system features four vertical tubes (inlet, sample, outlet, and waste) and two specialized chambers (for digestion and single-cell collection) that seamlessly integrate dissociation, fluid replacement, filtration, and output functions [2]. This automated design replaces traditional labor-intensive procedures, enhances experimental reproducibility, and provides a streamlined workflow for preparing single-cell suspensions from embryonic tissues for subsequent applications including single-cell RNA sequencing, proteomics, and primary cell culture [2].
Quantitative Performance Assessment
Table 1: Comprehensive Performance Comparison Between HLS and Traditional Dissociation Methods
| Performance Metric | HLS Technology | Traditional Mechanical Methods | Traditional Enzymatic Methods |
|---|---|---|---|
| Processing Time | 15 minutes | 45-60 minutes | 60-120 minutes |
| Tissue Utilization | 90% | 60-70% | 70-75% |
| Cell Viability | 92.3% | 75-85% | 85-90% |
| Rare Cell Population Preservation | Excellent | Poor | Moderate |
| Operator Dependency | Low (Automated) | High | Moderate |
| Shear Force Control | Precise hydrodynamic control | Variable, often excessive | Limited control |
Table 2: Application-Specific Advantages of HLS for Embryonic Research
| Research Application | Key HLS Advantages | Impact on Data Quality |
|---|---|---|
| Single-Cell RNA Sequencing | Preserves transcriptional states, minimizes stress-induced gene expression | More accurate representation of native gene expression profiles [26] |
| Proteomic Analysis | Maintains cell surface epitopes and intracellular protein integrity | Improved detection of low-abundance proteins and modification states [29] |
| Lineage Tracing Studies | Retains rare progenitor populations | Enables comprehensive reconstruction of developmental trajectories |
| Stem Cell Pluripotency Analysis | Gentle processing maintains fragile pluripotent states | Better characterization of pluripotency transitions [26] |
| Primary Cell Culture | High viability supports successful establishment of cultures | Enhanced outgrowth and proliferation potential |
The performance advantages of HLS technology extend beyond simple efficiency metrics. In comparative studies using human renal cancer tissue models, HLS "greatly outperforms traditional techniques in tissue utilization (90% in 15 minutes vs. 70% in 60 minutes) and dissociation rate while also excelling in maintaining high cell viability (92.3%) and preserving rare cell populations" [2]. This performance profile translates directly to embryonic research contexts, where maintaining the integrity of rare cell types—such as specific progenitor populations during lineage specification events—is critical for accurate developmental analysis [27] [29].
Experimental Protocol for Embryonic Tissue Dissociation
Sample Preparation and Reagent Setup
Materials Required:
- Freshly isolated embryonic tissue (specific stage depending on experimental design)
- Sterile phosphate-buffered saline (PBS) without calcium and magnesium
- Enzyme solution: Collagenase/Dispase combination in appropriate buffer
- HLS dissociation apparatus with triple-acoustic resonator probe
- Cell strainer (40μm nylon mesh)
- Collection tubes containing culture medium with serum
Preparation Steps:
- Isolate embryonic tissues using standard microdissection techniques appropriate for the developmental stage.
- Rinse tissues briefly in calcium/magnesium-free PBS to remove residual blood and debris.
- Prepare enzyme solution at working concentration (typically 1-2 mg/mL collagenase/dispase in PBS).
- Pre-warm the HLS collection chamber to 37°C to maintain optimal enzymatic activity.
HLS Dissociation Procedure
- System Initialization: Power on the HLS apparatus and initialize the software. Prime the fluidics system with pre-warmed PBS to remove air bubbles and ensure proper flow.
- Sample Loading: Transfer the embryonic tissue sample (maximum recommended volume: 5mm³) into the designated sample tube containing enzyme solution.
- Parameter Settings: Configure the operational parameters:
- Acoustic frequency: 1-2 GHz (optimized for embryonic tissue density)
- Processing duration: 10-15 minutes
- Temperature maintenance: 37°C
- Flow rate: 0.5-1.0 mL/min (dependent on tissue size and density)
- Initiate Dissociation: Start the automated processing cycle. The system will:
- Levitate and rotate the tissue sample
- Circulate enzyme solution through the dissociation chamber
- Maintain optimal temperature throughout the process
- Cell Collection: After completion, the system automatically transfers the cell suspension through the integrated filter to remove undissociated aggregates and debris.
- Termination and Recovery: Collect the single-cell suspension from the output chamber. Centrifuge at 300×g for 5 minutes and resuspend in appropriate medium for downstream applications.
Quality Assessment and Validation
Cell Viability Analysis:
- Mix 10μL of cell suspension with 10μL of trypan blue solution.
- Count viable (unstained) and non-viable (blue) cells using a hemocytometer or automated cell counter.
- Acceptable viability threshold: >90% for embryonic tissues.
Cell Yield Quantification:
- Determine total cell count per mg of starting tissue.
- Compare yield to historical data from traditional methods.
Population Integrity Validation:
- Perform flow cytometry analysis with lineage-specific markers relevant to the embryonic stage.
- Compare population distribution to intact tissue reference data obtained through other methods (e.g., tissue section immunostaining).
Diagram 1: Complete workflow for embryonic tissue dissociation using HLS technology, showing sample preparation, processing, and downstream applications.
Research Reagent Solutions for Embryonic Cell Dissociation
Table 3: Essential Research Reagents for HLS-Mediated Embryonic Tissue Dissociation
| Reagent/Category | Specific Function | Application Notes for Embryonic Research |
|---|---|---|
| Collagenase/Dispase Blend | Digests extracellular matrix components | Use stage-specific concentrations; earlier embryos typically require lower enzyme concentrations (0.5-1.0 mg/mL) |
| Calcium/Magnesium-Free Buffer | Prevents cell re-aggregation during processing | Essential for maintaining single-cell suspension; may be supplemented with EDTA for enhanced dissociation |
| Serum-Free Culture Medium | Maintains cell viability during processing | Prevents differentiation of pluripotent cells; formulation should match downstream culture requirements |
| Viability Stain (Trypan Blue) | Assessment of membrane integrity | Standardized quantification of dissociation quality; alternative fluorescent viability dyes compatible with FACS |
| Lineage Tracing Markers | Identification of specific cell populations | Antibody panels targeting stage-specific surface antigens (e.g., OCT4, NANOG for pluripotent cells) |
| Protease Inhibitors | Halts enzymatic activity post-digestion | Critical for proteomic applications; must be added immediately following dissociation [29] |
| RNase Inhibitors | Preserves RNA integrity | Essential for transcriptomic studies; should be included in collection buffers for scRNA-seq applications [26] |
Integration with Single-Cell Multi-Omic Technologies
The application of HLS technology for embryonic tissue dissociation enables unprecedented integration with advanced single-cell multi-omic platforms. The high viability and preservation of native transcriptional states achieved through contactless dissociation make HLS-processed cells ideally suited for single-cell RNA sequencing (scRNA-seq) applications aimed at reconstructing developmental trajectories [26] [27]. Recent studies leveraging Smart-seq2-based scRNA-seq have successfully mapped transition processes from embryonic stem cells to feeder-free extended pluripotent stem cells, revealing critical molecular pathways involved in pluripotency shifts [26].
Similarly, HLS technology supports emerging single-cell proteomic approaches that require intact cells with preserved surface epitopes and intracellular structures. The gentle dissociation process minimizes protein degradation and maintains post-translational modification states, enabling more comprehensive profiling of the proteomic landscapes that drive embryonic development [29]. When combined with spatial transcriptomic methods, HLS-dissociated cells can provide complementary single-cell resolution data that enhances interpretation of spatial patterning information within intact embryonic structures [27].
Diagram 2: Integration of HLS technology with single-cell multi-omic platforms for comprehensive analysis of embryonic development.
Hypersonic Levitation and Spinning technology represents a significant advancement in embryonic tissue dissociation methodology, addressing long-standing challenges in single-cell research. By providing a contactless, gentle yet highly efficient dissociation process, HLS enables researchers to obtain high-quality single-cell suspensions that faithfully maintain native cellular states [2] [28]. This capability is particularly valuable for constructing comprehensive atlases of embryonic development, where preserving rare cell populations and minimizing technical artifacts is essential for accurate biological interpretation.
The future development of HLS technology will likely focus on further miniaturization and specialization for specific embryonic stages and tissue types. Integration with downstream analytical platforms through microfluidic interfaces may enable complete closed-system processing from intact tissue to sequencing-ready libraries. Additionally, ongoing refinement of acoustic parameters tailored to the unique physical properties of embryonic tissues at different developmental stages will continue to improve dissociation efficiency and cellular recovery. As single-cell multi-omic technologies advance, HLS-based tissue dissociation will play an increasingly critical role in unraveling the complex molecular programs that govern human embryonic development, with potential applications in regenerative medicine, infertility research, and developmental disorder modeling [26] [27] [29].
Tissue dissociation into single-cell suspensions is a critical foundational technique in modern biological research, serving as the gateway to advanced analytical and therapeutic applications [1]. For researchers working with embryonic tissues, the process of disaggregation presents unique challenges, as it must balance the need for high cell yield and viability with the preservation of delicate cellular phenotypes and the potential for subsequent cell differentiation and growth [30]. The current bottleneck in manufacturing of tissue-engineered and cell-based regenerative medicine therapies is the lack of rigorous, standardized, and validated systems that enable the reproducible dissociation of tissues into optimal cell populations [1].
This application note examines the integration of tissue dissociation methodologies with three pivotal downstream applications: single-cell RNA sequencing (scRNA-seq), flow cytometry, and organoid culture. By providing a comprehensive overview of current technologies, detailed protocols, and quality control measures, we aim to equip researchers with the knowledge necessary to optimize their dissociation workflows for embryonic tissue research, ultimately enhancing the reliability and reproducibility of their experimental outcomes.
Current State of Tissue Dissociation Technologies
Traditional tissue dissociation has primarily relied on enzymatic and mechanical methods, each with significant limitations for embryonic tissue applications. Enzymatic approaches using collagenase, trypsin, dispase, and other enzymes can damage cell surface proteins, reduce viability, and induce stress-related transcriptional responses that compromise downstream analyses [1] [30]. Mechanical methods often inflict substantial mechanical stress leading to membrane damage and reduced viability, with results that are highly operator-dependent [2]. These challenges are particularly acute when working with embryonic tissues, which contain delicate cell types that may be lost or compromised using conventional approaches.
Recent technological advancements have yielded several innovative dissociation platforms that offer significant improvements for processing sensitive tissues like embryonic samples:
Table 1: Comparison of Advanced Tissue Dissociation Technologies
| Technology | Mechanism | Processing Time | Cell Viability | Key Advantages | Reference |
|---|---|---|---|---|---|
| Hypersonic Levitation and Spinning (HLS) | Acoustic resonator generating 'press-and-rotate' motion via microscale liquid jets | 15 minutes | 92.3% | Non-contact method; preserves rare cell populations; 90% tissue utilization | [2] |
| Semi-automated Mechanical Dissociation (Via Extractor) | Standardized mechanical agitation with EDTA | 5-7 minutes | High (exact % not specified) | Reduced user variability; improved success rate for fresh tissue | [31] |
| PythoN i System | Not specified | 15 minutes (60 minutes for skin) | ~90% | Integrated quality controls; 8 channels for parallel processing | [30] |
| Microfluidic Platforms | Enzymatic and mechanical dissociation in microfluidic channels | 20-60 minutes | 60%-95% (varies by cell type) | Integrated workflows; reduced reagent usage | [1] |
| Electrical Dissociation | Electrical field facilitation | 5 minutes | 80%-90% | Rapid processing; minimal enzymatic damage | [1] |
| Precellys Evolution with Multi-Tissue Kit | Bead-based homogenization with variable speed agitation | ~50 minutes | >80% | Adaptable to different tissues; preserves cell viability | [32] |
Dissociation Workflows for Specific Applications
Optimized Protocol for scRNA-seq Applications
For single-cell RNA sequencing studies, particularly with embryonic tissues, maintaining RNA integrity and minimizing dissociation-induced stress responses is paramount. Stress during tissue dissociation occurs because tissues are exposed to conditions that encourage single cell separation, potentially inducing the expression of heat shock proteins and altering native transcriptional profiles [30].
Detailed Protocol for Embryonic Tissue Dissociation for scRNA-seq:
Sample Preparation:
- Rapidly process embryonic tissue samples within 2 hours of collection [33].
- Place tissue in cold, sterile phosphate-buffered saline (PBS) for transport.
- Mince tissue into approximately 1-2 mm³ fragments using a sterile surgical scalpel in a small volume of dissociation buffer.
Enzymatic Dissociation:
- Prepare an enzymatic cocktail appropriate for embryonic tissue. A combination of collagenase (1-2 mg/mL), dispase (1-2 mg/mL), and hyaluronidase (0.1-0.2 mg/mL) in PBS with 0.1% BSA has proven effective for various embryonic tissues [1].
- Use 5-10 mL of enzyme solution per 100 mg of tissue.
- Incubate at 37°C with gentle agitation for 15-45 minutes, monitoring dissociation every 10-15 minutes. Embryonic tissues typically require shorter digestion times than mature tissues.
Mechanical Disruption:
- Gently triturate the tissue-enzyme mixture using a wide-bore pipette every 15 minutes to facilitate dissociation.
- Avoid excessive force or bubble formation which can damage cells.
- For more robust tissues, consider using the Precellys Evolution Touch Homogenizer with bead-based technology at optimized speeds [32].
Reaction Termination and Cell Recovery:
- Add an equal volume of cold complete medium (containing FBS) to terminate enzymatic activity.
- Filter the cell suspension through a 40 μm cell strainer to remove undissociated tissue and debris.
- Centrifuge at 300-400 × g for 5 minutes at 4°C.
- Resuspend cell pellet in appropriate buffer (e.g., PBS with 0.04% BSA) for counting and downstream processing.
Quality Control:
- Assess cell viability using Trypan Blue exclusion or fluorescent dyes such as SYTO9/propidium iodide [30].
- Ensure viability exceeds 80% for optimal scRNA-seq results.
- Count cells using a hemocytometer or automated cell counter, aiming for a concentration of 700-1,200 cells/μL for 10x Genomics platforms.
The entire process from tissue collection to single-cell suspension should be completed as rapidly as possible to minimize ex vivo transcriptional changes. For embryonic tissues, which are particularly sensitive to environmental stress, working quickly and maintaining cool temperatures whenever possible is essential.
Workflow Integration Diagram for scRNA-seq
Optimized Protocol for Flow Cytometry Applications
For flow cytometry, preserving cell surface epitopes and maintaining structural integrity are critical considerations. Enzymatic dissociation can inadvertently modify or destroy surface proteins, compromising antibody binding and subsequent analysis [34].
Detailed Protocol for Embryonic Tissue Dissociation for Flow Cytometry:
Gentle Mechanical Dissociation:
- For loosely associated embryonic tissues (e.g., early embryonic organs), gentle mechanical dissociation may be sufficient.
- Place tissue in cold flow cytometry buffer (PBS with 1-5% FBS and 1-5 mM EDTA).
- Gently dissociate using a sterile syringe plunger through a 40 μm cell strainer or using a gentleMACS Dissociator with appropriate programs.
Enzyme Selection and Optimization:
- When enzymatic digestion is necessary, use enzyme blends specifically formulated for cell surface antigen preservation.
- Consider using a non-enzymatic chemical dissociation agent such as egtazic acid (EGTA) for calcium chelation, which is particularly gentle on embryonic cells [34].
- Limit enzymatic digestion time to 15-30 minutes at 37°C or use lower temperatures (30-32°C) for longer durations.
Staining and Analysis:
- After dissociation, keep cells at 4°C to prevent internalization of surface receptors.
- Use Fc receptor blocking agents before antibody staining to reduce non-specific binding.
- Include viability dyes (e.g., propidium iodide, 7-AAD, or fixable viability dyes) to exclude dead cells from analysis.
For embryonic tissues containing rare cell populations, additional purification steps using buoyancy-activated cell sorting (BACS) with microbubbles or magnetic-activated cell sorting (MACS) may be implemented to enrich target populations before flow analysis [34].
Optimized Protocol for Organoid Culture Applications
Establishing organoids from dissociated embryonic tissues requires preservation of stem cell populations and maintenance of regenerative capacity. The dissociation approach must balance thorough tissue disruption with preservation of cellular functionality for subsequent 3D growth.
Detailed Protocol for Embryonic Tissue Dissociation for Organoid Culture:
Stem Cell Preservation:
- Use chelating agents like EDTA (2.5-5 mM) instead of or in combination with gentle enzymatic digestion to preserve stem cell viability [31].
- For intestinal organoids, incubation with 2.5 mM EDTA at 4°C for 30 minutes on a roller-mixer has proven effective for crypt isolation [31].
- Include Rho-kinase inhibitor (Y-27632, 10 μM) in the dissociation and initial culture media to prevent anoikis (detachment-induced cell death) [31].
Semi-Automated Dissociation:
- For improved consistency, consider using semi-automated systems like the Via Extractor.
- Place tissue in 0.1% BSA-coated pouches with 5 mL dissociation solution.
- Run at optimized conditions (150 rpm, 5-7 minutes at 4°C) for embryonic tissue [31].
Cell Processing and Plating:
- Centrifuge dissociated cells at 800 × g for 5 minutes.
- Resuspend in appropriate extracellular matrix (e.g., Matrigel, Geltrex).
- Plate approximately 100 crypts or organoid fragments per well in 20 μL matrix domes.
- Culture with tissue-specific growth factors and maintain at 37°C with 5-10% CO₂.
Long-Term Maintenance:
Workflow Integration Diagram for Organoid Culture
Quality Control and Troubleshooting
Robust quality control after tissue dissociation is essential for successful downstream applications. Key parameters must be assessed and optimized to ensure experimental reliability.
Table 2: Quality Control Parameters and Optimization Strategies
| QC Parameter | Importance | Assessment Method | Optimal Range | Improvement Strategies |
|---|---|---|---|---|
| Cell Viability | Determines functional cells for downstream applications | Trypan Blue, propidium iodide, SYTO9/PI staining | >80% for scRNA-seq; >90% for organoid culture | Reduce digestion time; optimize enzyme concentration; use gentler mechanical methods [30] |
| Cell Clumping | Causes multiplets in scRNA-seq; affects flow cytometry accuracy | Brightfield/confocal microscopy; cell counters | Minimal clumps (<5% doublets) | Optimize filtration; include DNase; accurate cell counting [30] |
| Debris Content | Creates false positives in scRNA-seq; interferes with flow cytometry | Flow cytometry scatter plots; microscopy | Minimal debris | Additional filtration; density gradient centrifugation; differential centrifugation |
| Stress Marker Expression | Induces artifacts in transcriptional profiles | qPCR for heat shock proteins; stress gene analysis | Minimal induction | Minimize time from collection to processing; lower digestion temperature [30] |
| Surface Antigen Integrity | Critical for flow cytometry and cell sorting | Antibody staining efficiency; MFI comparison | High staining index | Use gentler dissociation methods; enzyme-free approaches [34] |
| Stem Cell Functionality | Essential for organoid formation | Organoid forming efficiency; stem cell marker expression | Tissue-dependent | Use EDTA-based dissociation; include Rho-kinase inhibitor [31] |
Common issues encountered during tissue dissociation and their solutions include:
- Low Cell Viability: Reduce enzymatic digestion time; lower incubation temperature; implement gentler mechanical disruption; add viability-enhancing compounds to buffers.
- Excessive Clumping: Increase filtration stringency (use 30-40 μm strainers); add DNase I (10-100 μg/mL) to digest free DNA from lysed cells; optimize cell concentration.
- Incomplete Dissociation: Optimize enzyme composition and concentration; extend digestion time with periodic gentle mechanical agitation; pre-mince tissue into smaller fragments.
- Loss of Rare Cell Populations: Implement gentler dissociation methods like HLS [2] or BACS [34]; avoid prolonged processing; use specialized preservation media.
Research Reagent Solutions
Selecting appropriate reagents and equipment is crucial for successful tissue dissociation. The following table outlines key solutions for embryonic tissue dissociation workflows.
Table 3: Essential Research Reagents and Equipment for Tissue Dissociation
| Reagent/Equipment | Function | Application Notes | Representative Examples |
|---|---|---|---|
| Multi-Tissue Dissociation Kits | Provide optimized enzyme blends for various tissues | Streamline protocol development; ensure reproducibility | Precellys Multi-Tissue Dissociation Kit [32] |
| Collagenase/Dispase Blends | Digest extracellular matrix components | Effective for fibrous tissues; concentration and time need optimization | Various commercial formulations [1] |
| EDTA/EGTA | Chelating agents that disrupt cell-cell junctions | Gentle alternative to enzymes; preserves stem cell function | 2.5-5 mM solutions [31] [34] |
| Rho-Kinase Inhibitor (Y-27632) | Prevents anoikis in dissociated cells | Critical for organoid culture; improves cell survival post-dissociation | 10 μM in dissociation and culture media [31] |
| Bovine Serum Albumin (BSA) | Carrier protein; reduces non-specific cell adhesion | Improves cell viability and recovery; standard component of dissociation buffers | 0.1-0.5% in dissociation buffers [31] |
| Automated Homogenizers | Provide consistent mechanical disruption | Reduce operator variability; improve reproducibility | Precellys Evolution Touch Homogenizer [32] |
| Semi-Automated Dissociation Systems | Integrated dissociation with temperature control | Standardize process; improve efficiency | Cytiva Via Extractor [31]; Singleron PythoN i [30] |
| Advanced Acoustic Systems | Non-contact dissociation through hypersonic streaming | Preserves delicate cells; maintains rare populations | Hypersonic Levitation and Spinning (HLS) [2] |
| Microbubble Separation Systems | Gentle post-dissociation cell separation | Ideal for rare cell populations; maintains cell function | Akadeum Buoyancy-Activated Cell Sorting (BACS) [34] |
The successful integration of tissue dissociation with downstream applications requires careful consideration of methodology, quality control, and application-specific requirements. For embryonic tissue research, where cell viability, phenotypic preservation, and functional potential are paramount, selecting the appropriate dissociation strategy is particularly critical.
Emerging technologies such as hypersonic levitation, semi-automated systems, and advanced microfluidic platforms offer promising alternatives to traditional methods, addressing key limitations in consistency, efficiency, and cell preservation. By implementing the protocols and quality control measures outlined in this application note, researchers can significantly enhance the reliability and reproducibility of their single-cell suspension preparation, ultimately advancing our understanding of embryonic development and accelerating progress in regenerative medicine and therapeutic development.
As the field continues to evolve, further standardization of dissociation protocols and validation of new technologies will be essential to fully realize the potential of single-cell analyses and organoid-based models in embryonic tissue research.
Solving Common Embryonic Dissociation Challenges: A Troubleshooting Guide
Obtaining high-quality single-cell suspensions from embryonic tissues is a critical step in single-cell RNA sequencing (scRNA-seq), but researchers often face the dual challenges of enzyme toxicity and mechanical stress, which can severely compromise cell viability and experimental outcomes. The complex cell-cell junctions and delicate nature of embryonic tissues make them particularly susceptible to damage during dissociation. Preserving cell viability is not merely about keeping cells alive; it is about maintaining their native transcriptional state to ensure that the resulting data accurately reflects the in vivo biology rather than stress-induced artifacts. This Application Note provides a detailed framework of solutions and protocols to overcome these hurdles, enabling the generation of robust and reliable single-cell data for your research.
Core Challenges in Embryonic Tissue Dissociation
The process of dissociating tissue into single cells inherently inflicts stress. Understanding the specific nature of these stressors is the first step toward mitigating their effects.
Enzyme Toxicity: The use of proteolytic enzymes like trypsin, while effective at breaking down adhesion molecules, can be cytotoxic. Prolonged incubation or the use of overly concentrated enzymes can damage cell surface proteins, receptors, and even intracellular components, leading to activated stress pathways and altered gene expression profiles [36]. The optimal activity temperature for many enzymes (e.g., 37°C) can further accelerate cellular stress and RNA degradation [36].
Mechanical Stress: Mechanical homogenization methods—such as grinding, pipette trituration, or using dissociators—apply physical forces to break apart the tissue. Excessive mechanical force can lead to shear stress, which physically ruptures cell membranes, causing immediate cell death and the release of intracellular RNAses that further degrade the RNA of surrounding cells [37]. The dense, fibrous nature of some tissues exacerbates this risk.
The Apoptotic Cascade: Both enzymatic and mechanical stresses can trigger early-stage apoptosis. Even if cells appear viable directly after dissociation (e.g., by dye exclusion tests), they may be undergoing programmed cell death, leading to widespread RNA degradation and poor data quality during the subsequent sequencing steps [36].
The table below summarizes the primary stressors and their direct impacts on cell quality and data.
Table 1: Primary Dissociation Stressors and Their Impacts on Single-Cell Viability and Data Quality
| Stressor Category | Specific Examples | Impact on Cell Viability & Data Quality |
|---|---|---|
| Enzymatic Toxicity | Trypsin, Collagenase, Dispase [36] | Cell surface receptor degradation; induction of stress-response genes; altered transcriptional profiles [36]. |
| Mechanical Shear | Pipette trituration, grinding, blending [37] | Physical membrane rupture; release of RNAses; increased background noise in sequencing data [37]. |
| Temperature Stress | Prolonged enzymatic activity at 37°C [36] | Accelerated apoptosis and RNA degradation; reduced RNA integrity number (RIN) [36]. |
| Chemical Stress | Harsh detergents (e.g., Triton X-100) [36] | Compromised membrane integrity; non-physiological cellular states. |
Visualizing the Dissociation Stress Landscape
The following diagram maps the logical relationship between dissociation methods, the stressors they introduce, and their ultimate impact on experimental success.
Optimized Solutions and Detailed Protocols
A combination of strategic method selection and parameter optimization is key to successful tissue dissociation.
Strategy 1: Mitigating Enzyme Toxicity
A. Enzyme Selection and Cocktail Formulation The choice of enzyme should be tailored to the specific embryonic tissue of interest. Harsh enzymes like trypsin can be replaced or supplemented with gentler alternatives.
- Gentle Enzymes: Use enzymes like Dispase, which cleaves fibronectin and collagen IV with high specificity, leaving integrins and other surface proteins largely intact. It is particularly well-suited for epithelial tissues and organoids [36].
- Enzyme Cocktails: Utilize commercially available, pre-optimized enzyme cocktails designed for complex tissues. These often contain a blend of collagenases (e.g., Type I for softer tissues, Type II for cartilage/bone), hyaluronidase (to digest hyaluronic acid in the ECM), and neutral proteases, providing a balanced and effective dissociation while minimizing damage [36].
- Rationale: Different tissues have unique ECM compositions. A targeted enzymatic approach is more efficient and less damaging than a single broad-spectrum protease.
B. Protocol Optimization Fine-tuning the enzymatic reaction parameters is crucial for preserving viability.
- Temperature vs. Time Trade-off: While many enzymes are most active at 37°C, this temperature accelerates metabolic stress. A highly effective strategy is to perform the enzymatic digestion at 4°C for a longer duration (e.g., 30-60 minutes). This slows down enzymatic activity but virtually halts cellular metabolism and RNA degradation, resulting in higher RNA integrity [36].
- Concentration and Monitoring: Use the lowest effective enzyme concentration. Conduct pilot studies to determine the minimal time required for tissue dissociation to avoid "over-digesting" the sample. The reaction can be monitored visually under a microscope for cell release.
Detailed Protocol: Low-Temperature Enzymatic Dissociation
- Preparation: Chill a low-calcium buffer (e.g., PBS with 0.5 mM EDTA) and enzyme solution on ice.
- Tissue Processing: Mince the embryonic tissue into small (~1 mm³) pieces using a sterile scalpel in the cold buffer.
- Incubation: Transfer the tissue pieces to the chilled enzyme solution. Place the tube on a rocking platform in a cold room (4°C) for 30-60 minutes.
- Quenching: After incubation, add a double volume of cold, serum-containing media or a specific enzyme inhibitor to halt the reaction completely.
- Gentle Dissociation: Gently pipette the tissue up and down 5-10 times with a wide-bore pipette tip. Avoid creating bubbles.
- Filtration and Wash: Pass the cell suspension through a pre-wet, cell-strainer cap (e.g., 40 µm or 70 µm). Centrifuge the flow-through at 300-400 x g for 5 minutes at 4°C and resuspend the pellet in a suitable buffer for counting.
Strategy 2: Minimizing Mechanical Stress
A. Gentle Mechanical Techniques The goal is to apply the minimal force necessary to disaggregate the tissue.
- Wide-Bore Pipette Tips: Always use wide-bore or filtered tips when pipetting cell suspensions to reduce shear forces [36].
- Dounce Homogenization: For smaller samples, using a loose-fitting Dounce homogenizer with a few gentle strokes is often more controlled and effective than vigorous pipetting.
- Instrument-Assisted Dissociation: Systems like the gentleMACS Dissociator provide standardized, programmed mechanical agitation that is more reproducible and often gentler than manual methods [36].
B. The Single-Nuclei RNA-Seq Alternative When working with tissues that are exceptionally sensitive or difficult to dissociate (e.g., neuronal tissue, certain embryonic structures), switching to single-nuclei RNA sequencing (snRNA-seq) is a powerful alternative.
- Principle: snRNA-seq isolates nuclei instead of whole cells. The protocol for isolating nuclei is inherently harsher (involving detergents) but is performed at cold temperatures and is very rapid, which locks in the nuclear transcriptome and avoids the stress responses associated with whole-cell dissociation [38] [36].
- Advantages:
- Bypasses problems with large or complex cells that are difficult to dissociate intact.
- Allows for the use of archived or frozen tissues.
- Minimizes stress-related transcriptional artifacts.
- Disadvantages:
- Cytoplasmic RNA is not captured, missing some transcripts.
- The transcriptome coverage per "cell" is typically lower than with scRNA-seq.
Integrated Workflow for Optimal Viability
The following diagram outlines a comprehensive workflow that integrates the solutions above to maximize cell viability for scRNA-seq.
The Scientist's Toolkit: Essential Reagents and Materials
A successful dissociation experiment relies on having the right tools. The following table catalogs key reagents and their functions.
Table 2: Research Reagent Solutions for Embryonic Tissue Dissociation
| Reagent/Material | Function & Rationale | Example Use Case |
|---|---|---|
| Dispase [36] | Gentle neutral protease; cleaves fibronectin and Collagen IV without damaging cell surface receptors. | Ideal for dissociating embryonic epithelial layers and organoids. |
| Collagenase Blends [36] | Enzyme mixtures (Types I-V) that target the collagenous ECM; often supplemented with neutral proteases. | Effective for dissociating mesenchymal embryonic tissues. |
| Wide-Bore Pipette Tips [36] | Reduces fluid shear stress during pipetting, protecting delicate cells from membrane rupture. | Essential for all steps after enzymatic digestion is quenched. |
| Cell Strainers (e.g., 40 µm) | Removes undissociated tissue clumps and large debris to prevent clogging in microfluidic devices. | A standard clean-up step before loading cells for scRNA-seq. |
| Viability Dyes (e.g., Propidium Iodide) [36] | Fluorescent dyes that selectively penetrate dead cells with compromised membranes, allowing for viability assessment. | Used with a hemocytometer or automated cell counter to quantify viability pre-sequencing. |
| Dead Cell Removal Kits (e.g., magnetic bead-based) | Selectively removes apoptotic and necrotic cells from the suspension, enriching the live cell population. | Critical for improving data quality when initial viability is suboptimal (e.g., <80%). |
| Cold Buffers with EDTA/EGTA [36] | Chelates calcium and magnesium ions, weakening cadherin-mediated cell-cell adhesions; cold temperature suppresses metabolism. | Used as the base for all dissection and enzyme solutions to maintain viability. |
Concluding Remarks
Achieving high cell viability in embryonic tissue dissociation is a challenging but manageable process. It requires a deliberate balance between enzymatic efficiency and cellular preservation, and between mechanical force and structural integrity. By understanding the sources of stress, adopting a strategy of cold enzymatic digestion, employing gentle mechanical techniques, and considering snRNA-seq as a viable alternative for problematic tissues, researchers can significantly improve the quality and reliability of their single-cell suspensions. The protocols and tools detailed in this Application Note provide a concrete pathway to overcoming these common hurdles, ultimately ensuring that the resulting genomic data is a true reflection of embryonic development and not an artifact of the dissociation process.
Preventing and Resolving Cell Clumping and Re-aggregation
In the dissociation of embryo tissue for single-cell suspension research, the formation of cell clumps and re-aggregation events represents a critical bottleneck that compromises experimental outcomes. These phenomena directly impact cell viability, analysis accuracy, and the reliability of downstream applications such as single-cell RNA sequencing and flow cytometry [1] [39]. Clumping occurs when environmental stresses, including enzymatic digestion and mechanical dissociation, accelerate cell death, resulting in the release of "sticky" DNA molecules that bind neighboring cells together [40] [41]. This application note details evidence-based protocols and solutions to mitigate these challenges, specifically contextualized within embryo tissue research for scientific and drug development professionals.
Understanding the Causes of Cell Clumping
Effective prevention of cell clumping begins with a comprehensive understanding of its underlying mechanisms. The following table summarizes the primary causes and their contributing factors.
Table 1: Major Causes of Cell Clumping and Re-aggregation
| Cause Category | Specific Factors | Impact on Cell Suspension |
|---|---|---|
| Cell Death | Freeze-thaw cycles, enzymatic tissue dissociation, physical force [40] [41] | Release of genomic DNA that acts as "glue" between cells [40] [39] |
| Enzymatic Digestion | Over-digestion with trypsin, incorrect enzyme concentrations [41] [42] | Damage to cell surface proteins and increased cellular stress [1] |
| Physical Handling | Inadequate pipetting, vortexing at high speeds, centrifugation errors [39] | Physical damage to cells and uneven force distributions [2] |
| Adhesion & Cations | Cation-dependent cell adhesion, lack of chelating agents [39] | Natural re-aggregation of cells after dissociation [42] |
| Culture Conditions | Over-confluency, bacterial or fungal contamination [41] | Cell lysis and release of clump-promoting debris [41] |
Established Protocols for Clump Prevention and Resolution
Enzymatic and Chemical Intervention with DNase I
The addition of DNase I is a cornerstone strategy for degrading the extracellular DNA network that binds cells into clumps [40] [39]. The following optimized protocol is adapted for sensitive embryo tissues.
Materials:
- DNase I Solution (1 mg/mL) [40]
- Culture medium or buffer free of EDTA (e.g., HBSS or PBS) [40]
- Fetal Bovine Serum (FBS) [40]
- Polypropylene conical tubes (50 mL) [39]
- Cell strainer (70 µm) [40]
- PBS containing 2% FBS [40]
Procedure:
- Post-Thaw/Post-Dissociation: After thawing or initial tissue dissociation, transfer the cell suspension to a 50 mL conical tube. If clumping is observed, proceed to DNase treatment [40].
- DNase Application: Add DNase I Solution dropwise to the cell suspension while gently swirling the tube to achieve a final concentration of 100 µg/mL. [40]
- Incubation: Incubate the suspension at room temperature for 15 minutes [40].
- Washing: Add 25 mL of culture medium or buffer containing 2% FBS to wash the cells. Centrifuge at 300 x g for 10 minutes at room temperature. Carefully discard the supernatant [40].
- Final Resuspension and Filtration: Gently resuspend the cell pellet. If clumps persist, pass the entire sample through a 70 µm cell strainer into a fresh tube to remove remaining aggregates [40].
Critical Notes:
- Downstream Applications: DNase should be avoided if performing downstream DNA extraction. For RNA extraction, an RNase-free DNase I may be used [40].
- Sensitive Assays: For applications sensitive to DNase (e.g., hematopoietic colony assays), include an additional wash step with an appropriate assay buffer (without DNase) before continuing [40].
Optimized Mechanical Dissociation and Handling
Proper physical handling is crucial to prevent the formation of new clumps and to break apart weak aggregates without compromising cell integrity.
- Gentle Trituration: Use repetitive, gentle pipetting with a wide-bore pipette tip to physically disperse weak cell clusters. This method is particularly effective for fragile embryo cells without subjecting them to high-shear forces [41].
- Centrifugation Optimization: Resuspend the cell pellet by vortexing before adding wash buffer or media. Always use smaller volumes for more effective mixing, and avoid maximum vortex speeds to protect delicate cells [39].
- Strategic Filtration: When clumps are resistant to enzymatic and gentle mechanical dispersal, filtration through a 70 µm cell strainer provides a final purification step to achieve a true single-cell suspension prior to analysis or culture [40] [39].
- Non-Adhesive Labware: Use tubes and plates made of polypropylene instead of polystyrene to reduce cell adherence to plastic surfaces during processing and storage [39].
Chelating Agents for Cation-Dependent Clumping
For cell types whose adhesion is cation-dependent, the chelating agent EDTA can be highly effective.
- Application: Add 2 mM EDTA to cell preparation and staining buffers to chelate divalent cations like Ca²⁺ and Mg²⁺, thereby inhibiting cation-mediated cell adhesion [39].
- Exception: Omit EDTA from the protocol if the downstream application involves studying cation-dependent functions, such as integrin binding [39].
Advanced and Emerging Dissociation Technologies
Conventional enzymatic and mechanical methods face challenges regarding viability, yield, and the potential for introducing analytical artifacts [1]. Emerging technologies aim to overcome these hurdles.
Table 2: Advanced Tissue Dissociation Technologies
| Technology | Mechanism | Reported Efficacy | Viability | Time |
|---|---|---|---|---|
| Hypersonic Levitation & Spinning (HLS) | Non-contact, acoustic resonator probe generates micro-jets for dissociation [2] | 90% tissue utilization [2] | 92.3% [2] | 15 min [2] |
| Electric Field Dissociation | Application of electric fields to disrupt tissue [1] | >5x higher yield vs. traditional methods (Glioblastoma) [1] | ~80% (Glioblastoma) [1] | 5 min [1] |
| Microfluidic Platforms | Miniaturized channels combining enzymatic and mechanical shear [1] | ~20,000 cells/mg tissue (Mouse Kidney) [1] | ~95% (Mouse Kidney epithelial) [1] | 20-60 min [1] |
| Ultrasound Sonication | High-frequency sound waves for tissue disruption [1] | 72% (with enzyme, Bovine Liver) [1] | 91-98% (cell line) [1] | 30 min [1] |
The Scientist's Toolkit: Essential Reagents and Materials
Successful preparation of a single-cell suspension requires a curated set of reagents and tools. The following table details the essential components for preventing and resolving cell clumping.
Table 3: Research Reagent Solutions for Clump Prevention
| Reagent / Tool | Function | Application Notes |
|---|---|---|
| DNase I | Degrades extracellular DNA released by dead cells that causes clumping [40] [39] | Use at 100 µg/mL final concentration; avoid for downstream DNA extraction [40]. |
| EDTA | Chelates divalent cations (Ca²⁺, Mg²⁺) to prevent cation-dependent aggregation [39] | Use at 2 mM in buffers; omit for cation-dependent studies [39]. |
| Accutase | Enzyme blend with proteolytic, collagenolytic, and DNase activity; gentle on surface antigens [39] [42] | Preferred over trypsin for preserving cell surface markers [39]. |
| Cell Strainers | Physically removes persistent clumps via size-based filtration [40] [39] | 70 µm pore size is standard; use after enzymatic and mechanical dissociation [40]. |
| Serum (FBS) | Provides proteins that coat cells, improve viability, and reduce non-specific clumping [40] [39] | Use at 2-10% in buffers and media [40] [39]. |
| Polypropylene Tubes | Low-adherence surface minimizes cell loss to tube walls [39] | Use instead of polystyrene for all cell suspension work [39]. |
Experimental Workflow for Optimal Single-Cell Suspension
The following diagram illustrates a comprehensive, integrated workflow for obtaining a high-quality single-cell suspension from embryo tissue, incorporating both standard and advanced techniques.
Diagram 1: Single-Cell Suspension Workflow.
The prevention and resolution of cell clumping is a multifaceted challenge that requires a systematic approach combining enzymatic, chemical, and mechanical strategies. For researchers dissociating delicate embryo tissues, adherence to optimized protocols for DNase I application, gentle handling, and the strategic use of chelating agents and filtration is paramount. Furthermore, emerging technologies like Hypersonic Levitation and Spinning (HLS) offer promising, gentler alternatives to traditional methods, enabling higher cell viability and preservation of rare cell populations critical for developmental biology and drug discovery research. By integrating these proven and novel solutions, scientists can significantly enhance the quality and reliability of their single-cell suspension data.
Optimizing Incubation Times and Temperatures for Embryonic Cell Integrity
Within the broader scope of dissociating embryonic tissues into single-cell suspensions, the dissociation step itself is a critical bottleneck. The process of breaking down the extracellular matrix and cell–cell junctions is vital for downstream applications like single-cell RNA sequencing, flow cytometry, and establishing cell lines [1]. However, traditional methods face significant challenges in balancing dissociation efficiency with the preservation of cell integrity, where incubation time and temperature are key variables [1] [33]. This protocol details optimized parameters for embryonic tissue dissociation, providing a framework to maximize cell viability and yield while minimizing the introduction of technical artifacts that can distort downstream analyses.
Quantitative Data on Dissociation Parameters
The table below summarizes key metrics from various tissue dissociation studies, illustrating the relationship between methodology, time, and cellular outcomes.
Table 1: Comparative Performance of Tissue Dissociation Methods
| Technology / Protocol | Tissue Type | Key Incubation Parameters | Dissociation Efficacy (Live Cells/mg, unless noted) | Viability | Reference |
|---|---|---|---|---|---|
| Hypersonic Levitation & Spinning (HLS) | Human Renal Cancer | 15 minutes, Non-contact | 90% tissue utilization rate | 92.3% | [2] |
| Optimized Skin Protocol | Human Skin Biopsy | ~3 hours, Enzymatic/Mechanical | ~24,000 cells/4 mm punch | 92.75% | [33] |
| Enzymatic/Mechanical Workflow | Bovine Liver Tissue | 15 minutes, Enzymatic/Mechanical | 92% ± 8% (vs. 37%-42% enzymatic only) | >90% | [1] |
| Electric Field Dissociation | Bovine Liver Tissue | 5 minutes, Electrical | 95% ± 4% | 90% ± 8% | [1] |
| Ultrasound + Enzymatic | Bovine Liver Tissue | 30 minutes, Sonication plus Enzymatic | 72% ± 10% (sonication plus enzymatic) | 91%-98% | [1] |
| Enzyme-Free Acoustic Method | Mouse Heart Tissue | Cold-process | 3.6 × 10^4 live cells/mg | 36.7% | [1] |
Detailed Experimental Protocol for Embryonic Tissue Dissociation
This protocol is adapted from established methods for complex tissues and individual embryos, focusing on preserving embryonic cell integrity [33] [43] [44].
Reagents and Equipment
- Dissection Medium: A balanced salt solution (e.g., Dulbecco's Phosphate Buffered Saline - DPBS) without calcium and magnesium, kept on ice [18].
- Enzymatic Dissociation Cocktail: A mixture of collagenase (e.g., 50–200 U/mL) and dispase (e.g., 0.6–2.4 U/mL) in a calcium- and magnesium-containing buffer like HBSS to maintain enzyme activity [1] [18].
- Complete Growth Medium: Pre-warmed basal medium supplemented with serum or appropriate inhibitors to halt enzymatic activity.
- Sterile Tools: Fine scissors, scalpels, and forceps for mincing tissue.
- Cell Strainer: Sterile mesh (70-100 µm) for filtering the cell suspension.
- Centrifuge and Cell Counter.
Step-by-Step Methodology
Tissue Collection and Mincing:
- Isolate the embryonic tissue of interest in ice-cold dissection medium to minimize metabolic stress and transcriptional changes [45].
- Using a sterile scalpel or fine scissors, mince the tissue into small fragments of approximately 3–4 mm³ to increase the surface area for enzyme penetration [18].
- Wash the tissue fragments several times with a balanced salt solution to remove blood cells and debris [18].
Enzymatic Digestion (Critical Step):
- Transfer the minced tissue fragments into a tube containing the pre-warmed enzymatic dissociation cocktail. Use sufficient volume to fully submerge the tissue [18].
- Incubation: Place the tube in a 37°C incubator. The optimal incubation time must be determined empirically for each embryonic tissue type. Begin with 30-60 minutes, monitoring dissociation progress periodically [33] [18].
- Agitation: Gently agitate the tube using a rocker platform to ensure even digestion. Avoid vigorous shaking, which can shear cells [18].
Termination of Digestion and Cell Recovery:
- Once the tissue appears partially digested but before single cells are overly exposed to enzymes, add a volume of cold complete growth medium that is at least equal to the volume of the dissociation cocktail. This dilutes the enzymes and halts the reaction.
- Gently pipette the suspension up and down 5-10 times using a wide-bore pipette tip to further dissociate the remaining fragments mechanically.
- Pass the cell suspension through a sterile cell strainer into a new tube to remove any remaining tissue clumps and debris.
- Centrifuge the filtered suspension at 100–500 x g for 5–10 minutes to pellet the cells [18].
- Carefully decant the supernatant and resuspend the cell pellet in an appropriate volume of cold complete growth medium.
Quantification and Quality Control:
- Determine viable cell density and percent viability using an automated cell counter or hemocytometer with trypan blue exclusion [46] [18]. A viability of >90% is ideal for most downstream applications [18].
- Keep the cell suspension on ice until ready for use to preserve RNA integrity and prevent stress-induced gene expression.
Workflow and Parameter Relationship Diagrams
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Embryonic Tissue Dissociation
| Reagent | Function in Dissociation | Key Consideration |
|---|---|---|
| Collagenase | Digests collagen, a major component of the extracellular matrix [1] [18]. | Requires calcium and magnesium for optimal activity; concentration and type must be optimized [18]. |
| Dispase | A neutral protease that cleaves cell-cell junctions and the basement membrane with high specificity, often gentler on cell surface proteins [1] [18]. | Often used in combination with collagenase for more effective dissociation of complex tissues [18]. |
| Trypsin | A serine protease that cleaves peptide bonds, effective for detaching adherent cells and dissociating tissues [1] [18]. | Can be harsh and damage cell surface epitopes; requires inactivation with serum or inhibitors [1] [18]. |
| TrypLE Express | A recombinant enzyme alternative to trypsin, offering a gentler, more consistent, and animal-origin-free dissociation [18]. | A direct substitute for trypsin in many protocols, minimizing cell damage. |
| Cell Dissociation Buffer | A non-enzymatic, salt-based solution that chelates calcium and magnesium to disrupt cell adhesions [18]. | Ideal for preserving sensitive cell surface markers; less effective for dense tissues alone [18]. |
| Hyaluronidase | Degrades hyaluronic acid, another key constituent of the extracellular matrix [1]. | Typically used as a supplement in enzyme cocktails to enhance overall digestion efficiency [1]. |
| EDTA | A chelating agent that binds calcium and magnesium, disrupting cadherin-mediated cell-cell adhesions [1]. | Commonly added to enzymatic solutions to improve dissociation efficacy [1]. |
Optimizing incubation times and temperatures is a delicate balancing act central to the successful dissociation of embryonic tissues. As illustrated, shorter incubation times and lower temperatures generally favor cell viability and reduce stress-induced artifacts, but may compromise total cell yield [1] [33]. Emerging non-enzymatic and contactless technologies, such as hypersonic levitation and electrical dissociation, offer promising avenues to disrupt this trade-off, achieving high viability and efficiency within dramatically reduced timeframes [1] [2].
For researchers, a rigorous empirical approach is essential. The protocols and parameters outlined here provide a starting point, but must be validated and refined for each specific embryonic model and research objective. By systematically controlling these variables, scientists can generate high-quality single-cell suspensions that faithfully represent the in vivo cellular landscape, thereby ensuring the reliability and reproducibility of downstream analyses in developmental biology and drug discovery.
Strategy for Rare Cell Population Preservation During Dissociation
The dissociation of embryo tissue into a single-cell suspension is a critical first step for applications in single-cell RNA sequencing, drug screening, and developmental biology research. The primary challenge lies in effectively breaking down the complex tissue architecture and extracellular matrix (ECM) while maintaining the viability, transcriptional state, and functional integrity of all constituent cells, especially rare and fragile populations. These rare cells, such as progenitor or transient cell states in developing embryos, are often of high biological significance but are easily lost or damaged by suboptimal dissociation conditions. This application note details a strategic framework and optimized protocols designed to maximize the preservation of these valuable rare cell populations.
The Challenge of Tissue Dissociation for Rare Cells
Tissue dissociation is a necessary but disruptive process. Conventional enzymatic and mechanical methods often create a significant bottleneck, trading cell yield for viability and potentially skewing the resulting cell population. The very process of dissociation can be a major source of technical variation and artifacts in downstream single-cell analyses [1].
The challenges are particularly acute for rare cell populations:
- Enzymatic Damage: Prolonged exposure to enzymes like collagenase, trypsin, and dispase can damage cell surface proteins, reduce viability, and alter cellular transcriptomes [1] [19].
- Mechanical Stress: Traditional mincing, grinding, or pipetting inflicts mechanical shear forces that can lyse fragile cells or induce apoptosis [2].
- Time Sensitivity: Long processing times, including overnight digestions, increase the window for cellular stress, RNA degradation, and the introduction of contaminants [1] [19].
Therefore, an ideal strategy must balance dissociation efficiency with gentle handling, minimizing both enzymatic and mechanical stress to protect the most vulnerable cells.
Recent advancements have moved beyond conventional methods. The table below summarizes the performance of various dissociation technologies, highlighting their efficacy for preserving rare populations.
Table 1: Quantitative Comparison of Tissue Dissociation Methods
| Technology | Dissociation Type | Tissue Type | Cell Viability | Dissociation Time | Key Findings / Relevance to Rare Cells |
|---|---|---|---|---|---|
| Hypersonic Levitation & Spinning (HLS) | Non-contact, Acoustic | Human Renal Cancer | 92.3% | 15 min | Enhanced preservation of rare cell populations; 90% tissue utilization [2]. |
| Optimized Mixed Modal Microfluidic Platform | Microfluidic, Mechanical, Enzymatic | Mouse Kidney & Breast Tumor | 60%-95% (varies by type) | 20-60 min | Isolated ~3,500 leukocytes/mg tissue from kidney, demonstrating rare immune cell recovery [1]. |
| Electric Field Facilitated Dissociation | Electrical | Bovine Liver, Glioblastoma | ~80% (Glioblastoma) | 5 min | >5x higher cell yield from clinical glioblastoma tissue vs. traditional methods [1]. |
| Ultrasound Sonication | Ultrasound, Enzymatic | Bovine Liver | 91%-98% (cell line) | 30 min | Achieved 72% dissociation efficacy in liver tissue when combined with enzymes [1]. |
| Optimized Clinical Workflow | Enzymatic, Mechanical | Bovine Liver | >90% | 15 min | High viability, but enzymatic-only efficacy was low (37%-42%), showing need for mixed methods [1]. |
| Optimized Skin Dissociation Protocol | Enzymatic, Mechanical | Human Skin Biopsy | 92.75% | ~3 hours | Enabled identification of rare mast cells via scRNA-seq [19]. |
Workflow Diagram: From Tissue to Single Cells
The following diagram illustrates the core decision pathway for selecting a dissociation strategy aimed at rare cell preservation.
Detailed Experimental Protocols
Protocol 1: Optimized Enzymatic-Mechanical Dissociation for Embryonic Tissues
This protocol is adapted from established, high-viability methods for sensitive tissues like skin [19] and is suitable for most laboratory settings.
Research Reagent Solutions: Table 2: Essential Reagents for Enzymatic-Mechanical Dissociation
| Reagent / Material | Function | Example / Note |
|---|---|---|
| Dispase II | Proteolytic enzyme that cleaves fibronectin and collagen IV, effective for epithelial separation. | Preferred for preserving cell surface markers [19]. |
| Collagenase IV | Degrades native collagen, a major component of the extracellular matrix. | Worthington is a common source; concentration must be titrated [19]. |
| DNase I | Degrades extracellular DNA released by damaged cells, preventing cell clumping. | Critical for maintaining a single-cell suspension [19]. |
| RPMI 1640 Medium with HEPES | Tissue transport and digestion buffer. HEPES maintains pH during processing. | Can be supplemented with 10% FCS to protect cells [19]. |
| UltraPure BSA | Acts as a protective agent, reducing mechanical and enzymatic stress on cells. | Use in buffer formulations to enhance viability [19]. |
| Wide Bore Filtered Pipette Tips | For handling tissue fragments and cell suspensions without shearing cells. | Prevents mechanical damage to large or fragile cells [19]. |
Step-by-Step Methodology:
- Tissue Collection and Mincing:
- Immediately place the embryo tissue in cold, complete RPMI medium (with 10% FCS) after dissection.
- Using a scalpel in a small tissue culture dish, mince the tissue into fine fragments (<1 mm³) under sterile conditions. Use wide-bore pipette tips for all subsequent liquid transfers to minimize shear forces.
Enzymatic Digestion:
- Prepare a digestion enzyme cocktail in RPMI 1640. A recommended starting point for embryonic tissue is: 1-2 mg/mL Dispase II and 1 mg/mL Collagenase IV, with 10-20 µg/mL DNase I [19].
- Transfer the minced tissue to the enzyme solution and incubate at 37°C for 30-60 minutes with gentle agitation (e.g., on a rocking platform or using a low-speed orbital shaker). Do not exceed 60 minutes without assessing dissociation, as prolonged digestion harms viability.
- Monitor dissociation every 15-20 minutes by pipetting a small aliquot up and down with a wide-bore tip.
Reaction Termination and Cell Recovery:
- Halt the enzymatic reaction by adding a double volume of cold RPMI 1640 medium supplemented with 10% FCS.
- Pass the cell suspension through a 70 µm cell strainer to remove undigested fragments and debris.
- Centrifuge the filtrate at 300-400 x g for 5 minutes at 4°C. Carefully aspirate the supernatant.
Washing and Final Resuspension:
- Resuspend the cell pellet in a suitable buffer (e.g., PBS with 0.04% BSA) and pass it through a 40 µm cell strainer to ensure a single-cell suspension.
- Perform a cell count and viability assessment using an automated cell counter with Acridine Orange/Propidium Iodide (AO/PI) staining or Trypan Blue exclusion.
Protocol 2: Hypersonic Levitation and Spinning (HLS) Dissociation
HLS is a novel, non-contact method that uses high-frequency acoustic waves to generate precise hydrodynamic forces for tissue dissociation, showing exceptional promise for rare cell preservation [2].
Step-by-Step Methodology:
- Apparatus Setup:
- Utilize an automated HLS tissue dissociation apparatus that integrates a triple-acoustic resonator probe within a conical confinement structure, an inlet for enzyme solution, and chambers for digestion and single-cell collection [2].
Sample Loading and Levitation:
- Place a small piece of embryo tissue (up to a few mm³) into the digestion chamber.
- Introduce an appropriate, mild enzyme solution (e.g., low-concentration collagenase) into the chamber.
- Activate the hypersonic probe. The generated GHz-frequency acoustic field will create "liquid jets" that cause the tissue sample to levitate and execute a rapid self-rotation within the confined flow field.
Non-Contact Dissociation:
- The "press-and-rotate" operation, driven by hypersonic streaming, applies instant and precise hydrodynamic shear forces to the tissue surface. This enhances the dissociation of cell-cell and cell-matrix connections while protecting cell integrity.
- The method simultaneously enhances chemical processes, allowing the enzyme solution to permeate deeper into the tissue layers more effectively.
- The typical dissociation process is completed within 15 minutes [2].
Cell Collection:
- The automated apparatus facilitates fluid replacement and filtration. The detached cells, which revolve around the original tissue, are efficiently separated and collected in the output chamber.
- The resulting cell suspension is ready for counting and downstream applications.
Downstream Application: Single-Cell RNA-Sequencing
The quality of dissociation directly impacts scRNA-seq outcomes. Cells must maintain high RNA integrity and be free of stress-induced transcriptional artifacts. Following the optimized protocols above:
- Target approximately 6,000 cells for encapsulation using a platform like the 10x Genomics Chromium Controller [19].
- For library preparation, use a depth of at least 50,000 reads per cell to ensure sufficient coverage for detecting rare cell types [19].
- During bioinformatic analysis, apply rigorous quality control (QC) filters. Standard thresholds include: a minimum of 500 genes per cell, a library size of at least 1,500 counts, and exclusion of cells where mitochondrial counts exceed 25% of the library size, which indicates apoptosis or cellular stress [19].
The strategic preservation of rare cell populations during tissue dissociation requires a deliberate move away from harsh, one-size-fits-all protocols. The quantitative data and methodologies presented here demonstrate that the choice of dissociation strategy has a direct and measurable impact on cell viability, population representation, and the quality of subsequent data.
For researchers working with embryonic tissues, the following conclusions are critical:
- For maximal rare cell preservation, innovative non-contact methods like Hypersonic Levitation and Spinning (HLS) offer a superior alternative by minimizing all physical contact and completing dissociation rapidly [2].
- In standard laboratory settings, an optimized enzymatic-mechanical protocol that uses gentle enzymes like Dispase II, includes protective agents like BSA and DNase I, and strictly limits processing time can yield excellent results, as evidenced by the successful identification of rare mast cells in skin [19].
- Microfluidic and electrical dissociation platforms provide valuable alternatives, particularly for specific applications requiring integration with downstream processing or extremely fast processing times [1].
Ultimately, adopting these refined dissociation strategies is paramount for generating biologically accurate single-cell data from embryo tissue. It ensures that the full spectrum of cellular heterogeneity, including those critical but elusive rare populations, is captured for groundbreaking discoveries in developmental biology and drug development.
Tissue dissociation into single-cell suspensions is a critical first step in numerous downstream applications, from single-cell sequencing and flow cytometry to cell therapy manufacturing and the establishment of stem cell-derived embryo models. The ideal dissociation strategy must balance three core parameters: cell viability, process time, and final cell yield. However, the heterogeneous and delicate nature of embryonic tissues presents a significant challenge, as conventional methods often force trade-offs between these objectives. This Application Note provides a structured decision matrix and detailed protocols to guide researchers in selecting and optimizing dissociation methods for embryonic tissues, enabling robust and reproducible results in developmental biology research and drug development.
Quantitative Comparison of Dissociation Technologies
The performance of a dissociation method is primarily quantified by its cell yield, viability, and processing time. The table below summarizes these metrics for various technologies applied to different tissue types, providing a basis for initial method selection.
Table 1: Performance Metrics of Tissue Dissociation Methods
| Technology | Tissue Type | Dissociation Efficacy | Viability | Time |
|---|---|---|---|---|
| Electrical Dissociation [1] | Bovine Liver Tissue | 95% ± 4% | 90% ± 8% | 5 min |
| Human Clinical Glioblastoma | >5x higher than traditional methods | ~80% | ||
| Ultrasound + Enzymatic [1] | Bovine Liver Tissue | 72% ± 10% | Not Reported | 30 min |
| Ultrasound (Enzyme-Free) [1] | Mouse Heart Tissue | 3.6 x 10⁴ live cells/mg | 36.7% | Not Reported |
| Optimized Chemical-Mechanical [1] | Bovine Liver Tissue | 92% ± 8% | >90% | 15 min |
| Microfluidic Platform [1] | Mouse Kidney Tissue | ~20,000 epithelial cells/mg | ~95% (epithelial) | 20-60 min |
| Traditional Enzymatic [1] | Triple-negative Human Breast Cancer | 2.4 x 10⁶ viable cells | 83.5% ± 4.4% | >1 hour |
Detailed Experimental Protocols
Protocol: Chemical-Mechanical Dissociation for High-Yield Cell Isolation
This protocol, adapted from a reviewed workflow for bovine liver tissue, achieves high yield and viability through a combination of enzymatic and mechanical dissociation [1].
- Tissue Preparation: Minced fresh tissue into fragments smaller than 1 mm³ using a sterile scalpel or razor blade.
- Enzymatic Digestion: Incubate the tissue fragments in an appropriate enzyme cocktail (e.g., collagenase, dispase) at 37°C with gentle agitation for 15 minutes. The specific enzyme blend must be optimized for the embryonic tissue of interest.
- Mechanical Disruption: Following enzymatic digestion, gently pipette the tissue solution up and down 10-15 times with a serological pipette. Alternatively, pass the solution through a 1,000 µm filter followed by a 100 µm filter.
- Reaction Termination: Add a stop solution (e.g., culture medium containing serum or a specific enzyme inhibitor) to halt enzymatic activity.
- Cell Washing and Resuspension: Centrifuge the cell suspension at 300-500 x g for 5 minutes. Carefully aspirate the supernatant and resuspend the cell pellet in an appropriate buffer (e.g., PBS with 2% fetal bovine serum).
- Cell Filtration: Filter the cell suspension through a 40 µm cell strainer to remove any remaining cell clumps or debris. The single-cell suspension is now ready for counting and downstream applications.
Protocol: Enzyme-Free, Electrical Dissociation for Rapid Processing
This protocol utilizes electrical fields for rapid, enzyme-free dissociation, preserving cell surface epitopes and achieving high viability in minutes [1].
- Sample Loading: Place the minced embryonic tissue (approximately 0.5 cm³) into the chamber of an electrical dissociation device.
- Buffer Addition: Submerge the tissue in a low-conductivity, isotonic buffer.
- Parameter Application: Apply a pulsed electric field. Specific parameters (e.g., field strength, pulse duration, number of pulses) are instrument- and tissue-dependent and must be empirically determined.
- Cell Collection: Immediately after pulsing, gently agitate the chamber and collect the resulting single-cell suspension.
- Post-Processing: Centrifuge the cell suspension at 300 x g for 5 minutes to remove the buffer and resuspend in the desired culture medium or staining buffer.
Protocol: Isolation of Single Cells from Zebrafish Embryos
This protocol is specialized for the deyolking and dissociation of zebrafish embryos, a key model organism in developmental research [47].
- Embryo Collection and Dechorionation: Collect staged zebrafish embryos and manually remove the chorion using fine forceps.
- Anesthesia and Washing: Anesthetize embryos in ice-cold egg water for 20 minutes. Wash embryos twice with cold egg water.
- Deyolking:
- Replace the water with 1 mL of deyolking buffer (55 mM NaCl, 1.8 mM KCl, 1.25 mM NaHCO₃).
- Gently pipette the embryos up and down 8-12 times with a P1000 pipette until the yolk is dissolved and only the embryonic tissue remains.
- Centrifuge at 300 x g for 1 minute, remove the supernatant, and resuspend in egg water. Repeat this wash step three times.
- Enzymatic Dissociation:
- After the final wash, resuspend the tissue in 1 mL of cell dissociation reagent 1. Incubate at room temperature for 10 minutes, gently pipetting every 2-3 minutes to prevent clumping.
- Centrifuge at 300 x g for 3 minutes, remove the supernatant, and resuspend in 1 mL of cell dissociation reagent 2.
- Incubate at room temperature for 5-15 minutes, monitoring digestion progress every 5 minutes under a microscope until a majority of single cells and small clusters are present.
- Filtration and Collection:
- Centrifuge at 300 x g for 5 minutes. Resuspend the cell pellet in 1 mL of cold FACS buffer (Leibovitz's L-15 medium with 5% FBS).
- Filter the suspension through a 40 µm cell strainer, wash the strainer with an additional 1 mL of FACS buffer, and centrifuge again to collect the single cells for downstream analysis.
Signaling Pathways in Embryonic Cell Fate and Dissociation
Understanding the signaling environment of the source tissue is critical, as it can inform both the dissociation strategy and the subsequent culture conditions for the isolated cells.
Diagram 1: Signaling pathways regulating blastocyst lineage specification. Inhibition of Hippo, TGF-β, and ERK pathways is utilized to direct cells toward a trophectoderm fate, a principle applicable for deriving specific lineages in blastoid models [48].
The Scientist's Toolkit: Essential Reagents and Materials
Successful tissue dissociation relies on a core set of reagents and materials. The following table details key items and their functions in the process.
Table 2: Key Research Reagent Solutions for Tissue Dissociation
| Reagent / Material | Function / Application |
|---|---|
| Collagenase / Dispase | Enzyme blends that hydrolyze collagen and other proteins in the extracellular matrix, crucial for breaking down tissue structure [1]. |
| Trypsin / Papain | Proteolytic enzymes used for digesting tissue; papain is noted as particularly effective for neural tissue dissociation [1] [49]. |
| Percoll Solution | A medium for density gradient centrifugation, used to purify and isolate mononuclear cells from a heterogeneous cell suspension after dissociation [49]. |
| Ethylene Diamine Tetra-acetic Acid (EDTA) | A chelating agent that binds calcium ions, helping to disrupt cell-cell adhesions (e.g., cadherin-mediated junctions), often used in combination with enzymes [1]. |
| Fetal Bovine Serum (FBS) | Used in buffers (e.g., FACS buffer) to quench trypsin activity and as a supplement to protect cell viability during and after dissociation [47]. |
| ROCK Inhibitor (Y-27632) | A small molecule that significantly improves the survival and cloning efficiency of single cells, including stem cells, after dissociation [48]. |
| Hank's Balanced Salt Solution (HBSS) | A balanced salt solution used as a base for creating enzymatic and wash buffers to maintain physiological pH and osmotic balance [49]. |
| Lysophosphatidic Acid (LPA) | Acts as an inhibitor of the Hippo signaling pathway, promoting trophectoderm specification in blastoid models; an example of a pathway-specific reagent [48]. |
Integrated Workflow for Embryonic Tissue Dissociation
A generalized, integrated workflow for dissociating embryonic tissues into single-cell suspensions for downstream analysis is provided below.
Diagram 2: Integrated workflow for single-cell suspension preparation from embryonic tissue.
Validating Single-Cell Suspensions: From QC to Advanced scRNA-seq Analysis
In the field of developmental biology and regenerative medicine, research utilizing embryo-derived tissues has become a cornerstone for understanding cellular differentiation and function. A critical first step for many downstream analytical techniques, such as flow cytometry, is the effective dissociation of these complex three-dimensional tissues into a viable single-cell suspension. This process, however, poses significant challenges, including potential damage to cell surface markers, induction of apoptosis, and reduced overall cell viability, which can compromise data quality and reproducibility [42]. Therefore, implementing a robust quality control (QC) pipeline is not merely beneficial but essential. This application note details an integrated QC workflow, framing automated cell counting, viability staining, and flow cytometric analysis within the specific context of embryonic and complex tissue research. The protocols and data presented herein provide a framework for researchers to ensure that the cellular material used in their experiments—from drug screening to single-cell omics—is of the highest possible quality, thereby strengthening the validity of scientific findings.
The Critical Role of QC in Tissue Dissociation
The dissociation of embryo-derived tissues, such as brain organoids or other complex embryo-derived models, involves the deliberate breakdown of the extracellular matrix (ECM) and cell-cell junctions. The ECM is a sophisticated network of collagens, proteoglycans, and glycoproteins that provides structural and biochemical support to cells [42]. Successful dissociation requires a carefully optimized combination of enzymatic digestion and mechanical disruption to liberate individual cells without compromising their integrity or altering their surface phenotype.
Key challenges in this process include:
- Maintaining High Viability: Mechanical stress and enzymatic activity can readily damage the delicate cell membrane, leading to a high proportion of dead cells. These dead cells can later fragment and release DNA, increasing sample viscosity and promoting cell aggregation [42].
- Preserving Epitope Integrity: Over-digestion with proteolytic enzymes can cleave off cell surface antigens, leading to falsely negative results in subsequent immunophenotyping experiments [42]. For instance, dispase, while excellent for digesting collagen IV and fibronectin, is known to cleave certain surface markers relevant to T-cell analysis.
- Preventing Aggregation: DNA released from dead cells can entangle live cells, forming clumps that clog flow cytometer instruments and prevent accurate analysis of single cells.
Consequently, a multi-parameter QC assessment immediately post-dissociation is a non-negotiable step to assess the success of the tissue processing and to determine the sample's suitability for costly and time-consuming downstream applications.
Essential Reagent Solutions for Tissue Dissociation and QC
The following table catalogs key reagents and their specific functions in the process of creating and quality-controlling a single-cell suspension from embryonic tissues.
Table 1: Research Reagent Solutions for Tissue Dissociation and Quality Control
| Reagent Category | Example Reagents | Primary Function in Workflow |
|---|---|---|
| Enzymes for ECM Digestion | Dispase, Collagenase (Purified), Hyaluronidase | Degrades specific components of the extracellular matrix (e.g., collagen, fibronectin, hyaluronan) to release tissue structure [42]. |
| Enzymes for Cell-Cell Junction Cleavage | TrypLE, Papain | Cleaves proteins that form tight junctions and other intercellular connections between adjacent cells [42]. |
| DNA Degradation Reagents | DNase-I | Degrades free DNA released from dead cells, reducing sample viscosity and preventing cell aggregation [42]. |
| Viability Stains (Membrane-Impermeant) | Propidium Iodide (PI), 7-AAD | Labels dead cells with compromised membranes; used for live/dead discrimination in surface-staining protocols [50]. |
| Fixable Viability Dyes (FVDs) | eFluor 506, eFluor 780 | Amine-reactive dyes that covalently label dead cells, allowing for fixation and permeabilization without loss of signal; essential for intracellular staining [50]. |
| Metabolic Viability Dyes | Calcein AM | A cell-permeant dye converted by live-cell esterases into a fluorescent, cell-impermeant product, thereby labeling live cells [50]. |
| Flow Cytometry Staining Buffer | PBS with Fetal Calf Serum (FCS) | Provides a protein-rich, buffered medium to maintain cell health and block non-specific antibody binding during staining procedures [51]. |
| FcR Blocking Reagent | Human IgG, Mouse anti-CD16/CD32 | Blocks Fc receptors on cells to prevent non-specific binding of antibodies, thereby reducing background signal and improving data quality [51]. |
Automated Cell Counting as a Primary QC Check
Following tissue dissociation, the first and fastest QC metric is the determination of cell concentration and viability using an automated cell counter. This step is critical for normalizing downstream assays and ensuring staining efficiency.
Protocol: Determining Cell Concentration and Viability
Note: This protocol is adapted for use with an automated cell counter like the Countess II FL [52].
- Prepare Cell Sample: Gently mix the single-cell suspension to ensure a homogeneous sample.
- Stain with Trypan Blue: Combine 10 μL of the cell sample with 10 μL of trypan blue stain (or an alternative viability stain compatible with your instrument) in a microcentrifuge tube.
- Load Slide: Pipette 10 μL of the stained mixture into a chamber of a cell counting chamber slide.
- Insert and Count: Insert the slide into the instrument's sample port. The instrument will autofocus automatically. Press the "Count" button to initiate analysis.
- Record and Analyze: The instrument will display the concentrations (cells/mL) and percentages for total, live, and dead cells. Use the gating options for size and brightness to exclude debris and aggregates from the count [52].
Application and Data Interpretation
Automated cell counting provides a rapid assessment of the dissociation outcome. Research demonstrates that automated counters significantly reduce user-to-user variability compared to manual hemacytometry, which can exhibit over 20% variability between technicians [52]. This enhanced reproducibility is vital for longitudinal studies, such as tracking cell populations across a time course of organoid differentiation [53].
Table 2: Comparative Analysis of Cell Counting Methods [54]
| Method | Key Advantage | Key Disadvantage | Suitability for Embryonic Tissue Research |
|---|---|---|---|
| Hemacytometer (Manual) | Low cost; widely available. | High user variability (>20%); time-consuming [52] [54]. | Low, due to need for precision and reproducibility. |
| Automated Cell Counter | Good precision and speed; reduces user bias [52]. | May have limited accuracy with highly heterogeneous or clumpy samples [54]. | High, ideal for rapid, reproducible post-dissociation QC. |
| Flow Cytometry | High reproducibility; multi-parameter data. | Lower accuracy and precision for absolute counts; requires calibration [54]. | Medium, best for relative viability and complex immunophenotyping, not primary concentration checks. |
The following workflow diagram outlines the sequential steps for the integrated quality control process, from tissue dissociation to final analysis.
Viability Staining for Accurate Live/Dead Discrimination
Accurately distinguishing live cells from dead ones is paramount, as dead cells bind antibodies non-specifically and can severely compromise data integrity [51]. The choice of viability dye depends on the experimental design, particularly whether intracellular staining is required.
Protocol C: Staining with Fixable Viability Dyes (FVDs) for Intracellular Staining Compatibility
Note: This protocol is optimized for staining in 12 x 75 mm tubes and is compatible with subsequent intracellular staining procedures [50].
Materials:
- Phosphate-buffered saline (PBS), azide- and protein-free
- Flow Cytometry Staining Buffer (e.g., PBS with 2-5% FCS)
- Fixable Viability Dye (FVD), e.g., eFluor 660
- Centrifuge
Steps:
- Wash Cells: After dissociation and counting, wash cells twice in azide- and protein-free PBS by centrifuging at 200-400 x g for 5 minutes at 4°C. Discard the supernatant.
- Resuspend Cells: Resuspend the cell pellet at a concentration of 1-10 x 10^6 cells/mL in the protein-free PBS.
- Add FVD and Stain: Add 1 μL of FVD per 1 mL of cell suspension. Vortex the tube immediately to ensure rapid and uniform mixing.
- Incubate: Incubate the cells for 30 minutes at 2-8°C. Protect the tube from light throughout the incubation.
- Wash: Wash the cells 1-2 times with an excess of Flow Cytometry Staining Buffer to remove any unbound dye.
- Proceed: The cells are now ready for subsequent cell surface or intracellular antibody staining protocols [50].
Comparative Viability Stain Selection
Choosing the correct viability dye is a critical experimental decision. The table below compares the three main classes of viability dyes.
Table 3: Comparison of Common Viability Staining Dyes [50]
| Viability Dye Type | Mechanism of Action | Compatibility with Fixation/Permeabilization | Best Use Case |
|---|---|---|---|
| DNA-Binding Dyes (PI, 7-AAD) | Enters dead cells, intercalates into DNA. | Not compatible. Membrane integrity is lost upon fixation. | Simple live/dead discrimination during cell surface staining only. |
| Fixable Viability Dyes (FVDs) | Amine-reactive dyes bind to proteins in dead cells. | Fully compatible. Signal remains after fixation/permeabilization. | Gold standard for any experiment involving intracellular staining or sample archiving. |
| Metabolic Dyes (Calcein AM) | Converted to fluorescent product by esterases in live cells. | Not compatible with permeabilization. | Labeling and tracking of live cell populations in short-term assays. |
Integrated Flow Cytometry Analysis and Gating
Flow cytometry serves as the ultimate QC platform, allowing for multi-parameter verification of the single-cell suspension, including the confirmation of viability, identification of specific cell types, and detection of potential dissociation artifacts.
Workflow for Flow Cytometry QC
A generalized workflow for flow cytometric analysis of a dissociated embryonic tissue sample is as follows [51]:
- Sample Preparation: Use the single-cell suspension that has been processed through automated counting and viability staining.
- Blocking: To reduce non-specific antibody binding, resuspend the cell pellet in a blocking buffer (e.g., 2-10% goat serum or human IgG) and incubate for 30-60 minutes in the dark at 4°C.
- Antibody Staining: Wash cells and resuspend in staining buffer containing a pre-titrated antibody cocktail targeting cell surface markers of interest (e.g., CD24 for neurons, AQP4 for astrocytes in neural tissues) [53]. Incubate for 30 minutes on ice, protected from light.
- Wash and Fix: Wash cells twice to remove unbound antibody. If required, fix cells using a mild fixative like 1-4% PFA for 15-20 minutes on ice.
- Data Acquisition: Acquire data on the flow cytometer, collecting a sufficient number of events for robust statistical analysis. For rare populations, this may require >10^6 events.
- Data Analysis: Use software (e.g., FlowJo, or computational pipelines like CelltypeR [53]) to analyze the data. The core steps are visualized in the gating hierarchy below.
Application in Complex Tissue Research
In research involving complex embryo-derived models like human midbrain organoids (hMOs), flow cytometry moves beyond simple QC to become a powerful tool for quantitative cellular characterization. For example, the CelltypeR pipeline combines a 13-antibody panel with computational analysis to identify and quantify the major brain cell types (e.g., neurons, astrocytes, radial glia) within organoids, and can even track changes in their proportions over a differentiation time course [53]. This approach provides a robust and reproducible method to benchmark organoid models and assess the impact of experimental variables or disease states on cellular composition.
Within the context of embryo tissue dissociation research, the ultimate proof of a successful single-cell suspension lies in the functional validation of the isolated cells. This involves not just the survival of cells post-dissociation, but their demonstrated capacity to proliferate, self-organize, and form complex three-dimensional structures known as organoids. These organoids serve as powerful in vitro proxies for native tissues, enabling high-fidelity studies of development, disease, and drug response [55] [56]. This Application Note details the quantitative metrics and standardized protocols essential for validating the viability and functionality of cells following dissociation from embryonic tissues.
Quantitative Metrics for Validation Success
The following table summarizes key quantitative benchmarks for assessing the success of tissue dissociation and subsequent culture, derived from recent literature.
Table 1: Key Performance Metrics for Post-Dissociation Functional Validation
| Validation Parameter | Target Benchmark | Experimental Support & Context |
|---|---|---|
| Cell Viability | >90% [1] [2] [57] | Critical for downstream culture success; high viability is consistently reported with optimized enzymatic [1] and novel non-enzymatic methods like Hypersonic Levitation and Spinning (HLS) [2]. |
| Single-Cell Yield | ~2.4x10^6 viable cells (from triple-negative human breast cancer tissue) [1] | Highly tissue-dependent; serves as a reference point for optimization. Yields from complex tissues like mouse kidney can reach ~400,000 cells/mg [1]. |
| Organoid Formation Efficiency | Successful derivation from single Lgr5+ stem cells [56] | The gold standard for validating stem cell functionality post-dissociation, demonstrating retention of self-renewal and differentiation capacity. |
| Rare Cell Population Preservation | Enhanced preservation demonstrated via HLS [2] | A key advantage of gentler dissociation methods, crucial for maintaining heterogeneous tissue representation in downstream models. |
| Stress Response (Warm vs. Cold Dissociation) | Significant induction of immediate-early genes (e.g., Fos, Jun) in warm dissociation [57] | Cold-active protease dissociation on ice minimizes artifactual stress responses, preserving more accurate transcriptional profiles [57]. |
Detailed Experimental Protocols for Validation
Protocol A: Functional Validation via Primary Culture Initiation
This protocol assesses the basic health and proliferative capacity of dissociated cells.
Resuspension and Plating:
- Following dissociation and viability count, resuspend the single-cell pellet in an appropriate pre-warmed complete culture medium. The medium should be supplemented with necessary growth factors, antibiotics, and, for particularly sensitive cells (e.g., stem cells), a ROCK inhibitor (Y-27632) to mitigate anoikis [58].
- Seed cells at an optimized density (e.g., 1x10^5 cells/cm²) into standard 2D tissue culture plates coated with a relevant substrate (e.g., collagen, poly-L-lysine).
Monitoring and Analysis:
- Initial Adherence (24 hours): Assess the percentage of cells that have adhered and begun to spread using phase-contrast microscopy. Poor adherence can indicate membrane damage from harsh dissociation.
- Proliferation Rate (3-7 days): Monitor confluency and perform daily cell counts or use metabolic activity assays (e.g., MTT, Alamar Blue) to establish a growth curve.
- Morphology: Document cell morphology to ensure it is consistent with the expected cell type, indicating the cells have recovered their phenotypic characteristics.
Protocol B: Functional Validation via 3D Organoid Formation
This protocol validates the highest level of functionality—the capacity for self-organization.
Embedding in ECM:
- Centrifuge the dissociated single-cell suspension and carefully resuspend the pellet in a chilled extracellular matrix (ECM) such as Matrigel or BME at a density of 1x10^4 to 1x10^6 cells/mL, depending on the organoid type [55] [58].
- Plate 10-50 µL drops of the cell-ECM mixture into pre-warmed multi-well plates.
- Incubate the plates at 37°C for 15-30 minutes to allow the ECM to polymerize, forming a 3D scaffold.
Organoid Culture and Maintenance:
- Once solidified, overlay each ECM droplet with organoid-specific culture medium. The medium must be formulated to support stem cell maintenance and directed differentiation, often involving growth factors like EGF, Noggin, and R-spondin [55] [59] [56].
- Culture the plates at 37°C with 5% CO2, refreshing the medium every 2-4 days.
Validation of Organoid Success:
- Morphological Analysis (3-14 days): Observe regularly under a microscope for the formation of 3D, spherical structures. The emergence of visible, cystic or dense spheroids within 3-7 days indicates successful initiation.
- Lineage Markers (7-21 days): Fix organoids and perform immunofluorescence staining for tissue-specific markers (e.g., Sox9 for intestinal, TuJ1 for neuronal) to confirm differentiation along the correct lineage.
- Functional Assays: Depending on the tissue of origin, perform functional assays such as albumin secretion for hepatic organoids or electrophysiological measurements for neuronal organoids [55] [60].
Workflow Visualization
The diagram below illustrates the complete experimental workflow from tissue dissociation to functional validation.
The Scientist's Toolkit: Essential Reagents and Materials
Table 2: Key Research Reagent Solutions for Post-Dissociation Culture
| Item | Function | Examples & Notes |
|---|---|---|
| Extracellular Matrix (ECM) | Provides a 3D scaffold that mimics the native stem cell niche, essential for cell polarization, survival, and self-organization [55] [56]. | Matrigel, BME, Geltrex; also includes synthetic hydrogels for defined conditions [55] [58]. |
| ROCK Inhibitor (Y-27632) | Significantly improves survival of single cells, particularly stem cells, by inhibiting apoptosis induced by dissociation (anoikis) [58]. | Crucial in the first 24-48 hours of primary culture and organoid formation after passaging. |
| Stem Cell Niche Factors | Directs stem cell fate decisions (self-renewal vs. differentiation) to support long-term organoid growth and patterning. | EGF, Wnt agonists, R-spondin, Noggin, FGF10; combinations are tissue-specific [55] [59]. |
| Cold-Active Protease | Enables effective tissue dissociation at 0-4°C, minimizing artifact-inducing cellular stress responses seen in 37°C digestions [57]. | From Bacillus licheniformis; ideal for single-cell RNA-seq workflows. |
| Defined Culture Medium | Base medium providing nutrients, vitamins, and buffers, which is then supplemented with specific niche factors for the target tissue. | Advanced DMEM/F12 is common; allows for precise control over the chemical environment. |
Rigorous functional validation, through both primary culture and organoid formation, is the critical endpoint that confirms a tissue dissociation protocol has preserved not only cell life but also complex biological functions. By adhering to the quantitative benchmarks and detailed protocols outlined herein, researchers can confidently generate high-quality, biologically relevant systems from dissociated embryonic tissues. These systems are indispensable for advancing our understanding of developmental biology, disease modeling, and therapeutic discovery.
This application note provides a comprehensive benchmarking analysis for single-cell suspension preparation, focusing on the specific challenges of embryonic tissues. We evaluate three leading single-cell RNA sequencing (scRNA-seq) platforms—10x Genomics (Chromium), Parse Biosciences (Evercode), and BD (Rhapsody)—to guide researchers in selecting appropriate methodologies for developmental biology research. The dissociation of embryo tissues presents unique challenges including limited starting material, delicate cellular structures, and the need for high-resolution cellular mapping. By comparing platform performance metrics, optimization requirements, and compatibility with embryonic samples, this document provides a structured framework for establishing robust single-cell dissociation protocols in embryo research.
Single-cell RNA sequencing has revolutionized developmental biology by enabling the characterization of cellular heterogeneity and lineage specification during embryogenesis. However, the initial tissue dissociation step remains a critical bottleneck that profoundly impacts data quality and biological interpretation. For embryonic tissues, researchers must balance dissociation efficiency with preservation of cell viability and transcriptomic integrity. The choice of dissociation protocol and downstream scRNA-seq platform significantly influences cell yield, representation of fragile cell types, and detection of low-abundance transcripts—all crucial factors for constructing accurate embryonic developmental atlases.
Each major scRNA-seq platform employs distinct cellular partitioning mechanisms that interact differently with dissociated embryonic cell suspensions. Droplet-based systems (10x Genomics Chromium) face challenges with large embryonic cells and are sensitive to cell aggregation. Microwell-based systems (BD Rhapsody) offer advantages for low-RNA-content cells but may underrepresent certain populations. Combinatorial barcoding approaches (Parse Biosciences Evercode) bypass physical partitioning constraints but require optimization of fixation conditions. Understanding these platform-specific characteristics is essential for designing successful embryonic single-cell studies.
Comparative Platform Analysis
Table 1: Technical Specifications of Major scRNA-seq Platforms
| Parameter | 10x Genomics Chromium | Parse Biosciences Evercode | BD Rhapsody |
|---|---|---|---|
| Partitioning Method | Microfluidic droplets | Combinatorial barcoding in plates | Microwell array |
| Cell Size Limit | ~40 µm [61] [36] | Not size-restricted [61] [36] | ~30 µm recommended [36] |
| Cell Capture Efficiency | Moderate; biased against fragile cells [61] | High; preserves fragile cell types [61] | High for low-RNA-content cells [62] [63] |
| Typical Cell Input | 500-10,000 cells/sample | Up to 1 million cells/experiment [61] | 1,000-10,000 cells/cartridge |
| Platform Strengths | High throughput, standardized workflows | Scalability, flexible sample processing | Sensitivity for low-RNA cells |
| Embryonic Tissue Challenges | Size exclusion of large embryonic cells; shear stress on delicate cells | Requires fixation optimization for embryonic cells | Potential loss of epithelial-like embryonic cells [63] |
Embryonic Tissue Considerations Across Platforms
Embryonic tissues present unique challenges that interact significantly with platform technologies:
- Cell Size Heterogeneity: Developing embryos contain cells with diverse sizes, from small progenitor cells to large differentiating cells. Platform-specific size limitations can cause systematic exclusion of certain embryonic populations [61] [36].
- Cell Fragility: Embryonic cells are particularly sensitive to mechanical and enzymatic stress, potentially leading to RNA degradation and altered gene expression profiles during dissociation.
- Starting Material Limitations: Many embryonic studies, particularly those using valuable genetic models or rare clinical samples, face material limitations that constrain experimental design.
Table 2: Platform Performance with Challenging Cell Types Relevant to Embryonic Research
| Cell Type | 10x Genomics Chromium | Parse Biosciences Evercode | BD Rhapsody |
|---|---|---|---|
| Large Embryonic Cells | Limited by droplet size [36] | No restriction [36] | Limited by microwell size [36] |
| Fragile/Stress-Sensitive Cells | Moderate recovery; susceptible to shear stress [61] | High recovery; gentle processing [61] | High recovery; gentle capture [62] |
| Low-RNA-Content Cells | Underrepresented due to capture bias [63] | Good detection with optimized protocols | Excellent recovery and detection [62] [63] |
| Rare Cell Populations | Requires overloading; increases multiplet rate | High resolution with large-scale profiling [61] | Targeted screening capabilities |
Experimental Protocols for Embryonic Tissue Dissociation
Zebrafish Embryo Dissociation Protocol
Zebrafish represent a fundamental model organism in developmental biology research. The following optimized protocol generates high-quality single-cell suspensions from individual zebrafish embryos, enabling studies of inter-individual genetic differences and embryo-specific lineage tracing [64].
Critical Reagents and Equipment:
- Pronase Solution: 1 mg/ml in egg water for dechorionation [64]
- Stage-Specific Dissociation Media: FACSmax for early stages (10-24 hpf); 0.25% trypsin with 100 mg/ml collagenase for later stages (2-10 dpf) [64]
- Stop Solution: DMEM + 1% BSA to neutralize enzymatic activity [64]
- Resuspension Buffer: DPBS + 0.5% BSA + 18% Optiprep for maintaining cell viability [64]
- Specialized Equipment: Lo-bind Eppendorf tubes, 20 µm fitted mesh, temperature-controlled shaker [64]
Key Optimization Parameters:
- Developmental Stage Adaptation: Younger embryos (10-24 hpf) require gentler dissociation with FACSmax, while older embryos (2-10 dpf) need more aggressive enzymatic treatment with trypsin/collagenase [64].
- Temperature Control: All centrifugation steps should be performed at 4°C to preserve RNA integrity [64] [36].
- Mechanical Dissociation: Use trimmed pipette tips for gentle trituration to avoid cell lysis while achieving single-cell separation [64].
General Embryonic Tissue Best Practices
For embryonic tissues beyond zebrafish models, several universal principles apply:
Enzymatic Dissociation Optimization:
- Collagenase Applications: Type I collagenase for epithelial-rich embryonic tissues; Type II for cartilage and mesenchymal tissues [36].
- Gentle Proteases: TrypLE serves as a less cytotoxic alternative to trypsin for delicate embryonic cells [36].
- Enzyme Combinations: Hybrid approaches using collagenase with dispase or hyaluronidase improve dissociation of complex embryonic matrices [36].
Viability Preservation Strategies:
- Cold-Active Enzymes: Utilize enzymes with high activity at reduced temperatures (4-10°C) to minimize stress responses [36].
- Rapid Processing: Limit total dissociation time to under 60 minutes to preserve transcriptomic fidelity [64] [36].
- Metabolic Inhibitors: Consider RNA-stabilizing additives for particularly sensitive embryonic cell types.
Platform Selection Framework for Embryonic Research
Decision Matrix for Embryonic Applications
Platform-Specific Dissociation Optimization
For 10x Genomics Chromium Applications:
- Implement stringent filtering through flow cytometry or magnetic activation to remove doublets and small aggregates
- Optimize cell concentration (500-1,000 cells/μL) to minimize multiplets while maintaining capture efficiency
- Include RNA-stabilizing additives during dissociation to counter stress-induced transcriptional responses
For Parse Biosciences Evercode Applications:
- Optimize fixation conditions (methanol vs. formaldehyde) for different embryonic cell types
- Leverage the platform's scalability to process multiple embryos individually for developmental time series
- Utilize the high cell recovery rate to preserve rare progenitor populations
For BD Rhapsody Applications:
- Capitalize on the platform's sensitivity for low-RNA-content cells to capture early embryonic progenitors
- Implement targeted mRNA panels for specific embryonic lineages combined with whole transcriptome analysis
- Use the platform's compatibility with low-input samples for precious embryonic materials
Research Reagent Solutions
Table 3: Essential Reagents for Embryonic Tissue Dissociation
| Reagent Category | Specific Examples | Application in Embryonic Dissociation | Considerations |
|---|---|---|---|
| Proteolytic Enzymes | Pronase, Collagenase I/II, TrypLE | Tissue-specific matrix degradation; Pronase for dechorionation [64]; Collagenase for ECM-rich tissues [36] | Stage-specific concentration optimization; temperature sensitivity |
| Mechanical Dissociation Aids | Trimmed pipette tips, 20µm meshes [64], Dounce homogenizers | Gentle trituration; aggregate removal; cell size selection | Calibration required for embryonic tissue sensitivity |
| Viability Maintenance | BSA (0.5-2%) [64], Optiprep [64], FACSmax [64] | Membrane stabilization; osmotic balance; reduce mechanical stress | Concentration optimization for different embryonic stages |
| Cell Storage/Preservation | Methanol, formaldehyde, commercial preservation media | Fixation for combinatorial barcoding; pause points in workflows | Impact on RNA quality and antigen preservation |
| Quality Assessment | Trypan blue [64], propidium iodide, fluorescent viability dyes | Viability quantification; RNA integrity assessment | Establishment of embryonic-specific viability thresholds |
Successful single-cell analysis of embryonic tissues requires careful matching of dissociation protocols with platform-specific strengths and constraints. Researchers must consider the unique characteristics of their embryonic system—including developmental stage, cell size distribution, and target cell population fragility—when selecting both dissociation methodology and sequencing platform. The protocols and benchmarking data presented here provide a foundation for optimizing single-cell suspension preparation from embryonic tissues, enabling more accurate and comprehensive mapping of developmental processes across model systems and therapeutic applications.
As single-cell technologies continue to evolve, future methodology development will likely focus on integrated multi-omic approaches that preserve spatial information while capturing full transcriptomic diversity. The recommendations provided herein represent current best practices while establishing a framework for incorporating emerging technologies into embryonic research pipelines.
Single-cell RNA sequencing (scRNA-seq) has revolutionized the study of early development by enabling the unbiased transcriptional profiling of individual cells within complex tissues. For embryonic research, this technology is indispensable for mapping cell lineage specification, understanding cellular heterogeneity, and validating stem cell-based embryo models [65]. The usefulness of these embryo models hinges entirely on their molecular, cellular, and structural fidelity to their in vivo counterparts, making scRNA-seq an essential authentication tool [65]. However, embryonic tissues present unique challenges for single-cell analysis due to their small cell sizes, sensitivity to dissociation-induced stress, and the rapid transcriptional dynamics that characterize early development. This application note provides a comprehensive framework for optimizing scRNA-seq workflows specifically for embryonic cells, with detailed protocols designed to preserve transcriptomic integrity throughout the experimental process.
Experimental Design and Planning
Platform Selection for Embryonic Cell Analysis
Choosing an appropriate scRNA-seq platform is critical for success with embryonic cells. The decision must balance capture efficiency, sensitivity, and practical considerations related to cell size and sample availability. The table below summarizes key commercial platforms and their suitability for embryonic research:
Table 1: Comparison of scRNA-seq Platforms for Embryonic Cell Applications
| Commercial Solution | Capture Platform | Throughput (Cells/Run) | Max Cell Size | Fixed Cell Support | Suitability for Embryonic Cells |
|---|---|---|---|---|---|
| 10X Genomics Chromium | Microfluidic oil partitioning | 500–20,000 | 30 µm | Yes | Excellent for standard embryonic cells; size-limited |
| BD Rhapsody | Microwell partitioning | 100–20,000 | 30 µm | Yes | Good flexibility for various embryonic cell types |
| Singleron SCOPE-seq | Microwell partitioning | 500–30,000 | < 100 µm | Yes | Ideal for larger or irregular embryonic cells |
| Parse Evercode | Multiwell-plate | 1,000–1M | No strict limit | Yes | Excellent for complex experiments requiring multiplexing |
| Fluent/PIPseq (Illumina) | Vortex-based oil partitioning | 1,000–1M | No strict limit | Yes | Superior for delicate cells sensitive to microfluidics |
For embryonic tissues containing particularly fragile cells or those with unusual morphologies, technologies that avoid strict size restrictions (e.g., Singleron, Parse Evercode, Fluent/PIPseq) offer significant advantages [45]. Plate-based combinatorial indexing methods are especially valuable for large-scale studies involving multiple embryo models or time courses, as they enable massive multiplexing while minimizing batch effects [66].
The Scientist's Toolkit: Essential Research Reagents
Successful scRNA-seq of embryonic tissues requires carefully selected reagents at each stage of the workflow:
Table 2: Essential Research Reagents for Embryonic Cell scRNA-seq
| Reagent Category | Specific Examples | Function in Workflow | Considerations for Embryonic Cells |
|---|---|---|---|
| Tissue Dissociation | Collagenase, Trypsin-EDTA, Accumax | Break down extracellular matrix | Test multiple enzymes; prefer gentle conditions; perform on ice to minimize stress responses [45] |
| Cell Viability Stains | Propidium iodide, DAPI, Calcein AM | Identify dead/dying cells | Critical for sorting viable embryonic cells; use at minimum effective concentration |
| Fixation Agents | Methanol, DSP (dithio-bis(succinimidyl propionate)) | Preserve cellular RNA content | Enables workflow flexibility; methanol optimized for ACME protocol [45] |
| Cell Capture Beads | Barcoded oligo-dT beads | Bind polyadenylated mRNA | Ensure compatibility with chosen platform; check binding capacity |
| Reverse Transcription Mix | Template-switching enzymes, nucleotides | cDNA synthesis from captured mRNA | Quality critical for library complexity; use fresh aliquots |
| Library Preparation | Amplification primers, Cleanup beads | Prepare sequencing libraries | Optimize cycle number to avoid amplification bias |
Step-by-Step Experimental Protocols
Embryonic Tissue Dissociation and Cell Preparation
The dissociation of embryonic tissues into high-quality single-cell suspensions represents the most technically challenging aspect of the workflow. The following optimized protocol minimizes transcriptional stress responses:
Materials Required:
- Cold dissection buffer (e.g., PBS with 0.04% BSA)
- Pre-tested dissociation enzymes (concentration optimized for specific embryonic stage)
- Ice-cold collection medium with RNase inhibitors
- Cell strainers (30-40µm)
- Fluorescence-activated cell sorter (FACS) with large nozzle (up to 130µm)
Procedure:
- Rapid Tissue Collection: Immediately place dissected embryonic tissues in ice-cold, oxygenated dissection buffer to minimize hypoxia-induced stress responses.
- Enzymatic Dissociation: Transfer tissue to pre-cooled enzyme solution. For many embryonic tissues, a combination of collagenase (1-2 mg/mL) and gentle protease (0.05-0.1%) provides optimal dissociation. Perform digestion on ice or at 4°C for 30-60 minutes with gentle agitation [45].
- Mechanical Dissociation: Gently triturate tissue using wide-bore pipette tips. Avoid excessive force that could damage cell membranes.
- Reaction Quenching: Add excess ice-cold collection medium with 2-5% FBS to neutralize enzymes.
- Filtration and Debris Removal: Pass cell suspension through 30-40µm cell strainers. Use density gradient centrifugation or magnetic bead-based debris removal if necessary.
- Viability Staining and Sorting: Stain with viability dye (e.g., propidium iodide) and sort using FACS with a 100-130µm nozzle to minimize shear stress on fragile embryonic cells [67]. Collect into collection medium with RNase inhibitors.
Troubleshooting Note: For particularly sensitive embryonic cells, consider fixation-based approaches such as the ACME (methanol maceration) method or reversible DSP fixation immediately following dissociation to preserve authentic transcriptional states [45].
Library Preparation and Quality Control
Once high-quality single-cell suspensions are obtained, proceed with library preparation using these embryo-optimized methods:
Cell Capture and Barcoding:
- Cell Density Optimization: Adjust input cell concentration to 800-1,200 cells/µL to maximize capture efficiency while minimizing multiplets.
- Platform-Specific Protocol: Follow manufacturer instructions for your chosen platform with these embryo-specific modifications:
- For droplet-based systems: Include supplemental RNase inhibitors in the lysis buffer
- For combinatorial barcoding: Process fixed cells to maintain transcriptional fidelity
cDNA Amplification and Library Construction:
- Reverse Transcription: Use template-switching oligos for full-length cDNA capture when studying isoform diversity in developing embryos.
- cDNA Amplification: Limit PCR cycles to the minimum necessary (typically 10-14 cycles) to maintain representation of low-abundance transcripts.
- Library QC: Assess library quality using Bioanalyzer or TapeStation; aim for broad smear between 300-5,000 bp.
Quality Control Parameters for Embryonic Cells:
- Cell Viability: >90% by trypan blue exclusion or calcein AM staining
- Cell Integrity: Minimal cytoplasmic leakage (maintained refractive index)
- RNA Integrity: RIN >8.5 if assessing bulk RNA (though not always possible with limited embryonic material)
Data Analysis and Interpretation
Quality Control and Preprocessing
The unique characteristics of embryonic scRNA-seq data require specialized QC approaches:
Table 3: Quality Control Thresholds for Embryonic scRNA-seq Data
| QC Metric | Typical Range (Embryonic Cells) | Exclusion Criteria | Biological Significance |
|---|---|---|---|
| Transcripts per Cell | 500-5,000 (varies by developmental stage) | <200 or >10,000 | Low counts indicate poor capture; high counts suggest multiplets |
| Genes per Cell | 250-3,000 | <200 | Measures library complexity |
| Mitochondrial Read % | 3-10% (intact cells); <1% (nuclei) | >15-20% | Indicates cell stress or apoptosis |
| Ribosomal Read % | 5-20% | >40% | May indicate stressed cells |
| Doublet Rate | Platform-dependent (0.5-8%) | Algorithmically identified | Artificial cell type signatures |
Critical QC Steps:
- Empty Droplet Removal: Use knee plots or classifier filters to distinguish biological cells from background [68].
- Dead Cell Identification: Filter based on mitochondrial percentage (≥15-20% threshold) [68].
- Doublet Detection: Employ Scrublet (Python) or DoubletFinder (R) with expected doublet rates adjusted for embryonic cell concentration [68].
- Batch Effect Correction: Apply integration tools (Seurat, FastMNN, SCVI) when analyzing multiple embryos or time points [65] [68].
Embryonic Cell Authentication Using Reference Datasets
A critical application of scRNA-seq in embryonic research is validating stem cell-based embryo models against authentic in vivo references. The recently developed integrated human embryo reference enables robust authentication:
Reference-Based Validation Workflow:
- Dataset Projection: Project query embryo model data onto the standardized reference using stabilized UMAP [65].
- Lineage Annotation: Assign predicted cell identities based on transcriptional similarity to reference cell states.
- Fidelity Assessment: Quantify molecular fidelity by calculating correlation coefficients between model cells and their in vivo counterparts.
- Misannotation Risk Mitigation: Identify and correct for potential misannotations that occur when relevant human embryo references are not utilized [65].
Available Reference Resources:
- Comprehensive human embryo reference (zygote to gastrula) [65]
- Primate-specific comparative datasets [65]
- Shiny interfaces for exploratory data analysis [65]
Visualizing Experimental Workflows and Analytical Processes
End-to-End scRNA-seq Workflow for Embryonic Cells
The following diagram illustrates the complete experimental and computational workflow for scRNA-seq analysis of embryonic cells:
Embryonic Reference Mapping Process
This diagram details the critical process of authenticating embryo models against reference datasets:
Applications in Embryonic Research and Drug Development
The integration of scRNA-seq with embryonic research has created powerful new paradigms for understanding human development and advancing therapeutic discovery:
Embryo Model Validation
Stem cell-based embryo models require rigorous validation to ensure they faithfully recapitulate in vivo development. The comprehensive human embryo reference tool enables unbiased assessment of molecular fidelity across developmental stages from zygote to gastrula [65]. This approach has revealed risks of misannotation when relevant references are not utilized for benchmarking, highlighting the critical importance of proper authentication practices [65].
Lineage Trajectory Reconstruction
Advanced computational methods can reconstruct developmental trajectories from scRNA-seq data of embryonic cells. Pseudotime analysis using tools like Slingshot enables the identification of key transcription factors driving lineage specification [65]. For example, analysis of human embryogenesis has revealed modulated expression of 367 transcription factors along the epiblast trajectory, 326 along the hypoblast trajectory, and 254 along the trophectoderm trajectory [65].
Toxicological and Drug Screening Applications
As regulatory agencies phase out animal testing requirements for drug development, human embryo-derived models screened with scRNA-seq offer a physiologically relevant alternative [66]. Organoids derived from embryonic stem cells can recapitulate organ architecture and cellular heterogeneity, providing human-specific systems for toxicity assessment and therapeutic testing [66]. The scalability of combinatorial barcoding approaches makes them particularly suitable for high-throughput screening of multiple compounds across countless organoids [66].
Successful scRNA-seq of embryonic cells requires meticulous attention to each step of the experimental workflow, from gentle tissue dissociation to appropriate computational analysis. The protocols outlined in this application note provide a foundation for obtaining high-quality transcriptomic data that faithfully represents the in vivo state of embryonic cells. As reference datasets continue to expand and computational methods become more sophisticated, scRNA-seq will play an increasingly central role in validating embryo models, reconstructing lineage relationships, and advancing human-based drug development platforms. By adhering to these optimized protocols and leveraging the latest analytical tools, researchers can maximize the biological insights gained from precious embryonic samples while ensuring the transcriptional integrity of their single-cell data.
The dissociation of embryonic tissues into viable single-cell suspensions represents a critical gateway to understanding cellular heterogeneity, lineage commitment, and developmental trajectories. Modern single-cell technologies have revolutionized our ability to deconstruct embryogenesis at unprecedented resolution, but present researchers with a complex landscape of platforms and methods, each with distinct trade-offs in throughput, cost, and data quality. This application note provides a structured comparative analysis of these technologies within the specific context of embryonic research, offering detailed protocols and strategic guidance for selecting optimal experimental approaches.
Comparative Technology Analysis
The following tables provide a quantitative overview of the current single-cell analysis and sequencing markets, followed by a detailed comparison of leading technology platforms.
Table 1: Single-Cell Market Size and Growth Projections
| Market Segment | 2024/2025 Market Size | 2032/2034/2035 Projected Market Size | Projected CAGR | Key Drivers |
|---|---|---|---|---|
| Single-Cell Analysis Market | USD 4.90-5.19 Billion (2024) [69] [70] | USD 19.90-29.15 Billion (2034/2035) [69] [71] | 14.6%-18.74% [69] [71] | Precision medicine, cancer research, technological advancements [69] [70] [72] |
| Single-Cell Sequencing Market | USD 1.63-1.88 Billion (2024/2025) [73] | USD 6.65 Billion (2034) [73] | 15.05% [73] | Demand for high-resolution cellular insights, dropping costs [73] |
Table 2: Market Share by Application and End User (2024)
| Segment | Leading Category | Market Share | Fastest-Growing Category |
|---|---|---|---|
| Application | Cancer Research [69] [70] [74] | 30.1%-33.2% [70] [74] | Immunology [69] [70] [72] |
| End User | Academic & Research Laboratories [69] [70] [74] | 51%-73.7% [74] [73] [71] | Pharmaceutical & Biotechnology Companies [73] |
Technology Platform Comparison
Table 3: Throughput, Cost, and Data Quality of Single-Cell Platforms
| Technology / Platform | Typical Throughput (Cells per Run) | Estimated Cost per Cell | Key Data Quality Metrics | Best Suited for Embryonic Research Applications |
|---|---|---|---|---|
| Droplet-Based (e.g., 10x Genomics) | 10,000 - 80,000 cells [73] | ~$0.01 (newest assays) [73] | High gene detection sensitivity [73] | Large-scale embryonic cell atlases, heterogeneous tissue analysis |
| Combinatorial Barcoding (e.g., SPLiT-seq) | Up to millions of cells [75] | Lower cost for scaling [75] | Compatible with fixed cells [75] | Very large-scale studies, time-course experiments with sample pooling |
| Microwell-Based (e.g., BD Rhapsody) | Up to 800,000 cells [72] | Moderate | Preserves cell integrity [72] | Sensitive cell types, studies requiring high cell viability |
| Single-Nucleus RNA-seq (snRNA-seq) | Varies with platform | Lower sample prep cost [76] | Lower coverage of mature mRNA [76] | Archived frozen embryo samples, difficult-to-dissociate tissues |
| Spatial Transcriptomics | Tissue area-dependent | High | Retains spatial location information [70] | Mapping progenitor zones, understanding tissue context in development |
Experimental Protocols for Embryonic Tissue Dissociation
The choice of dissociation protocol is pivotal for the success of any single-cell experiment, as it directly impacts cell viability, yield, and transcriptional fidelity.
ACME HS Dissociation Protocol for Challenging Embryonic Tissues
The ACetic acid-MEthanol High Salt (ACME HS) protocol is a chemical fixation-dissociation method ideal for preserving RNA integrity in sensitive tissues [76] [75].
Workflow Diagram: ACME HS Dissociation Protocol
Reagents and Materials:
- ACME Solution: Acetic Acid, Methanol, Glycerol in Nuclease-free Water [75]
- Wash Buffer: 3xSSC* High-Salt Buffer (stabilizes RNA, inhibits RNases) [76]
- Resuspension Buffer: PBS with 1% Bovine Serum Albumin (BSA) [75]
- Equipment: Centrifuge, Ice bucket, Pipettes, Cell Strainer
Key Advantages for Embryonic Research:
- Simultaneously fixes and dissociates cells, "freezing" transcriptional state [75]
- Permits cryopreservation of dissociated cells for batch processing [76] [75]
- High RNA Integrity Number (RIN) maintained [76]
- Minimizes stress-induced transcriptional artifacts common in enzymatic methods [76] [75]
Enzymatic Dissociation Protocol with Cold-Active Protease
This protocol uses a cold-active protease to minimize dissociation artifacts for fresh embryonic tissues [77] [76].
Workflow Diagram: Cold-Active Enzymatic Dissociation
Reagents and Materials:
- Cold-Active Protease: Active at 4°C (e.g., from psychrophilic organisms) [76]
- Wash Buffer: Ice-cold Phosphate Buffered Saline (PBS)
- Quenching Medium: Fetal Bovine Serum (FBS) or serum-containing culture medium
- Equipment: Refrigerated centrifuge, Wide-bore pipette tips, Cell strainer
Key Advantages for Embryonic Research:
- Dramatically reduces stress-induced gene expression (e.g., FOS/JUN genes) [76]
- Maintains high cell viability for live-cell applications [77]
- Better preservation of surface epitopes for protein co-detection [77]
Protocol Selection Guidance for Embryonic Tissues
Decision Framework: Selecting a Dissociation Protocol
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Key Reagents and Their Functions in Single-Cell Embryonic Research
| Reagent / Material | Function | Protocol Application | Technical Notes for Embryonic Tissues |
|---|---|---|---|
| Cold-Active Protease | Tissue digestion at low temperatures (2-6°C) [76] | Enzymatic Dissociation | Minimizes stress responses; ideal for fresh, sensitive embryonic tissues [76] |
| ACME Solution | Simultaneous chemical fixation and dissociation [75] | ACME HS | Acetic acid-methanol-glycerol mix; preserves RNA and morphology [75] |
| 3xSSC* High-Salt Buffer | RNA stabilization during rehydration [76] | ACME HS | High ionic strength inhibits RNases; critical for RNA integrity [76] |
| Actinomycin D (ActD) | Transcription inhibitor [76] | Optional add-on | Blocks new RNA synthesis during dissociation; use with toxicity caution [76] |
| N-Acetyl-L-cysteine (NAC) | Mucus removal [75] | Pre-treatment | For mucosal embryonic tissues; reduces sample loss and clogging [75] |
| DMSO-based Cryopreservation Media | Long-term cell storage [75] | Post-dissociation | Compatible with ACME-fixed cells; enables batch processing [75] |
| Bovine Serum Albumin (BSA) | Reduce cell adhesion and buffer non-specific binding [75] | Resuspension Buffer | Standard component (0.1-1%) in resuspension buffers for all protocols [75] |
The integration of multi-omics approaches at the single-cell level represents the next frontier in embryonic research. The combination of single-cell transcriptomics with epigenomic, proteomic, and spatial data provides a holistic view of cellular functions and lineage decisions [73] [72]. Furthermore, artificial intelligence and machine learning are increasingly critical for interpreting the vast, complex datasets generated by these technologies, enabling the identification of novel developmental trajectories and rare progenitor populations [69] [73] [72].
As the field progresses, the convergence of improved dissociation methods, declining costs, and enhanced computational power will continue to deepen our understanding of embryonic development at its most fundamental level—the single cell.
Conclusion
The dissociation of embryonic tissue into single-cell suspensions is a rapidly evolving field, moving beyond basic disaggregation to a sophisticated process that prioritizes cellular integrity and functional potential. The integration of gentle, novel methods like hypersonic levitation and cold-activated proteases with rigorous validation protocols ensures the reliable production of high-quality single cells. This capability is fundamental for unlocking deeper insights into developmental biology, enhancing the precision of disease modeling, and accelerating the development of cell-based therapies. Future progress hinges on standardizing these protocols, increasing the accessibility of automated platforms, and further developing non-destructive methods that perfectly balance high yield with the complete preservation of native cell states, thereby fully realizing the promise of single-cell technologies in biomedical research.
References
- 1. Recent advancements in tissue dissociation techniques for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12622302/]
- 2. Hypersonic levitation and spinning: paving the way for ... [https://www.nature.com/articles/s44172-025-00497-0]
- 3. Cell Isolation Methods 2025: Single-Cell Technologies Guide [https://semionics.com/cell-isolation-methods-2025-guide/]
- 4. Guidelines for preparation and flow cytometry analysis of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11739683/]
- 5. Comparison of methods for the preparation of single-cell ... [https://www.sciencedirect.com/science/article/abs/pii/S0165027025000251]
- 6. Brain Single Cell Isolation for Flow Cytometry [https://www.protocols.io/view/brain-single-cell-isolation-for-flow-cytometry-c3euyjew.html]
- 7. Comparison of single-cell RNA-seq methods to enable ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12539239/]
- 8. Editorial: Extracellular matrix modifications in development ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2025.1562296/full]
- 9. Mechanisms of Assembly and Remodeling ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11931590/]
- 10. Desmosomal cadherin tension loss in pemphigus vulgaris ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12307676/]
- 11. Overview of junctional complexes during mammalian early ... [https://www.frontiersin.org/journals/endocrinology/articles/10.3389/fendo.2023.1150017/full]
- 12. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOooyJJqw2g3Nf1NFDJYjkjXnwYN3ICIW-8oKfssiNICbGONMc7jF]
- 13. A Cold-Active Protease Tissue Dissociation Protocol for the ... [https://en.bio-protocol.org/en/bpdetail?id=5293&type=0]
- 14. An Optimized Tissue Dissociation Protocol for Single-Cell ... [https://explore.data.humancellatlas.org/projects/849ed38c-5917-43c4-a8f9-0782241cf10c]
- 15. Protocol to achieve high-resolution single-cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11345562/]
- 16. An optimized workflow for single-cell transcriptomics and ... [https://www.nature.com/articles/s41598-020-58939-y]
- 17. Dynamic thresholding and tissue dissociation optimization ... [https://www.nature.com/articles/s42003-023-05182-6]
- 18. Enzymatic and Non-Enzymatic Cell Dissociation Protocols [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/cell-dissociation-protocol.html]
- 19. An Optimized Tissue Dissociation Protocol for Single-Cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9096112/]
- 20. How to Dissociate Tissue [https://singleron.bio/what-is-tissue-dissociation/]
- 21. TIGR Tissue Grinder and Dissociator | Your Partner in Cell ... [https://www.ols-bio.com/tigr-tissue-grinder-dissociator-enzyme-free-gentle-tissue-dissociation]
- 22. The pros and cons of mechanical and dissociation ... enzymatic [https://www.sciopen.com/article/10.26599/CO.2024.9410009]
- 23. - PMC Organoids [https://pmc.ncbi.nlm.nih.gov/articles/PMC10270325/]
- 24. A Cold-Active Protease Tissue Dissociation Protocol for the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12067300/]
- 25. Dissociation of solid tumor tissues with cold active protease for ... [https://genomebiology.biomedcentral.com/articles/10.1186/s13059-019-1830-0]
- 26. High-resolution single-cell RNA-seq data and ... [https://www.nature.com/articles/s41597-025-05024-6]
- 27. Decoding the blueprints of embryo development with ... [https://www.sciencedirect.com/science/article/pii/S1084952125000059]
- 28. paving the way for enhanced single-cell analysis via ... [https://pubmed.ncbi.nlm.nih.gov/41006575/]
- 29. Comparative proteomic landscapes elucidate human ... [https://www.sciencedirect.com/science/article/abs/pii/S0092867424014703]
- 30. Quality controls after tissue dissociation for single cell RNA ... [https://singleron.bio/quality-controls-after-tissue-dissociation-for-single-cell-rna-sequencing/]
- 31. Automated tissue dissociation for the establishment of ... [https://www.nature.com/articles/s41598-025-03905-9]
- 32. Optimizing Tissue Dissociation for High-Yield Single Cell ... [https://www.the-scientist.com/optimizing-tissue-dissociation-for-high-yield-single-cell-recovery-72399]
- 33. An Optimized Tissue Dissociation Protocol for Single-Cell ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2022.872688/full]
- 34. Cell Dissociation Methods for Disaggregation of Tissue [https://www.akadeum.com/blog/cell-dissociation/?srsltid=AfmBOoqgyCnxylOhofifmmcH-wIlTVKPCDt1HXqbn05Ul8LRK5QFr6XM]
- 35. An Efficient Organoid Cutting Method for Long-Term ... [https://link.springer.com/article/10.1007/s13770-025-00731-y]
- 36. 7 Things to Consider When Preparing a Cell Suspension ... [https://www.parsebiosciences.com/blog/cell-prep-for-single-cell-sequenci/]
- 37. Single-cell sequencing reveals cellular heterogeneity and ... [https://www.frontiersin.org/journals/physiology/articles/10.3389/fphys.2025.1685955/full]
- 38. snCED-seq: high-fidelity cryogenic enzymatic dissociation ... [https://www.nature.com/articles/s41467-025-59464-0]
- 39. How To Prepare a Good Single Cell Suspension [https://www.appliedcytometry.com/articles/good-single-cell-suspension/]
- 40. How to Reduce Cell Clumping in Single Cell Suspensions [https://www.stemcell.com/how-to-reduce-cell-clumping-in-single-cell-suspensions-with-dnase.html]
- 41. How to Reduce Cell Clumping in Cell Cultures [https://www.akadeum.com/blog/cell-clumping/?srsltid=AfmBOopw_WKgss_AsK2PwKOOCmHZaw9WWeieOd4FEFR1-pykx-JtGtC-]
- 42. Best Practices for Preparing a Single Cell Suspension from ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6375754/]
- 43. Maximizing single cell dissociation protocol for individual ... [https://www.sciencedirect.com/science/article/pii/S2215016124004096]
- 44. Protocol for the preparation of zebrafish whole heart cell ... [https://www.sciencedirect.com/science/article/pii/S2666166724007512]
- 45. Establishing single cell RNA transcriptomics: a brief guide [https://pmc.ncbi.nlm.nih.gov/articles/PMC12403637/]
- 46. Suspension Cell Culture Protocol [https://www.thermofisher.com/us/en/home/references/gibco-cell-culture-basics/cell-culture-protocols/subculturing-suspension-cells.html]
- 47. 分离和斑马鱼胚胎单细胞的表征 [https://www.jove.com/cn/t/53877/isolation-and-characterization-of-single-cells-from-zebrafish-embryos]
- 48. Human blastoids model blastocyst development and ... [https://www.nature.com/articles/s41586-021-04267-8]
- 49. WO2024078647A2 - 一种神经组织单细胞悬液的制备方法 [https://patents.google.com/patent/WO2024078647A2/zh]
- 50. BestProtocols: Viability Staining Protocol for Flow Cytometry [https://www.thermofisher.com/us/en/home/references/protocols/cell-and-tissue-analysis/protocols/viability-staining-flow-cytometry.html]
- 51. Flow Cytometry Protocol | Abcam [https://www.abcam.com/en-us/technical-resources/protocols/flow-cytometry-for-intracellular-and-extracellular-targets]
- 52. The Importance Of Accurate Cell Counting In Flow ... [https://www.thermofisher.com/us/en/home/life-science/cell-analysis/cell-analysis-learning-center/cell-analysis-resource-library/cell-analysis-application-notes/importance-accurate-cell-counting-flow-cytometry-cell-sorting.html]
- 53. CelltypeR: A flow cytometry pipeline to characterize single ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11367488/]
- 54. Comparison of six different methods to calculate cell ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5901878/]
- 55. Stem Cell Organoid Engineering - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4728053/]
- 56. Organoids | Nature Reviews Methods Primers [https://www.nature.com/articles/s43586-022-00174-y]
- 57. Systematic assessment of tissue dissociation and storage ... [https://genomebiology.biomedcentral.com/articles/10.1186/s13059-020-02048-6]
- 58. Modeling methods of different tumor organoids and their ... [https://www.oaepublish.com/articles/cdr.2025.34]
- 59. Self-organized yolk sac-like organoids allow for scalable ... [https://www.sciencedirect.com/science/article/pii/S2667237523000796]
- 60. Stem Cells and Organoids: A Paradigm Shift in Preclinical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12299584/]
- 61. The Single Cell Solution Immunology Has Been Waiting For [https://www.parsebiosciences.com/blog/the-single-cell-solution-immunology-has-been-waiting-for/]
- 62. Profiling low-mRNA content cells in complex human ... [https://pubmed.ncbi.nlm.nih.gov/39661509/]
- 63. Comparative analysis of 10X Chromium vs. BD Rhapsody ... [https://www.sciencedirect.com/science/article/pii/S2405844024043895]
- 64. Maximizing single cell dissociation protocol for individual ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11426156/]
- 65. A comprehensive human embryo reference tool using ... [https://www.nature.com/articles/s41592-024-02493-2]
- 66. From Animal Testing to Organoids: The New Era of Human ... [https://www.parsebiosciences.com/blog/from-animal-testing-to-organoids-the-new-era-of-human-based-drug-development/]
- 67. Optimizing single cell RNA sequencing of stem ... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2025.1590889/full]
- 68. Getting Started with scRNA-seq: Post-Sequencing Data ... [https://www.parsebiosciences.com/blog/getting-started-with-scrna-seq-post-sequencing-data-analysis/]
- 69. Single Cell Analysis Market Drives at 18.74% CAGR by 2034 [https://www.towardshealthcare.com/insights/single-cell-analysis-market-sizing]
- 70. Single Cell Analysis Market: Trends, Dynamics, ... [https://www.pharmiweb.com/press-release/2025-07-14/single-cell-analysis-market-trends-dynamics-segmentation-and-future-outlook]
- 71. Single Cell Analysis Market Size, Growth & Trends [2035] [https://www.rootsanalysis.com/reports/single-cell-analysis-market.html]
- 72. Single-Cell Analysis Market Size & Growth 2025 - 2032 [https://www.persistencemarketresearch.com/market-research/single-cell-analysis-market.asp]
- 73. Single Cell Sequencing Market Grows at 15.05% CAGR ... [https://www.towardshealthcare.com/insights/single-cell-sequencing-market-sizing]
- 74. Single-cell Analysis Market to Reach USD 12.29 Billion by ... [https://finance.yahoo.com/news/single-cell-analysis-market-reach-085100637.html]
- 75. ACME dissociation : a versatile cell fixation- dissociation method for... [https://genomebiology.biomedcentral.com/articles/10.1186/s13059-021-02302-5]
- 76. Frontiers | Comparative evaluation of ACetic - MEthanol high salt... [https://www.frontiersin.org/journals/cell-and-developmental-biology/articles/10.3389/fcell.2024.1469955/full]
- 77. One protocol does not rule them all: A comparison of three... [https://communities.springernature.com/posts/one-protocol-does-not-rule-them-all-a-comparison-of-three-single-cell-dissociation-protocols-for-gut-tissue]