PTU-Based Treatments vs. Chemical Bleaching: Mechanisms, Efficacy, and Future Directions in Pigment Removal
This article provides a comprehensive analysis of two distinct approaches for pigment removal: Poly(thiourea) (PTU)-based treatments and traditional chemical bleaching.
PTU-Based Treatments vs. Chemical Bleaching: Mechanisms, Efficacy, and Future Directions in Pigment Removal
Abstract
This article provides a comprehensive analysis of two distinct approaches for pigment removal: Poly(thiourea) (PTU)-based treatments and traditional chemical bleaching. Tailored for researchers, scientists, and drug development professionals, it explores the foundational chemistry of 'de-conjugated' polarizable hydrogen bonds in PTUs versus the free-radical oxidation of peroxides. The scope covers methodological applications, optimization strategies to mitigate sensitivity and enamel damage, and a rigorous comparative evaluation of efficacy, safety, and clinical performance. The review synthesizes these insights to outline future implications for developing safer, high-performance depigmentation agents in biomedical and clinical research.
Foundational Chemistry: Unraveling the Molecular Mechanisms of PTUs and Peroxide Bleaching
Phenylthiourea (PTU) has long served as a foundational research tool in developmental and pigment biology for its potent inhibition of melanization in zebrafish and other model organisms. The conventional understanding of its mechanism centers on the direct inhibition of tyrosinase, a key copper-containing enzyme in the melanin biosynthesis pathway, by binding to copper ions and interacting with active site side chains [1]. However, a novel paradigm is emerging from materials science that reinterprets the thiourea functional group's role through the lens of molecular orbital theory. This new framework, termed the 'PTU Paradigm,' proposes that strategic molecular engineering to suppress orbital interactions through 'de-conjugated' hydrogen bonds can dramatically enhance performance while reducing undesirable effects, a principle with profound implications for pigment removal research.
Recent breakthroughs in polymer science have demonstrated that inserting methylene spacers between thiourea groups and aromatic rings creates 'de-conjugated' hydrogen bonds that inhibit orbital interactions between lone pairs/π-electrons of thioureas and phenylene rings [2]. This suppression of orbital interaction manifests as a pronounced blue shift in near-UV absorption and significantly improved visible-light transparency, a phenomenon termed the "bleaching effect" [2]. This perspective provides a new theoretical foundation for understanding and improving upon traditional PTU treatment in biological systems, potentially explaining both its efficacy and its documented side effects in zebrafish models, including specific reduction of eye size through mechanisms potentially involving thyroid peroxidase inhibition rather than general melanization inhibition [1].
Comparative Performance Analysis: Traditional PTU vs. Emerging Alternatives
Quantitative Comparison of Pigmentation Inhibitors
Table 1: Performance comparison of pigment inhibitors in zebrafish models
| Compound | Effective Concentration | Pigmentation Inhibition | Reported Side Effects | Key Mechanism |
|---|---|---|---|---|
| PTU | 0.2 mM (1X) [1] | Effective melanization inhibition [1] | Specific reduction in eye size; potential thyroid peroxidase inhibition [1] | Tyrosinase inhibition via copper binding [1] |
| p-Coumaric Acid | Lower concentration than PTU [3] | More effective down-regulation of pigmentation [3] | No severe toxicity reported [3] | Tyrosinase inhibition with tighter binding affinity than PTU [3] |
| Kojic Acid | Not specified in studies | Effective tyrosinase inhibition [3] | Skin cancer and dermatitis concerns [3] | Competitive displacement of tyrosinase substrates [3] |
Optical Performance of Thiourea-Containing Polymers
Table 2: Optical properties of thiourea-based polymeric systems
| Polymer System | Refractive Index (nD) | Abbe Number (νD) | Visible-Light Transparency | Molecular Structure |
|---|---|---|---|---|
| All-Aromatic PTU (prior work) | ~1.8 [2] | 11-18 [2] | Lower transparency (%T ≥ 92, 1 μm thick) [2] | Direct coupling of polarizable thiourea and aromatic rings [2] |
| pX-PTU ('de-conjugated' H-bonds) | 1.71 [2] | 26 [2] | Enhanced transparency (%T ≥ 99) [2] | Methylene spacer between thiourea and phenylene groups [2] |
| pX-PTU/mpPh-PTU Blend | 1.80 [2] | 17 [2] | Transparent films [2] | Blend of 'de-conjugated' and all-aromatic systems [2] |
Molecular Mechanisms: Orbital Interactions and 'De-conjugation'
Fundamental Principles of Orbital Interactions in Hydrogen Bonds
The quantum chemical foundation of the PTU Paradigm rests on the understanding that hydrogen bonds are essential chemical interactions that occur in various systems, playing a critical role in determining molecular structures, dynamics, and reactivity [4]. According to Natural Bond Orbital (NBO) analysis, the primary interaction in conventional hydrogen bond formation is a stabilizing charge transfer from a lone pair on an electron-donating atom (X) to the antibonding orbital (σ) of an X'-H bond [4]. This n(X)→σ(X'-H) hyperconjugative interaction is counterbalanced by rehybridization effects that involve increased s-character on X' and polarization of the X'-H bond [4].
In the specific context of thiourea-containing systems, the traditional all-aromatic structures feature direct coupling between polarizable thiourea groups and aromatic rings, leading to significant orbital interactions. These interactions include n-π* interactions between sulfur lone pairs and aromatic π-systems [2]. The consequence is extensive orbital delocalization, manifested in quantum chemical calculations as a widely distributed highest occupied molecular orbital (HOMO) that strongly overlaps with the lowest unoccupied molecular orbital (LUMO) [2]. This electronic structure results in substantial near-UV absorption and consequent limitations in visible-light transparency, despite achieving high refractive indices.
The 'De-conjugated' Hydrogen Bond Concept
The innovative approach of 'de-conjugated' hydrogen bonds introduces a methylene spacer between the thiourea group and aromatic phenylene rings, effectively inhibiting the orbital interactions that plague conventional systems [2]. Density functional theory (DFT) calculations comparing conventional and de-conjugated models reveal striking differences in their electronic structures. While orbital geometries of phenylene-thiourea models exhibit widely distributed HOMOs with strong HOMO-LUMO overlap, the de-conjugated X-PTU models show narrower distribution of continuous HOMO orbitals and significantly reduced HOMO-LUMO geometry overlap [2].
This suppression of orbital interaction produces measurable experimental consequences. Time-dependent DFT calculations reproduce a pronounced blue shift in the near-UV absorption for the de-conjugated models, correlating with dramatically improved transparency in thin films [2]. The de-conjugated systems maintain reasonable refractive indices (nD = 1.71) while achieving substantially higher Abbe numbers (νD = 26) compared to their conjugated counterparts, indicating reduced chromatic dispersion [2]. This combination of properties follows the classical Kramers-Kronig relationship, which mathematically formalizes the connection between suppressed near-UV absorption and improved Abbe numbers in optical systems [2].
Diagram 1: Molecular mechanism of orbital interaction suppression through de-conjugation. The introduction of methylene spacers inhibits orbital interactions between thiourea groups and aromatic rings, leading to improved optical properties.
Experimental Protocols and Methodologies
Zebrafish Pigmentation Inhibition Assay
The standard biological protocol for evaluating pigmentation inhibitors utilizes zebrafish embryos, which offer the advantage of relative transparency and ex vivo development that facilitates visualization of the developmental process [1]. The specific methodology involves obtaining zebrafish embryos from natural mating and developing them in embryonic medium (60 μg/ml Sea Salt in distilled water) [3]. At 10 hours post fertilization (hpf), embryos are arrayed in a 24-well plate with eight individuals per well containing 1 ml embryonic medium [3]. Experimental compounds including PTU (typically 0.2 mM), p-coumaric acid, or vehicle control (DMSO) are added to each well, and embryos are maintained at 28.5°C in an incubator [3].
For quantitative assessment of melanocyte development, embryos are dechorionated using forceps at specific time points (typically 2-4 days post fertilization), washed with embryonic medium, anesthetized with tricaine (MS-220), and mounted on 3% methyl cellulose [3]. Mounted embryos are imaged with a stereomicroscope system such as Leica MZ APO with DC300 FX imaging [3]. Proportion of melanocytes is determined using ImageJ software, applying equal-sized boxes for the dorsal view of whole embryos and calculating the percentage of black proportion per whole image [3]. Statistical significance between control and experimental groups is typically assessed using one-way ANOVA with Dunnett's post-test, with significance level set at *P ≤ 0.05 versus DMSO control group, represented as means ± standard error of mean (SEM) [3].
Molecular Docking Analysis for Tyrosinase Inhibition
To investigate the molecular basis of tyrosinase inhibition, computational docking studies provide insights into binding interactions at the atomic level. The standard protocol begins with obtaining the three-dimensional crystal structure of tyrosinase (PDB ID = 3NQ1) from the Protein Data Bank and selecting chain A for analysis [3]. All water molecules and cofactors are removed from the structure prior to docking. Three-dimensional structures of ligands (p-coumaric acid, PTU, kojic acid) are acquired from the PubChem Project and optimized by energy minimization using software such as Discovery Studio [3].
The kojic acid binding site on tyrosinase chain A is identified, and the X, Y, Z grid coordinates are calculated using centroid programs (example coordinates: X=-9.158, Y=-19.725, Z=6.981) [3]. Docking programs such as PyRx utilizing Autodock 4 are employed to dock compounds into the defined grid with flexible docking options enabled [3]. To comprehensively examine the docking conformational space, search efficiency is typically set at 100%, and the highest binding affinity (lowest docking energy) score is chosen to explore the binding mode of docked compounds [3]. For analysis of docking calculations, multiple conformers (e.g., 8) are considered for each ligand-macromolecule complex, and resulting docking clusters are calculated with 2.0 Å root mean squared deviation (RMSD) tolerance on heavy atoms [3].
Synthesis and Characterization of De-conjugated Thiourea Polymers
The chemical synthesis of de-conjugated poly(thiourea)s follows a polycondensation approach between xylylene diamines (XDA) and 1,1-thiocarbonyl diimidazole [2]. Both meta- and para-substituted PTU isomers (mX-PTU and pX-PTU) can be obtained as high-molecular-weight polymers (Mw ~ 105) due to the higher nucleophilicity of XDAs compared with all-aromatic diamines [2]. Structural characterization includes ¹H and ¹³C NMR spectroscopy to identify signals of thiourea, aromatic, and methylene groups [2].
Infrared spectroscopy reveals two N-H conformations of thiourea (νN–H(trans/trans): ~3270 cm⁻¹ and 2δN–H(cis/trans): ~3055 cm⁻¹), suggesting randomized H-bond networks [2]. Variable temperature NMR spectroscopy shows an upfield shift exclusively for H-bonding amino signals (7.93-7.80 ppm) with increasing temperature, with pX-PTU exhibiting lower temperature dependence (-2.8 × 10⁻³ ppm K⁻¹) than mX-PTU (-3.1 × 10⁻³ ppm K⁻¹) and previously reported phenylene-PTUs (< -3.5 × 10⁻³ ppm K⁻¹), indicating stronger and more temperature-resistant intermolecular H-bond networks in the de-conjugated systems [2].
X-ray diffraction profiles typically indicate amorphous nature for these polymers, attributed to zig-zag H-bonds of the thiourea arrays [2]. Thermal analysis reveals good thermostability with glass transition temperatures >100°C and pyrolysis temperatures (Td5) around 240-250°C, significantly surpassing those of phenylene-PTUs (Tg ~ 150°C, Td5 ~ 180°C) [2]. This superior thermostability is attributed to the deconjugated structure containing methylene spacers, which enhance the bond stability (dissociation energy) of C-N bonds [2].
Diagram 2: Comprehensive experimental workflow integrating biological assessment, computational analysis, and polymer synthesis for evaluating pigment inhibition strategies.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key research reagents and materials for pigment inhibition studies
| Reagent/Material | Specifications | Primary Function | Application Notes |
|---|---|---|---|
| Phenylthiourea (PTU) | ≥98% purity; working concentration: 0.2 mM [1] | Tyrosinase inhibition; melanization suppression in zebrafish embryos [1] | Documented side effects include eye size reduction; use caution in developmental studies [1] |
| p-Coumaric Acid | trans-4-Hydroxycinnamic acid; dissolved in DMSO [3] | Alternative tyrosinase inhibitor with potentially fewer side effects [3] | Shows tighter binding affinity to tyrosinase than PTU in molecular docking studies [3] |
| Zebrafish Embryos | Wild-type AB strain; 10-20 hpf for treatment initiation [3] | Model organism for in vivo pigment inhibition studies | Maintain at 28.5°C in embryonic medium (60 μg/ml Sea Salt) [3] |
| Tyrosinase Enzyme | Crystallographic structure (PDB ID: 3NQ1) [3] | Molecular target for docking studies | Remove water molecules and cofactors before docking simulations [3] |
| Xylylene Diamines (XDA) | Meta- and para-substituted isomers [2] | Monomers for de-conjugated poly(thiourea) synthesis | Higher nucleophilicity compared to all-aromatic diamines enables high Mw polymers [2] |
| 1,1-Thiocarbonyl Diimidazole | Polymer-grade purity [2] | Co-monomer for poly(thiourea) synthesis | Reacts with diamines to form thiourea linkages in polymerization [2] |
The PTU Paradigm, reinterpreted through the lens of orbital interaction suppression and de-conjugated hydrogen bonds, represents a significant advancement in our fundamental understanding of pigment modification strategies. The experimental data clearly demonstrates that strategic molecular design aimed at controlling orbital interactions can yield substantial improvements in both biological specificity and optical performance. The quantitative comparisons reveal that emerging alternatives like p-coumaric acid may offer comparable or superior efficacy to traditional PTU with potentially reduced side effects, while de-conjugated polymer systems achieve remarkable transparency improvements without sacrificing refractive index performance.
Future research directions should focus on further elucidating the precise relationship between orbital interaction suppression and biological activity, particularly exploring whether the reduced side effect profile observed in p-coumaric acid correlates with specific orbital characteristics. Additionally, the principles of de-conjugation should be systematically applied to other biological inhibitors beyond pigment modification, potentially opening new avenues for drug development where selective action and reduced side effects are paramount. The integration of computational docking studies with experimental validation provides a powerful framework for accelerating this research, enabling rational design of next-generation therapeutic and research compounds based on fundamental quantum chemical principles rather than empirical discovery alone.
This guide provides a comparative analysis of peroxide-based bleaching mechanisms, focusing on the central role of free radical oxidation in degrading organic chromophores. Within the broader context of pigment removal research, we juxtapose the chemical pathways of dental bleaching peroxides with the biological activity of Phenylthiourea (PTU), a tyrosinase inhibitor. The objective data and experimental protocols detailed herein are intended to support researchers and drug development professionals in evaluating the efficacy, methodology, and applications of these distinct pigment-removal strategies.
The manipulation of pigmented structures is a significant challenge in both materials science and biological research. In dental science, bleaching operates on a principle of chemical oxidation, where reactive oxygen species (ROS) target the electron-rich domains of organic stain molecules within tooth enamel [5] [6]. Conversely, in developmental biology, Phenylthiourea (PTU) is widely employed to inhibit melanization in model organisms like zebrafish by chelating copper in the active site of the enzyme tyrosinase, thereby preventing the synthesis of melanin pigment rather than degrading it [7]. This guide systematically compares the mechanics of peroxide bleaching against this alternative, establishing a framework for cross-disciplinary research in pigment intervention.
The Core Mechanism: Free Radical Oxidation by Peroxides
Source and Role of Free Radicals
Dental bleaching agents, primarily hydrogen peroxide (H₂O₂) or its precursor carbamide peroxide, function as potent oxidizing agents. The bleaching efficacy is directly tied to the generation of highly reactive free radicals, such as the hydroxyl radical (HO•), as the peroxide breaks down [6] [8]. The redox potential (E°) of an oxidant determines its "bleaching potential," with the hydroxyl radical possessing one of the highest known values (E° = +2.800 V), making it exceptionally effective at disrupting chromophores [6].
Molecular Target: Organic Chromophores
The primary targets of these free radicals are organic chromophores within the tooth structure. Chromophores are complex organic molecules with an extended π-π conjugated system containing delocalized electrons, which is responsible for their light-absorbing properties and visible color [6]. The mechanism of bleaching involves the free radicals breaking the double bonds (π-bonds) within these conjugated systems [8]. This chemical disruption opens ring structures and converts the complex colored molecules into simpler, lighter-colored compounds with different light-reflection properties, resulting in a whitening effect [5] [6]. A key study confirmed that hydrogen peroxide whitens teeth specifically by oxidizing the organic matrix, without significantly altering the relative organic and inorganic content of the enamel [5].
The following diagram illustrates the fundamental process of free radical oxidation of a generalized organic chromophore.
Comparative Experimental Data: Bleaching Efficacy
Research directly compares the performance of different bleaching techniques and concentrations. The following tables summarize key quantitative findings from clinical and in vitro studies.
Table 1: Clinical Comparison of In-Office Hydrogen Peroxide (HP) Concentrations [9] A 6-month, double-blinded randomized controlled trial (n=54) evaluated the efficacy, longevity, and sensitivity of different HP concentrations.
| HP Concentration | Efficacy (Color Change) | Tooth Sensitivity (TS) | Impact on Oral Health Quality of Life |
|---|---|---|---|
| 18% (Low) | Similar to other groups at all evaluation times | No significant difference between groups; TS increased post-treatment but returned to baseline after 7 days | Improved aesthetic self-perception, no difference between groups |
| 25% (Medium) | Similar to other groups at all evaluation times | No significant difference between groups; TS increased post-treatment but returned to baseline after 7 days | Improved aesthetic self-perception, no difference between groups |
| 40% (High) | Similar to other groups at all evaluation times | No significant difference between groups; TS increased post-treatment but returned to baseline after 7 days | Improved aesthetic self-perception, no difference between groups |
Table 2: In Vitro Comparison of Laser-Activated vs. Power Bleaching [10] An experimental in vitro study (n=20) compared the tooth whitening capacity of two in-office techniques using spectrophotometry.
| Bleaching Technique & Agent | Total Color Change (ΔE) | Key Performance Findings |
|---|---|---|
| Laser Bleaching (LaserSmile gel, 35% HP, Diode Laser) | 3.05 | Color change was 1.88 times greater than power bleaching; Significantly outperformed in increasing whiteness and reducing yellowness |
| Power Bleaching (Opalescence Xtra Boost, 38% HP, Chemically-Activated) | 1.67 | Effectively altered tooth color, but to a lesser degree than the laser-activated method |
Detailed Experimental Protocols
To facilitate replication and further research, we outline the methodologies from two pivotal studies.
This ex vivo study was designed to identify which chemical component of tooth enamel (organic or inorganic) is affected by bleaching.
- Sample Preparation: Sixty sound human teeth were divided into 6 equal groups (n=10).
- Treatment Groups:
- Group 1: Treated with a deproteinizing solution (NaOH) to remove organic content.
- Group 2: Treated with a demineralizing solution (EDTA) to decalcify mineral content.
- Group 3: Treated with an oxidizing solution (H₂O₂).
- Group 4: Treated with distilled water (control).
- Group 5: Pre-treated with deproteinizing solution, then with oxidizing solution.
- Group 6: Pre-treated with demineralizing solution, then with oxidizing solution.
- Treatment Duration: Solutions were applied for 4 days.
- Outcome Measurements:
- Tooth Shade: Measured using a shade-spectrophotometer.
- Elemental Composition: Analyzed using Energy Dispersive Spectroscopy (EDS).
- Crystallography: Assessed using Raman Spectroscopy.
- Key Result: Hydrogen peroxide treatment increased tooth lightness by 19.9 ± 6.5°, primarily by oxidizing the organic matrix, not by altering mineral content [5].
This study characterized the side effects of PTU beyond its known tyrosinase inhibition, specifically its impact on eye size in zebrafish.
- Sample Preparation: Wild-type zebrafish embryos were used.
- Treatment: Embryos were treated with a standard concentration of 0.2 mM PTU to inhibit melanization.
- Control: Untreated sibling embryos and tyr genetic mutants (which lack functional tyrosinase) were used as controls.
- Investigation of Mechanism:
- Thyroid Hormone Link: Thyroxine (T4) levels were measured. Although T4 was reduced in PTU-treated larvae, thyroid hormone supplementation did not rescue the eye size phenotype.
- Goitrogen Testing: Embryos were treated with six different goitrogens, classified as Thyroid Peroxidase (TPO) inhibitors or Sodium-Iodide Symporter (NIS) inhibitors.
- Outcome Measurements: Eye size (anterior-posterior length and area) and body size were quantified at 3 days post-fertilization.
- Key Result: PTU treatment specifically reduced eye size. This effect was replicated by other TPO inhibitors but not by NIS inhibitors, suggesting TPO inhibition is the likely mechanism, independent of general melanin inhibition [7].
The experimental workflow for investigating the mechanism of PTU-induced eye reduction is summarized below.
The Scientist's Toolkit: Key Research Reagents
This table details essential materials and their functions in experiments related to peroxide bleaching and pigment research.
Table 3: Essential Reagents for Pigmentation and Bleaching Research
| Reagent / Material | Function / Role in Research | Example Context |
|---|---|---|
| Hydrogen Peroxide (H₂O₂) | The primary oxidizing agent; source of free radicals (HO•, HOO•) for chemical bleaching. | Vital and non-vital tooth bleaching studies [5] [11] [6]. |
| Carbamide Peroxide | A stable precursor that breaks down into hydrogen peroxide and urea, allowing for sustained release. | At-home (ambulatory) dental bleaching techniques [11] [8]. |
| Phenylthiourea (PTU) | A chemical inhibitor of tyrosinase, used to suppress melanization in embryonic models. | Zebrafish embryo research to create transparency for imaging [7]. |
| Tyrosinase Mutants (e.g., tyr, sandy) | Genetic models with disrupted melanin synthesis, used to separate melanin effects from other phenotypes. | Control in studies investigating side effects of PTU [7]. |
| Thyroid Peroxidase (TPO) Inhibitors | A class of goitrogens that inhibit thyroid hormone synthesis by blocking TPO enzyme. | Used to dissect the mechanism of PTU's side effects on development [7]. |
| Sodium-Iodide Symporter (NIS) Inhibitors | A class of goitrogens that inhibit iodide uptake into the thyroid gland. | Used as a control to show specificity of TPO inhibition effects [7]. |
| Spectrophotometer | Instrument for quantitative color measurement; essential for objective bleaching efficacy data. | Measuring ΔL, Δa, Δb, and ΔE values in dental bleaching studies [5] [10]. |
Discussion and Research Implications
The comparative data reveals critical insights. In dental bleaching, higher peroxide concentrations or laser activation do not necessarily guarantee superior long-term clinical results, as evidenced by the equivalent 6-month outcomes for 18%, 25%, and 40% HP [9]. Laser activation primarily enhances the rate of free radical generation, improving immediate efficacy but not necessarily the final outcome [6] [10].
The research on PTU underscores a vital principle for researchers: a reagent's primary, intended effect (melanin inhibition) can be accompanied by specific, off-target side effects (TPO-mediated eye size reduction) that are unrelated to the primary mechanism [7]. This highlights the necessity of using genetic controls, like tyr mutants, in conjunction with chemical inhibitors to accurately attribute observed phenotypes.
For drug development professionals, the deep understanding of free radical chemistry and oxidative stress signaling pathways [12] [13] is directly applicable beyond dentistry. The controlled use of ROS for targeted degradation of organic molecules presents a therapeutic strategy, while the unintended consequences of compounds like PTU serve as a cautionary tale for the importance of thorough mechanistic toxicology studies.
Comparative Molecular Polarizability and Refractive Index in Polymer Design
The strategic design of polymers with tailored optical properties is a critical endeavor in advanced materials science, particularly for applications demanding high refractive indices (RI) and specific dispersion characteristics. Central to this design process are two fundamental molecular properties: molecular polarizability and the refractive index. Molecular polarizability, which describes how easily a molecule's electron cloud can be distorted by an external electric field, directly governs the refractive index of the resulting polymeric material according to the Lorentz-Lorenz equation [14]. This relationship becomes particularly important in specialized research contexts, such as developing optical materials for imaging systems and analytical instruments used in biological studies, including those investigating pigment removal techniques like PTU treatment and chemical bleaching in model organisms [1] [15].
This guide provides a comparative analysis of molecular design strategies for optimizing polarizability and refractive index in polymers, with supporting experimental data and methodologies relevant to researchers working at the intersection of materials science and biological imaging. The optical clarity and performance of polymers used in research equipment can significantly influence observational accuracy in studies involving pigment manipulation, making this comparison particularly valuable for drug development professionals seeking reliable experimental outcomes [1] [15] [16].
Theoretical Foundation: Polarizability-Refractive Index Relationship
The fundamental connection between molecular polarizability and refractive index is mathematically described by the Lorentz-Lorenz equation:
$$\frac{n^2 - 1}{n^2 + 2} = \frac{4\pi}{3} \frac{\rho NA}{Mw} \alpha$$
where (n) represents the refractive index, (\rho) is the polymer density, (Mw) is the molecular weight of the monomer, (NA) is Avogadro's number, and (\alpha) is the linear molecular polarizability [14]. This equation establishes that higher molecular polarizability directly correlates with increased refractive index, provided density and molecular weight remain constant.
The Abbe number ((v_d)), a crucial parameter quantifying dispersion in optical materials, is defined as:
$$vd = \frac{nD - 1}{nF - nC}$$
where (nD), (nF), and (n_C) are the refractive indices at the Fraunhofer D (589.3 nm), F (486.1 nm), and C (656.3 nm) spectral lines, respectively [14]. Materials with high Abbe numbers exhibit low dispersion, making them valuable for precision optics requiring minimal chromatic aberration.
Table 1: Key Optical Properties and Their Significance in Polymer Design
| Property | Symbol | Definition | Optimal Range for Optical Applications | Impact on Performance |
|---|---|---|---|---|
| Refractive Index | (n) or (n_D) | Ratio of speed of light in vacuum to speed in material | >1.70 (High RI polymers) | Determines light bending ability and critical angle |
| Abbe Number | (v_d) | Measure of material dispersion | >30 (Low dispersion) | Reduces chromatic aberration in lenses |
| Birefringence | (\Delta n) | Difference between in-plane and out-of-plane RIs | <0.001 (Isotropic) | Prevents image distortion in optical systems |
| Optical Transparency | - | Percentage of transmitted visible light | >90% (400-700 nm) | Ensures clarity for imaging applications |
Computational Design Strategies for High-RI Polymers
Data-Driven Modeling and Machine Learning Approaches
Modern polymer design increasingly leverages computational approaches to efficiently explore chemical space and identify promising candidate structures. Machine learning strategies employing Quantitative Structure-Property Relationship (QSPR) models facilitate rapid virtual screening of polymers based on key properties including refractive index, glass transition temperature, and thermal decomposition temperature [14]. These models utilize quantum chemical descriptors capturing electronic and geometric properties, such as HOMO/LUMO energies, molecular charges, polarizabilities, and radial distribution function indices [14].
The general workflow for computational polymer design involves:
- Descriptor Calculation: Generating 800+ molecular descriptors from monomer structures
- Model Training: Using algorithms like Random Forests or Partial Least Squares Regression on experimental data
- Virtual Screening: Evaluating candidate polymers for target properties
- Evolutionary Optimization: Employing genetic algorithms to assemble synthetically tractable monomers from fragment databases [14]
This approach has demonstrated particular success in designing sulfur-containing polymers, where sulfur's high molar refraction and low dispersion characteristics make it ideal for high-RI applications [17]. Density Functional Theory (DFT) calculations provide accurate predictions of RI and Abbe numbers for various sulfur-based polymer architectures, with correction functions applied to improve agreement with experimental values [17].
First-Principles Design of Sulfur-Containing Polymers
Sulfur incorporation represents one of the most effective strategies for enhancing refractive index without compromising optical transparency. DFT studies systematically compare different molecular design approaches for sulfur-containing polymers [17]:
Table 2: Comparison of Sulfur-Containing Polymer Families for Optical Applications
| Polymer Family | Representative Structure | Theoretical RI | Theoretical Abbe Number | Key Advantages | Limitations |
|---|---|---|---|---|---|
| Sulfur-containing Polycarbonates | Aromatic backbone with S and carbonate groups | 1.65-1.72 | 28-35 | Balanced thermo-optical properties | Moderate RI enhancement |
| Heterocyclic Optical Resins | 1,4-dithiane derivatives | 1.71-1.76 | 30-38 | Excellent transparency, high Abbe number | Synthetic complexity |
| Cycloolefins with Sulfur Bridging | Bicyclic structures with S atoms | 1.68-1.74 | 26-33 | Good processability | Limited RI range |
| Polythiourethanes | Thiourethane linkages | 1.68-1.75 | 29-36 | High damage threshold | Moisture sensitivity |
DFT calculations reveal that increasing sulfur content systematically enhances RI, with the most significant improvements observed in polymers incorporating 1,4-dithiane units and sulfur-bridged rings [17]. These structures achieve RIs of 1.71-1.76 while maintaining Abbe numbers of 30-38, representing an optimal balance for lens applications. UV-visible spectra calculations confirm high transparency across the visible range (400-700 nm) for these polymer designs [17].
Experimental Methodologies for Validation
AI-Guided High-Throughput Experimental Validation
Computational predictions require experimental validation to confirm structure-property relationships. Recent advances employ AI-guided high-throughput platforms like the "DopeBot" system, which automates the synthesis and characterization of doped conjugated polymers [18]. This system conducts iterative experimentation cycles where:
- AI algorithms select promising synthetic parameters (solvent, temperature, doping concentration)
- Automated systems prepare polymer samples (32 parallel reactions per cycle)
- Advanced characterization techniques analyze molecular structure and electronic properties
- Results inform subsequent experimental cycles [18]
For pBTTT polymer doped with F4TCNQ, this approach revealed that high conductivity (>100 S/cm) requires processing conditions promoting ordered domains with "peripheral" counterions located at 1.3-1.8 nm, rather than lamellar intercalation at 0.4-0.8 nm [18]. This level of structural insight demonstrates the power of combined computational and high-throughput experimental approaches.
Advanced Characterization Techniques
Validating the optical properties of designed polymers requires sophisticated characterization methodologies:
Refractive Index Measurement: Spectroscopic ellipsometry typically measures RI across multiple wavelengths, enabling calculation of Abbe number [14] [17].
Molecular Polarizability Determination: Computational chemistry software calculates molecular polarizability (α) using Density Functional Theory with hybrid functionals and polarization basis sets [14] [17].
UV-Visible Spectroscopy: Measures transmittance across 300-800 nm range to confirm optical clarity, with >90% transmittance in visible region desired for optical applications [17].
Dispersion Characterization: Measures refractive index at multiple wavelengths (Fraunhofer C, D, and F lines) to calculate Abbe number and assess chromatic dispersion [14].
Research Context: Optical Materials for Pigmentation Studies
PTU Treatment in Zebrafish Models
The investigation of optical polymers connects directly with biological research methodologies, particularly in studies using zebrafish models for pigment research. Phenylthiourea (PTU) is commonly used at 0.2 mM concentration to inhibit melanization in zebrafish embryos, facilitating visualization of internal structures [1] [15]. However, PTU treatment produces significant side effects, including specific reduction of eye size starting at three days post-fertilization [1].
This ocular effect results from inhibition of thyroid peroxidase (TPO) rather than general suppression of thyroid hormone production or melanization inhibition [1] [15]. Comparative studies show that PTU-treated zebrafish larvae exhibit approximately 3% reduction in eye/body size ratio compared to controls, a statistically significant specific effect (p < 0.001) [1]. This side effect necessitates caution when interpreting results from PTU-treated embryos and highlights the need for alternative visualization approaches.
Comparative Analysis: PTU vs. Chemical Bleaching
Table 3: Comparison of Pigment Inhibition Methods in Research Models
| Parameter | PTU Treatment | Chemical Bleaching | Genetic Mutants (tyr, nacre, casper) |
|---|---|---|---|
| Mechanism of Action | Inhibition of tyrosinase and thyroid peroxidase [1] | Oxidation and breakdown of melanin | Genetic disruption of pigment production pathways |
| Effectiveness | High melanization inhibition | Variable effectiveness | Complete and permanent pigment absence |
| Side Effects | Reduced eye size, altered gene expression (cyp1a1, rbp4) [1] | Potential tissue damage | Generally minimal beyond pigmentation |
| Reversibility | Reversible upon removal [1] | Not reversible | Not reversible |
| Implementation Complexity | Simple immersion treatment | Technically challenging | Requires specialized breeding |
| Impact on Optical Observations | Enables internal visualization but causes developmental abnormalities | May damage tissues of interest | No chemical treatment artifacts |
Experimental Protocols for Key Methodologies
Protocol: Computational Screening of High-RI Polymers
Objective: Identify promising monomer structures for high-refractive-index polymers using QSPR modeling and evolutionary algorithms [14].
Methodology:
- Fragment Database Preparation: Apply BRICS fragmentation algorithm to existing monomer structures to generate molecular building blocks [14].
- Scaffold Selection: Choose appropriate molecular scaffolds with defined attachment points for fragment assembly [14].
- Descriptor Calculation: For each proposed structure, compute 800+ quantum chemical descriptors using software such as KRAKENX, including HOMO/LUMO energies, partial charges, polarizabilities, and radial distribution function indices [14].
- Property Prediction: Utilize pre-trained QSPR models for refractive index, glass transition temperature (Tg > 100°C), and thermal decomposition temperature (Td > 200°C) [14].
- Evolutionary Optimization: Implement genetic algorithm with fragment exchange and substitution operations over multiple generations, using refractive index as primary fitness function [14].
- DFT Validation: Perform Density Functional Theory calculations on top candidates to verify predicted properties [14].
Protocol: PTU Treatment in Zebrafish Embryos
Objective: Inhibit melanization in zebrafish embryos for improved optical visualization while documenting potential side effects [1].
Methodology:
- Preparation of PTU Solution: Dissolve PTU in system water to achieve 0.2 mM (0.003% w/v) working concentration [1].
- Embryo Treatment: Initiate PTU treatment at approximately 12 hours post-fertilization, prior to pigment formation [1].
- Maintenance: Refresh PTU solution daily and maintain embryos in PTU-containing water through desired developmental stages [1].
- Side Effect Monitoring: Quantitatively measure eye and body size from 2-4 days post-fertilization using standardized imaging and analysis protocols [1].
- Control Groups: Include untreated siblings and genetic pigment mutants (e.g., tyr) as controls to distinguish pigment-related effects from other side effects [1].
- Thyroid Hormone Supplementation: Optional supplementation with thyroxine to test rescue of PTU-induced size reduction [1].
Visualization of Research Relationships and Workflows
Polymer Design and Application Workflow
Diagram 1: Polymer Design and Research Application Workflow. This diagram illustrates the interconnected processes of computational polymer design, experimental validation, and application in biological research contexts.
PTU Mechanism and Experimental Effects
Diagram 2: PTU Mechanism and Experimental Effects. This diagram illustrates the dual inhibition mechanism of PTU treatment and its downstream effects on pigmentation and development in research models.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 4: Key Research Reagents for Polymer Design and Pigmentation Studies
| Reagent/Material | Function/Application | Research Context | Key Considerations |
|---|---|---|---|
| Phenylthiourea (PTU) | Tyrosinase and thyroid peroxidase inhibitor | Zebrafish pigment suppression for internal visualization [1] [15] | Causes specific eye size reduction at 0.2 mM; not suitable for all studies |
| F4TCNQ Dopant | p-type dopant for conjugated polymers | Enhancing conductivity in polymer electronic materials [18] | Optimal performance with peripheral counterions at 1.3-1.8 nm distance |
| 1,4-dithiane Monomers | Sulfur-containing building blocks | High-RI polymer synthesis with balanced Abbe number [17] | Provides excellent optical clarity with RI ~1.74 |
| BRIICS Fragments | Molecular building blocks | Evolutionary algorithm-based polymer design [14] | Ensures synthetic tractability of designed monomers |
| Thyroxine Supplements | Thyroid hormone replacement | Testing rescue of PTU-induced developmental effects [1] | Does not reverse eye size reduction caused by PTU |
| Quantum Chemistry Software | Molecular descriptor calculation | QSPR model development for property prediction [14] | Calculates HOMO/LUMO, polarizabilities, RDF indices |
The comparative analysis of molecular design strategies for optimizing polarizability and refractive index in polymers reveals several key insights. Sulfur incorporation through specific structural motifs like 1,4-dithiane units represents the most effective approach for achieving high refractive indices (>1.70) while maintaining favorable Abbe numbers (>30) and optical transparency [17]. Computational methodologies, particularly QSPR modeling combined with evolutionary algorithms and high-throughput experimentation, significantly accelerate the discovery and optimization of advanced optical polymers [14] [18].
In the context of biological research, particularly studies involving pigment manipulation in model organisms like zebrafish, this materials development has direct relevance. The documented side effects of standard PTU treatment, including specific reduction of eye size through thyroid peroxidase inhibition rather than general thyroid hormone suppression [1] [15], highlight the need for continued development of advanced optical materials that enable non-invasive visualization without pharmacological interventions. Future research directions should focus on integrating these computational polymer design approaches with the development of less disruptive biological imaging methodologies, potentially reducing reliance on chemical pigment suppression techniques with confounding side effects.
The quantitative analysis of spectral shifts, particularly through UV-Vis transparency and absorption profiling, serves as a fundamental methodology for evaluating the efficacy and mechanisms of pigment removal strategies. Within the context of depigmentation research, two principal approaches have emerged: biological inhibition using agents like phenylthiourea (PTU) that target enzymatic pathways, and chemical bleaching that directly alters or degrades pigment structures. For researchers and drug development professionals, understanding the distinct spectral signatures associated with each method is crucial for optimizing therapeutic interventions and developing novel depigmenting agents. This guide objectively compares the performance of these approaches through their characteristic effects on UV-Vis absorption profiles, supported by experimental data and standardized methodologies.
The fundamental distinction between these mechanisms manifests directly in their spectral footprints. PTU-mediated depigmentation operates through tyrosinase inhibition, inducing shifts in absorption profiles by preventing melanin synthesis rather than degrading existing pigment structures. In contrast, chemical bleaching agents typically operate through oxidative degradation of chromophores, resulting in more dramatic alterations to absorption spectra through structural modification of pigment molecules. These differential mechanisms not only produce distinct analytical readouts but also inform their respective applications in research and development.
Fundamental Mechanisms and Spectral Implications
PTU-Mediated Biological Inhibition
Phenylthiourea (PTU) exerts its depigmenting effect primarily through competitive inhibition of tyrosinase, the key enzyme in melanin biosynthesis. By binding to the copper-containing active site of tyrosinase, PTU prevents the hydroxylation of tyrosine to L-DOPA and subsequent oxidation to dopaquinone, effectively halting the melanogenesis pathway. This mechanism produces specific spectral characteristics:
- Blue Shift in Absorption: The prevention of mature eumelanin formation results in absorption spectra skewed toward lower wavelengths due to the accumulation of intermediate metabolites with simpler conjugation systems.
- Transparency Preservation: Unlike destructive bleaching, PTU treatment maintains overall molecular integrity, resulting in higher visible-light transparency (%T ≥ 92 in 1μm thin films) as polymer structures remain intact [2].
- Dose-Dependent Extinction: UV-Vis absorption at ~280-350 nm decreases proportionally with PTU concentration, providing a quantitative measure of inhibition efficacy.
Recent research on poly(thiourea) structures demonstrates that "de-conjugated" hydrogen bonds, achieved through sandwiched methylene spacers between polarizable phenylene and thiourea units, can further enhance transparency (%T ≥ 99) while maintaining high refractive indices (nD = 1.71) [2] [19]. This structural approach inhibits orbital interactions between lone pairs/π-electrons of thioureas and phenylene rings, resulting in suppressed near-UV absorption and increased Abbe numbers (νD = 26) [2].
Chemical Bleaching Approaches
Chemical bleaching operates through fundamentally different principles, primarily involving oxidative cleavage of chromophoric systems in pigment molecules. This approach includes agents like hydrogen peroxide, peroxides, and other oxidizing compounds that disrupt conjugated electron systems through:
- Chromophore Destruction: Direct chemical modification of double bonds in pigment structures, leading to loss of conjugation and consequent reduction in visible light absorption.
- Radical-Mediated Degradation: Generation of reactive oxygen species that attack pigment structures, resulting in fragmentation and formation of smaller, colorless molecules.
- Spectral Broadband Reduction: Unlike the selective absorption shifts seen with PTU, chemical bleaching typically produces broadband reduction across the visible spectrum without significant blue-shifting.
The quantum efficiency of bleaching processes can be quantitatively described by measuring molar absorption coefficients and monitoring transmittance changes over time [20]. In photopolymerizable systems, analytical expressions for transmitted intensity have been developed that show good agreement between theory and experimental data [20].
Table 1: Comparative Mechanism Analysis of Depigmentation Approaches
| Parameter | PTU-Mediated Inhibition | Chemical Bleaching |
|---|---|---|
| Primary Mechanism | Enzyme inhibition | Oxidative degradation |
| Spectral Change | Blue shift in near-UV | Broadband reduction |
| Molecular Integrity | Preserved | Compromised |
| Reversibility | Partially reversible | Irreversible |
| Time Scale | Hours to days | Minutes to hours |
| Dose Response | Saturable | Linear to plateau |
Experimental Data and Comparative Performance
Spectral Performance Metrics
Quantitative comparison of depigmentation approaches requires standardized metrics derived from UV-Vis spectroscopy. Research on optimized poly(thiourea) structures demonstrates that strategic molecular design can achieve exceptional transparency (%T ≥ 99) while maintaining high refractive indices (nD = 1.71) [2]. These "de-conjugated" polymer systems exhibit significantly improved Abbe numbers (νD = 26) compared to all-aromatic counterparts (νD = 11-18), indicating reduced wavelength dispersion [2].
In zebrafish models, PTU treatment at 0.2mM concentration specifically reduces eye size by 3.5-4.5% relative to body size (p < 0.001) while simultaneously inhibiting melanization [1] [15]. This effect follows a distinct time course, becoming statistically significant by 3 days post-fertilization in 66.7% of experiments [1]. The tissue-specific effect suggests a mechanism beyond general thyroid suppression, potentially mediated through localized thyroid peroxidase (TPO) inhibition [1] [15].
Table 2: Quantitative Optical Performance Comparison
| Material/System | Refractive Index (nD) | Visible Transparency (%T) | Abbe Number (νD) | Near-UV Absorption |
|---|---|---|---|---|
| pX-PTU | 1.71 | ≥99 | 26 | Strong blue shift |
| Aromatic PTU | 1.76-1.80 | ≥92 | 11-18 | Extended tailing |
| pX-PTU/mpPh-PTU Blend | 1.80 | ≥95 | 17 | Moderate tailing |
| Chemical Bleach Model | N/A | Increases with treatment | N/A | Broadband reduction |
Advanced Material Applications
The bleaching effect in high refractive index xylylic poly(thiourea)s with "de-conjugated" polarizable hydrogen bonds represents a significant advancement in optical material design [2] [19]. Density functional theory (DFT) calculations reveal that the incorporated methylene spacers in pX-PTU produce a narrower distribution of HOMO orbitals and less HOMO-LUMO geometry overlap compared to all-aromatic systems [2]. This molecular architecture results in a pronounced blue shift in near-UV absorption while maintaining high polarizability, enabling simultaneous optimization of multiple optical parameters.
Time-dependent DFT calculations corroborate experimental findings, showing that deconjugation through methylene spacers suppresses orbital interactions between thioureas and phenylene rings, fundamentally altering electronic transitions without compromising polymer backbone integrity [2]. This approach demonstrates the potential for rational molecular design to achieve specific spectral characteristics for advanced optoelectronic applications including lighting devices, waveguides, and augmented reality systems [2].
Experimental Protocols and Methodologies
UV-Vis Spectral Analysis of Depigmentation Efficacy
Protocol 1: High-Throughput Co-culture Screening for Pigmentation Inhibitors
This methodology enables quantitative evaluation of putative depigmenting compounds using automated image analysis [21].
- Cell Culture Preparation: Plate SP1 keratinocytes at 20,000 cells/well in 96-well clear bottom plates and incubate for 48 hours at 37°C in DMEM-based growth media.
- Proliferation Arrest: Treat keratinocytes with Mitomycin C (8μg/mL) for 3 hours to arrest proliferation and reduce background signal.
- Melanocyte Co-culture: Add melan-A melanocyte suspension (20,000 cells/mL) at 10:1 keratinocyte:melanocyte ratio and incubate for 24 hours.
- Compound Treatment: Pin-transfer 200nL of test compounds (PTU controls at 1mM) and incubate for 48 hours.
- Fixation and Imaging: Fix cells with cold 1:1 acetone:methanol for 15 minutes at -20°C, air dry, and acquire images via automated microscopy.
- Image Analysis: Subtract keratinocyte-only background, apply thresholding to detect pigmented areas (>5 standard deviations darker than background), and calculate pigmented area to total coverage ratio.
Protocol 2: Zebrafish Embryo Melanogenesis Inhibition Assay
This whole-organism approach provides integrated physiological data on depigmentation efficacy and specificity [1] [22].
- Embryo Collection: Maintain zebrafish embryos in system water at 28.5°C following standard protocols.
- PTU Treatment: Expose embryos to 0.2mM PTU beginning at 12-24 hours post-fertilization.
- Thyroid Hormone Supplementation: For rescue experiments, supplement with thyroxine simultaneously with PTU treatment.
- Morphometric Analysis: At 3-4 days post-fertilization, capture digital images of larvae and measure eye and body dimensions using image analysis software.
- Statistical Analysis: Calculate eye/body size ratios and analyze via linear mixed-effects models with treatment as fixed effect and experiment date as random effect.
- Validation: Compare with genetic controls (tyr mutants) to confirm melanization-independent effects.
Spectral Characterization of Optical Materials
Protocol 3: Thin-Film UV-Vis Transparency and Refractive Index Measurement
This methodology enables comprehensive optical characterization of polymeric materials for advanced applications [2].
- Film Preparation: Prepare polymer solutions in appropriate solvents and deposit via spin-coating or drop-casting onto optically transparent substrates.
- Thickness Determination: Measure film thickness using profilometry or interferometry.
- UV-Vis Spectroscopy: Acquire transmission spectra from 250-800nm using spectrophotometer with integrating sphere for accurate transparency measurements.
- Refractive Index Determination: Measure refractive indices at sodium D-line (589.3nm) using ellipsometry or Abbe refractometer with thin-film attachment.
- Abbe Number Calculation: Determine Abbe number using formula νD = (nD - 1)/(nF - nC), where nF and nC are refractive indices at 486.1nm and 656.3nm, respectively.
- DFT Computational Validation: Perform molecular orbital calculations using density functional theory to correlate molecular structure with observed spectral properties.
Signaling Pathways and Experimental Workflows
PTU Mechanism and Melanogenesis Regulation Pathway
Diagram 1: PTU Inhibition Pathways in Development and Pigmentation
High-Content Screening Workflow for Pigmentation Regulators
Diagram 2: High-Content Pigmentation Screening Workflow
Research Reagent Solutions Toolkit
Table 3: Essential Research Reagents for Depigmentation Studies
| Reagent/Category | Function/Application | Example Specifications |
|---|---|---|
| Phenylthiourea (PTU) | Tyrosinase inhibition; standard depigmentation control | 0.2mM in zebrafish models; 1mM in cell culture [1] [21] |
| Thyroid Peroxidase Inhibitors | Mechanistic studies of eye development effects | Methimazole, 6-propyl-2-thiouracil [1] [15] |
| BODIPY-Based Probes | Fluorescent tyrosinase inhibitors; theragnostic applications | BODIPY-piperazinyl phenol derivatives (IC50 ~17μM) [22] |
| High-Refractive Index Polymers | Optical material development; transparency studies | pX-PTU (nD=1.71, %T≥99) [2] [19] |
| Ginsenoside Formulations | Natural product alternatives; formulation optimization | Protopanaxtriol saponins in ethosomes (93% EE) [23] |
| Spectrophotometry Standards | UV-Vis instrument calibration; quantitative comparison | Phthalocyanine references (Reactive Blue 21) [24] |
The systematic analysis of spectral shifts through UV-Vis transparency and absorption profiling provides critical insights for optimizing pigment removal strategies in both biomedical and material science applications. PTU-mediated biological inhibition offers specificity and mechanistic clarity but presents challenges due to off-target effects on developmental processes, particularly through thyroid peroxidase inhibition. Chemical bleaching approaches, while potentially less specific, provide robust depigmentation with distinct spectral signatures characterized by broadband absorption reduction.
For research and drug development applications, the choice between these approaches depends critically on the specific application requirements. PTU and its structural analogs remain valuable tools for fundamental studies of melanogenesis, while emerging materials like deconjugated poly(thiourea)s demonstrate how molecular engineering can achieve specific optical properties for advanced applications. The continued refinement of high-throughput screening methodologies will accelerate the discovery of next-generation depigmenting agents with improved specificity and safety profiles.
Future research directions should focus on developing more specific tyrosinase inhibitors that minimize off-target effects, optimizing formulation strategies for enhanced delivery, and establishing standardized spectral classification systems for quantitative comparison of depigmentation efficacy across studies and material systems.
Synthesis and Application: Protocols for PTU Development and Peroxide-Based Treatments
Synthetic Routes for High-Molecular-Weight Xylylic Poly(thiourea)s
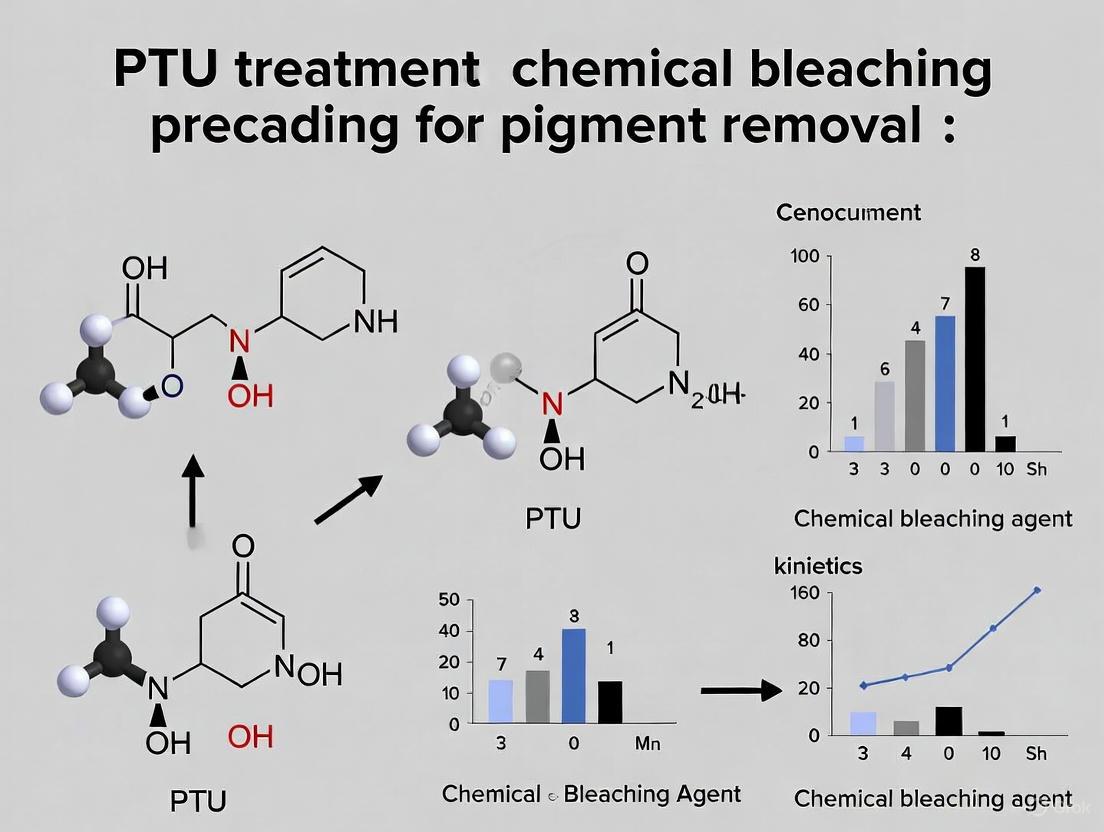
The investigation of advanced polymeric materials for pigment removal represents a frontier in therapeutic and cosmetic research. Within this field, xylylic poly(thiourea)s (pX-PTU) have emerged as promising candidates due to their unique structural properties and functional capabilities. These polymers are characterized by a poly(p-xylylene thiourea) backbone that incorporates "de-conjugated" hydrogen bonds, which inhibit orbital interactions between the highly polarizable phenylene and thiourea units through strategically placed methylene spacers [25]. This specific architectural arrangement confers exceptional material properties, including high visible-light transparency (%T ≥ 99) and a remarkably high refractive index (nD = 1.71) with a reasonable Abbe number (νD = 26) [25].
The relevance of pX-PTU to pigment removal research stems from the growing interest in non-invasive, chemical-based approaches as alternatives to traditional treatments like 1-phenyl-2-thiourea (PTU). PTU functions as a potent tyrosinase inhibitor—the rate-limiting enzyme in melanin biosynthesis—by chelating copper ions at the enzyme's active site, thereby preventing the catalytic conversion of L-tyrosine to L-DOPA and subsequent oxidation to dopaquinone [26] [27]. While effective, PTU treatment raises concerns regarding potential toxicity and long-term stability, driving the exploration of polymeric systems that can offer improved safety profiles and sustained efficacy. High-molecular-weight xylylic poly(thiourea)s present an innovative approach through their bleaching effect and material properties that may be engineered for specific therapeutic applications, including controlled release systems or as active components in topical formulations [25].
Synthesis and Structural Characterization of Xylylic Poly(thiourea)s
Synthetic Approaches
The synthesis of high-molecular-weight xylylic poly(thiourea)s employs controlled polymerization techniques to achieve precise architectural control. While specific synthetic protocols for pX-PTU are not fully detailed in the available literature, analogous approaches for high-performance polymers provide instructive methodologies. The synthesis of ultra-high molecular weight polymers via topochemical routes represents a particularly promising strategy, as these solid-state transformations enable production of polymers with number-average molecular weights (Mn) above 10^6 Dalton in a stereospecific, regioregular, solvent-free, and catalyst-free manner [28].
Advanced polymerization techniques like atom transfer radical polymerization (ATRP) combined with 'click' chemistry have been successfully employed for creating architecturally complex polymers with low polydispersities [29]. These methods allow for precise control over molecular weight and functionality, which are critical parameters for optimizing the performance of pX-PTU in pigment removal applications. The functional group tolerance displayed in topochemical polymerization reactions is particularly valuable, as it accommodates both solubilizing side chains and reactive groups that permit post-polymerization functionalization—a key consideration for tailoring polymer properties to specific biological applications [28].
Structural Analysis and Characterization
Rigorous structural characterization is essential for correlating polymer architecture with functional performance in pigment removal applications. For pX-PTU, spectroscopic confirmation of structure includes solution-phase ¹H, ¹³C, and ¹⁹F NMR characterizations, which show broadened peaks typical of polymers along with chemical shifts and integrals consistent with the proposed structure [28]. The distinctive "de-conjugated" hydrogen bonding pattern in pX-PTU can be verified through infrared spectroscopic studies, particularly examining vibrational features in the 1570–1635 cm⁻¹ range associated with specific structural motifs [28].
Molecular weight determination for soluble ultra-high molecular weight polymers like pX-PTU can be accomplished via size-exclusion chromatography (SEC). For instance, topochemically produced polymers of comparable systems have demonstrated molecular weights of Mn = 1.9 × 10⁶ and Mw = 3.9 × 10⁶ with polydispersity indices of approximately 1.99 [28]. These structural characteristics directly influence performance parameters including solubility, film-forming capability, and compatibility with biological systems—all critical factors for pigment removal applications.
Table 1: Key Characterization Techniques for Xylylic Poly(thiourea)s
| Characterization Method | Parameters Analyzed | Significance for Pigment Research |
|---|---|---|
| Size-Exclusion Chromatography (SEC) | Molecular weight (Mn, Mw), polydispersity index (PDI) | Determines solubility and diffusion characteristics in biological environments |
| NMR Spectroscopy (¹H, ¹³C, ¹⁹F) | Chemical structure, functional group integrity, purity | Confirms structural motifs responsible for deconjugated hydrogen bonding |
| Infrared (IR) Spectroscopy | Hydrogen bonding patterns, vibrational features | Verifies deconjugation between phenylene and thiourea units |
| X-ray Diffraction (XRD) | Crystallinity, molecular packing | Correlates solid-state structure with bleaching efficacy |
| Refractometry | Refractive index (nD), Abbe number (νD) | Quantifies optical properties relevant to skin applications |
Experimental Protocols for Polymer Synthesis and Evaluation
Polymer Synthesis Methodology
The synthesis of xylylic poly(thiourea)s follows a multi-step protocol designed to achieve precise control over molecular architecture and functionality. While the exact synthetic route for pX-PTU is proprietary, the following generalized procedure adapted from analogous high-performance polymer systems can be employed:
Monomer Synthesis and Purification:
- Begin with synthesis of AQM (azaquinodimethane) ditriflate monomers following established procedures with modifications [28].
- Purify monomers via recrystallization from appropriate solvent systems (e.g., toluene, tetrahydrofuran) to achieve high-purity crystals suitable for polymerization.
- Characterize monomers using ¹H NMR, ¹³C NMR, and elemental analysis to verify structure and purity before polymerization.
Topochemical Polymerization:
- Dissolve monomer (e.g., 500 mg) in minimal appropriate solvent (e.g., toluene) and allow slow evaporation at controlled temperature (20-25°C) to form crystalline aggregates.
- Expose monomer crystals to visible light irradiation or thermal initiation (80°C) for solid-state polymerization [28].
- Monitor reaction progress by color change from yellow to colorless, indicating polymerization completion.
- Isolate polymer product and wash with cold solvent to remove any unreacted monomer.
Post-Polymerization Functionalization:
- For functionalized derivatives, dissolve polymer in anhydrous dimethylformamide (DMF) under inert atmosphere.
- Add appropriate functionalizing agents (e.g., alkyl halides, acyl chlorides) with base catalyst.
- React for 12-24 hours at 60-80°C with continuous stirring.
- Precipitate functionalized polymer into methanol, filter, and dry under vacuum until constant weight.
Evaluation of Pigment Removal Efficacy
The assessment of pX-PTU for pigment removal applications requires specialized biological models and analytical techniques. Zebrafish embryos have emerged as a powerful phenotype-based screening platform due to genetic similarity to humans (approximately 70-87%) and transparent embryos that facilitate direct observation of pigment changes [30] [31]. The following protocol details the evaluation procedure:
Zebrafish Melanogenesis Assay:
- Obtain wild-type AB zebrafish eggs and maintain at 28°C in E3 embryo medium (5.03 mM NaCl, 0.17 mM KCl, 0.33 mM CaCl₂, 0.33 mM MgSO₄) [31].
- At 9 hours post-fertilization (hpf), transfer 20 fertilized eggs to each well of 12-well plates containing 1.5 mL of conditioned medium with test concentrations of pX-PTU (1-100 μg/mL).
- Include appropriate controls: blank control (E3 medium with 1% ethanol or DMSO), positive control (25 μM 8-methoxypsoralen or known tyrosinase inhibitors like PTU).
- Incubate plates at 28°C for 63 hours, replenishing test solutions every 24 hours.
- Document melanin distribution and development daily using stereomicroscopy with standardized imaging conditions.
Tyrosinase Activity Assessment:
- Harvest zebrafish larvae and homogenize in ice-cold lysis buffer.
- Centrifuge at 11,000 rpm at 4°C for 10 minutes using Sorvall ST16 centrifuges or equivalent [30].
- Determine protein concentration in supernatant using BCA protein assay kit with bovine serum albumin as standard.
- Assess tyrosinase activity using commercial tyrosinase kit according to manufacturer instructions, measuring optical density at 475 nm.
- Normalize tyrosinase activity to total protein content and express as percentage inhibition relative to control.
Cytotoxicity Evaluation:
- Assess embryo mortality, malformation rate, and overall development throughout exposure period.
- For cell-based assays, utilize B16F10 melanoma cells cultured under standard conditions.
- Treat cells with pX-PTU extracts at varying concentrations (1-100 μg/mL) for 72 hours.
- Evaluate cell viability using MTT assay or equivalent metabolic activity measurement.
- Calculate IC₅₀ values for both anti-melanogenic effects and cytotoxicity to establish therapeutic index.
Table 2: Key Research Reagent Solutions for Polymer Synthesis and Biological Evaluation
| Reagent/Material | Function/Application | Experimental Considerations |
|---|---|---|
| AQM Ditriflate Monomers | Polymer building blocks | Require strict purification; sensitive to moisture and light |
| Dimethylformamide (DMF) | Polymerization solvent | Must be anhydrous for molecular weight control |
| Tyrosinase Enzyme Kit | Enzymatic activity assessment | Measure monophenolase and diphenolase activities separately |
| B16F10 Melanoma Cells | In vitro melanogenesis model | Require α-MSH stimulation for melanin production |
| Zebrafish Embryos (AB line) | In vivo pigment screening | 9-72 hpf optimal for melanogenesis studies |
| PTU (1-phenyl-2-thiourea) | Positive control inhibitor | Use at 50-200 μM for complete melanin inhibition |
| MTT Assay Reagents | Cytotoxicity determination | Measure at 570 nm reference wavelength |
Comparative Performance Analysis: pX-PTU vs. Alternative Approaches
Efficacy in Pigment Reduction
The performance of xylylic poly(thiourea)s in pigment removal applications must be evaluated against established treatments, particularly PTU-based approaches and other tyrosinase inhibitors. Quantitative comparison reveals distinct advantages and limitations of each system:
Table 3: Performance Comparison of Pigment Removal Agents
| Parameter | Xylylic Poly(thiourea)s (pX-PTU) | PTU (1-phenyl-2-thiourea) | Natural Extracts (e.g., Mulberry) | Commercial Inhibitors (e.g., Kojic Acid) |
|---|---|---|---|---|
| Melanin Reduction Efficacy | Moderate to high (dose-dependent) | High (>80% at 200 μM) [31] | Variable (dose-dependent) [27] | Moderate (40-60% at typical concentrations) |
| Tyrosinase Inhibition Mechanism | Physical barrier and potential copper chelation | Copper chelation at active site [26] | Competitive inhibition, copper chelation [27] | Competitive inhibition, copper chelation |
| Cytotoxicity | Low (high molecular weight limits cellular uptake) | Moderate (dose-dependent toxicity) [26] | Low to moderate (extract-dependent) [27] | Moderate (potential sensitization) |
| Structural Stability | High (resistant to degradation) | Moderate (sensitive to oxidation) | Variable (extract-dependent stability) | Low (prone to oxidation and degradation) |
| Optical Properties | High transparency (%T ≥ 99) [25] | N/A | Variable (often colored extracts) | N/A |
| Therapeutic Index | Potentially high (separation of efficacy and toxicity) | Moderate | Moderate to high | Low to moderate |
The data illustrates that while PTU demonstrates potent melanin reduction capability, achieving over 80% reduction at 200 μM concentration in zebrafish models [31], it presents toxicity concerns that limit its therapeutic application. In contrast, pX-PTU offers a potentially more favorable safety profile due to its high molecular weight, which limits systemic absorption while maintaining efficacy through surface activity and potential barrier functions.
Molecular Mechanisms and Signaling Pathways
The mechanism of action for pX-PTU in pigment removal appears distinct from traditional tyrosinase inhibitors like PTU. While PTU directly targets the enzyme's copper-containing active site, pX-PTU likely operates through multiple pathways:
Diagram 1: Proposed multi-target mechanism of xylylic poly(thiourea)s in pigment removal
In comparison, PTU and similar small-molecule inhibitors function through direct enzyme inhibition, as illustrated in the following pathway:
Diagram 2: PTU mechanism through direct tyrosinase inhibition in melanogenesis pathway
The comparative analysis reveals that pX-PTU operates through a physical, surface-level mechanism potentially complemented by secondary signaling effects, while PTU directly interrupts the biochemical pathway of melanin synthesis. This fundamental difference in mechanism underlies the distinct efficacy and safety profiles of these approaches.
The development of high-molecular-weight xylylic poly(thiourea)s represents an innovative approach in pigment removal research, bridging materials science with therapeutic applications. While traditional PTU treatment relies on potent biochemical inhibition of tyrosinase, pX-PTU offers a alternative mechanism centered on physical barrier properties, optical characteristics, and potentially multi-factorial modulation of melanogenic pathways. The distinct "de-conjugated" hydrogen bonding architecture of pX-PTU confers exceptional material properties including high visible-light transparency and refractive index, which may be advantageous for cosmetic and therapeutic formulations where visual appearance is crucial [25].
For researchers and drug development professionals, the comparative analysis presented herein suggests several promising research directions. First, the potential for synergistic combinations of pX-PTU with low concentrations of conventional tyrosinase inhibitors should be explored, possibly enabling enhanced efficacy while minimizing toxicity concerns. Second, structure-activity relationships within the poly(thiourea) family warrant systematic investigation to optimize pigment removal capability while maintaining favorable safety profiles. Finally, advanced delivery systems leveraging the film-forming properties of pX-PTU could enable sustained, localized activity with reduced risk of systemic exposure.
As pigment removal research evolves beyond simple enzyme inhibition toward multi-modal approaches, materials like xylylic poly(thiourea)s offer versatile platforms that can be engineered for specific applications. The ongoing challenge remains balancing efficacy, safety, and stability—a tripartite requirement that underscores the need for continued fundamental research into the structure-property relationships of these advanced polymeric systems.
The formulation of peroxide-based gels represents a critical area of research in cosmetic and therapeutic applications, particularly for pigment removal. These formulations balance efficacy with safety, manipulating variables such as peroxide concentration, pH, and activation systems to optimize performance. Within broader research on depigmenting agents, peroxide-based gels offer a contrasting mechanism to chemical alternatives like phenylthiourea (PTU). While PTU inhibits melanization by suppressing tyrosinase activity and potentially affecting thyroid peroxidase, peroxide gels function primarily through oxidative degradation of pigment molecules [1] [15]. This guide systematically compares the performance characteristics of peroxide-based gel formulations against other alternatives, providing researchers and drug development professionals with consolidated experimental data and methodologies critical for product development and application strategy.
Comparative Performance of Bleaching Agents
Whitening Efficacy
The effectiveness of a bleaching agent is primarily quantified by color change (ΔE) and the whitening index (WID). Research consistently demonstrates that hydrogen peroxide concentration directly influences the degree of whitening achieved.
Table 1: Whitening Efficacy of Different Agents
| Bleaching Agent | Concentration | Color Change (ΔE) | Whitening Index (WID) | Study Duration | Citation |
|---|---|---|---|---|---|
| Hydrogen Peroxide | 35% | 9.6 | Highest among tested agents | 14 days | [32] |
| Hydrogen Peroxide | 10% Carbamide (~3.5% HP) | Significant ΔE00 | Effective whitening | 14 days | [33] |
| PAP | Not Specified | 6.6 | Moderate | 7 days | [32] |
| Bromelain | 1% | 5.3 | Substantial clinical potential | 4 weeks | [34] |
| Sodium Bicarbonate | Not Specified | 7.5 | Moderate | 7 days | [32] |
| Ficin | 1% | Similar to carbamide peroxide | Similar to carbamide peroxide | 4 weeks | [34] |
Hydrogen peroxide (H₂O₂) remains the most effective agent, with 35% H₂O₂ producing the strongest color change (ΔE=9.6) [32]. Its efficacy is concentration-dependent; a 2.5% H₂O₂ mouthrinse required 60 days of use to produce significant visual alteration, whereas a 10% carbamide peroxide gel (equivalent to ~3.5% H₂O₂) produced more pronounced results in just 14 days [33]. Alternative agents like PAP and bromelain show measurable but reduced efficacy. Bromelain, ficin, and papain-based gels demonstrate substantial clinical potential, with ficin and bromelain showing color change comparable to carbamide peroxide in vitro [34].
Safety and Side Effects
Safety profiles, including effects on dental tissues and cytotoxicity, vary significantly among bleaching agents.
Table 2: Safety Profile and Side Effects of Bleaching Agents
| Bleaching Agent | Enamel Surface Alterations | Cytotoxicity (Cell Viability) | Tooth Sensitivity | Citation |
|---|---|---|---|---|
| 35% Hydrogen Peroxide | Mild interprismatic dissolution | Cytotoxic (Viability <70%) | Highest reported sensitivity | [32] [35] [36] |
| 10% Carbamide Peroxide | Increased roughness | Not Specified | Affects 51% of patients (approx.) | [37] |
| 6% Hydrogen Peroxide (with additives) | Less damage | Reduced cytotoxicity with additives | Significantly lower | [36] |
| Bromelain | No surface changes | Non-cytotoxic (>70% viability) | Not Specified | [32] [34] |
| PAP (pH adjusted) | No surface changes | Cytotoxic | Not Specified | [32] |
| 3% Hydrogen Peroxide | Noticeable surface undulations | Not Specified | Mild | [37] |
| 8% Carbamide Peroxide | Less alteration than 3% HP | Not Specified | Statistically lower than 3% HP | [37] |
Higher peroxide concentrations correlate with increased adverse effects. Enamel treated with 35% H₂O₂ showed mild interprismatic dissolution, while low-concentration gels (≤6% HP) caused less damaging effects [32] [35]. Cytotoxicity is a significant concern; H₂O₂, PAP, and sodium bicarbonate reduced fibroblast viability below 70%, whereas bromelain was the only non-cytotoxic agent tested [32]. Tooth sensitivity is the most common side effect, reported by up to 51% of patients using 10% carbamide peroxide. Lower concentrations directly reduce this sensitivity; 6% H₂O₂ gels with additives like hyaluronic acid and NF_TiO₂ nanoparticles caused significantly lower sensitivity than 35% H₂O₂ gels [36] [37].
The Impact of Formulation Variables
Peroxide Concentration
Hydrogen peroxide concentration fundamentally influences the diffusion kinetics through biological tissues and the final bleaching outcome. Research demonstrates that 35% H₂O₂ diffuses through enamel and dentin significantly faster than 20% H₂O₂, leading to quicker pulpal chamber penetration [38]. However, this increased diffusion also elevates the risk of cytotoxic effects on pulpal cells and tooth sensitivity [38] [35]. The trend in formulation is toward low-concentration peroxides (e.g., ≤6% HP), which are considered effective with fewer damaging effects on enamel and cells [35]. A 2025 clinical trial confirmed that 6% H₂O₂ gel, when enhanced with NF_TiO₂ nanoparticles and activated by violet LED, achieved comparable efficacy to 35% H₂O₂ after six months, with markedly reduced tooth sensitivity [36].
pH of the Formulation
The pH of a bleaching gel affects both its chemical stability and its interaction with dental hard tissues. Many high-concentration peroxide gels are acidic, contributing to enamel demineralization and surface roughness [37]. Formulators adjust pH to optimize the reactivity of the active ingredient. For enzyme-based agents like papain, the gel's pH is adjusted to the optimum for proteolytic activity to maximize function [34]. The pH also influences the byproducts of the bleaching reaction; carbamide peroxide decomposes into urea, which increases pH, potentially mitigating some acidic damage [37]. In vitro tests show that an 8% carbamide peroxide gel started alkaline and turned neutral, while a 3% H₂O₂ gel remained acidic, corresponding to the former causing less enamel alteration [37].
Activation Systems
Activation systems are employed to accelerate the decomposition of hydrogen peroxide into reactive oxygen radicals, enhancing the bleaching process.
- Chemical Activation: Manganese gluconate (0.025%) can be added as a catalyst. For 20% H₂O₂, chemical activation significantly increased the diffusion time compared to light activation, suggesting a more controlled reaction [38].
- Light Activation: Halogen lamps, LEDs, and lasers transfer energy to the peroxide. Light activation significantly decreases the diffusion time of HP through dental tissues, leading to a faster onset of action [38]. However, this may also increase the risk of pulpal irritation due to temperature rise and accelerated diffusion [38].
- Nanoparticle and Violet LED Activation: A modern approach uses NF_TiO₂ (nitrogen-fluoride co-doped titanium dioxide) nanoparticles in combination with violet LED light. This system activates low-concentration (6%) H₂O₂ gels, enabling them to achieve long-term whitening results similar to high-concentration gels but with drastically reduced tooth sensitivity [36].
Experimental Protocols for Key Assays
Protocol: Evaluating Hydrogen Peroxide Diffusion
This protocol assesses the penetration rate of bleaching gels through dental hard tissues [38].
- Tooth Specimen Preparation: 120 cylindrical bovine enamel-dentin specimens (6 mm diameter, 2 mm height: 1 mm enamel/1 mm dentin) are prepared using a trephine mill. Specimens are stored in 0.1% thymol solution.
- Simulated Pulpal Chamber Setup: Each specimen is fixed over a transparent epoxy well with a 20 μL internal cavity. The chamber is filled with 10 μL of an enzymatic reagent (4-aminoantipyrin and phenol with peroxidase) that changes color from transparent to pink upon contact with H₂O₂.
- Gel Application & Data Acquisition: Bleaching gels (20 μL) are applied to the enamel surface according to experimental groups (e.g., with/without activation). A digital camera records the time required for the enzymatic reagent to change color.
- Data Analysis: Diffusion time data is analyzed using two-way ANOVA and Tukey's tests to determine statistical significance between different concentrations and activation methods.
Protocol: Assessing Whitening Efficacy and Enamel Surface
This in vitro protocol evaluates color change and enamel alterations [34].
- Specimen Preparation and Staining: Bovine enamel-dentin discs are prepared, polished, and stained in a coffee solution for one week.
- Bleaching Treatment: stained specimens are treated with experimental gels. A typical application involves placing the gel on enamel for 15 minutes, then rinsing. This cycle is repeated multiple times over a set period.
- Color Measurement: A spectrophotometer measures CIELAB color parameters (L, a, b*) before and after treatment. The color difference (ΔE) is calculated using the CIEDE2000 formula. The Whiteness Index (WID) is also determined.
- Surface Analysis: Enamel microhardness is measured using a Knoop microhardness tester. Surface roughness is quantified with a profilometer. Enamel morphology is directly observed using Scanning Electron Microscopy (SEM).
Protocol: Determining Cytotoxicity
This protocol evaluates the biological safety of bleaching agents [32].
- Cell Culture Preparation: Primary human fibroblasts or other relevant cell lines (e.g., L929 mouse fibroblasts) are cultured in standard medium.
- Preparation of Eluates: Bleaching agents are prepared and, if necessary, mixed with culture medium to create an extract for testing.
- Exposure and Incubation: Cells are exposed to the eluates for a predetermined period (e.g., 24 hours).
- Viability Assay: Cell viability is assessed using a standardized assay like WST-1. Metabolic activity is measured, and viability is expressed as a percentage relative to an untreated control group. According to ISO 10993-5, cell viability >70% is generally considered non-cytotoxic.
Signaling Pathways and Experimental Workflows
Peroxide Activation and Bleaching Pathway
The following diagram illustrates the core chemical pathway of peroxide-based bleaching and its contrasting mechanism with PTU.
Peroxide Bleaching vs. PTU Pigment Inhibition Pathway
Experimental Workflow for Gel Evaluation
This workflow outlines the key phases in a comprehensive evaluation of a peroxide-based gel formulation.
Gel Formulation and Testing Workflow
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for Peroxide Gel Research
| Reagent / Material | Function in Research | Example Use Case | Citation |
|---|---|---|---|
| Bovine Tooth Specimens | Standardized substrate for bleaching efficacy and safety tests. | Used as an enamel model for diffusion, whitening, and surface alteration studies. | [38] [35] [34] |
| Spectrophotometer | Objectively measures color parameters (L, a, b*) to calculate ΔE and WID. | Quantifying color change after gel application in vitro. | [32] [34] |
| Scanning Electron Microscope (SEM) | High-resolution imaging of surface morphology and alterations. | Visualizing interprismatic enamel dissolution after bleaching. | [32] [34] [37] |
| Enzymatic Reagent (4-aminoantipyrin/phenol/peroxidase) | Detects hydrogen peroxide penetration by colorimetric change. | Simulating pulpal fluid to measure H₂O₂ diffusion time through dentin. | [38] |
| Primary Human Fibroblasts | In vitro model for assessing cytotoxicity and biocompatibility. | Determining cell viability (WST-1 assay) after exposure to gel eluates. | [32] |
| Manganese Gluconate | Chemical activator that catalyzes the decomposition of H₂O₂. | Added to bleaching gel to accelerate the production of free radicals. | [38] |
| NF_TiO₂ Nanoparticles | Photo-catalyst that enhances the efficacy of low-concentration H₂O₂ when activated by light. | Formulated with 6% H₂O₂ and activated by violet LED for low-sensitivity bleaching. | [36] |
| Violet LED Light Source | Provides specific wavelength light to activate photocatalytic nanoparticles. | Used to energize NF_TiO₂ nanoparticles in low-concentration peroxide gels. | [36] |
In pigment removal research, particularly in zebrafish models used for developmental biology and drug screening, achieving specimen transparency is crucial for clear visualization. The choice between phenylthiourea (PTU) treatment and chemical bleaching represents a critical methodological crossroads, each with distinct implications for experimental outcomes. PTU, a tyrosinase inhibitor, actively prevents melanin synthesis during development, while chemical bleaching removes existing pigment post-fixation. This guide objectively compares these approaches within advanced delivery frameworks, providing researchers with experimental data and protocols to inform their methodological selections for precise and reliable results.
Comparative Analysis: PTU Treatment vs. Chemical Bleaching
The following table summarizes the core performance characteristics of PTU treatment and chemical bleaching based on current research findings.
Table 1: Performance Comparison of Pigment Removal Methods
| Parameter | PTU Treatment | Chemical Bleaching |
|---|---|---|
| Mechanism of Action | Inhibits tyrosinase enzyme, preventing melanogenesis [1] | Oxidizes and removes pre-formed melanin [39] |
| Efficacy of Pigment Removal | High; effectively prevents melanization [1] | Effective, but may require optimization [39] |
| Treatment Timeline | From ~12 hours post-fertilization (hpf) onward [1] | Post-fixation of embryos [39] |
| Developmental Side Effects | Specifically reduces eye size in larval zebrafish [1] [15] | Not reported to cause specific morphological defects [39] |
| Impact on Thyroid Hormone | Reduces thyroxine level; inhibits thyroid peroxidase (TPO) [1] | No known impact on thyroid pathway |
| Key Experimental Consideration | Side effects are not due to melanization inhibition but TPO inhibition; requires cautious data interpretation [1] | Avoids pharmacological side effects associated with PTU [39] |
Detailed Experimental Protocols
PTU Treatment Protocol
The standard protocol for PTU treatment involves continuous exposure of zebrafish embryos to the chemical to prevent pigment formation.
- Solution Preparation: A 0.2 mM (0.003% w/v) PTU solution is prepared in a 30% Danieau solution [1].
- Treatment Initiation: Embryos are placed in the PTU solution beginning at gastrulation (approximately 12 hpf) [1]. Some protocols report initiating treatment at 24 hpf with 0.075 mM PTU, though this is less effective [1].
- Maintenance: The PTU solution should be refreshed daily to maintain efficacy throughout the developmental period under study [1].
- Key Notes: The eye size reduction phenotype typically becomes measurable at 3 days post-fertilization (dpf) [1]. This effect is likely mediated through the inhibition of thyroid peroxidase (TPO) rather than a general suppression of thyroid hormone synthesis or the inhibition of tyrosinase [1].
Chemical Bleaching Protocol
Chemical bleaching offers an alternative method that removes pigment after fixation, avoiding the developmental side effects of PTU.
- Solution Preparation: A bleaching solution is prepared using 3% hydrogen peroxide (H₂O₂) and 1.79 mM potassium hydroxide (KOH) [39].
- Procedure: Fixed embryos are incubated in the bleaching solution for approximately 5 minutes to remove melanin [39].
- Key Notes: This method is performed on fixed specimens, eliminating the risk of pharmacological side effects during development [39]. However, its effectiveness compared to PTU can vary.
Signaling Pathways and Experimental Workflow
The diagram below illustrates the mechanistic pathways of both pigment removal methods and their physiological consequences, highlighting the key difference that PTU treatment acts during development while bleaching acts post-fixation.
The Scientist's Toolkit: Essential Research Reagents
This table details key reagents used in zebrafish pigment removal research, providing researchers with a quick reference for their experimental setup.
Table 2: Essential Reagents for Pigment Removal Research
| Reagent | Function/Application | Key Considerations |
|---|---|---|
| Phenylthiourea (PTU) | Tyrosinase inhibitor used to prevent melanization in developing zebrafish embryos [1]. | Causes specific reduction in eye size via TPO inhibition; requires caution in interpretation of morphological data [1] [15]. |
| Hydrogen Peroxide (H₂O₂) | Oxidizing agent used in chemical bleaching solution to remove pre-formed melanin in fixed specimens [39]. | Used in combination with KOH; a post-fixation method that avoids developmental side effects [39]. |
| Potassium Hydroxide (KOH) | Base used in bleaching solution to facilitate the oxidation of melanin by H₂O₂ [39]. | Concentration is critical for effective and safe bleaching; standard protocol uses 1.79 mM [39]. |
| Proteinase K | Protease used to increase permeability of embryos for better probe penetration in protocols like in situ hybridization [39]. | Digestion time (e.g., 5 min) must be carefully controlled to avoid tissue damage [39]. |
| Paraformaldehyde (PFA) | Fixative used to preserve tissue morphology prior to bleaching or other staining procedures [39]. | Standard concentration is 4%; fixation time is typically 20 minutes at room temperature [39]. |
In the field of pigment removal research, particularly in studies comparing PTU (1-phenyl-2-thiourea) treatment with chemical bleaching, advanced characterization techniques are indispensable for understanding molecular mechanisms and evaluating efficacy. Nuclear Magnetic Resonance (NMR), Infrared (IR), and X-ray Diffraction (XRD) spectroscopy, complemented by Density Functional Theory (DFT) calculations, provide critical insights into structural, electronic, and dynamic processes at the molecular level. This guide objectively compares the performance of these analytical techniques in the context of pigment research, supported by experimental data from relevant studies, to aid researchers and drug development professionals in selecting appropriate methodologies for their investigations.
Table 1: Comparison of Key Characterization Techniques in Pigment Research
| Technique | Key Measurable Parameters | Applications in Pigment Research | Advantages | Limitations |
|---|---|---|---|---|
| NMR Spectroscopy | Chemical structure, molecular dynamics, conformational changes, interaction sites [40] [41] | Study of ligand-binding interactions, structural verification of synthesized compounds [42] [40] | Non-destructive, quantitative, provides atomic-level detail [41] | Lower sensitivity compared to other techniques; solid-state requires Magic Angle Spinning [43] |
| IR Spectroscopy | Functional group identification, molecular vibrations, chemical bonding [44] [40] | Monitoring chemical changes during bleaching, verifying compound formation [42] | High sensitivity to functional groups, can be used under various conditions [44] | Complex data interpretation for large molecules, overlapping bands |
| X-ray Diffraction (XRD) | Crystal structure, phase identification, unit cell parameters, atomic arrangement [42] | Determining crystal structure of novel organic-inorganic hybrid materials [42] | Direct structural determination, high information content [42] | Requires single crystals, insensitive to amorphous phases |
| DFT Calculations | Optimized molecular geometry, electronic properties, vibrational frequencies, reaction pathways [42] [44] [40] | Predicting NMR chemical shifts, IR vibrational modes, and UV-Vis spectra [44] [40] | Provides theoretical foundation for experimental data, predicts properties [40] | Computationally expensive for large systems, accuracy depends on functional |
Experimental Protocols for Pigment Research
Protocol for PTU-Induced Pigmentation Studies in Zebrafish
The PTU-induced zebrafish model is a well-established system for studying depigmentation mechanisms and screening potential therapeutic agents [30] [45].
- Animal Model and Maintenance: Wild-type AB zebrafish are maintained at 28°C under a 14-hour light/10-hour dark cycle. Embryos are obtained from spawning and raised in a zebrafish medium [30].
- Depigmentation Model Generation: At 24 hours post-fertilization (hpf), zebrafish embryos are treated with PTU (typically 50-200 µM) for 24 hours to inhibit tyrosinase and induce depigmentation. The success of the model is confirmed by observing a reduction in melanin granules under a microscope [30].
- Drug Administration: After successful modeling, the experimental groups are treated with different concentrations of the test compound (e.g., galangin at 1 and 2 µM [30] or ML233 at ~20 µM [45]) for 48 hours. A positive control group (e.g., 25 µM 8-methoxypsoralen) and a model control group (only PTU) are included.
- Tyrosinase Activity and Melanin Content Assay: Zebrafish larvae are homogenized in lysis buffer at 4°C. The lysate is centrifuged, and the supernatant is used to determine protein concentration. Tyrosinase activity is measured using a commercial kit by monitoring the optical density at 475 nm. Melanin content can be quantified by dissolving melanin granules and spectrophotometrically measuring the absorbance [30].
- Pathway Analysis: Transcriptomic analysis (RNA sequencing) and molecular docking can identify potential pathways and targets. Key protein expression (e.g., TYR, MITF, TRP-1, TRP-2, MAPK pathway proteins) is validated by Western blot [30].
Protocol for Chemical Bleaching Efficacy Studies
Chemical bleaching agents act through direct oxidation or inhibition of pigment formation.
- Sample Preparation: For tooth bleaching studies, enamel-dentine blocks are prepared from bovine incisors. Cylindrical cavities are drilled, and a bleaching agent or a discoloring material like Mineral Trioxide Aggregate (MTA) is applied. Samples are incubated to simulate the oral environment [46].
- Bleaching Agent Application: Common agents include hydrogen peroxide (e.g., 35%, applied for 30 minutes), carbamide peroxide (e.g., 20%, applied for 2-4 hours/nightly), and sodium perborate (applied once every three days). Application times vary based on manufacturer instructions and desired simulation of clinical conditions [46].
- Color Measurement: A spectrophotometer is used to measure color parameters (L, a, b) according to the CIE Lab system at baseline and various time points. The color change (∆E) is calculated using the formula: ∆E = [(Li - L0)^2 + (ai - a0)^2 + (bi - b0)^2]^½, where ∆E > 3.3 is typically considered clinically significant [46].
- Direct Tyrosinase Inhibition Assay: For agents like ML233, molecular docking and in vitro enzymatic assays are performed to predict and confirm direct binding and inhibition of tyrosinase function [45].
Protocol for DFT-Assisted Spectroscopic Analysis
DFT calculations provide a theoretical basis for interpreting experimental spectroscopic data.
- Computational Details:
- Software: Calculations are performed using programs like Gaussian [40].
- Method and Basis Set: The geometry of the molecule is fully optimized in the gas phase using a DFT method, commonly the B3LYP functional, with a basis set such as 6-311++G(d,p) [40].
- Frequency Calculation: The optimized geometry is confirmed as an energy minimum by performing a frequency calculation, which yields no imaginary frequencies. The results also provide theoretical IR spectra [44].
- Scaling Factor: A scaling factor (e.g., 0.967) is often applied to the calculated vibrational frequencies to correct for systematic overestimation and achieve better agreement with experimental data [40].
- Spectroscopic Predictions:
- NMR Chemical Shifts: Calculated using the Gauge-Independent Atomic Orbital (GIAO) method at a level like B3LYP/6-311++G(2d,p) [42] [40].
- UV-Vis Spectrum: Predicted using Time-Dependent DFT (TD-DFT) calculations, which can be performed in both the gas phase and solution to simulate environmental effects [40].
Signaling Pathways in Melanogenesis and Inhibition
The following diagrams illustrate the key molecular pathways involved in melanin synthesis and the points of inhibition for PTU and chemical agents.
Experimental Workflow for Pigment Removal Research
The typical workflow for a comparative study integrating biological models and material characterization is outlined below.
Essential Research Reagents and Materials
Table 2: Key Research Reagent Solutions for Pigment Removal Studies
| Reagent/Material | Function in Research | Example Application in Context |
|---|---|---|
| PTU (1-Phenyl-2-Thiourea) | Tyrosinase inhibitor used to induce depigmentation in animal models [30] [45]. | Creating a vitiligo model in zebrafish embryos for drug screening [30]. |
| Galangin | Flavonoid tested for its potential to ameliorate depigmentation by activating MAPK pathways and increasing melanin content [30]. | Used as a therapeutic agent in PTU-treated zebrafish to study melanogenesis restoration [30]. |
| ML233 | Small molecule identified as a direct tyrosinase inhibitor, reducing melanin production [45]. | Applied in zebrafish and murine cells to study direct inhibition of melanogenesis as an alternative to bleaching [45]. |
| Hydrogen Peroxide (H₂O₂) | Oxidizing bleaching agent that directly degrades melanin pigments [46] [47]. | Used in tooth bleaching studies to correct MTA-induced discoloration [46]. |
| Carbamide Peroxide | A stable complex that releases hydrogen peroxide, used as a slower-acting bleaching agent [46]. | Compared with other agents for efficacy in external tooth bleaching protocols [46]. |
| Sodium Perborate | A source of active oxygen used as a bleaching agent in various applications [46]. | Evaluated for tooth bleaching efficacy, often showing lower performance than H₂O₂ [46]. |
| Mineral Trioxide Aggregate (MTA) | Dental material known to cause tooth discoloration, used to create a model for bleaching studies [46]. | Applied in bovine tooth blocks to induce staining for testing bleaching agent efficacy [46]. |
| B16F10 Cells | Murine melanoma cell line used for in-vitro studies of melanogenesis and tyrosinase activity [30]. | Used to validate the effects of compounds like galangin on melanin synthesis and related protein expression in a controlled cellular environment [30]. |
The synergistic application of NMR, IR, XRD, and DFT calculations provides a powerful framework for advancing pigment removal research. PTU treatment operates through specific inhibition of tyrosinase and modulation of the MAPK signaling pathway, as demonstrated by combined zebrafish phenotyping, transcriptomics, and Western blot analysis. In contrast, chemical bleaching agents like hydrogen peroxide act primarily via direct oxidation of melanin pigments. The choice of characterization technique should be guided by the research question: NMR for solution-state structure and dynamics, IR for functional group monitoring, XRD for definitive crystal structure determination, and DFT for theoretical modeling and spectral prediction. A multi-technique approach, integrating experimental data with computational models, offers the most comprehensive strategy for elucidating complex mechanisms in pigment research and guiding the development of novel therapeutics.
Challenges and Innovations: Optimizing for Sensitivity, Stability, and Efficacy
Addressing Tooth Sensitivity and Enamel Hardness in Peroxide Formulations
Tooth bleaching and dermatological depigmentation represent two distinct approaches to managing pigmentation: one targeting extrinsic stains on dental surfaces and the other addressing intrinsic melanin in skin. Peroxide-based chemical bleaching serves as a cornerstone for clinical tooth whitening, yet its effects on enamel integrity and the associated patient sensitivity are significant considerations in formulation science. This guide objectively compares the performance of various peroxide-based dental whitening formulations, focusing on their efficacy and their impact on enamel microhardness and sensitivity, providing a contrast to the mechanistic approach of PTU (1-phenyl-2-thiourea), a tyrosinase inhibitor used in melanogenesis research.
Performance Comparison of Peroxide Formulations
The following tables summarize key experimental data from clinical and in vitro studies, comparing the efficacy and side effects of different bleaching formulations.
Table 1: Clinical Whitening Efficacy and Sensitivity Profile of At-Home Formulations
| Formulation Type | Key Components | Reported Whitening Efficacy (ΔE*) | Reported Sensitivity Incidence / Level | Color Stability | Primary Study Reference |
|---|---|---|---|---|---|
| Conventional At-Home (20% Carbamide Peroxide) | 20% CP, custom trays | 9.54 ± 3.86 (at 14 days) | Significantly higher VAS scores [48] | Good, significantly better than OTC at 6 months [48] | [48] |
| OTC Ready-to-Use Gel Trays | Lower concentration CP (exact % often proprietary) | 4.24 ± 2.97 (at 14 days) | Lower than conventional 20% CP [48] | Significant color relapse after 6 months [48] | [48] |
| OTC Paint-On Gel | Lower concentration CP (exact % often proprietary) | 2.74 ± 2.70 (at 14 days) | Lower than conventional 20% CP [48] | Significant color relapse after 6 months [48] | [48] |
| 10% CP with 3% Potassium Nitrate | 10% CP, 3% KNO₃ (desensitizer) | No significant difference vs. 10% CP without desensitizer | Did not experience more sensitivity than formulations with 3% KNO₃ [49] | Not specified | [49] |
| 10% CP with 0.5% Potassium Nitrate | 10% CP, 0.5% KNO₃ (desensitizer) | No significant difference vs. other active 10% CP groups | Not associated with more sensitivity than placebo [49] | Not specified | [49] |
Table 2: In-Vitro Impact of Bleaching Agents on Enamel Microhardness and Structure
| Bleaching Agent | Impact on Enamel Microhardness | Impact on Surface Roughness & Morphology | Protective/Remineralizing Strategies | Primary Study Reference |
|---|---|---|---|---|
| 30% Hydrogen Peroxide (HP) | Significant decrease in microhardness [50] | Increased enamel porosity & surface area; reduced average pore size; protein denaturation [50] | Incorporation of 10% Strontium-Fluorapatite (Sr-FAp) increased microhardness vs. HP alone [51] | [50] [51] |
| 10% Carbamide Peroxide (Fluoride-Free) | Decreased microhardness [52] | Morphological changes observed [52] | Post-bleaching application of Anticay (Calcium Sucrose Phosphate) significantly increased microhardness [52] | [52] |
| 10% Carbamide Peroxide (with Fluoride) | Higher microhardness than fluoride-free 10% CP [52] | Minor erosive patterns without crystal deposition [52] | Fluoride itself acts as a protective component [52] | [52] |
| High-Concentration CP & HP (20%, 38%, 45%) | All promoted a significant reduction in hardness; CP 45% showed the lowest values [53] | No significant effect on roughness (Ra) found [53] | Not assessed in the study [53] | [53] |
Detailed Experimental Protocols
To ensure reproducibility and provide context for the data presented, this section outlines the key methodologies from the cited research.
Protocol for Evaluating Microhardness and Remineralization Agents
This methodology is adapted from studies investigating the effect of bleaching and subsequent remineralization on enamel microhardness [52].
- Sample Preparation: Twenty freshly extracted human premolars are decoronated and sectioned mesiodistally into two halves. The enamel surfaces are ground flat and polished. Samples are embedded in acrylic resin.
- Group Allocation: Specimens are randomly divided into groups, for example:
- Group 1: Control (no treatment)
- Group 2: Fluoride-free 10% CP bleaching agent (e.g., Opalescence 10%)
- Group 3: Fluoride-containing 10% CP bleaching agent (e.g., Opalescence PF 10%)
- Group 4: Fluoride-free 10% CP bleaching agent followed by application of a remineralizing agent (e.g., Anticay/ToothMin)
- Treatment Protocol: The bleaching agent is applied to the enamel surface for a set period (e.g., 8 hours daily for 7 days). In the remineralization group, the agent is applied for a specific duration (e.g., 1 minute) after each bleaching treatment. All samples are stored in distilled water or artificial saliva when not undergoing treatment.
- Microhardness Testing: The surface hardness of each sample is determined using a Vickers or Knoop microhardness tester. A diamond indenter is applied to the enamel surface under a specific load (e.g., 200g for Vickers) and dwell time (e.g., 15 seconds). The size of the indentation is measured to calculate the hardness number.
Protocol for Clinical Assessment of Whitening Efficacy and Sensitivity
This protocol summarizes the design of a randomized controlled trial comparing at-home bleaching systems [48].
- Study Design: A randomized, controlled, double-blind clinical trial.
- Participants & Groups: Participants are randomly assigned to different whitening system groups (e.g., conventional 20% CP with custom trays, OTC ready-to-use gel trays, OTC paint-on gel).
- Intervention: Participants use the assigned whitening system according to the manufacturer's or study protocol (e.g., 20% CP for 2 weeks with custom trays worn several hours per day).
- Efficacy Measurement: Tooth shade is measured using a digital spectrophotometer at baseline, during, and after the treatment period (e.g., at 7 days, 14 days, 2 weeks post-bleaching, and 6 months post-bleaching). The color change (ΔE) is calculated based on the CIE Lab color system.
- Sensitivity Measurement: Participants record tooth sensitivity daily using a visual analogue scale (VAS), typically ranging from 0 (no sensitivity) to 10 (unbearable sensitivity). The data is collected throughout the active bleaching phase.
Signaling Pathways and Experimental Workflows
The following diagrams illustrate the key biological pathways affected by bleaching agents and the experimental workflow for comparative studies.
Figure 1: Proposed pathway of hydrogen peroxide-induced enamel alterations. HP penetration leads to mineral loss and organic matrix protein denaturation, resulting in increased enamel porosity. This increased porosity is a primary factor in the reduction of microhardness and the increase in clinical sensitivity [50].
Figure 2: Standard in-vitro workflow for evaluating bleaching formulations. This flowchart outlines the common experimental sequence used to compare the effects of different bleaching agents and protective additives on enamel properties [52] [51] [53].
The Scientist's Toolkit: Key Research Reagents and Materials
Table 3: Essential Materials for Bleaching and Enamel Property Research
| Reagent / Material | Function in Experimental Context |
|---|---|
| Carbamide Peroxide (CP) | Common bleaching agent; decomposes to hydrogen peroxide and urea. Used in various concentrations (e.g., 10%, 20%, 45%) for at-home and in-office bleaching simulations [52] [49] [53]. |
| Hydrogen Peroxide (HP) | The active oxidizing agent in many whitening products. Used in concentrations from ~9.5% to 38% for evaluating efficacy and enamel effects [50] [51] [53]. |
| Potassium Nitrate | A desensitizing agent incorporated into bleaching gels (e.g., 0.5% to 3%) to reduce postoperative sensitivity without affecting efficacy [49]. |
| Strontium-containing Fluorapatite (Sr-FAp) | A remineralizing filler added to high-concentration HP gels (e.g., 30%) to form a protective layer on enamel, mitigating hardness loss and structural damage while maintaining whitening efficacy [51]. |
| Calcium Sucrose Phosphate (Anticay/ToothMin) | A remineralizing agent applied post-bleaching. It provides high concentrations of calcium and phosphate ions to promote remineralization and recover microhardness lost during bleaching [52]. |
| Sodium Fluoride | A common remineralizing agent; can be incorporated into bleaching gels (e.g., Opalescence PF) to help preserve enamel microhardness compared to fluoride-free formulations [52]. |
| Artificial Saliva | A storage medium used to maintain specimen hydration and mimic the oral environment's mineral equilibrium between experimental treatments [53]. |
| Vickers/Knoop Hardness Tester | Instrument used to measure enamel microhardness by indenting the surface with a diamond pyramid (Vickers) or rhombic-based pyramid (Knoop) under a known load [52] [53]. |
| Spectrophotometer | Instrument used for objective color measurement (CIE Lab* values) to quantitatively assess bleaching efficacy (ΔE*) [48] [53]. |
Enhancing PTU Thermostability and Intermolecular H-Bond Networks
In the field of pigment removal research, the scientific community continuously seeks more effective and stable treatment modalities. This guide provides a comprehensive comparison between PTU-based polymer treatments and chemical bleaching agents, focusing on their fundamental mechanisms, thermal stability, and optical performance. For researchers and drug development professionals, understanding the enhanced thermostability and robust hydrogen-bond networks in advanced materials like poly(thiourea)s is crucial for developing next-generation treatments. The data and methodologies presented herein establish a foundational framework for evaluating these competing approaches, with particular emphasis on structural integrity and functional efficacy under various conditions.
The broader thesis context of this analysis revolves around the paradigm shift from conventional chemical bleaching—which often relies on aggressive, sometimes cytotoxic agents—toward more sophisticated polymer-based systems that offer superior control over material properties. Recent advances in polymer science have demonstrated that strategically engineered hydrogen-bond networks can simultaneously enhance multiple material properties, including thermal stability and optical characteristics, which are paramount for both industrial and biomedical applications.
Comparative Performance Analysis: PTU Polymers vs. Chemical Bleaching Agents
The following analysis compares the performance of advanced PTU polymers against traditional chemical bleaching agents and other bioactive compounds used in pigment-related research. The data, drawn from recent studies, highlights key parameters including thermal stability, optical properties, and biological activity.
Table 1: Performance Comparison of PTU Polymers and Alternative Agents
| Material/Agent | Key Performance Indicators | Experimental Values | Research Context |
|---|---|---|---|
| pX-PTU (Polymer) | Refractive Index ((nD)) / Abbe Number ((νD)) / Glass Transition Temp. ((T_g)) / Transparency (%T, 1µm) | 1.71 / 26 / 150 °C / ≥ 99% | Optical polymers; High thermostability and transparency [2]. |
| pX-PTU + mpPh-PTU Blend | Refractive Index ((nD)) / Abbe Number ((νD)) / Glass Transition Temp. ((T_g)) | 1.80 / 17 / 164 °C | Polymer blends for enhanced optical properties [2]. |
| PTS Ginsenosides (Natural) | Tyrosinase Inhibition (IC50) / In Vivo Effect | Not specified / Significant inhibition of UVB-induced melanin in mice | Natural skin whitening agent [23]. |
| Bee Pollen Peptide VY-9 (Natural) | Mono-phenolase IC50 / Di-phenolase IC50 / In Vivo Effect (Zebrafish) | 0.55 ± 0.03 µM / 2.54 ± 0.06 µM / Anti-melanogenic at 4 µM | Competitive tyrosinase inhibitor; depigmentation agent [54]. |
| PAE-ONLs (Liposomal) | Encapsulation Efficiency (EE) / Anti-inflammatory Effect (Zebrafish) | 64.61 ± 0.42% / Reduced inflammatory cell migration | Drug delivery system for anti-inflammatory compounds [55]. |
The data reveals a clear functional distinction between the two categories. PTU polymers, such as pX-PTU, excel in material science applications requiring high thermal stability and superior optical properties, with a glass transition temperature of 150 °C and exceptional light transparency [2]. In contrast, chemical and natural agents like bee pollen peptides and ginsenosides are specialized for biological activity, demonstrating potent tyrosinase inhibition for depigmentation [23] [54]. This dichotomy underscores the importance of selecting the appropriate technology based on the target application, whether it is for advanced optical devices or bioactive formulations.
Experimental Protocols for Key Analyses
To ensure the reproducibility of comparative studies in pigment removal research, this section outlines detailed methodologies for critical experimental procedures cited in this guide.
Protocol for Synthesizing and Characterizing Xylylic Poly(Thiourea)s (X-PTUs)
The synthesis and analysis of high-performance PTUs follow a meticulously controlled protocol [2].
- Synthesis Procedure: High-molecular-weight ((M_w \sim 10^5)) poly(thiourea)s are synthesized via polycondensation of xylylene diamines (XDA) and 1,1-thiocarbonyl diimidazole. The reaction is conducted under inert atmosphere conditions to prevent oxidation of sensitive thiourea groups. The resulting polymers are purified through repeated precipitation in a non-solvent such as methanol or hexane, followed by vacuum drying to remove any residual solvents.
- Structural Characterization: The chemical structure of the synthesized X-PTUs is confirmed using (^1)H and (^{13})C Nuclear Magnetic Resonance (NMR) spectroscopy. The presence of randomized H-bond networks is verified by Fourier-Transform Infrared (FTIR) Spectroscopy, specifically identifying N-H stretching vibrations at ~3270 cm(^{-1}) (ν({N-H(trans/trans)})) and ~3055 cm(^{-1}) (2δ({N-H(cis/trans)})) [2].
- Hydrogen-Bond Network Analysis: The strength and thermal resistance of intermolecular hydrogen bonds are investigated using Variable-Temperature (VT) (^1)H NMR. Chemical shifts of the H-bonding amino protons (around 7.8-7.9 ppm) are monitored over a temperature gradient. A lower temperature dependence coefficient (e.g., -2.8 × 10(^{-3}) ppm K(^{-1}) for pX-PTU) indicates a stronger, more resilient H-bond network [2].
- Thermal and Optical Properties:
- Thermal Stability: The glass transition temperature ((Tg)) and pyrolysis temperature ((Td^5), temperature at 5% weight loss) are determined using Differential Scanning Calorimetry (DSC) and Thermogravimetric Analysis (TGA), respectively, under a nitrogen atmosphere [2].
- Optical Properties: Thin films for UV-vis transparency measurements are prepared via drop-casting or spin-coating. The refractive index ((nD)) and Abbe number ((νD)) are measured using an ellipsometer at the sodium D-line [2].
Protocol for Evaluating Anti-Melanogenesis Activity in Zebrafish
The zebrafish model is a powerful tool for high-throughput screening of depigmenting agents, as utilized in studies on bee pollen peptides and ginsenosides [23] [54].
- Zebrafish Embryo Exposure: Fertilized zebrafish eggs are collected and incubated in E3 medium. At 4 hours post-fertilization (hpf), embryos are placed in multi-well plates and exposed to the test compound, which is dissolved in the medium (e.g., with a final DMSO concentration not exceeding 0.1%). The exposure medium is replaced daily, and the embryos are maintained until the desired observation stage [54].
- Melanin Synthesis Assessment: After exposure, embryos are anesthetized and mounted for imaging. The anti-melanogenic effect is typically quantified by visualizing a reduction in melanin pigmentation in the body or eyes of the embryos under a microscope. Image analysis software can be used to measure the relative area or intensity of melanin spots for quantitative comparison between treatment and control groups [54].
- Cytotoxicity Screening: A critical parallel assay is performed to rule out non-specific toxicity. Embryos are observed for lethality and developmental malformations, such as coagulation, lack of somite formation, non-detachment of the tail, and absence of heartbeat [54].
Table 2: Essential Research Reagents for Pigmentation and Polymer Studies
| Reagent/Material | Function and Application | Research Context |
|---|---|---|
| 1,1-Thiocarbonyl Diimidazole | Monomer for synthesizing poly(thiourea) chains via polycondensation. | Polymer synthesis [2]. |
| Xylylene Diamines (XDA) | Comonomers (m- and p-substituted) that determine polymer backbone structure and H-bond strength. | Polymer synthesis [2]. |
| Tyrosinase Enzyme | Key enzyme in melanin synthesis pathway; target for inhibitory activity testing. | Evaluation of depigmenting agents [54]. |
| Kojic Acid | Standard, potent tyrosinase inhibitor used as a positive control in anti-melanogenesis assays. | Bioactivity testing [54]. |
| Zebrafish Embryos | In vivo model for high-throughput screening of compound toxicity and anti-melanogenic efficacy. | Bioactivity testing [23] [54]. |
| B16F10 Murine Melanoma Cells | In vitro model for studying melanogenesis and testing the effects of compounds on melanin production and tyrosinase activity. | Bioactivity testing [54]. |
| Liposomes (e.g., PAE-ONLs) | Drug delivery vehicle to enhance the stability, solubility, and bioavailability of encapsulated active compounds. | Formulation optimization [55]. |
Signaling Pathways and Experimental Workflows
Understanding the mechanistic pathways of pigment formation and the logical flow of experimentation is vital for advancing research.
Melanogenesis Signaling Pathway
The following diagram illustrates the core biochemical pathway of melanin production within melanocytes, which is the primary target for depigmenting agents.
Diagram Title: Core Melanogenesis Pathway and Inhibition
Workflow for Developing PTU Polymers
This flowchart outlines the integrated research process for developing and evaluating advanced PTU polymers, from molecular design to application testing.
Diagram Title: PTU Polymer Development Workflow
This comparison guide objectively presents the distinct performance profiles of PTU-based polymers and chemical bleaching agents. The experimental data confirms that xylylic poly(thiourea)s, particularly pX-PTU, establish a new benchmark for thermostability and robust hydrogen-bond networks in material science, achieving a glass transition temperature of 150 °C and exceptional optical transparency [2]. Meanwhile, alternative agents like bee pollen peptides operate through highly specific biological mechanisms, such as competitive tyrosinase inhibition, to achieve depigmentation [54].
For researchers in pigment removal, the choice between these technologies is not one of superiority but of application alignment. PTU polymers are the definitive choice for applications demanding high thermal resilience and specific optical properties, whereas biological agents are suited for direct intervention in melanin synthesis pathways. The continued independent development of both fields—driven by structured experimental protocols and a deep understanding of underlying mechanisms—will provide the scientific community with an expanding toolkit for addressing diverse challenges in pigment research and beyond.
Optimizing Bleaching Gel Concentration and Treatment Duration for Minimal Tissue Irritation
The quest for effective pigment removal, whether in clinical dermatology, cosmetic applications, or research models, necessitates a delicate balance between achieving sufficient bleaching efficacy and minimizing undesirable tissue irritation. This guide objectively compares the performance of various bleaching approaches, with particular focus on how gel concentration and treatment duration parameters influence this critical balance. The context is framed within methodological considerations for pigment research, specifically contrasting physical/chemical bleaching with the biological inhibition of pigment synthesis using phenylthiourea (PTU).
Excessive exposure to bleaching agents remains a significant concern across applications, with documented cases of adverse effects including skin allergies, eye irritation, and respiratory issues following improper use [47]. Similarly, in dental bleaching, tooth sensitivity emerges as the most prevalent side effect, affecting approximately 51% of patients undergoing at-home treatments, often due to the penetration of active agents into sensitive tissue layers [37]. The optimization of bleaching protocols is therefore not merely a matter of efficiency but a fundamental requirement for safety and patient comfort. This guide synthesizes recent clinical and experimental data to provide evidence-based recommendations for achieving optimal bleaching outcomes while prioritizing tissue integrity.
Quantitative Comparison of Bleaching Formulations and Outcomes
Dental Bleaching Gels: Clinical Efficacy and Sensitivity Data
The following table summarizes key findings from recent clinical trials on hydrogen peroxide (HP) and carbamide peroxide (CP) based bleaching gels, highlighting the relationship between concentration, pH, treatment duration, and clinical outcomes.
Table 1: Clinical Performance of Dental Bleaching Gels
| Bleaching Gel Formulation | Concentration & pH Profile | Treatment Protocol | Bleaching Efficacy (ΔE00/ΔWID) | Tissue Irritation / Tooth Sensitivity | Source (Clinical Trial) |
|---|---|---|---|---|---|
| Whiteness HP Automixx Plus (In-office) | 35% HP, Neutral & stable pH | 1 application of 50 min (combined with at-home bleaching) | Significant whitening (comparable to acidic gel) | Significantly lower risk and intensity of TS | Verde et al., 2025 [56] |
| Whiteness HP Maxx (In-office) | 35% HP, Acidic & unstable pH | 3 applications of 15 min (combined with at-home bleaching) | Significant whitening (comparable to neutral gel) | Higher risk and intensity of TS | Verde et al., 2025 [56] |
| Experimental Gel (HA-NP-HP6+LED) | 6% HP, with NF_TiO₂ nanoparticles & Hyaluronic Acid | 3 sessions (1×30 min/week), violet LED activation | ΔE00: >1.8 (acceptable) after 14 days; comparable to 35% HP at 6 months | Significantly lower TS; stable pulpal oxygen saturation | de Melo et al., 2025 [57] |
| Invisalign (At-home) | 3% HP, Acidic pH | Daily for 6–8 h, over two weeks | ΔE00 = 5.39 ± 2.50 | Mild sensitivity (0 | BMC Oral Health, 2025 [37] |
| Beyke iWhite (At-home) | 8% CP (~2.9% HP), Initially alkaline, turns neutral | Daily for 6–8 h, over two weeks | ΔE00 = 4.75 ± 2.25 | Statistically lower TS than 3% HP; mild sensitivity | BMC Oral Health, 2025 [37] |
Research Model Bleaching: Protocol and Considerations for Zebrafish Embryos
Bleaching in research contexts, such as clarifying zebrafish embryos post-in situ hybridization, requires different efficacy and tissue integrity metrics, primarily focused on pigment removal without damaging morphological integrity or target signals.
Table 2: Bleaching Protocols in Zebrafish Research Models
| Method | Chemical Composition | Protocol Details | Efficacy & Purpose | Considerations & Tissue Impact |
|---|---|---|---|---|
| Post-Fixation Bleaching | 3% H₂O₂, 1% KOH in dH₂O | 10-15 min for 36 hpf embryos; ~45 min for 5 dpf embryos; monitor closely [58]. | Effective pigment removal for imaging/WMISH. | Over-exposure can "chew up" embryos; less developmental impact than PTU [58]. |
| Post-Fixation Bleaching (Alternative) | 3% H₂O₂, 0.5% KOH | ~20 minutes for 48 hpf embryos [58]. | Effective pigment removal. | Protocol noted as not altering ISH staining, unlike post-ISH bleaching [58]. |
| PTU (Biological Inhibition) | Phenylthiourea | Raise embryos in PTU-containing water (e.g., 0.003%-0.2%) from early stages. | Prevents melanin formation in vivo. | Avoids post-fixation chemical treatment; may affect development and gene expression [58]. |
Experimental Protocols for Irritation and Efficacy Assessment
Clinical Assessment of Tooth Sensitivity and Pulpal Response
The following methodology, adapted from recent randomized controlled trials, provides a framework for quantitatively assessing irritation in dental bleaching studies [56] [57] [37].
- Participant Selection and Randomization: Recruit adult participants with good oral health and baseline tooth shade of A2 or darker. Use a randomized, split-mouth, or parallel-group study design to ensure comparability. Standardized inclusion/exclusion criteria are critical [37].
- Bleaching Procedure: Apply the bleaching gel according to the experimental or manufacturer's protocol. For in-office procedures, soft tissue must be protected with a light-cured resin or petroleum jelly. For at-home treatments, participants use custom-fitted trays [56] [57].
- Irritation Measurement (Tooth Sensitivity):
- Real-time Sensitivity: Assess immediately after and at predetermined intervals post-bleaching using a Visual Analogue Scale (VAS) or Numerical Rating Scale (NRS). Participants mark their sensitivity level on a 10 cm line (VAS) or select a number from 0-10 (NRS) [56] [37].
- Spontaneous Sensitivity: Participants record any sensitivity experienced at home in a diary [57].
- Objective Pulpal Measurement: Use pulse oximetry to monitor pulpal oxygen saturation (SaO₂) before and after each bleaching session. Stable SaO₂ levels indicate minimal inflammatory response or oxidative stress within the pulp [57].
- Efficacy Measurement: Measure tooth color at baseline, post-treatment, and follow-up (e.g., 14 days, 6 months) using a spectrophotometer. Report color change in the perceptually uniform ΔE00 metric and whiteness index (ΔWID) [57] [37].
- Statistical Analysis: Perform appropriate statistical tests (e.g., ANOVA, Wilcoxon Signed Rank) to compare sensitivity and efficacy outcomes between different gel formulations and concentrations [56] [37].
In Vitro Skin Irritation Testing Using Reconstructed Human Epidermis (RHE)
The OECD TG 439 guideline provides a standardized method for assessing the potential of chemicals to cause skin irritation, replacing traditional animal testing [59] [60].
- Model Preparation: Use a validated Reconstructed Human Epidermis (RHE) model, such as EpiSkin, EpiDerm SIT, or the novel GB-RHE model. These are multilayered, differentiated tissues grown at the air-liquid interface [60].
- Test Substance Application: Apply the bleaching substance or formulation (e.g., 20 μL of liquid or 20 mg of solid) uniformly onto the surface of the RHE model. For nanoparticles (e.g., TiO₂), ensure suspensions are prepared appropriately [60].
- Exposure and Incubation: Incubate the treated tissues for a defined period (e.g., 60 minutes) at controlled conditions (e.g., 37°C, 5% CO₂) [60].
- Post-Treatment Viability Assessment:
- Carefully clean the tissue surface post-incubation.
- Assess tissue viability using the MTT assay. MTT is converted to a blue formazan product by viable cells. Extract the formazan and measure absorbance spectrophotometrically.
- Calculate cell viability as a percentage relative to the negative control.
- Irritancy Classification: According to OECD TG 439, a test substance is classified as an irritant if it reduces tissue viability below 50% of the negative control. Non-irritants maintain viability above this threshold [60].
- Histological and Ultrastructural Analysis: For a deeper understanding of tissue health and nanoparticle internalization, perform histological analysis (e.g., H&E staining) and transmission electron microscopy (TEM) on post-exposure tissues [60].
Zebrafish Embryo Bleaching Protocol for Pigment Removal
This protocol is optimized for depigmenting fixed zebrafish embryos to improve clarity for imaging or whole-mount in situ hybridization (WMISH) [58].
- Fixation: Fix embryos in 4% paraformaldehyde (PFA) in phosphate-buffered saline (PBS) overnight at 4°C.
- Bleaching Solution Preparation: Prepare a fresh bleaching solution for each use. A standard formulation is 3% hydrogen peroxide and 1% potassium hydroxide (KOH) in distilled water [58]. For example:
- 100 μL of 30% H₂O₂
- 100 μL of 10% KOH
- 800 μL dH₂O
- Total: 1 mL per tube of embryos.
- Bleaching Process:
- Remove the PFA and wash embryos 3 times for 5 minutes each with PBT (PBS with 0.1% Tween-20).
- Add 1 mL of bleaching solution to the embryos in an open tube to allow gas escape.
- Incubate at room temperature, monitoring closely. Typical durations are:
- 10-15 minutes for 36 hpf embryos.
- ~45 minutes for 5 dpf embryos.
- Critical Step: Monitor the reaction closely. The process is complete when the black pigment spots are no longer visible. Over-bleaching can degrade embryonic tissues.
- Post-Bleaching Wash: Once bleached, immediately remove the bleaching solution and rinse the embryos 2-3 times with PBT. The embryos may float initially but will sink after washes.
- Storage or Proceeding: Embryos can be stored in methanol at -20°C or proceed directly to WMISH or imaging.
Signaling Pathways and Experimental Workflows
Melanin Biosynthesis and Inhibition Pathways
The following diagram illustrates the core pathway of melanin synthesis within melanocytes and the points of inhibition by common agents, including bleaching chemicals and PTU.
Workflow for Bleaching Gel Irritation and Efficacy Assessment
This diagram outlines a comprehensive experimental workflow integrating clinical and in vitro methods to evaluate bleaching treatments.
The Scientist's Toolkit: Essential Reagents and Materials
Table 3: Key Research Reagents and Materials for Bleaching Studies
| Reagent / Material | Function / Application | Specific Examples & Notes |
|---|---|---|
| Hydrogen Peroxide (H₂O₂) | Primary active oxidizing agent in bleaching gels; also used in research model depigmentation. | In-office dental gels (35%), at-home gels (3-6%), zebrafish bleaching solution (3%) [56] [57] [58]. |
| Carbamide Peroxide (CP) | Stable complex that decomposes to urea and H₂O₂; provides a slower, sustained release of peroxide. | Common in at-home dental bleaching (e.g., 8%, 10%) [37]. |
| Phenylthiourea (PTU) | Tyrosinase inhibitor; used in research to prevent melanogenesis in vivo in model organisms. | Alternative to chemical bleaching in zebrafish; may have developmental side-effects [58]. |
| Reconstructed Human Epidermis (RHE) | In vitro 3D tissue model for assessing skin irritation and corrosion potential of chemicals and formulations. | EpiSkin, EpiDerm SIT, GB-RHE; used per OECD TG 439 [59] [60]. |
| Titanium Dioxide Nanoparticles (TiO₂ NPs) | Photocatalyst; enhances bleaching efficacy of low-concentration H₂O₂ when activated by light (e.g., violet LED). | NF_TiO₂ (N and F co-doped) allows activation by visible light, improving safety profile [57]. |
| Hyaluronic Acid (HA) | Biopolymer used as a thickening/gelling agent; offers biocompatibility and may help reduce tissue sensitivity. | Used in experimental low-concentration dental gels as an alternative to synthetic carbomers [57]. |
| Violet LED Light Source | Activates specific photocatalysts (e.g., NF_TiO₂) to accelerate H₂O₂ decomposition, enabling lower H₂O₂ concentrations. | Wavelength ~405-410 nm; used in advanced in-office dental bleaching systems [57]. |
| Potassium Hydroxide (KOH) | Alkaline agent used in depigmentation solutions for research models; enhances the bleaching action of H₂O₂. | Component of standard zebrafish embryo bleaching solution (with H₂O₂) [58]. |
| Spectrophotometer | Instrument for objective, quantitative measurement of color change (efficacy) in dental and material studies. | Reports metrics such as ΔE00 and ΔWID for high accuracy [56] [57] [37]. |
The pursuit of aesthetic dentistry has catalyzed significant innovation in tooth whitening technologies, with a pronounced shift towards enhancing efficacy while minimizing adverse effects. Traditional chemical bleaching, primarily using hydrogen peroxide (HP) or carbamide peroxide (CP), operates through an oxidative mechanism that can compromise enamel integrity and cause pulpal sensitivity [61]. In response, two groundbreaking approaches have emerged: the use of physical activation via Violet Light-Emitting Diodes (LED) and the incorporation of remineralizing components into bleaching formulations. This guide objectively compares the performance of these innovations against conventional alternatives and other emerging technologies, providing researchers and drug development professionals with synthesized experimental data and methodologies. The analysis is framed within the broader context of developing safer, more controlled depigmentation strategies that complement traditional chemical pathways.
Violet LED as an Activating Agent
Mechanism of Action and Performance Data
Violet LED technology (λ 405–410 nm) represents a paradigm shift, functioning through a physical interaction with dental pigments rather than solely acting as a catalyst for peroxide-based gels. Its mechanism involves emitting light energy that is absorbed by organic, pigmented macromolecules within the enamel, leading to their breakdown into smaller, less colored components [62] [63]. This process can whiten teeth without the mandatory use of peroxide gels, thereby avoiding the chemical aggression and oxidative damage associated with them.
Recent clinical and in-vitro studies provide robust data on its performance, both as a standalone treatment and in conjunction with peroxides. The following table summarizes key findings from comparative studies:
Table 1: Performance Data of Violet LED in Tooth Whitening
| Study Type | Groups / Protocol | Key Outcome Measure | Result | Reported Significance |
|---|---|---|---|---|
| Clinical Trial [62] | vLED (2x25' cycles, no gel) | Color Change (ΔE) over 12 months | Effective whitening observed, but ΔE lower than vLED/CP group | p < 0.0001 (difference between groups over time) |
| vLED/CP (37% Carbamide Peroxide) | Superior ΔE at all time points vs. vLED alone | |||
| In-Vitro Study [63] | Violet LED only (4 sessions) | Color Change (ΔE) | ΔE=15.37; similar whitening to peroxide-only groups | No significant difference vs. HP 35% or CP 16% groups (p > 0.05) |
| HP 35% (3 sessions, no light) | ΔE=21.73-22.89 | |||
| Clinical Trial [64] | 35% HP (15') + Violet LED | Color Change (ΔE00) | Whitening efficacy significantly enhanced by LED in this group | LED effect was protocol-dependent |
The data indicates that violet LED alone can produce clinically significant whitening, achieving ΔE values that surpass the perceptibility threshold (ΔE>3.3) [63]. However, its combination with high-concentration peroxides (e.g., 37% CP) yields the most potent bleaching effect over the long term [62]. A critical finding is that the light's synergistic effect is not universal but appears to be more pronounced in specific protocols, such as those with reduced application times [64].
Diagram 1: Mechanism of Violet LED Whitening
Detailed Experimental Protocol
To evaluate the efficacy of violet LED, researchers often employ controlled in-vitro designs. The following methodology, adapted from a published in-vitro study, provides a template for comparative testing [63]:
- Tooth Specimen Preparation: Seventy bovine incisors are cleaned and stained in a black tea solution for six days to create a standardized staining baseline.
- Group Allocation: Specimens are randomly divided into groups (n=10), including:
- Negative Control: Artificial saliva.
- Positive Controls: HP 35% or CP 16% applied without light activation.
- Test Groups: Violet LED alone; HP or CP gels photocatalyzed with violet LED.
- Bleaching Procedure:
- For the Violet LED-only group, specimens are irradiated for multiple sessions according to the device manufacturer's guidelines (e.g., four sessions).
- For combination groups, the peroxide gel is applied and immediately irradiated with the violet LED. A typical irradiation cycle is 2 minutes, repeated with 30-second intervals to prevent overheating.
- Color Measurement: Baseline (Lab*) and post-treatment color coordinates are measured using a calibrated digital spectrophotometer. The color change (ΔE) is calculated using the CIEDE2000 formula for enhanced accuracy.
Remineralizing Components in Whitening Formulations
Mechanism of Action and Performance Data
A major innovation to counter the enamel demineralization and microhardness reduction from peroxides is the incorporation of remineralizing fillers. These components, such as Strontium-containing Fluorapatite (Sr-FAp) and nano-Hydroxyapatite (n-HAp), function by forming a protective layer on the enamel surface. This layer acts as a physical barrier against oxidative damage and simultaneously releases calcium, phosphate, and fluoride ions that promote remineralization [61] [51].
Table 2: Performance Data of Remineralizing Components in Whitening
| Remineralizing Agent | Study Model | Bleaching Efficacy | Impact on Enamel | Key Statistical Finding |
|---|---|---|---|---|
| Strontium-Fluorapatite (Sr-FAp) [51] | In-Vitro (Bovine teeth, 30% HP) | ΔEab, ΔWID, ΔE00: No significant difference from HP alone | Microhardness: Highest in 10 wt% Sr-FAp group (p < 0.05). \newline Gloss: Decreased with 10% Sr-FAp. | Whitening efficacy preserved while microhardness was significantly improved. |
| Nano-Hydroxyapatite (n-HAp) & Polymers [61] | Literature Review | Effective whitening maintained. | Reduces mineral loss and peroxide diffusion. Improves enamel integrity. | A key strategy for mitigating adverse effects of HP. |
| Calcium Compounds & Fluoride [61] | Literature Review | Supports whitening as an adjunct. | Reduces demineralization and tooth sensitivity. Preserves enamel. | Valued as adjuncts for patients with high sensitivity risks. |
The data demonstrates that the primary benefit of remineralizing agents is their protective function. For instance, incorporating 10% Sr-FAp into a 30% HP solution significantly increased enamel microhardness post-bleaching compared to HP alone, without compromising the whitening efficacy (ΔE values were not significantly different) [51]. Polymer-based carriers like Carbopol and Hydroxypropyl Methylcellulose (HPMC) further enhance this safety profile by modulating the release and diffusion of active peroxide, potentially reducing its penetration into deeper dental tissues [61].
Diagram 2: Protective Mechanism of Remineralizing Agents
Detailed Experimental Protocol
A typical in-vitro protocol for testing the efficacy of remineralizing agents, as seen in a recent study, involves the following steps [51]:
- Synthesis of Remineralizing Agent: Sr-FAp can be synthesized via a hydrothermal method, where calcium and strontium nitrates are mixed with ammonium phosphate and fluoride, then subjected to heat and pressure. The resulting crystals are characterized using XRD and SEM.
- Specimen Preparation and Staining: Bovine incisors are embedded in resin, polished, and stained in a black tea solution for several days.
- Experimental Group Formation: Teeth are randomly assigned to groups (e.g., n=12):
- Negative Control: Distilled water.
- Positive Control: 30% HP solution.
- Test Groups: 30% HP incorporated with 1 wt%, 5 wt%, and 10 wt% Sr-FAp.
- Bleaching Procedure: The respective solutions are applied to the enamel surface for multiple cycles (e.g., three applications of 20 minutes each).
- Post-Treatment Analysis:
- Efficacy: Color change (ΔEab, ΔE00, ΔWID) is measured with a spectrophotometer.
- Safety: Enamel surface microhardness (Vickers or Knoop test), surface roughness, and gloss are quantified. The enamel microstructure is examined using Scanning Electron Microscopy (SEM).
Comparative Analysis with Alternative Technologies
Performance Against Other Whitening Systems
To contextualize the innovations of Violet LED and remineralizing agents, it is essential to compare them with other available and emerging technologies.
Table 3: Comparison of Whitening Technologies and Innovations
| Technology | Mechanism | Whitening Efficacy | Safety & enamel impact | Key Clinical Findings |
|---|---|---|---|---|
| Violet LED + HP/CP [62] [63] | Physical (light) + Chemical (oxidation) | High (Superior to vLED alone) | Mild sensitivity; requires gingival protection. | Most effective overall; standard for in-office power bleaching. |
| Violet LED Alone [62] [63] | Physical pigment breakdown | Moderate (Clinically significant) | Very low sensitivity; no gel-related risks. | Promising for sensitive patients; less variation in brightness (ΔL). |
| HP with Sr-FAp/ n-HAp [61] [51] | Chemical oxidation + remineralization | High (Preserved vs. HP alone) | Improved microhardness; reduced demineralization. | Effectively whitens while protecting enamel surface. |
| PAP+ [64] [65] | Peroxide-free oxidation | Comparable to conventional HP | Lower sensitivity; no enamel dehydration. | Effective, safe, less sensitive; major market growth. |
| Purple Toothpaste/Color Correctors [66] [65] | Optical color masking (cosmetic) | Temporary (lasts ~1 hour) | Safe for daily use; no active chemistry. | Does not alter intrinsic tooth color; temporary cosmetic fix. |
| Natural Abrasives (Charcoal) [67] [65] | Mechanical abrasion | Low (surface stains only) | High risk of enamel abrasion and surface roughness. | Limited long-term efficacy; potentially damaging. |
This comparison reveals a clear efficacy-safety spectrum. Violet LED with high-concentration peroxide sits at the high-efficacy end, ideal for maximum whitening impact. Peroxide-free systems like PAP+ and Violet LED alone occupy a middle ground, offering a favorable balance of efficacy and markedly improved comfort. Remineralizing agents are not standalone alternatives but are highly effective as adjuncts that shift conventional peroxide treatments toward the safer end of the spectrum. In contrast, purple color correctors and abrasive natural agents offer minimal, often temporary or potentially harmful, effects.
The Scientist's Toolkit: Essential Research Reagents and Materials
For researchers aiming to replicate or build upon these studies, the following table catalogues key materials and their functions as derived from the cited experimental protocols.
Table 4: Essential Research Reagents and Materials for Whitening Studies
| Reagent / Material | Function in Research | Example Use Case |
|---|---|---|
| Violet LED Device (400-410 nm) | To provide light energy for physical pigment breakdown or photocatalysis of peroxide. | Standalone whitening [63] or activation of 37% carbamide peroxide [62]. |
| Hydrogen Peroxide (30-37%) | The standard oxidative bleaching agent for establishing positive control groups. | Conventional in-office bleaching control [63] [51]. |
| Carbamide Peroxide (37%) | High-concentration bleaching agent that decomposes into hydrogen peroxide and urea. | Control group for in-office power bleaching [62]. |
| Strontium-containing Fluorapatite (Sr-FAp) | Remineralizing filler to be incorporated into HP gels to protect enamel. | Test groups evaluating enamel microhardness and whitening efficacy [51]. |
| Bovine Incisors | Standardized tooth substrate for in-vitro studies due to similarity to human enamel. | Staining and testing efficacy across multiple experimental groups [63] [51]. |
| Digital Spectrophotometer | To objectively measure color coordinates (Lab*) and calculate ΔE/ΔE00. | Primary outcome measure for all efficacy studies [62] [63] [51]. |
| Microhardness Tester | To quantify changes in enamel surface microhardness (Vickers/Knoop). | Key safety endpoint to assess enamel damage or protection [51]. |
| Scanning Electron Microscope (SEM) | To visualize and analyze surface morphology and the presence of protective layers. | Observing Sr-FAp nanoparticles on enamel [51]. |
| Polymer Carriers (e.g., Carbopol, HPMC) | To modify gel viscosity, adhesion, and control release of active ingredients. | Formulating bleaching gels to improve handling and potentially reduce cytotoxicity [61]. |
| Visual Analog Scale (VAS) | To subjectively assess patient-reported dental sensitivity. | Clinical trial outcome for comparing sensitivity between protocols [62]. |
Efficacy and Safety Validation: Clinical and Laboratory Performance Metrics
This comparison guide provides a systematic analysis of pigment removal efficacy between 1-phenyl-2-thiourea (PTU) treatment and chemical bleaching agents, focusing on standardized color measurement metrics (ΔSGU, ΔE00, WID), underlying mechanisms, and experimental protocols. While PTU remains widely used in zebrafish research for melanin inhibition, quantitative clinical data demonstrates that peroxide-based bleaching technologies achieve significantly superior shade change outcomes in dental applications. Comprehensive tables present direct comparison of efficacy data, tooth sensitivity outcomes, and longevity measures across randomized controlled trials. The analysis reveals that PTU's pigment inhibition extends beyond melanogenesis to include unintended biological effects, whereas chemical bleaching agents provide more predictable and controlled depigmentation through targeted oxidative pathways.
Pigment removal strategies in biological research and clinical applications operate through distinct mechanistic pathways. 1-phenyl-2-thiourea (PTU) functions primarily as a tyrosinase inhibitor that blocks melanogenesis by chelating copper ions at the enzyme's active site, thereby preventing the conversion of tyrosine to melanin [7] [68]. This mechanism makes PTU particularly valuable in zebrafish embryo research where optical clarity is essential for microscopic imaging. In contrast, chemical bleaching agents employ oxidative chemistry to degrade pigment structures. Peroxide-based whiteners (hydrogen peroxide and carbamide peroxide) decompose into reactive oxygen species that diffuse through substrate matrices and break chromophore bonds through oxidation, resulting in color lightening [69] [70]. Enzyme-based alternatives like papain and bromelain utilize proteolytic activity to dismantle protein-based stain structures [71].
The research community requires clear efficacy comparisons between these approaches to inform experimental design and clinical applications. This analysis directly compares these technologies using standardized color measurement metrics and provides detailed experimental protocols for reproducibility.
Quantitative Efficacy Comparison
Color Change Metrics Across Technologies
Table 1: Comparative Efficacy of Pigment Removal Technologies
| Technology | ΔE00 (Mean) | ΔSGU (Mean) | ΔWID | Treatment Duration | Study Reference |
|---|---|---|---|---|---|
| PTU (0.003%) | Not reported | Not reported | Not reported | 24 hpf onward | [7] [68] |
| 10% Carbamide Peroxide | 9.24 | 6.27 | Significant increase | 14 days | [69] [70] |
| 35% Hydrogen Peroxide | 7.89 | 5.62 | Significant increase | Single session | [69] |
| Activated Charcoal | 1.42 (vs. RT) | Minimal change | Minimal change | 14 days | [70] |
| Enzyme-based Gels | Not quantified | Effective stain removal | Not reported | 10 days | [71] |
Side Effects and Longevity Measures
Table 2: Adverse Effects and Longevity Profiles
| Technology | Tooth Sensitivity Incidence | Other Adverse Effects | Longevity Assessment | Study Reference |
|---|---|---|---|---|
| PTU (0.003%) | Not applicable | Reduced zebrafish eye size, autophagy activation, altered thyroid function | Duration-dependent effects | [7] [68] |
| 10% Carbamide Peroxide | Higher prevalence (7-14 days) | Mild transient sensitivity | Maintained up to 2 years | [69] [70] |
| 35% Hydrogen Peroxide | Significant on first day | Higher initial sensitivity | Similar long-term results | [69] |
| Activated Charcoal | Similar to regular toothpaste | Low acceptability, difficulty of use | Short-term only | [70] |
| Enzyme-based Gels | None reported | No cytotoxicity observed | Not assessed | [71] |
Clinical trials demonstrate that 10% carbamide peroxide produces significantly greater color improvement (Δb* p=0.003, Δa* p=0.014) compared to 35% hydrogen peroxide, with ΔE00 values (>9.0) indicating excellent effectiveness according to established classification systems [69] [70]. Both peroxide approaches substantially outperform activated charcoal-based products, which show ΔE00 values of only 1.42 - classifying them as marginally effective despite significant marketing claims [70].
For PTU treatments, efficacy assessment differs substantially as it is measured through biological endpoints rather than color space parameters. The standard 0.003% PTU treatment effectively inhibits melanization in zebrafish embryos but induces significant morphological alterations including reduced eye size beginning at 3 days post-fertilization [7]. This effect correlates with thyroid peroxidase inhibition rather than general thyroid hormone suppression, indicating a specific mechanistic pathway distinct from pigment inhibition [7].
Experimental Protocols and Methodologies
PTU Treatment in Zebrafish Models
Embryo Preparation and Treatment:
- Collect zebrafish embryos and raise in standard E3 embryo medium until approximately 24 hours post-fertilization (hpf)
- Prepare 0.2 mM (0.003% w/v) PTU solution in embryo medium
- Treat embryos from 24 hpf onward, replacing PTU solution daily
- Maintain control group in PTU-free medium under identical conditions
- Continue treatment through desired developmental stages (typically 3-5 days post-fertilization) [7] [68]
Assessment Endpoints:
- Melanization Inhibition: Visual inspection of pigment reduction compared to controls
- Morphometric Analysis: Measure eye size (anterior-posterior length and area) using digital imaging software
- Thyroid Function: Assess thyroxine levels via immunoassays or transgenic reporters
- Molecular Analysis: Evaluate autophagy markers (LC3-I, LC3-II, Sqstm1) via western blotting [7] [68]
Validation Controls:
- Include genetic melanization mutants (tyr, nacre, casper) as alternative pigment controls
- Test thyroid hormone supplementation for rescue experiments
- Compare with other goitrogens to isolate mechanism [7]
Chemical Bleaching Clinical Protocols
In-Office Power Bleaching (35% Hydrogen Peroxide):
- Pre-treatment: Apply 2% potassium nitrate desensitizing gel for 10 minutes
- Application: Apply 35% hydrogen peroxide gel (Whiteness HP Blue, FGM) to tooth surfaces
- Activation: No light activation required for chemically-activated formulas
- Treatment Duration: Two applications of 15-20 minutes each per session
- Post-treatment: Remove gel thoroughly and advise on postoperative sensitivity management [69]
At-Home Bleaching (10% Carbamide Peroxide):
- Custom Tray Fabrication: Create vinyl mouthguards from dental impressions
- Gel Application: Dispense 10% carbamide peroxide gel into tray reservoirs
- Wear Protocol: Daily application for 2-8 hours depending on sensitivity
- Treatment Duration: 14-day continuous use with daily assessments
- Color Measurement: Baseline, 7-day, 14-day, and 2-week post-treatment assessments [69] [70]
Color Measurement Methodology:
- Objective Measurement: Use pre-calibrated spectrophotometers (Vita Easyshade) with custom positioning trays for consistency
- Measurement Parameters: Record L* (lightness), a* (red-green), b* (yellow-blue) values
- Calculation: Compute ΔE00 using CIEDE2000 formula: ΔE00 = [(ΔL′/KₗSₗ)² + (ΔC′/KᶜSᶜ)² + (ΔH′/KₕSₕ)² + Rₜ(ΔC′/KᶜSᶜ)(ΔH′/KₕSₕ)²]¹/²
- Subjective Measurement: Use VITA classical shade guide units (SGU) with standardized lighting conditions [69] [70]
Mechanism and Pathway Analysis
Diagram Title: Pigment Removal Mechanisms Comparison
The mechanistic pathways reveal fundamental differences between PTU and chemical bleaching approaches. PTU's effects extend beyond its intended tyrosinase inhibition to include thyroid peroxidase (TPO) inhibition, which reduces thyroid hormone production and correlates with specific developmental abnormalities in zebrafish models [7]. Additionally, PTU treatment activates autophagic pathways, characterized by increased LC3-I, LC3-II, and autolysosome formation across multiple tissues [68]. These unintended effects complicate the interpretation of research results when PTU is used solely for pigment inhibition.
Chemical bleaching agents operate through more direct oxidative pathways. Hydrogen peroxide and carbamide peroxide decompose into reactive oxygen species that diffuse through enamel and dentin, breaking down organic chromophores through oxidation without the systemic effects associated with PTU [69] [70]. Enzyme-based approaches provide an alternative mechanism through targeted proteolysis of stain components without oxidative chemistry [71].
Research Reagent Solutions
Table 3: Essential Research Materials for Pigment Removal Studies
| Reagent/Equipment | Specifications | Research Application | Key Considerations |
|---|---|---|---|
| 1-phenyl-2-thiourea (PTU) | 0.2 mM (0.003% w/v) in embryo medium | Zebrafish pigment inhibition for imaging | Causes reduced eye size, autophagy activation; use tyr mutants as alternative [7] [68] |
| Carbamide Peroxide | 10% in custom trays | At-home dental whitening clinical trials | Gold standard for efficacy comparison; causes transient sensitivity [69] [70] |
| Hydrogen Peroxide | 35% for in-office use | Professional dental whitening studies | Higher initial sensitivity; comparable long-term results [69] |
| Spectrophotometer | Vita Easyshade or equivalent | Objective color measurement (ΔE00) | Requires custom positioning trays for measurement consistency [69] [70] |
| VITA Shade Guide | Classical A1-D4 units | Subjective color assessment (ΔSGU) | Standardized lighting conditions essential [69] |
| Activated Charcoal | Powder or toothpaste formulations | OTC product efficacy assessment | Minimal efficacy per clinical evidence; low user satisfaction [70] |
| Enzyme Preparations | Papain, bromelain (6-8% microcapsules) | Alternative stain removal systems | Effective for specific stain types; minimal adverse effects [71] |
The comparative analysis reveals a significant efficacy-efficacy gap between PTU-mediated biological pigment inhibition and chemical bleaching technologies. While PTU remains valuable for specific research applications where optical clarity is paramount, its unintended biological effects necessitate careful experimental design and appropriate controls. Chemical bleaching technologies, particularly peroxide-based systems, demonstrate superior and more predictable performance in clinical shade change parameters when measured using standardized metrics (ΔE00, ΔSGU, WID).
For research applications requiring pigment inhibition, genetic alternatives like tyr mutants may provide preferable options to PTU by eliminating chemical treatment variables [7] [68]. In clinical tooth whitening, peroxide-based systems remain the efficacy benchmark against which emerging technologies should be compared using the rigorous methodological frameworks and standardized metrics outlined in this analysis.
Laboratory Assessment of Enamel Microhardness and Pulpal Response (SaO₂)
The pursuit of aesthetic dentistry has led to the development of numerous treatments for pigment removal, each with distinct effects on dental hard tissues and the pulp. This guide provides a comparative analysis of two predominant approaches: chemical bleaching using hydrogen peroxide (HP) or carbamide peroxide (CP) gels, and an emerging alternative, poly(thiourea) (PTU)-based treatment, which leverages a "de-conjugated" hydrogen bond mechanism for a "bleaching" effect on material transparency rather than direct tooth whitening. For researchers and drug development professionals, understanding the laboratory methodologies for quantifying treatment efficacy and safety is paramount. Key metrics include enamel microhardness, which reflects the structural integrity of the enamel surface, and pulp oxygen saturation (SaO₂), a direct indicator of pulpal vascular health and vitality. This guide objectively compares the performance of these treatments based on experimental data, detailing the protocols essential for rigorous laboratory assessment.
Comparative Performance Data
The following tables synthesize quantitative data from recent in vitro studies, comparing the effects of various treatments on enamel properties and pulpal safety parameters.
Table 1: Impact of Treatments on Enamel Microhardness and Surface Properties
| Treatment Modality | Specific Agent/Protocol | Enamel Microhardness Change (Vickers HV) | Surface Roughness & Morphology | Key Experimental Findings |
|---|---|---|---|---|
| Chemical Bleaching (High-Concentration HP) | Opalescence Boost (40% HP) | Significant reduction: 37.21 ± 1.74 Hv (control) to 34.63 ± 1.70 Hv (treated); p = 0.00 [72] | N/A | FTIR spectroscopy revealed notable alterations in phosphate and carbonate bands [72]. |
| Opalescence Quick (45% CP) | Significant reduction: 45.82 ± 1.71 Hv (control) to 39.34 ± 1.94 Hv (treated); p < 0.0001 [72] | N/A | FTIR showed chemical alterations. Carbamide peroxide-based product also reduced hardness [72]. | |
| 35% HP (Bovine Model) | Significant decrease post-bleaching; recovery after 1-month saliva immersion [73] | Increased surface roughness post-bleaching, decreasing over 1-month saliva immersion [73] | Gloss and microhardness increased with immersion time in artificial saliva, indicating natural remineralization can reverse some damage [73]. | |
| Chemical Bleaching (Low-Concentration HP + Additives) | 1.5% HP with Hyaluronic Acid & NF_TiO₂ | N/A | N/A | Achieved similar color change (ΔE00) to 35% HP commercial gel, with significantly higher cell viability and lower H₂O₂ diffusion [74]. |
| Laser Treatment (Preventive) | 445 nm Diode Laser | No significant reduction post-treatment [75] | Smoother surface morphology and reduced acid-induced erosion compared to controls [75] | XRD analysis confirmed formation of a more acid-resistant pyrophosphate phase and lattice compaction [75]. |
| PTU-Based Material (Optical Application) | Poly(p-xylylene thiourea) (pX-PTU) | Not Applicable (Polymer property assessment) | Not Applicable (Polymer property assessment) | Exhibits high transparency (%T ≧ 99) and high refractive index (nD = 1.71) for optoelectronic applications, not direct tooth whitening [2]. |
Table 2: Pulpal Response and Safety Assessment Metrics
| Assessment Parameter | Treatment / Intervention | Key Findings & Safety Data | Clinical Implication |
|---|---|---|---|
| Pulp Oxygen Saturation (SaO₂) | Normal Vital Pulp (Baseline) | Mean SpO₂ in vital maxillary anterior teeth: 84.94% - 89.29% [76] | Provides a reference range for assessing pulp vitality; a significant deviation may indicate compromised blood flow. |
| Pulp Chamber Temperature | 445 nm Diode Laser (1.25W, pulsed) | Minimal temperature elevation: 0.05667 ± 0.04131 °C [75] | The protocol is considered thermally safe for the pulp, a critical consideration for any energy-based treatment. |
| Cytotoxicity & H₂O₂ Diffusion | 35% HP (Commercial Gel) | N/A | High H₂O₂ diffusion linked to low cell viability and high oxidative stress in pulp cell models [74]. |
| 6% HP with Hyaluronic Acid & NF_TiO₂ | N/A | Significantly reduced H₂O₂ diffusion and oxidative stress, leading to higher cell viability compared to 35% HP [74]. |
Detailed Experimental Protocols
To ensure reproducibility and standardization in research, the following section elaborates on key laboratory methodologies cited in the performance data.
Vickers Microhardness Testing
This is a primary method for evaluating the mechanical property of enamel post-treatment [72] [73] [50].
- Principle: A diamond indenter in the shape of a right pyramid with a square base is pressed into the enamel surface under a predefined load. The size of the resulting permanent impression is measured and used to calculate the Vickers hardness number (Hv).
- Detailed Protocol:
- Sample Preparation: Extracted human or bovine teeth are sectioned and embedded in acrylic resin. The enamel surface is polished sequentially with silicon carbide papers (e.g., 600, 800, and 1200 grit) under running water to create a flat, standardized surface [72] [73].
- Treatment Application: The bleaching or other treatment agents are applied according to manufacturer instructions or experimental design.
- Indentation: A microhardness tester (e.g., DMH-2) is used with a standard load (typically 200 g for enamel) and a dwell time of 10 seconds [73] [77].
- Measurement: The two diagonals of the square impression are measured using a microscope attached to the tester. The average diagonal length is used to calculate the Vickers hardness number.
- Replication: Multiple indentations (e.g., 5 per specimen) are made on different areas of the treated and control surfaces to ensure reliability [73].
Fourier Transform Infrared (FTIR) Spectroscopy
This technique is used to detect chemical alterations in the enamel structure, such as changes in organic matrix or mineral content [72].
- Principle: It measures the absorption of infrared light by a material, identifying specific molecular bonds and functional groups based on their characteristic vibrational frequencies.
- Application in Enamel Analysis: FTIR can monitor changes in the phosphate (∼960-1100 cm⁻¹) and carbonate (∼1450 cm⁻¹, ∼870 cm⁻¹) bands of hydroxyapatite. A study on bleached enamel showed significant alterations in these bands, indicating demineralization or chemical changes to the enamel crystal structure [72].
In Vitro Pulp Chamber Temperature Assessment
This protocol evaluates the thermal safety of energy-based treatments like lasers [75].
- Principle: A thermocouple probe is placed within the pulp chamber of an extracted tooth to measure temperature changes during the procedure.
- Detailed Protocol:
- Tooth Preparation: Extracted mono-rooted teeth with intact root structures are selected. The apical portion is removed, and root canals are instrumented.
- Probe Placement: A fast-response K-type thermocouple is positioned at the ceiling of the pulp chamber. A thermoconductive paste is used to ensure optimal thermal coupling.
- Simulation: The root is immersed in a 37°C water bath to simulate body temperature, with the cervical portion above the waterline.
- Irradiation & Recording: The laser is applied to the external enamel surface using predefined parameters, and the temperature change inside the pulp chamber is recorded in real-time [75].
Cell Viability and Oxidative Stress Assays
These in vitro tests are crucial for pre-clinical safety assessment of bleaching agents [74].
- Model System: Bovine enamel/dentin discs are placed in artificial pulp chambers (APCs). The treatment gel is applied to the enamel surface.
- Cytotoxicity Assessment: The culture medium (extract) that has diffused through the dentin is collected and applied to odontoblast-like cells (e.g., MDPC-23). Cell viability (CV) is then assessed using standard assays like MTT or Alamar Blue.
- Oxidative Stress (OxS) Measurement: The same extracts can be used to measure markers of oxidative stress in the cell cultures, indicating the level of reactive oxygen species that penetrated the dentin.
Research Reagent Solutions
The table below lists key materials and their functions as used in the cited studies, serving as a reference for setting up similar experiments.
Table 3: Essential Research Reagents and Materials
| Reagent / Material | Function in Research | Example from Literature |
|---|---|---|
| Hydrogen Peroxide (HP) / Carbamide Peroxide (CP) | Active bleaching agent in chemical whitening studies. Concentration and pH are key variables. | Opalescence Boost (40% HP), Opalescence Quick (45% CP) [72]. |
| Artificial Saliva | Simulates the oral environment for in vitro studies, allowing for remineralization and providing a more clinically relevant medium. | Used as a storage medium to observe recovery of microhardness and surface gloss over time [73] [77]. |
| Vickers Microhardness Tester | Instrument to measure the resistance of enamel to plastic deformation, quantifying treatment-induced softening or hardening. | Used with a 200g load to compare bleached and unbleached enamel [72] [73]. |
| FTIR Spectrometer | Analyzes chemical and structural changes in the enamel's inorganic and organic components post-treatment. | Used to detect alterations in phosphate and carbonate bands of hydroxyapatite after bleaching [72]. |
| Pulse Oximeter (Dental) | Measures oxygen saturation (SpO₂) in the dental pulp vasculature, providing a objective assessment of pulp vitality. | Used to establish reference SpO₂ values (84.94%-89.29%) in vital maxillary anterior teeth [76]. |
| Hyaluronic Acid (HA) & NF_TiO₂ | Components of experimental low-concentration bleaching gels. HA may modulate gel viscosity and biocompatibility, while NF_TiO₂ nanoparticles may act as catalysts. | Used in gels with 1.5% and 6% HP to achieve efficacy with reduced cytotoxicity [74]. |
| Diode Laser (445 nm) | Energy source used to pre-treat enamel, enhancing its resistance to acid demineralization through surface modification. | Applied with specific parameters (1.25 W, 800 J/cm²) to safely enhance acid resistance [75]. |
Experimental Workflow and Signaling Pathways
The following diagrams, generated using Graphviz, illustrate the logical workflow for laboratory assessment and the conceptual mechanism of a novel PTU-based material.
Enamel Microhardness and Pulpal Response Assessment Workflow
This diagram outlines the core experimental pathway for evaluating dental treatments.
Mechanism of "De-conjugated" H-bonds in PTU vs Aromatic PTU
This diagram contrasts the molecular design of traditional aromatic PTUs with the novel pX-PTU, explaining its enhanced optical properties.
Within the broader thesis of PTU treatment versus chemical bleaching, the data reveals a clear dichotomy. Chemical bleaching, particularly with high-concentration HP (35-45%), demonstrates significant efficacy but at a cost to enamel integrity (reduced microhardness, chemical alterations) and pulpal safety (cytotoxicity from H₂O₂ diffusion) [72] [50] [74]. The trajectory of research is aimed at mitigating these drawbacks, as seen with the development of low-concentration HP gels augmented with hyaluronic acid and NF_TiO₂, which show promising efficacy with markedly reduced side effects [74].
Conversely, the PTU-based approach described in the literature represents a fundamentally different principle. It is not a direct tooth whitener but a polymer engineered for high refractive index and transparency in optoelectronics through a "de-conjugated" hydrogen bond design [2]. Its relevance to dental research lies in its novel material science concept—achieving a desired optical property ("bleaching" meaning whitening of the material itself, not teeth) by strategically inhibiting damaging molecular interactions. This presents a conceptual model for future biomaterial development: can a similar "de-conjugating" or protective strategy be designed into a topical agent that prevents pigment formation or removes stains without aggressively interacting with and damaging the enamel crystal structure?
In conclusion, the laboratory assessment of enamel microhardness and pulpal response remains critical for advancing aesthetic dentistry. While current chemical bleaching evolves towards safer formulations, emerging concepts from material science, like the design principles of PTUs, offer inspiring alternative pathways for future research into non-invasive pigment removal strategies. The choice for researchers and drug developers is not merely between existing products, but between evolving a conventional technology and pioneering a new one based on novel biomimetic or bio-protective mechanisms.
Within the scope of pigment removal research, two distinct approaches emerge: direct chemical bleaching and the modulation of biological pigment synthesis. This guide provides a direct efficacy comparison between these strategies, focusing on the bleaching power of chemical agents against the optical performance, characterized by transparency and Abbe number, of restorative materials. The context is framed by a broader thesis investigating 1-phenyl-2-thiourea (PTU) as a tyrosinase inhibitor for inducing depigmentation, in contrast to conventional chemical bleaching methods. Chemical bleaching, utilizing agents like hydrogen peroxide, operates by oxidizing and breaking down chromogenic compounds, directly lightening the substrate. Conversely, in restorative dentistry, the success of a treatment is not solely dependent on color matching but also on the optical characteristics of the materials used, such as their transparency and their Abbe value, which quantifies chromatic aberration. This article objectively compares the performance of these different approaches and materials by synthesizing current experimental data, providing researchers and drug development professionals with clear, quantitative comparisons and standardized experimental protocols.
Bleaching Power: Mechanisms and Efficacy Comparison
Chemical bleaching agents function primarily through oxidation. Hydrogen peroxide (HP) and carbamide peroxide (CP) are potent oxidizing agents that degrade large, colored chromogen molecules into smaller, lighter compounds by releasing oxygen free radicals. Sodium perborate (SP) acts as a slower-releasing bleaching agent, decomposing in water to yield hydrogen peroxide and sodium metaborate [78] [79].
Quantitative Efficacy of Bleaching Agents
A recent comparative study evaluated the efficacy of three bleaching agents on blood-stained, devitalized teeth over time, measuring color change using ΔE00 and Whiteness Index (ΔWID) [78]. The findings are summarized in the table below.
Table 1: Comparative Efficacy of Bleaching Agents on Devitalized Teeth
| Bleaching Agent | Concentration | Key Findings on Efficacy | Noted Advantages/Disadvantages |
|---|---|---|---|
| Carbamide Peroxide (CP) | 37% | Most significant whitening effect during a 6-day process [78]. | Considered the most effective agent in the study [78]. |
| Hydrogen Peroxide (HP) | 35% | Effective whitening, second to CP in the study [78]. | Potential for demineralization and increased surface roughness; corrosive to soft tissues [78] [35]. |
| Sodium Perborate (SP) | N/S | Less effective and more time-consuming than HP or CP [78]. | Lower incidence of adverse effects due to higher pH; often used in walking bleach technique [78]. |
The underlying mechanism involves the release of free radicals, which, while effective for bleaching, can have unintended consequences. Studies note that these radicals can interfere with the bonding process of dental resins and may damage dental tissues, leading to increased surface roughness and mineral loss [78] [35]. Furthermore, teeth bleached with these agents demonstrate a heightened propensity for recoloration compared to unbleached teeth [78].
Experimental Protocol: Evaluating Bleaching Efficacy
The following methodology, adapted from a 2025 study, outlines a standard protocol for evaluating bleaching efficacy in vitro [78]:
- Sample Preparation: Caries-free, unfilled human incisors are selected. Endodontic access cavities are created, and root canals are shaped, disinfected, and filled. The apical area is sealed with glass ionomer cement.
- Staining Protocol: A blood-staining protocol is employed. Teeth are immersed in a plasma-removed erythrocyte suspension and subjected to centrifugation cycles until they reach a color of A3 or darker on the VITA scale.
- Group Allocation and Bleaching: Stained samples are stratified and randomly allocated into groups (e.g., control, HP-treated, CP-treated, SP-treated). Bleaching gels are applied according to manufacturer instructions.
- Color Measurement: Color measurements are taken using a dental spectrophotometer at baseline and post-bleaching. Color differences are calculated using the formulas for ΔE00 and ΔWID to quantify the whitening effect.
Transparency and Abbe Number in Material Science
In restorative and optical materials, achieving the right appearance involves more than just color matching; it requires controlling specific optical properties.
- Transparency is the physical property of a material that allows light to pass through without being scattered. In materials like dental ceramics and transparent polymers, high transparency is often desirable. However, transparency can be compromised by scattering sites such as residual pores, grain boundaries, second phases, and surface roughness [80]. For a material to achieve high transparency, it must have a high density (>99.9%), minimal pore volume, no second phases at grain boundaries, and a smooth surface finish [80].
- Abbe Value (or Abbe number) is a dimensionless number that measures the dispersion of light as it passes through a transparent material. Dispersion is the phenomenon where different wavelengths (colors) of light are bent by different amounts, leading to chromatic aberration, which manifests as colored fringes around objects [81]. A higher Abbe value indicates lower dispersion and better optical clarity. The Abbe value is crucial in lens design, as a low Abbe value can cause visual distortions and color fringing, especially in higher-power prescriptions [81].
Table 2: Optical Properties of Common Lens Materials
| Lens Material | Index of Refraction | Abbe Value | Key Characteristics |
|---|---|---|---|
| CR-39 | 1.498 | 58 | Highest Abbe value, excellent optics, least expensive [81]. |
| Trivex | 1.53 | 45 | Good optical clarity, impact-resistant [81]. |
| Polycarbonate | 1.586 | 30 | Low Abbe value, high impact resistance [81]. |
| 1.74 High-Index | 1.74 | 33 | Thinner lenses, lower Abbe value than CR-39 [81]. |
There is a general correlation in optical materials where a higher index of refraction (allowing for thinner lenses) often comes with a lower Abbe value, meaning increased chromatic aberration [81]. This creates a trade-off that must be managed based on the application's requirements.
PTU-Induced Depigmentation: An Alternative Pathway
In contrast to direct chemical bleaching, an alternative approach for managing pigmentation is to target the biological synthesis of melanin. The compound 1-phenyl-2-thiourea (PTU) is a well-known tyrosinase inhibitor used in research to create depigmentation models.
Mechanism of PTU
PTU inhibits tyrosinase, the key and rate-limiting enzyme in the melanin synthesis pathway [82] [30]. By blocking tyrosinase activity, PTU effectively prevents the formation of melanin pigments, leading to depigmentation. This model is widely used in zebrafish and cell cultures to study conditions like vitiligo and to screen potential therapeutic compounds [82].
Galangin as a Counter-Mechanism to PTU
Research into treatments for depigmentation disorders has investigated compounds that can reverse the effects of PTU. Galangin (GA), a flavonoid isolated from Alpinia officinarum, has been shown to ameliorate PTU-induced vitiligo in zebrafish and B16F10 melanoma cells [82] [30]. The mechanism involves the activation of the mitogen-activated protein kinase (MAPK) signaling pathway, specifically p38 and JNK. This activation leads to an upregulation of the microphthalmia-associated transcription factor (MITF), which in turn increases the expression of tyrosinase (TYR) and other melanogenic enzymes (TRP1, TRP2), ultimately restoring melanin production [82] [30]. Additionally, GA demonstrates antioxidant activity by reducing reactive oxygen species (ROS) and malondialdehyde (MDA) levels while increasing glutathione (GSH), catalase (CAT), and total superoxide dismutase (T-SOD) [82].
Figure 1: PTU Inhibition and Galangin's Mechanism for Ameliorating Vitiligo.
Experimental Protocol: Zebrafish Depigmentation Model
The following workflow details the standard method for establishing a PTU-induced depigmentation model and evaluating potential treatments like galangin [82] [30]:
- Zebrafish Maintenance: Wild-type AB zebrafish are maintained at 28°C under a 14/10-hour light/dark cycle. Embryos are collected and raised in zebrafish medium.
- PTU Modeling: At 24 hours post-fertilization (hpf), zebrafish embryos are treated with PTU (e.g., 200 μM) for 24 hours to induce depigmentation.
- Administration of Test Compound: After successful modeling, the PTU solution is replaced with fish medium containing different concentrations of the test compound (e.g., Galangin at 1 and 2 μM) and a positive control (e.g., 8-MOP) for 48 hours. A blank control and model group are maintained.
- Analysis:
- Melanin Content: Melanin is extracted from zebrafish larvae, and content is measured spectrophotometrically.
- Tyrosinase Activity: Larval protein lysates are analyzed using a tyrosinase activity kit.
- Transcriptomic Analysis & Western Blot: RNA sequencing and molecular docking identify potential pathways and targets. Protein expression of TYR, MITF, TRP1, TRP2, and MAPK pathway components is detected via Western blot.
Figure 2: Experimental Workflow for Zebrafish Depigmentation Model.
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Reagents for Pigmentation and Bleaching Research
| Reagent/Material | Function/Application in Research |
|---|---|
| 1-Phenyl-2-thiourea (PTU) | A tyrosinase inhibitor used to establish chemical-induced depigmentation models in zebrafish and cell cultures [82] [30]. |
| Galangin (GA) | A natural flavonoid studied for its ability to counteract depigmentation by activating the MAPK pathway and upregulating melanogenesis [82] [30]. |
| Hydrogen Peroxide (HP) | A strong oxidizing agent and the active component in many bleaching gels. Used to study direct chemical bleaching efficacy and its side effects on tissues and materials [78] [35]. |
| Carbamide Peroxide (CP) | A common bleaching agent that decomposes into hydrogen peroxide and urea. Used in comparative studies of bleaching speed and overall effect [78]. |
| Sodium Perborate (SP) | A bleaching agent often used in "walking bleach" techniques for endodontically treated teeth. Serves as a slower-release alternative to HP [78] [79]. |
| Universal Resin Composites | Dental restorative materials (e.g., Filtek Universal, Omnichroma) used to evaluate the effects of bleaching agents on color stability (ΔE), translucency (TP), and surface gloss [83]. |
| VITA Easyshade Spectrophotometer | An instrument used for objective, quantitative measurement of tooth and material color, essential for calculating ΔE00 and Whiteness Index (WID) [78] [35]. |
This comparison guide delineates the distinct efficacies and applications of chemical bleaching versus biologically-targeted depigmentation. Chemical bleaching with HP, CP, and SP offers rapid and potent direct oxidation of chromogens but can compromise material surfaces and lead to susceptibility to recoloration. In the realm of material science, properties like transparency and Abbe value are critical performance metrics that are influenced by material composition and processing, independent of bleaching power. Conversely, the PTU model provides a controlled research platform for studying depigmentation pathophysiology at a biological level, with compounds like galangin demonstrating the potential to reverse this process by activating intrinsic melanogenic pathways. The choice between these strategies is fundamentally dictated by the research or clinical objective: direct cosmetic alteration versus the investigation and modulation of the underlying biological mechanisms of pigmentation.
The pursuit of an improved dental appearance through tooth whitening is a significant aspect of cosmetic dentistry, driven by the strong association between a white smile and perceptions of health, attractiveness, and confidence [84]. The efficacy and safety of these treatments are paramount, with patient-centered outcomes—specifically, satisfaction, sensitivity rates, and compliance—serving as critical metrics for evaluating success. This guide provides an objective comparison of the performance of various professional tooth whitening systems, focusing on these key outcomes and the experimental data that underpin their clinical profiles. The analysis is framed within the broader research context of depigmentation strategies, offering a vital comparison for chemical bleaching against which emerging treatments, such as potential PTU-based therapies, can be evaluated.
Comparative Analysis of Whitening Modalities
Tooth whitening procedures primarily utilize peroxides, either hydrogen peroxide (HP) or carbamide peroxide (CP), which function by releasing oxygen molecules that penetrate enamel and dentin to oxidize and break down complex organic pigment molecules (chromophores) responsible for discoloration [84]. The concentration of the active agent, the application protocol (in-office versus at-home), and the physicochemical properties of the gel (e.g., pH) are key variables influencing both efficacy and adverse effects [56] [85].
Table 1: Key Patient-Centered Outcomes Across Bleaching Modalities
| Bleaching Modality | Typical Agent & Concentration | Reported Sensitivity Incidence | Key Satisfaction & Efficacy Findings | Compliance & Stability Notes |
|---|---|---|---|---|
| In-Office HP (High Conc.) | 35-40% HP [86] | Up to 78%; can reach 100% with 35%+ HP [87] | High, immediate color change (ΔE ~5.3-5.9) [86]; Significant OHRQoL improvement [88] | Shorter chair time; higher risk of color relapse compared to some at-home techniques [88] [84] |
| At-Home CP | 10-16% CP [88] [86] | Significantly lower; as low as 15% with 10% CP [87] | Comparable long-term whitening to high-conc. HP (ΔE ~11.4 at 6 mo) [86]; Significant OHRQoL improvement [88] | Excellent color stability over 6 months [88] [86]; Requires high patient compliance over 1-2 weeks [86] |
| At-Home HP (Low Conc.) | 6% HP [88] | Minimal sensitivity reported [88] | Effective whitening and positive OHRQoL impact [88] | Good long-term color stability; requires patient adherence [88] |
| Combined Bleaching | In-office HP + At-home CP [56] | Risk and intensity highly dependent on in-office gel pH; higher than at-home alone [56] [84] | Significant whitening achieved; aesthetic self-perception and psychosocial impact improved post-treatment [56] | Neutral pH in-office gel reduces TS risk without compromising efficacy, aiding compliance [56] |
The Critical Role of Protocol and Formulation
Experimental data highlights how modifications to standard protocols can optimize patient outcomes. For instance, the pH of in-office bleaching gels is a significant factor in tooth sensitivity (TS). A 2025 randomized clinical trial demonstrated that using a 35% HP gel with a neutral, stable pH significantly reduced the risk and intensity of TS during combined bleaching treatments, compared to an acidic, unstable pH gel, without compromising whitening efficacy [56].
Furthermore, in-office application protocols influence both efficacy and safety. An in-vitro study testing various protocols for HP gels found that shorter application sequences (e.g., 2 x 8 minutes) or a single prolonged application (1 x 24 minutes) provided an optimal balance, achieving significant whitening while minimizing the penetration of HP into the pulp chamber—a key factor linked to clinical sensitivity [85].
Experimental Data and Methodologies
The comparative data presented are derived from rigorous clinical and in-vitro studies. Understanding their methodologies is crucial for interpreting the results and designing future research, including for novel PTU-based depigmentation agents.
Representative Clinical Trial Protocol
A randomized controlled trial (RCT) design is the gold standard for comparing bleaching systems [88] [56]. A typical methodology includes:
- Participants: Healthy adults (e.g., ≥18 years) with anterior teeth darker than a predefined shade (e.g., A3) and without active caries, severe periodontal disease, or extensive restorations [88] [89].
- Randomization & Blinding: Participants are randomly allocated to treatment groups (e.g., in-office paint-on varnish, at-home tray). While operator blinding can be challenging due to different product formats, outcome assessors (e.g., color measurement) are often blinded [88].
- Intervention Groups: Groups are defined by the bleaching system, concentration, and protocol. For example:
- Group A: In-office 6% HP paint-on varnish.
- Group B: At-home 6% HP with a pre-filled disposable tray.
- Group C: At-home 16% CP with a custom tray [88].
- Outcome Measurement:
- Efficacy: Tooth color is measured objectively at baseline, immediately post-treatment, and at follow-ups (e.g., 6 months) using a spectrophotometer to obtain CIE Lab* values. The color difference (ΔE00) and whiteness index difference (WID) are calculated [88] [86].
- Sensitivity: Often assessed using a Visual Analogue Scale (VAS) or numerical rating scale, recorded during and/or after each bleaching session [56] [89].
- Patient-Reported Outcomes (PROs): Validated questionnaires, such as the Oral Health Impact Profile (OHIP-14), are used to evaluate changes in Oral Health-Related Quality of Life (OHRQoL). Aesthetic self-perception and psychosocial impact are also measured with specific scales [88] [56].
In-Vitro Penetration and Efficacy Protocol
To assess the safety mechanism of bleaching protocols, in-vitro studies on extracted teeth are conducted.
- Tooth Preparation: Extracted human teeth (e.g., premolars) are selected, cleaned, and inspected for defects. A baseline whiteness index is recorded [85].
- Experimental Groups: Teeth are randomized into groups based on the bleaching gel and application protocol (e.g., 1x8min, 2x8min, 3x8min) [85].
- HP Penetration Measurement: The pulp chamber is sealed and filled with a solution that reacts with permeated HP. After bleaching, the solution is analyzed using UV-Vis spectrophotometry to quantify the amount of HP that penetrated the tooth structure [85].
- Efficacy Measurement: A spectrophotometer is used to measure the whiteness index (WID) before and after bleaching to quantify efficacy [85].
The diagram below illustrates the logical relationship between bleaching protocols and their measured outcomes in such a study.
Diagram 1: In-vitro study logic for bleaching safety and efficacy.
The Scientist's Toolkit: Essential Reagents and Materials
Table 2: Key Research Reagents and Materials for Bleaching Studies
| Reagent / Material | Function in Experimental Protocol |
|---|---|
| Hydrogen Peroxide (HP) / Carbamide Peroxide (CP) Gels | The active bleaching agents under investigation. Concentrations are verified via titration (e.g., with potassium permanganate) [85]. |
| Spectrophotometer | The primary objective instrument for measuring tooth color in CIE Lab* color space, enabling calculation of color difference (ΔE) and whiteness index (WID) [88] [86]. |
| VITA Classical / 3D-Master Shade Guides | Standardized visual aids for clinical shade matching by calibrated examiners, providing a common clinical reference [88] [89]. |
| Artificial Saliva | A chemically defined solution used in vitro to maintain hydration and simulate oral environment conditions during and after bleaching treatments [86]. |
| Visual Analogue Scale (VAS) | A validated patient-reported outcome tool (typically a 100mm line) used to quantify the subjective intensity of tooth sensitivity and gingival irritation [56] [89]. |
| OHIP-14 Questionnaire | A validated 14-question instrument for assessing Oral Health-Related Quality of Life across seven domains, measuring the impact of bleaching treatment from the patient's perspective [88]. |
Synthesis for Research and Development
The data clearly demonstrates a trade-off in chemical bleaching: higher concentrations and acidic formulations accelerate depigmentation but at the cost of significantly increased sensitivity, which can impact patient compliance and satisfaction. The emergence of alternative agents like Phthalimidoperoxycaproic acid (PAP), which is reported to achieve whitening with a lower incidence of sensitivity by avoiding free radical generation, represents one approach to breaking this paradigm [90]. This establishes a clear benchmark for any new depigmentation agent, such as those potentially based on PTU. A successful alternative would need to demonstrate non-inferiority in efficacy (ΔE/WID) and color stability while showing a superior profile in reducing tooth sensitivity, as measured by standardized VAS and PROs. Future research should focus on standardized protocols for direct comparison and explore combination therapies that maximize patient comfort alongside aesthetic results.
Conclusion
The comparative analysis between PTU-based treatments and chemical bleaching reveals a clear trade-off: while peroxide-based bleaching offers potent, rapid depigmentation, it carries inherent risks of sensitivity and enamel damage. PTUs, characterized by their 'de-conjugated' polarizable hydrogen bonds, present a innovative pathway to high refractive index and transparency with potentially superior biocompatibility. The future of pigment removal lies in hybrid strategies that leverage the strengths of both approaches. Research should prioritize the development of PTU-inspired biomaterials for clinical depigmentation, the integration of nanoparticle enhancers to reduce required peroxide concentrations, and the application of smart, activated delivery systems for personalized treatment protocols. These directions promise a new generation of depigmentation agents that are both highly effective and exceptionally safe for broad biomedical application.
References
- 1. Phenylthiourea Specifically Reduces Zebrafish Eye Size [https://pmc.ncbi.nlm.nih.gov/articles/PMC3384602/]
- 2. Bleaching effect of high refractive index xylylic poly(thiourea)s ... [https://pubs.rsc.org/ja/content/articlehtml/2025/cc/d5cc03785a]
- 3. Coumaric Acid Potently Down-regulates Zebrafish Embryo ... [https://www.bslonline.org/journal/view.html?doi=10.15616/BSL.2017.23.1.8]
- 4. Orbital Interactions in Hydrogen Bonds: A Perspective ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12242117/]
- 5. Hydrogen peroxide whitens teeth by oxidizing the organic ... [https://www.sciencedirect.com/science/article/abs/pii/S0300571212002266]
- 6. Insight in the Chemistry of Laser-Activated Dental Bleaching [https://pmc.ncbi.nlm.nih.gov/articles/PMC4385672/]
- 7. Phenylthiourea Specifically Reduces Zebrafish Eye Size [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0040132]
- 8. What is the Difference Between Whitening and Bleaching? [https://www.dentalcare.com/en-us/ce-courses/ce491/what-is-the-difference-between-whitening-and-bleaching]
- 9. Clinical evaluation of in-office bleaching with low, medium ... [https://pubmed.ncbi.nlm.nih.gov/40259078/]
- 10. Comparison of laser and power bleaching techniques in tooth ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5410669/]
- 11. Dental Bleaching Techniques; Hydrogen-carbamide ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4311381/]
- 12. Free radicals and their impact on health and antioxidant ... [https://www.nature.com/articles/s41420-024-02278-8]
- 13. Oxidative stress, free radicals and antioxidants [https://pmc.ncbi.nlm.nih.gov/articles/PMC10206224/]
- 14. Designing High-Refractive Index Polymers Using Materials ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6415069/]
- 15. Researcher's fish-eye view could offer insights for human ... [https://www.purdue.edu/newsroom/releases/2012/Q3/researchers-fish-eye-view-could-offer-insights-for-human-vision.html]
- 16. Downregulation of Melanin Synthesis by Haginin A and Its ... [https://www.sciencedirect.com/science/article/pii/S0022202X1533832X]
- 17. Molecular Designs of Sulfur-containing High Refractive ... [https://link.springer.com/article/10.1007/s10118-025-3334-6]
- 18. Designing Polymers for Use in Next-Generation Bioelectronics [https://research.ncsu.edu/designing-polymers-for-use-in-next-generation-bioelectronics/]
- 19. Bleaching effect of high refractive index xylylic poly ... [https://www.sciencedirect.com/org/science/article/pii/S1359734525020798]
- 20. Theoretical and experimental study of the bleaching ... [https://pubmed.ncbi.nlm.nih.gov/18285901/]
- 21. High Throughput, High Content Screening for Novel ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3977999/]
- 22. Depigmenting action and bioimaging of luminescent ... [https://www.sciencedirect.com/science/article/pii/S0045206825011514]
- 23. Investigation of the whitening activity of ginsenosides from ... [https://www.sciencedirect.com/science/article/pii/S122684532300194X]
- 24. Identification of Phthalocyanine Textile Dye Reactive Blue ... [https://www.mdpi.com/2079-6447/4/4/34]
- 25. Bleaching effect of high refractive index xylylic poly ... [https://pubs.rsc.org/en/content/articlelanding/2025/cc/d5cc03785a]
- 26. Small-Molecule Tyrosinase Inhibitors for Treatment of ... [https://www.mdpi.com/1420-3049/30/4/788]
- 27. Inhibition of Tyrosinase and Melanogenesis by a White ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12346983/]
- 28. Solution-processable and functionalizable ultra-high ... [https://www.nature.com/articles/s41467-021-27090-1]
- 29. Differences Between Cyclic and Linear PEGylated Poly ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2788102/]
- 30. Galangin ameliorates PTU-induced vitiligo in zebrafish and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11931063/]
- 31. Assessing the skin-whitening property of plant extracts from ... [https://arabjchem.org/assessing-the-skin-whitening-property-of-plant-extracts-from-taiwanese-species-using-zebrafish-as-a-rapid-screening-platform/]
- 32. Effectiveness and Safety of Over-the-Counter Tooth ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9915942/]
- 33. Efficacy and safety of peroxide-based mouthrinse on ... [https://www.sciencedirect.com/science/article/abs/pii/S0300571225000302]
- 34. Novel in-office peroxide-free tooth-whitening gels [https://www.nature.com/articles/s41598-020-66733-z]
- 35. Comparison of Bleaching Products With Up to 6% and With ... [https://www.frontiersin.org/journals/physiology/articles/10.3389/fphys.2019.00919/full]
- 36. Clinical Performance of Low-Concentration Bleaching Gels ... [https://pubmed.ncbi.nlm.nih.gov/40935235/]
- 37. Effect of low-concentration at-home bleaching gels on ... [https://bmcoralhealth.biomedcentral.com/articles/10.1186/s12903-025-07026-7]
- 38. Influence of Concentration and Activation on Hydrogen ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3791642/]
- 39. Comparative in situ hybridization protocols in zebrafish [https://pmc.ncbi.nlm.nih.gov/articles/PMC9490454/]
- 40. Spectroscopic properties (FT-IR, NMR and UV) and DFT ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10709190/]
- 41. NMR in Material Science [https://www.bruker.com/en/products-and-solutions/mr/make-mr-more-relevant/nmr-in-material-science.html]
- 42. XRD, NMR, FT-IR and DFT structural characterization of a ... [https://www.sciencedirect.com/science/article/abs/pii/S0022286021003586]
- 43. Solid-State Nuclear Magnetic Resonance (NMR) Spectroscopy [https://camcor.uoregon.edu/facilities-and-instrumentation/solid-NMR]
- 44. Density Functional Theory Calculations for Interpretation of ... [https://www.mdpi.com/2073-4352/14/7/645]
- 45. The small molecule ML233 is a direct inhibitor of tyrosinase ... [https://www.nature.com/articles/s42003-025-07973-5]
- 46. The Effects of Three Bleaching Agents on Tooth Discoloration Caused by Mineral Trioxide Aggregate - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC9923412/]
- 47. Bleaching Agents: A Review of Their Utilization and Management [https://www.mdpi.com/2071-1050/16/20/9084]
- 48. Evaluating the efficiency of two different over-the-counter ... [https://www.nature.com/articles/s41405-024-00227-2]
- 49. Comparison of traditional and low sensitivity whiteners [https://pubmed.ncbi.nlm.nih.gov/18666494/]
- 50. The effect of Hydrogen Peroxide treatments on dental ... [https://www.sciencedirect.com/science/article/pii/S0300571225001599]
- 51. Whitening efficacy and enamel properties of 30% hydrogen ... [https://www.nature.com/articles/s41598-025-18529-2]
- 52. Evaluation and comparison of the microhardness ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4632217/]
- 53. Effect of bleaching agents on hardness, surface roughness ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7462371/]
- 54. Bee pollen peptides as potent tyrosinase inhibitors with ... [https://www.nature.com/articles/s41598-024-81495-8]
- 55. Study of the Stability and Anti-Inflammatory Activity ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12191921/]
- 56. Impact of the in-office bleaching gels pH on tooth sensitivity ... [https://pubmed.ncbi.nlm.nih.gov/40257513/]
- 57. Clinical Performance of Low-Concentration Bleaching Gels ... [https://www.sciencedirect.com/science/article/abs/pii/S030057122500541X]
- 58. Bleaching embryos after In Situ Hybridization [https://groups.google.com/g/bionet.organisms.zebrafish/c/lnHLcbN7GvI]
- 59. Applicability of the in vitro skin irritation methods (EpiSkin™ ... [https://www.sciencedirect.com/science/article/pii/S027323002500008X]
- 60. Pre-validation of a novel reconstructed skin equivalent model ... [https://pubs.rsc.org/en/content/articlehtml/2025/na/d4na00804a]
- 61. Tooth-Whitening Agents and Polymer-Based Carriers [https://pmc.ncbi.nlm.nih.gov/articles/PMC12473685/]
- 62. Effectiveness of violet LED with or without a bleaching gel [https://www.frontiersin.org/journals/dental-medicine/articles/10.3389/fdmed.2024.1427301/full]
- 63. The influence of violet LED light on tooth bleaching protocols [https://www.sciencedirect.com/science/article/abs/pii/S1572100020304063]
- 64. Bleaching with biocatalysts and violet LED: a randomized ... [https://pubmed.ncbi.nlm.nih.gov/40846796/]
- 65. Purple Toothpaste vs Professional Whitening - Good Tooth [https://goodtoothdentalcare.com/purple-toothpaste-vs-professional-whitening-what-actually-works-for-whiter-teeth-in-2025/]
- 66. Hydrogen Peroxide-Free Color Correctors for Tooth ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12384575/]
- 67. A review on natural teeth whitening [https://www.sciencedirect.com/science/article/abs/pii/S134900792100178X]
- 68. 1-phenyl 2-thiourea (PTU) activates autophagy in zebrafish ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8143244/]
- 69. Triple-blinded randomized clinical trial comparing efficacy and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8523096/]
- 70. Whitening efficacy of activated charcoal-based products [https://www.sciencedirect.com/science/article/abs/pii/S0300571224000472]
- 71. Effect of Experimental Bleaching Gels With Enzymes on ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11976588/]
- 72. In Vitro Evaluation of Chemical and Microhardness ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12385387/]
- 73. Analysis of Enamel Surface and Shear Bond Strength with ... [https://www.jkdhs.org/journal/view.html?doi=10.17135/jdhs.2025.25.2.130]
- 74. Hyaluronic Acid-Based Bleaching Gels With NF_TiO 2 and ... [https://pubmed.ncbi.nlm.nih.gov/40342264/]
- 75. Microscopic and Crystallographic Analysis of Increased ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12385602/]
- 76. Reference values for pulp oxygen saturation as a diagnostic ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7691259/]
- 77. Effect of Fluoride Varnish on Enamel Microhardness [https://www.dovepress.com/effect-of-fluoride-varnish-on-enamel-microhardness-an-in-vitro-study-peer-reviewed-fulltext-article-CCIDE]
- 78. Comparative study of the effectiveness of different ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12415242/]
- 79. Effect of bleaching materials on optical properties and ... [https://www.nature.com/articles/s41598-025-21258-1]
- 80. Transparence - an overview [https://www.sciencedirect.com/topics/physics-and-astronomy/transparence]
- 81. The Ultimate Guide to Understanding Abbe Value [https://eyesoneyecare.com/resources/ultimate-guide-to-understanding-abbe-value/]
- 82. Galangin ameliorates PTU-induced vitiligo in zebrafish and ... [https://www.frontiersin.org/journals/pharmacology/articles/10.3389/fphar.2025.1521097/full]
- 83. Impact of in-office bleaching agents on the optical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12225077/]
- 84. Evaluation of the Effectiveness of Different Types ... [https://www.mdpi.com/2306-5354/11/12/1178]
- 85. Influence of Different Protocols on In‐Office Bleaching [https://pmc.ncbi.nlm.nih.gov/articles/PMC12457872/]
- 86. Treatment Durations and Whitening Outcomes of Different ... [https://www.mdpi.com/1648-9144/59/6/1130?]
- 87. Teeth Whitening: Related Statistics on Effectiveness and ... [https://www.loudfamilydentistry.com/blog/teeth-whitening-related-statistics-on-effectiveness-and-sensitivity-incidence]
- 88. Bleaching efficacy and quality of life of different bleaching ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9383653/]
- 89. Effect of Various Vital Bleaching Systems on Clinical ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11351963/]
- 90. PAP Teeth Whitening vs Traditional Peroxide - Good Tooth [https://goodtoothdentalcare.com/pap-teeth-whitening-vs-traditional-peroxide-whats-safer-and-more-effective-in-2025/]