RNAscope vs. Conventional ISH: A Comprehensive Sensitivity and Technical Comparison for Researchers
This article provides a detailed comparison of RNAscope in situ hybridization (ISH) and conventional ISH methods, focusing on analytical sensitivity, specificity, and practical applications for researchers and drug development professionals.
RNAscope vs. Conventional ISH: A Comprehensive Sensitivity and Technical Comparison for Researchers
Abstract
This article provides a detailed comparison of RNAscope in situ hybridization (ISH) and conventional ISH methods, focusing on analytical sensitivity, specificity, and practical applications for researchers and drug development professionals. It covers the foundational principles of RNAscope's patented 'Z' probe design that enables single-molecule detection, methodological workflows for assay implementation, troubleshooting guidelines for optimization, and validation data from systematic reviews. The content synthesizes current technical specifications and performance metrics to guide scientists in selecting appropriate molecular detection techniques for their specific research contexts, particularly in oncology, infectious disease, and biomarker development.
Understanding the Technology: How RNAscope's Novel Design Enables Superior Sensitivity
For researchers investigating gene expression, the ability to visually localize specific RNA molecules within the intact cellular environment provides invaluable biological insights. In situ hybridization (ISH) has served as a fundamental histological method for nucleic acid detection for over five decades, enabling the visualization of DNA or RNA within tissues and cells [1] [2]. However, conventional ISH techniques have been hampered by significant limitations, particularly regarding sensitivity, specificity, and technical complexity. These constraints have impeded research progress, especially in detecting low-abundance transcripts, analyzing highly homologous gene families, and validating sequencing data with spatial context.
The emergence of single-molecule detection technologies represents a paradigm shift in molecular analysis, overcoming the limitations of ensemble-averaged measurements by enabling direct observation of individual molecular events [3] [4]. Unlike conventional techniques that examine large groups of molecules and provide averaged data, single-molecule detection focuses on individual molecular behavior, unlocking insights into heterogeneity and dynamic processes often hidden in ensemble measurements [3]. This granularity is particularly crucial for visualizing low-expression genes, identifying rare cellular events, and precisely quantifying biomarker expression within their morphological context.
This technical guide examines the transformative impact of single-molecule detection technologies, with particular focus on the RNAscope platform, which has established new standards for sensitivity and specificity in RNA ISH applications. By comparing conventional ISH limitations with RNAscope's innovative methodology, we provide researchers, scientists, and drug development professionals with a comprehensive framework for implementing these advanced detection strategies in their experimental designs.
Conventional ISH: Limitations and Technical Constraints
Fundamental Methodological Constraints
Conventional in situ hybridization, particularly methods utilizing digoxigenin (DIG)-labeled RNA probes, has been widely employed for transcript localization analysis [2]. While these methods represented a significant advancement over earlier radiolabeled approaches, they possess several inherent limitations that restrict their application in contemporary research settings:
Sensitivity Limitations: Traditional ISH often lacks sufficient sensitivity to detect low-expression genes or short transcripts, even with optimized probe design and signal amplification strategies [2]. Sensitivity can be increased to some extent by designing multiple probes against different regions of the target transcript or by employing signal amplification methods such as Tyramide Signal Amplification (TSA) [2]. However, these approaches have practical constraints, as probes that are too long result in reduced cell penetration, typically requiring optimization between 200-1000 base pairs for RNA probes [2].
Specificity Challenges: For highly homologous gene pairs, difficulties are faced when designing probes with appropriate lengths that ensure sensitivity while maintaining specificity [2]. This limitation is particularly problematic for gene families with high sequence similarity, where distinguishing between individual members becomes technically challenging.
Technical Complexity: Conventional ISH procedures are notably complex and time-consuming, often requiring 2-3 days for complete staining and extensive optimization of experimental conditions [2]. The multistep process demands significant technical expertise, contributing to variability between experiments and laboratories.
Practical Implementation Challenges
From an application standpoint, conventional ISH presents several practical challenges that limit its utility in modern research environments:
Limited Multiplexing Capability: Double ISH for two gene transcripts is difficult for some gene combinations, particularly when genes have widely different GC percentages, making it challenging to design probes with similar dissociation temperatures and appropriate probe lengths [2].
Compatibility Issues with Immunostaining: Combining ISH with immunostaining is problematic due to decreased antigen reactivity caused by proteinase treatment or hybridization at denaturing temperatures [2]. This limitation restricts correlative analysis of RNA and protein expression within the same sample.
Reproducibility Concerns: The dependence on user-intensive optimization and the complexity of experimental procedures contribute to variability between experiments and laboratories, undermining the reproducibility of results [2].
Table 1: Key Limitations of Conventional In Situ Hybridization
| Parameter | Limitation in Conventional ISH | Impact on Research Applications |
|---|---|---|
| Sensitivity | Often insufficient for low-expression genes or short transcripts | Inability to detect biologically relevant but low-abundance targets |
| Specificity | Challenging for highly homologous genes | Difficulty distinguishing between closely related gene family members |
| Multiplexing | Technically difficult for multiple targets | Limited capacity for complex gene expression profiling |
| Immunostaining Combination | Problematic due to antigen degradation | Restricted correlative RNA-protein analysis |
| Experimental Time | 2-3 days for complete procedure | Reduced throughput and efficiency |
| Technical Expertise | Requires significant optimization | Barriers to adoption and reproducibility issues |
RNAscope Technology: Principles and Workflow
Core Technological Innovation
The RNAscope platform represents a groundbreaking advance in in situ hybridization technology, employing a novel probe design strategy that fundamentally addresses the sensitivity and specificity limitations of conventional ISH. The core innovation lies in its proprietary "double Z" probe design, which functions similarly to a molecular mechanism requiring dual verification for signal generation [5] [6].
This double Z probe architecture consists of three key elements [5]:
- The lower region (18-25 bases) complementary to the target RNA
- A spacer sequence linking the two components
- The upper region (14-base tail sequence) that forms a binding site
The mechanism ensures specific amplification only when two independent Z probes bind adjacent sites on the target RNA, forming a 28-base binding site for the pre-amplifier [5]. This design makes it highly improbable that non-specific binding events will generate amplified signals, as single Z probes binding to non-specific sites cannot form the required binding site for subsequent amplification steps [5].
Signal Amplification Cascade
The RNAscope assay employs a sophisticated multistep amplification cascade that enables single-molecule detection [5]:
- Hybridization: Approximately 20 double Z target probe pairs specifically hybridize to the target RNA molecule
- Pre-Amplifier Binding: Pre-amplifiers hybridize to the 28-base binding site formed by each double Z probe pair
- Amplifier Binding: Multiple amplifiers bind to each pre-amplifier, dramatically increasing signal capacity
- Label Probe Binding: Labeled probes containing fluorescent molecules or chromogenic enzymes bind to numerous sites on each amplifier
This 20×20×20 probe design and signal amplification strategy provides sufficient sensitivity to visualize individual RNA molecules as distinct punctate dots under a standard microscope [5] [6]. The requirement for multiple independent probe pairs to bind adjacent sites ensures exceptional specificity, while the branched DNA amplification architecture provides the necessary signal intensity for detection.
Experimental Workflow
The standard RNAscope assay follows a streamlined, single-day workflow that significantly reduces technical complexity compared to conventional ISH [7] [5]:
The streamlined process from sample preparation to quantification can be completed within a single day, a significant improvement over conventional ISH methods that typically require 2-3 days [5] [2]. This efficiency gain, combined with reduced need for extensive optimization, makes the technology particularly valuable for research environments requiring rapid turnaround and high reproducibility.
Comparative Analysis: Technical Specifications and Performance Metrics
Direct Performance Comparison
When evaluated against conventional ISH methods and other high-sensitivity variants, RNAscope demonstrates distinct advantages across multiple performance parameters:
Table 2: Comprehensive Comparison of ISH Methodologies
| Parameter | Conventional DIG-RNA ISH | RNAscope | HCR ISH | clampFISH | SABER FISH |
|---|---|---|---|---|---|
| Sensitivity | Moderate | Single-molecule detection | Single-molecule detection | Single-molecule detection | Single-molecule detection |
| Experimental Difficulty | Difficult | Easy | Moderate | Moderate | Moderate |
| Staining Time | 2-3 days | 1 day | 1-3 days | 1-3 days | 2-3 days |
| Multiplex Staining | Difficult | Easy | Easy | Easy | Easy |
| Immunostaining Compatibility | Limited due to protein degradation | Excellent with high antigen retention | Good | Good | Good |
| Probe Design | By user (can be outsourced) | Provided by manufacturer | By user (can be outsourced) | By user | By user |
| Monetary Cost | Low per sample | High per sample | Moderate (decreases with scale) | Moderate (decreases with scale) | Moderate (decreases with scale) |
| Time Cost for Optimization | Significant | Minimal | Significant | Significant | Significant |
| Automated Staining | Applicable | Applicable | Not reported | Not reported | Not reported |
| microRNA Detection | Difficult | Applicable (with miRNAscope) | Applicable | Not reported | Not reported |
Advantages for Specific Research Applications
The unique characteristics of RNAscope technology provide particular benefits for specialized research applications:
Viral Pathogenesis Research: RNAscope enables direct detection of viral RNA in human or animal cells with exceptional sensitivity, identifying individual viral particles despite low viral loads [7]. The platform's proprietary probe design strategy allows accurate detection among highly related viral species/strains, providing precise detection even with contaminating viruses [7].
Neuroscience Research: The technology has become an essential tool in neuroscience, allowing simultaneous detection of between three and forty-eight low-abundance mRNAs in single cells (multiplexing or hiplexing) [8]. This capability is crucial for characterizing different cell types in rodent brains, a widely used approach in neuroscience [8].
Oncology and Biomarker Validation: RNAscope provides a powerful multiomic method to detect gene and protein expression within the spatial and morphological tissue context, making it invaluable for biomarker validation in oncology research [6] [9]. The technology's ability to work with formalin-fixed, paraffin-embedded (FFPE) tissues enhances its utility for clinical specimen analysis [5].
Single-cell RNAseq Validation: The single-molecule sensitivity and cellular resolution of RNAscope make it an ideal platform for validating findings from single-cell RNA sequencing studies, providing spatial context that sequencing approaches cannot offer [9].
Quantitative Analysis and Image Processing Methodologies
Automated Quantification Approaches
The quantitative analysis of RNAscope data has evolved from manual counting to sophisticated automated image analysis pipelines, significantly enhancing reproducibility and throughput. Manual quantification of transcripts remains a laborious and time-consuming task even for small tissue sections, creating bottlenecks in research workflows [8]. To address this challenge, automated analysis methods have been developed:
QuPath Analysis Pipeline: An open-source bioimage analysis platform that includes annotation tools, built-in algorithms for cell detection, and interactive machine learning [8]. Custom scripts within QuPath can create automated workflows for optimizing cell detection parameters and establishing mRNA signal thresholds using negative controls [8].
HALO Software Integration: Commercial image analysis platform specifically adapted for RNAscope quantification, enabling high-throughput analysis of transcript-positive cells across entire tissue sections [9]. The software provides capabilities for spatial biology analysis, allowing researchers to examine expression patterns within specific tissue architectures [9].
Threshold Determination: Standardized protocols establish criteria for quantifying positive cells using negative controls, improving consistency across studies [8]. This approach includes methodical optimization of fluorescence intensity thresholds and other cell detection parameters to ensure accurate classification of transcript-positive cells.
Standardization and Reproducibility Framework
The implementation of standardized analysis protocols addresses a critical challenge in single-molecule RNA detection - the lack of consistent methodology for quantifying RNA transcripts and transcript-positive cells across different laboratories [8]. Key elements of this standardization include:
- Threshold Establishment: Using negative control probes to derive objective mRNA signal thresholds for distinguishing positive from negative cells [8]
- Parameter Optimization: Systematic optimization of cell detection parameters rather than relying on subjective manual counting
- Validation Procedures: Implementing rigorous validation steps to ensure automated quantification matches manual assessment
- Data Output Customization: Providing flexible data outputs tailored to specific research questions, including cell-by-cell expression data and spatial distribution metrics [9]
This standardized approach to quantification enables more reliable comparison of results across different studies and laboratories, enhancing the reproducibility of single-molecule detection studies [8].
Research Reagent Solutions and Experimental Implementation
Essential Research Tools and Applications
Successful implementation of RNAscope technology requires specific reagents and equipment optimized for the platform:
Table 3: Essential Research Reagents and Equipment for RNAscope Implementation
| Reagent/Equipment Category | Specific Examples | Function and Application Notes |
|---|---|---|
| RNAscope Kits | RNAscope Fluorescent Multiplex reagent kit v1 (Fresh frozen) | Core detection reagents for fresh frozen tissue applications [8] |
| RNAscope Universal Pretreatment Kit (FFPE) | Target retrieval and protease treatment for formalin-fixed tissues [8] | |
| Probe Systems | Target-specific probes (e.g., Rn-Hcrtr1-C1, Rn-Th-C2, Rn-Fos-C3) | Target detection in different channels (C1, C2, C3) for multiplexing [8] |
| Negative control probes (e.g., RNAscope 3-plex negative control) | Essential for establishing background thresholds and validation [8] | |
| Sample Preparation | RNAscope RTU Protease IV reagent | Enzyme treatment for tissue permeabilization and target unmasking [8] |
| Immedge hydrophobic barrier pen | Creates liquid barrier around tissue sections on slides [8] | |
| Equipment | HybEZ II system hybridization oven | Provides precise temperature control for hybridization steps [8] |
| Slide scanner (e.g., Carl Zeiss AxioScan Z.1) | High-resolution digitization of fluorescent slides for quantification [8] | |
| Image Analysis | QuPath open-source software | Automated cell detection and transcript quantification [8] |
| HALO Software | Commercial platform for high-throughput quantitative analysis [9] |
Implementation Considerations for Different Sample Types
The optimal implementation of RNAscope varies depending on sample type and research objectives:
Fresh Frozen vs. FFPE Tissues: RNAscope kits for fresh frozen and fixed tissues have different preparation requirements [8]. Fixed tissues require additional pretreatment steps including RNAscope Target Retrieval and RNAscope Protease III, available in the RNAscope Universal Pretreatment kit [8].
Multiplex Experiment Planning: For multiplex assays, each target probe must be in a different channel (C1, C2, or C3), with one target probe always in the C1 channel [8]. Channel C1 target probes are ready-to-use (RTU), while channel C2 and C3 probes are shipped as 50× concentrated stock that must be mixed with a C1 RTU probe [8].
Probe Selection Strategy: ACD offers RNAscope probes for >100 viruses, with new probes or made-to-order probes designed and manufactured within two weeks [7]. This rapid probe development capability makes the platform particularly valuable for emerging infectious disease research.
Tissue Preparation Protocols: For fresh frozen brain collection, optimal preservation requires snap-freezing in 2-methylbutane at -30°C to protect tissue from cracking and prevent mRNA degradation [8]. For fixed preparations, transcardial perfusion with formalin followed by overnight post-fixation and cryoprotection in 30% sucrose is recommended [8].
The advent of single-molecule detection technologies, particularly the RNAscope platform, has fundamentally transformed the landscape of in situ hybridization, enabling unprecedented sensitivity and specificity in RNA visualization within intact cells and tissues. The proprietary double Z probe design, coupled with a sophisticated signal amplification system, has effectively addressed the key limitations of conventional ISH methods while providing a streamlined workflow that enhances experimental efficiency and reproducibility.
For researchers investigating viral pathogenesis, neuroscience, oncology, and numerous other fields, these advances provide powerful tools for spatial genomic analysis that complement and extend capabilities offered by other molecular detection methods. While real-time PCR remains a sensitive, rapid, and cost-effective viral detection method, it does not preserve morphological context and provides expression levels averaged across cell populations [7]. Similarly, next-generation sequencing offers powerful a priori discovery capabilities but lacks spatial resolution [7]. RNAscope technology bridges this critical gap by providing molecular detection coupled with morphological context, enabling visualization of viruses in different infected tissues and cell types [7].
As single-molecule detection technologies continue to evolve, future developments will likely focus on increasing multiplexing capabilities, enhancing quantification algorithms, and improving integration with complementary analytical methods such as proteomics and single-cell sequencing. These advances will further solidify the role of single-molecule detection as an indispensable tool for biomedical research and diagnostic applications, providing increasingly sophisticated insights into gene expression patterns within their native cellular and tissue environments.
In situ hybridization (ISH) has long been a cornerstone technique for spatial genomic analysis, yet conventional methods often fall short in sensitivity and specificity, particularly for detecting low-abundance RNA targets. The RNAscope platform represents a paradigm shift in ISH technology, overcoming these limitations through a proprietary 'double Z' probe design and a sophisticated signal amplification system. This whitepaper delineates the core mechanism of the RNAscope assay, explaining how its unique architecture enables single-molecule RNA visualization while effectively suppressing background noise. Framed within a broader thesis on ISH sensitivity, we present quantitative comparisons and methodological protocols that substantiate RNAscope's superior performance over conventional ISH techniques, providing researchers and drug development professionals with a comprehensive technical guide.
In situ hybridization enables the detection of nucleic acids within intact cells and tissues, preserving crucial spatial context that is lost in grind-and-bind methods like RT-PCR [2]. However, conventional ISH techniques have been hampered by significant technical challenges that limit their sensitivity and reliability. Conventional RNA ISH methodologies, often utilizing digoxigenin (DIG)-labeled RNA probes, struggle with low sensitivity for detecting low-expression genes or short transcripts [2]. Their reliability is highly dependent on factors such as probe design, coverage of the target mRNA sequence, and the stringency of hybridization conditions [2]. A primary limitation is the high background noise resulting from non-specific probe binding, which obscures genuine signals and compromises assay specificity [5] [10].
The RNAscope technology, developed by Advanced Cell Diagnostics (ACDBio), introduces a novel approach designed to overcome these fundamental limitations. This platform employs a unique 'double Z' probe design and a cascading signal amplification system that work in concert to achieve unparalleled sensitivity and specificity [5] [10]. The system is engineered to visually output individual RNA molecules as distinct, punctate dots under a standard microscope, enabling both qualitative localization and quantitative analysis at a single-cell level [11] [5]. This technical advance has established RNAscope as a new standard in high-sensitivity ISH, cited in over 10,000 publications and widely adopted for spatial gene expression analysis [11] [2].
The Architectural Core: 'Double Z' Probe Design
The foundational innovation of the RNAscope platform lies in its proprietary 'double Z' probe design. This architecture is meticulously engineered to maximize specificity by ensuring that signal amplification occurs only when two independent probes bind adjacent sites on the target RNA.
Structural Components and Binding Mechanism
Each 'double Z' probe set is composed of multiple probe pairs designed to hybridize to the target RNA. Individually, each probe within a pair has a distinct structure and function, as detailed below.
- Target Hybridization Region: The lower arm of each Z probe contains an 18- to 25-base sequence that is complementary to the target RNA. This relatively short length is optimal for accessing partially degraded RNA in archival FFPE tissues, a common challenge in molecular pathology [5] [12].
- Spacer Sequence: A linker region that connects the target-binding region to the tail sequence, facilitating the distinct Z-shaped conformation of the probe [5].
- Tail Sequence: The upper region of the Z is a 14-base tail sequence. Critically, when two Z probes hybridize adjacently to the target RNA, their combined 28-base tail sequences create a contiguous binding site for the subsequent pre-amplifier molecule [5].
This design functions on a principle of cooperative binding akin to fluorescence resonance energy transfer (FRET) assays [5]. The system is engineered such that a single probe binding non-specifically to an off-target sequence will not yield a detectable signal. It is only when both probes in a pair bind in tandem to their respective adjacent target sites that the complete 28-base pre-amplifier binding site is formed [5] [10]. This requirement drastically reduces the probability of amplifying background noise from non-specific hybridization, which is a major limitation of conventional ISH [2].
Redundancy and Robustness
To ensure robust detection of single RNA molecules, the RNAscope assay utilizes approximately 20 such double Z probe pairs for each target RNA [5]. This high level of redundancy means that even if some probe binding sites are inaccessible due to RNA secondary structure, partial degradation, or protein binding, the remaining probes can still generate a sufficient signal. The technology is so sensitive that the binding of only three double Z probe pairs is theoretically required to detect a single RNA molecule [5]. This robust design makes RNAscope particularly effective for analyzing challenging samples, such as formalin-fixed, paraffin-embedded (FFPE) tissues, where RNA integrity is often compromised [5] [10].
The Amplification Cascade: From Hybridization to Visualization
Following the specific hybridization of double Z probe pairs, the RNAscope system employs a multi-step, branched DNA signal amplification cascade. This process dramatically enhances the signal without increasing background noise, enabling the visualization of individual RNA molecules.
Sequential Amplification Stages
The amplification process occurs through a precise sequence of hybridization events, each building upon the previous to create a large signaling complex.
The diagram above illustrates the sequential hybridization cascade of the RNAscope signal amplification system. Each step is critical for transforming a single RNA molecule into a detectable signal dot.
- Pre-Amplifier Binding: The pre-amplifier molecule hybridizes specifically to the 28-base binding site formed by the tail sequences of two adjacently bound Z probes [5]. This step is crucial for specificity, as the pre-amplifier requires the full contiguous site and will not bind to single Z probes that may have attached non-specifically [12].
- Amplifier Assembly: Multiple amplifier molecules then hybridize to the numerous binding sites present on each pre-amplifier [5]. A single pre-amplifier can bind several amplifier molecules, creating the first major level of signal amplification.
- Label Probe Attachment: Each amplifier molecule contains multiple binding sites for enzyme-labeled or fluorescently-labeled probes [5] [12]. The binding of numerous label probes to each amplifier constitutes the second major level of amplification, resulting in a large complex that can be easily visualized.
Signal Output and Quantification
The final output of this amplification cascade is the formation of a punctate dot for each target RNA molecule, which can be visualized using either fluorescence or bright-field microscopy, depending on the label used [5] [10]. This discrete dot-like signal is a direct consequence of the localized amplification at the site of each target RNA molecule. Unlike the diffuse staining often encountered in conventional ISH, these distinct dots enable precise quantification. Researchers can count dots on a cell-by-cell basis, either manually or using automated image analysis software like HALO or QuPath, to determine the absolute copy number of RNA transcripts within individual cells [5] [12].
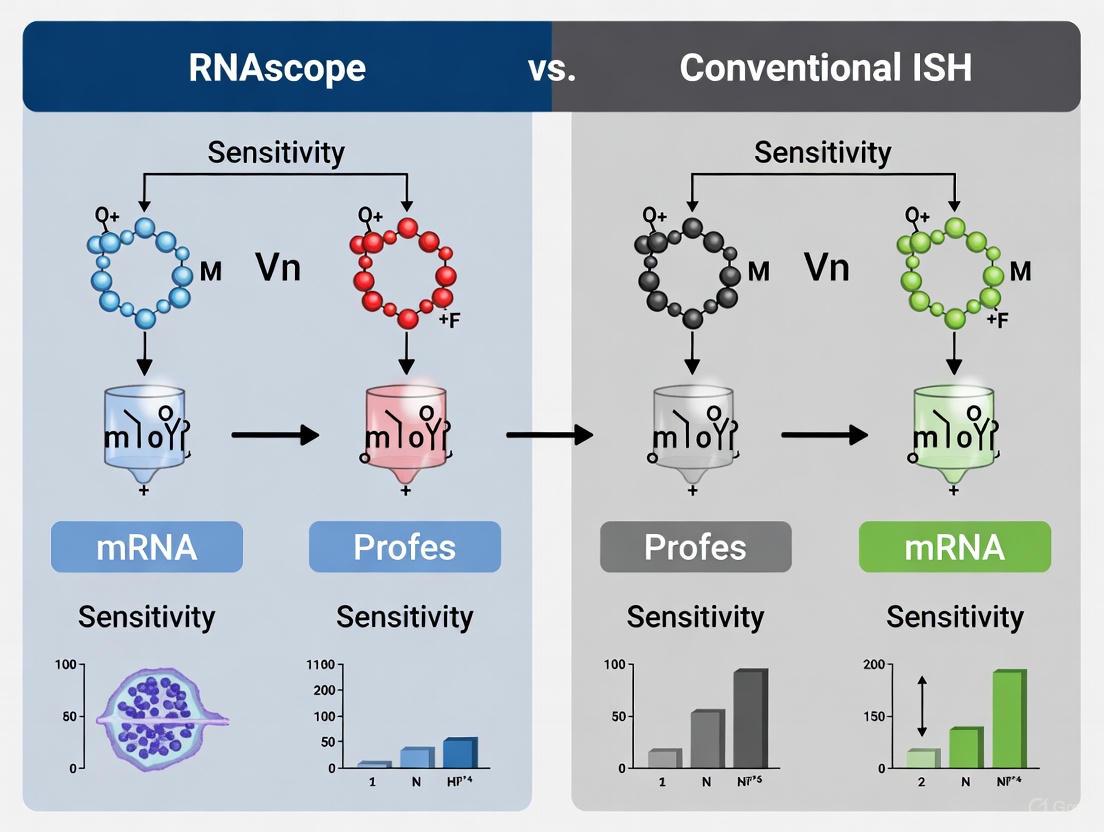
Comparative Analysis: RNAscope vs. Conventional ISH
To objectively evaluate the advancement represented by the double Z probe design, a systematic comparison with conventional ISH methodologies is essential. The following tables summarize key qualitative and quantitative differences.
Table 1: Technical and Performance Characteristics Comparison
| Characteristic | Conventional DIG-ISH | RNAscope (Double Z) |
|---|---|---|
| Probe Design | Long RNA probes (200-1000 bp) [2] | Short oligonucleotide ZZ pairs (~50 bp combined) [5] |
| Signal Amplification | Tyramide Signal Amplification (TSA), if used [2] | Proprietary cascading DNA branched amplification [5] |
| Specificity Mechanism | Hybridization stringency [2] | Dual-probe recognition & cooperative binding [5] |
| Single-Molecule Sensitivity | Difficult or impossible for low-copy targets [2] | Yes, standard outcome [5] [10] |
| Background Noise | Often high, requires careful optimization [2] | Inherently low due to design [5] |
| Multiplexing Ability | Difficult [2] | Easy (multiplex fluorescent assays available) [2] |
| Compatibility with FFPE | Moderate, sensitive to degradation [2] | High, works well with partially degraded RNA [5] |
Table 2: Practical Implementation and Cost Comparison [2]
| Factor | Conventional DIG-ISH | RNAscope |
|---|---|---|
| Experimental Difficulty | Difficult | Easy |
| Staining Time | 2–3 days | 1 day |
| Probe Design & Synthesis | Done by user (can be outsourced) | Provided by manufacturer only |
| Monetary Cost (Total) | Low | High |
| Monetary Cost (Per Sample) | Low | High |
| Automated Staining | Applicable | Applicable (optimized for platforms) |
| Major Advantage | Cost-effective for large studies | Efficiency, ease of use, reliability |
The data reveals a clear trade-off. While conventional ISH has a lower monetary cost, RNAscope offers significant advantages in sensitivity, specificity, speed, and reliability. The double Z design's requirement for dual recognition fundamentally reduces false positives, and its signal amplification system provides the sensitivity needed for low-abundance targets. The simplified, faster workflow (one day versus several days) and ease of use make RNAscope particularly valuable for diagnostic applications and high-value research where result reliability is paramount [2].
The Scientist's Toolkit: Essential Reagents and Materials
Successful implementation of the RNAscope assay requires specific reagents and materials. The following table outlines the core components of the RNAscope workflow and their functions.
Table 3: Essential Research Reagents for RNAscope Assays [12]
| Reagent/Material | Function in the Workflow |
|---|---|
| RNAscope Target Retrieval | A buffer used with heat to partially reverse formalin-induced cross-links in FFPE samples, making target RNA more accessible [12]. |
| RNAscope Hydrogen Peroxide | Blocks endogenous peroxidase activity to prevent background in chromogenic detection [12]. |
| RNAscope Protease (Plus, III, or IV) | Enzymatically permeabilizes cell membranes and degrades proteins bound to RNA to further unmask the target sequence [12]. |
| RNAscope Specific Probe Probes | The custom-designed ~20 double Z probe pairs targeted to the specific RNA of interest. |
| RNAscope Amplification Reagents | The proprietary set of pre-amplifier, amplifier, and label probes that execute the cascading signal amplification [5] [12]. |
| Control Probes (PPIB, dapB) | Essential validation tools. PPIB (a housekeeping gene) is a positive control; dapB (a bacterial gene) is a negative control for background assessment [12]. |
| Chromogenic or Fluorescent Detection Kit | Provides the enzyme (e.g., HRP) and substrate (e.g., DAB) for bright-field detection, or the fluorescent dyes for multiplex fluorescence microscopy [5] [12]. |
Experimental Protocol: A Standard Workflow for FFPE Tissues
The following detailed methodology outlines a standard RNAscope assay for formalin-fixed, paraffin-embedded (FFPE) tissue sections, as derived from the cited technical resources [5] [12].
Sample Preparation and Pretreatment
- Sectioning and Baking: Cut FFPE tissue sections at 5-10 μm thickness and mount on positively charged slides. Bake slides at 60°C for 1 hour to ensure tissue adhesion.
- Dewaxing and Dehydration: Deparaffinize slides by immersing in xylene (or xylene substitute), followed by sequential dehydration in 100% ethanol and air drying.
- Target Retrieval: Immerse slides in RNAscope Target Retrieval reagent and heat using a steamer or water bath (e.g., 98-100°C for 15 minutes). This critical step reverses cross-links. Cool slides briefly in distilled water.
- Protease Digestion: Treat slides with RNAscope Protease (e.g., Protease Plus for 30 minutes at 40°C) to permeabilize the tissue and unmask the target RNA. The protease type and incubation time may require optimization based on tissue type and fixation.
Probe Hybridization and Amplification
- Hybridize Probes: Apply the target-specific RNAscope probe mixture to the tissue section and incubate at 40°C for 2 hours in a hybridization oven (e.g., ACD's HybEZ Oven). This allows the double Z probes to bind specifically to their target RNA.
- Signal Amplification: Perform a series of sequential wash and amplification steps according to the kit protocol. This typically involves:
- Hybridization of the pre-amplifier.
- Hybridization of the amplifier.
- Hybridization of the label probes (enzymatic or fluorescent). Each hybridization step is followed by stringent washes to remove unbound reagents.
- Signal Detection:
- For chromogenic detection: Develop the signal using an enzyme substrate (e.g., DAB for brown stain). Counterstain with hematoxylin, dehydrate, and mount with a permanent mounting medium.
- For fluorescent detection: Apply a fluorescent label, counterstain with DAPI or another nuclear stain, and mount with an anti-fade mounting medium.
Data Interpretation and Quantification
- Scoring: RNAscope results are evaluated semi-quantitatively by counting the number of punctate dots per cell, not by signal intensity [12]. Compare the target gene signal with the positive control (PPIB) and negative control (dapB). A successful assay has a PPIB score ≥2 and a dapB score <1 [12].
- Quantification: Single-molecule signals can be quantified manually or using automated image analysis software such as HALO, QuPath, or ImageJ [5] [12].
The 'double Z' probe design and its accompanying signal amplification system represent a fundamental technological breakthrough in the field of in situ hybridization. By enforcing a dual-recognition requirement for signal generation, the RNAscope platform achieves an exceptional signal-to-noise ratio that enables the visualization of individual RNA molecules within the morphological context of tissues. When framed within the context of sensitivity research, this core mechanism provides a definitive explanation for RNAscope's superior performance over conventional ISH methods, particularly for low-abundance targets, multiplexed analyses, and challenging sample types like FFPE tissues. For researchers and drug development professionals, understanding this mechanism is key to leveraging its full potential in spatial biology and molecular pathology.
This technical guide provides a comprehensive analysis of the performance metrics that establish RNAscope in situ hybridization (ISH) as a superior technology for RNA detection compared to conventional methods. Through systematic evaluation of peer-reviewed studies and technical data, we quantify the substantial enhancements in sensitivity and specificity that RNAscope delivers. The patented double Z-probe design enables single-molecule visualization while preserving tissue morphology, achieving near-perfect sensitivity and specificity rates of up to 100% in controlled studies. This whitepaper details the experimental protocols, validation methodologies, and quantitative benchmarks that demonstrate RNAscope's transformative impact across research and clinical diagnostic applications, particularly in biomarker development, gene therapy analysis, and spatial transcriptomics.
The accurate in situ detection of RNA molecules has presented persistent challenges in molecular pathology and research. Conventional RNA ISH techniques, utilizing digoxigenin (DIG)-labeled or radioactive probes, have been hampered by significant limitations including insufficient sensitivity for low-abundance targets, high background noise from non-specific binding, and inability to provide reliable single-cell resolution data [13]. Before advanced ISH technologies emerged, researchers primarily relied on techniques like Northern blotting and PCR-based methods that require RNA extraction, resulting in loss of spatial context and potential degradation of labile RNA molecules [13].
The introduction of RNAscope in 2012 represented a paradigm shift in RNA visualization, bringing a novel ISH technology with patented probe design that simultaneously delivers signal amplification and background suppression [13] [10]. This technical guide examines the key performance metrics that quantify RNAscope's improvements over conventional methods, providing researchers with comprehensive data to inform their experimental design and technology selection for RNA detection applications.
Core Principle and Mechanism
RNAscope employs a unique double Z-probe design that fundamentally differs from conventional linear probes used in traditional ISH. This design features two distinct probe segments ("Z probes") that bind adjacent regions on the target RNA, creating a docking site for pre-amplifier molecules only when both probes hybridize correctly [13]. The requirement for this dual hybridization is the foundation of RNAscope's exceptional specificity, as it virtually eliminates false-positive signals from non-specific binding.
The signal amplification system employs a multi-level branching DNA architecture that generates substantial signal amplification without increasing background noise. Each successfully bound Z-probe pair can accommodate multiple pre-amplifier molecules, each of which in turn binds numerous amplifier molecules, finally recruiting many label-bearing probes [13]. This cascading amplification can generate up to 8,000-fold signal intensification per RNA molecule, enabling visualization of individual transcripts as distinct dots under standard microscopy [13].
Assay Portfolio for Diverse Applications
The RNAscope technology platform comprises multiple assay formats optimized for different target types and applications:
- RNAscope: Designed for standard mRNA targets longer than 300 nucleotides, enabling detection of coding genes, non-coding RNAs, and viral sequences [14]
- BaseScope: Optimized for shorter targets of 50-300 nucleotides, ideal for detecting splice variants, point mutations, and highly homologous sequences [14] [15]
- miRNAscope: Specialized for small RNA detection (17-50 nucleotides) including microRNAs, siRNAs, and ASOs [14]
- ProximityScope: Engineered to detect functional protein interactions through proximity-dependent signal generation [14]
Table 1: RNAscope Technology Portfolio and Applications
| Assay Type | Target Length | Primary Applications | Key Advantages |
|---|---|---|---|
| RNAscope | >300 nucleotides | mRNA, lncRNA, viral RNA, bacterial RNA | Single-molecule sensitivity, high multiplexing capability |
| BaseScope | 50-300 nucleotides | Splice variants, point mutations, short homologous sequences | Enhanced sensitivity for short targets |
| miRNAscope | 17-50 nucleotides | miRNAs, siRNAs, ASOs | Specialized for small RNA detection |
| ProximityScope | Protein complexes | Protein-protein interactions | Proximity-dependent signaling |
Comparative Performance Metrics
Sensitivity and Specificity Benchmarks
A systematic review conducted in 2021 evaluated RNAscope against established gold standard methods across human tissue samples, providing robust quantitative performance data [13]. The analysis encompassed 27 retrospective studies, primarily in cancer samples, comparing RNAscope with immunohistochemistry (IHC), quantitative PCR (qPCR), quantitative reverse transcriptase PCR (qRT-PCR), and DNA ISH.
The results demonstrated that RNAscope achieves exceptionally high concordance with molecular detection methods, with concordance rates ranging from 81.8% to 100% when compared to qPCR, qRT-PCR, and DNA ISH [13]. This near-perfect alignment with amplification-based methods highlights RNAscope's exceptional sensitivity in detecting RNA targets within their native tissue context.
The comparison with IHC revealed more variable concordance (58.7%-95.3%), reflecting the fundamental differences in what each technology measures [13]. This discrepancy underscores that RNAscope detects RNA transcripts while IHC detects protein products, capturing different stages of the gene expression pathway. The lower concordance ranges likely reflect post-transcriptional regulation events and technical limitations of antibody-based detection.
Quantitative Comparison with Conventional ISH
Traditional RNA ISH methods have been limited by poor sensitivity and high background noise, restricting their application to highly expressed targets. The unique double Z-probe design of RNAscope fundamentally addresses these limitations through several mechanisms:
- Background Suppression: The requirement for simultaneous binding of two adjacent probes dramatically reduces non-specific signal generation
- Signal Amplification: The branched DNA amplification system enables detection of individual RNA molecules without radioactive labels
- Single-Molecule Resolution: Each detected dot represents a single RNA molecule, enabling precise quantification at cellular and subcellular levels [10]
The systematic review confirmed that RNAscope achieves "highly sensitive and specific method" status, with performance characteristics surpassing conventional ISH across all evaluated parameters [13].
Table 2: Performance Comparison Between RNAscope and Conventional Methods
| Performance Metric | RNAscope | Conventional ISH | IHC | qPCR/qRT-PCR |
|---|---|---|---|---|
| Sensitivity | Up to 100% [13] | Limited to highly expressed genes [13] | Variable (antibody-dependent) | High (but loses spatial context) |
| Specificity | Up to 100% [13] | Moderate to low (high background) [13] | Variable (antibody-dependent) | High |
| Spatial Context | Excellent (single-cell resolution) | Moderate (limited by sensitivity) | Excellent (single-cell resolution) | None (tissue homogenate) |
| Target Range | Any RNA >50 nt | Limited by sensitivity constraints | Protein targets only | Any RNA (degrades during extraction) |
| Quantification Capability | Direct transcript counting | Semi-quantitative | Semi-quantitative | Highly quantitative |
Experimental Protocols and Methodologies
Standardized Workflow for Consistent Results
The RNAscope assay follows a standardized three-step workflow that can be applied to various sample types including formalin-fixed paraffin-embedded (FFPE) tissues, fresh frozen tissues, and fixed cells [13]. The protocol consists of three critical stages after sample preparation:
- Permeabilization: Controlled enzymatic treatment to enable probe access while preserving RNA integrity and tissue morphology
- Hybridization: Target-specific double Z-probes are hybridized to the RNA of interest
- Signal Amplification: Multi-step amplification process builds the detection complex for visualization
The entire process can be completed in approximately 6 hours for fluorescent detection and 3 hours for chromogenic detection, significantly faster than traditional ISH protocols which often require overnight hybridization [13]. The workflow is compatible with automated platforms including Leica BOND RX, Roche DISCOVERY ULTRA, and Lunaphore COMET systems, enabling standardization and high-throughput applications [14].
Quality Control Measures
RNAscope incorporates rigorous control systems to validate assay performance:
- Positive Control Probes: Target housekeeping genes with known expression patterns (PPIB for moderate expression, Polr2A for low expression, UBC for high expression) to verify RNA integrity and assay functionality [13]
- Negative Control Probes: Utilize the bacterial dapB gene, which should not hybridize to human or animal tissues, to assess background noise and non-specific binding [13]
These controls are essential for interpreting experimental results and ensuring data reliability, particularly in clinical diagnostic applications where false positives or negatives could impact patient management decisions.
Diagram 1: RNAscope Experimental Workflow with Quality Control
Quantitative Analysis Methods
RNAscope enables precise quantification of RNA molecules through distinct dot enumeration, with each dot representing an individual RNA transcript [13]. Analysis can be performed through multiple approaches:
- Manual Scoring: Counting dots in multiple representative regions following manufacturer guidelines
- Computational Analysis: Utilizing specialized software platforms including Halo, QuPath, and Aperio for automated dot recognition and quantification [13] [16]
For automated analysis, whole slide scanning generates comprehensive digital images that software algorithms process to identify and count signals while accounting for tissue architecture and cell boundaries. The open-source software QuPath has been specifically adapted for RNAscope analysis in complex tissues like brain, incorporating custom scripts for cell detection parameter optimization and mRNA signal thresholding using negative controls [16].
Research Applications and Validation Studies
Validation of High-Throughput Transcriptomic Data
RNAscope has emerged as a powerful validation tool for high-throughput transcriptomic technologies including RNA sequencing (RNA-Seq), microarrays, and NanoString nCounter analyses [17]. While these technologies generate comprehensive expression data, they typically lack spatial context and require confirmation through orthogonal methods.
In multiple published studies, RNAscope has confirmed RNA-Seq findings while adding crucial spatial information. For example, Silberstein et al. used RNAscope to validate IL18 expression proximal to transplantation sites in single-cell RNA-Seq experiments investigating stem cell niche regulators [17]. Similarly, Chen et al. applied RNAscope to validate NanoString nCounter results demonstrating LINC00473 association with LKB1 inactivation in lung cancer, providing a potential surrogate biomarker where antibody-based LKB1 detection showed poor specificity [17].
Antibody Validation and Alternative Detection
The technology serves as a robust orthogonal method for validating antibody specificity, addressing growing concerns about antibody reliability in research and diagnostics [18]. With an estimated 50% of commercial antibodies demonstrating insufficient specificity for their intended applications, RNAscope provides an mRNA-based validation standard that is not subject to the same epitope-dependent limitations as IHC.
Researchers have successfully employed RNAscope to resolve ambiguous IHC results, with one group reporting that "We actually tested 13 different antibodies with different conditions and didn't get trustworthy results - so the RNAscope assay saved us" [18]. The technology is particularly valuable for detecting targets where antibodies perform poorly, including low-abundance antigens, secreted proteins, and targets without available antibodies [14].
Essential Research Reagent Solutions
The successful implementation of RNAscope technology requires specific reagent systems and analytical tools. The following table details essential components for establishing RNAscope in a research setting.
Table 3: Essential Research Reagent Solutions for RNAscope Implementation
| Reagent/Tool | Function | Application Notes |
|---|---|---|
| Target-Specific Probes | Hybridize to RNA of interest | Designed for targets >300 nt; custom designs available for any species |
| Positive Control Probes | Verify assay performance | PPIB (moderate expression), Polr2A (low expression), UBC (high expression) |
| Negative Control Probes | Assess background signal | Bacterial dapB gene for human/animal tissues |
| Detection Kits | Signal generation | Chromogenic (brightfield) or fluorescent (multiplexing) options |
| Protease Reagents | Tissue permeabilization | Optimized for FFPE, frozen, or cell samples |
| Hybridization Buffers | Enable specific probe binding | Stringency-controlled for optimal signal-to-noise |
| Amplification Reagents | Signal amplification | Multi-step system for 8000x signal enhancement |
| Analysis Software | Image quantification | Halo, QuPath, or Aperio for automated dot counting |
Discussion and Future Perspectives
The quantitative performance metrics establish RNAscope as a transformative technology for in situ RNA analysis, delivering substantial improvements in sensitivity, specificity, and reproducibility compared to conventional ISH methods. The systematic review data confirms that RNAscope achieves concordance rates exceeding 80% with gold standard molecular techniques while providing unparalleled spatial context preservation [13].
While the evidence supporting RNAscope's analytical performance is robust, the systematic review notes that "there were not enough data to suggest that RNAscope could stand alone in the clinical diagnostic setting," indicating need for further prospective studies to validate diagnostic accuracy values in compliance with regulatory standards [13]. Additional cost-benefit analyses would strengthen the case for widespread clinical adoption.
Future developments will likely expand RNAscope's capabilities in spatial transcriptomics, particularly through enhanced multiplexing approaches. The compatibility with automated platforms and standardized analysis pipelines will further improve reproducibility and throughput, positioning RNAscope as an essential tool in both basic research and translational applications.
For research and drug development professionals, RNAscope offers a validated platform with quantified performance advantages that justify its implementation for critical applications including biomarker development, gene therapy evaluation, and diagnostic assay development. The technology's ability to provide spatially resolved, single-cell expression data with high sensitivity and specificity makes it uniquely positioned to address fundamental questions in biology and disease pathogenesis.
The accurate detection of RNA within intact tissues using in situ hybridization (ISH) has been historically plagued by two fundamental obstacles: the inherent instability of RNA molecules and the persistent background noise that complicates interpretation. RNA is notoriously susceptible to degradation by ubiquitous RNases, a problem exacerbated in archival formalin-fixed paraffin-embedded (FFPET) tissues where formalin fixation causes RNA cross-linking and fragmentation [19]. Concurrently, conventional ISH methods often suffer from high background noise due to non-specific probe binding, which obscures true signal and undermines assay specificity and reliability. This technical guide examines the evolution of the RNAscope assay, detailing the proprietary technologies that empower it to overcome these enduring challenges, thereby providing sensitive and specific single-molecule detection of RNA within its morphological context.
Core Technology: The RNAscope Assay Design
The RNAscope technology is built upon a foundational innovation that simultaneously addresses both sensitivity and specificity: the proprietary double Z (ZZ) probe design [20] [21].
Probe Design and Signal Amplification
This design employs pairs of "Z" probes that must bind adjacent to each other on the same target RNA molecule for signal amplification to initiate. This paired binding requirement is the first critical barrier against non-specific hybridization. The complete RNAscope probe is composed of a longer sequence that hybridizes to the target RNA, flanked by two tail sequences that serve as binding sites for pre-amplifiers [22]. This architecture forms the basis for a branched DNA (bDNA) signal amplification system.
- Specificity Layer: The double Z probe design ensures that a full signal is generated only when two independent probe fragments bind correctly to their target sequences in close proximity. This dramatically reduces background from probes binding to off-target sequences with partial complementarity [21].
- Amplification Layer: Once hybridized, the ZZ probes create a scaffold for the sequential binding of pre-amplifier and amplifier molecules, ultimately culminating in the binding of enzyme-labeled oligonucleotides (e.g., horseradish peroxidase). Each target RNA molecule can then be visualized as a distinct, punctate dot after the enzyme catalyzes a chromogenic or fluorescent reaction [20] [23].
This combination of signal amplification and simultaneous background suppression is the cornerstone of the technology, enabling single-molecule detection with a high signal-to-noise ratio [24] [21].
Quantitative Performance and Experimental Validation
The performance of RNAscope, particularly in challenging sample types like FFPE tissues, has been rigorously quantified. A 2025 systematic study assessed RNA-FISH signal integrity in 62 archived breast cancer samples (30 FFPETs and 32 Fresh Frozen Tissues, FFTs) using the RNAscope Multiplex Fluorescent v2 assay [19].
Impact of Archival Duration on RNA Detection
The study confirmed that RNAscope signals in FFPETs are lower than in FFTs in an archival duration-dependent fashion [19]. However, the degradation was not uniform across all RNA species. It was most pronounced in high-expressing housekeeping genes (HKGs) like UBC and PPIB, compared to low-to-moderate expressors like POLR2A and HPRT1 (p < 0.0001) [19]. This highlights that while the probes are designed to detect fragmented RNA, the initial expression level of the target influences signal retention over time.
Table 1: RNAscope Signal Degradation in FFPE Tissues Over Time (Adapted from [19])
| Housekeeping Gene (HKG) | Expression Level Category | Impact of Archival Duration on Signal | Quantitative Degradation (R² value) |
|---|---|---|---|
| PPIB | High Expressor | Most Pronounced Degradation | R² = 0.35 (Adjusted Transcript) |
| UBC | High Expressor | Significant Degradation | Data not specified in excerpt |
| POLR2A | Low-to-Moderate Expressor | Less Affected | Less significant than high expressors |
| HPRT1 | Low-to-Moderate Expressor | Less Affected | Less significant than high expressors |
Protocol for Sample Quality Assessment
The study underscores the necessity of performing a sample quality check using a panel of HKGs before analyzing target genes of interest [19]. The recommended experimental protocol is as follows:
- Sample Selection & Sectioning: Select FFPET blocks with >50% target cells (e.g., cancer cells) to minimize bias. Cut tissue sections at 5 ±1 μm for FFPET and mount on charged slides (e.g., Fisher Scientific SuperFrost Plus) [25] [19].
- Pretreatment:
- RNAscope Multiplex Fluorescent Assay:
- Use the RNAscope Multiplex Fluorescent v2 Kit.
- Hybridize with a panel of positive control HKG probes (e.g., UBC, PPIB, POLR2A, HPRT1) and the negative control bacterial dapB probe [19].
- Perform sequential signal amplification and development with fluorophores (e.g., Opal 520, 570, 620, 690) [22] [19].
- Image Acquisition & Analysis:
- Quality Control Criteria: Successful staining and adequate RNA quality are indicated by a PPIB/POLR2A score ≥2 or UBC score ≥3, and a dapB score <1 [25].
Table 2: The Scientist's Toolkit: Essential Reagents for RNAscope Multiplex Assays
| Item Category | Specific Examples & Catalog Numbers | Critical Function |
|---|---|---|
| Core Detection Kit | RNAscope Multiplex Fluorescent Reagent Kit v2 (Cat. No. 323100) | Provides essential pretreatment reagents, detection reagents, and buffers for the assay workflow. |
| Control Probes | Positive Control: PPIB, POLR2A, UBC; Negative Control: dapB (Cat. No. 320871) | Validate assay performance and RNA quality; distinguish specific signal from background noise. |
| Target Probes | Catalog or Made-to-Order C1-C4 Probes (For Multiplex Assays) | Gene-specific probes designed with the double-Z architecture for targeted RNA detection. |
| Fluorophores | TSA Vivid Dyes (ACD) or Opal Dyes (Akoya Biosciences) | Fluorophores for signal visualization, assigned to different probe channels (C1-C4). |
| Supporting Equipment | HybEZ Hybridization System; Fluorescent Microscope with DAPI/FITC/TRITC/Cy5 filters | Provides a controlled environment for hybridization and enables high-resolution image capture. |
Advanced Workflows and Evolving Applications
The core RNAscope technology has evolved to support increasingly sophisticated research applications through specialized workflows and robust analytical support.
Protease-Free Workflow for Sensitive Epitopes
A significant recent advancement is the development of a protease-free workflow, now available on automated platforms like the Roche DISCOVERY ULTRA [26]. This workflow is crucial for experiments combining RNAscope ISH with immunohistochemistry (IHC) or immunofluorescence (IF) to detect proteins with protease-sensitive epitopes. By eliminating the protease digestion step that could destroy sensitive protein antigens, this workflow enables seamless spatial multiomics, allowing researchers to visualize RNA and protein targets concurrently in the same tissue section with unparalleled morphological context [26].
Professional Image Analysis and Quantification
To transform the distinct punctate dots generated by RNAscope into quantitative data, both semi-quantitative and fully quantitative digital image analysis methods are employed [23]. Bio-Techne's Professional Assay Services (PAS) leverages powerful tools like HALO software to provide comprehensive quantitative analysis [9]. This includes:
- Spatial Biology Analysis: Quantifying gene expression within specific cell types or tissue compartments.
- Custom Data Outputs: Tailoring analysis to specific research needs, such as validating single-cell RNA sequencing data or performing efficacy assessments for therapeutic development [9].
RNAscope technology represents a paradigm shift in RNA in situ hybridization. Its proprietary double Z probe design fundamentally overcomes the traditional limitations of RNA instability and background noise by ensuring that signal amplification occurs only upon specific hybridization. The technology's robustness is validated by its ability to provide quantifiable, single-molecule data even from suboptimal archived FFPE samples, as demonstrated by rigorous, recent research. With ongoing innovations such as protease-free automated workflows and sophisticated image analysis solutions, RNAscope continues to evolve, solidifying its role as an indispensable tool for researchers and drug development professionals pursuing precise spatial gene expression analysis.
Practical Implementation: RNAscope Workflows, Assay Types, and Research Applications
This guide details the standardized workflow for the RNAscope in situ hybridization (ISH) assay, a highly sensitive method that enables single-molecule RNA visualization within an intact morphological context. The protocol is framed within research comparing its sensitivity to conventional ISH, highlighting key methodological advancements that allow for precise, quantitative spatial biology analysis.
RNAscope is a novel RNA ISH technology that employs a unique probe design strategy, enabling simultaneous signal amplification and background suppression to achieve single-molecule visualization while preserving tissue morphology [10]. This represents a significant advancement over conventional ISH methods.
The table below summarizes the core comparative advantages:
- Table 1: Key Technical Advantages of RNAscope over Conventional ISH
| Feature | RNAscope | Conventional ISH | Impact on Sensitivity and Specificity |
|---|---|---|---|
| Probe Design | Proprietary ZZ probe pairs targeting a single RNA molecule [10] | Traditional single-stranded DNA or RNA probes | Preers nonspecific probe binding and suppresses background [10] |
| Signal Amplification | Proprietary multistep amplification [10] | Limited or no amplification | Enables single-molecule detection without increased noise [10] |
| Sample Compatibility | Optimized for routine FFPE tissues [10] [19] | Often challenging for FFPE due to RNA cross-linking | Reliable performance on the most common clinical archives [19] |
| Assay Time | Can be completed within a single day [27] | Often requires multiple days [10] | Higher throughput and efficiency |
| Quantification | Single RNA molecules appear as distinct, quantifiable punctate dots [27] [8] | Often diffuse signal, challenging to quantify | Enables precise manual or digital image analysis [27] |
Figure 1: The complete RNAscope workflow, from sample preparation to quantitative analysis. Key steps like protease digestion and probe hybridization are critical for assay performance [27] [28] [19].
Detailed Experimental Protocols
Sample Preparation and Pre-treatment
Proper sample preparation is the foundation for a successful RNAscope assay. The protocol varies based on tissue type and archival method.
For Fresh-Frozen Tissues (e.g., Rodent Brain):
- Perfusion & Collection: Deeply anesthetize the animal and perform transcardial perfusion with ice-cold PBS followed by 4% Paraformaldehyde (PFA). Rapidly remove the tissue [8].
- Snap-Freezing: Drop the tissue into chilled 2-methylbutane (-30°C to -40°C) for 20-30 seconds to snap-freeze. Avoid thawing and store at -80°C [8].
- Sectioning: Embed tissue in O.C.T. compound and section at a 10-20 µm thickness using a cryostat. Mount sections on charged slides (e.g., Superfrost Plus) [8].
- Fixation: Fix slides in 4% PFA at room temperature for 20 minutes [19].
For Formalin-Fixed Paraffin-Embedded (FFPE) Tissues:
- Fixation: Fix tissue in 10% Neutral Buffered Formalin (NBF) for 12-24 hours. Prolonged fixation can compromise RNA integrity [19].
- Processing & Embedding: Process through graded alcohols and xylene using standard histology protocols, then embed in paraffin.
- Sectioning: Section at 4-5 µm thickness and mount on charged slides.
Critical Pre-treatment for FFPE Sections:
- Baking: Bake slides at 60°C to melt paraffin and ensure adhesion.
- Deparaffinization: Immerse slides in xylene and graded ethanol series to remove paraffin.
- Antigen Retrieval: Incubate slides in RNAscope Target Retrieval Reagent at 98-102°C for 15-30 minutes [19]. This reverses formalin-induced cross-links.
- Protease Digestion: Apply Protease III or IV to the sections for 15-30 minutes at 40°C. This step is critical: under-digestion results in low signal and background, while over-digestion damages morphology and RNA [28].
Special Protocol for Calcified Tissues (e.g., Bone, Teeth): Decalcification is required but can severely damage RNA. A 2025 study on mouse teeth identified optimal methods [29].
- Fixation: Perfuse and post-fix intact jaw segments in 4% PFA for 72 hours.
- Decalcification: Use a gentle decalcification agent. The study found ACD Bone Decalcification Buffer or Morse's Solution (10% sodium citrate, 20% formic acid) for 12 hours at 4°C best preserved RNA integrity for RNAscope in dental pulp [29].
- Post-decalcification Processing: Proceed with standard dehydration, embedding, and sectioning as for FFPE tissues.
Probe Hybridization and Signal Amplification
This is the core of the RNAscope assay, performed in a HybEZ Oven to control temperature and humidity, which are critical for performance [28].
- Probe Hybridization: Apply the target-specific RNAscope probe mixture to the tissue section and incubate at 40°C for 2 hours. The proprietary ZZ probes bind specifically to the target RNA [10].
- Signal Amplification: A series of sequential amplifier molecules are hybridized to the bound probes. This multi-step cascade achieves significant signal amplification, with each target RNA molecule generating a distinct punctate dot [10].
Signal Detection, Visualization, and Quantitative Analysis
The amplified signal can be detected using chromogenic or fluorescent methods.
- Chromogenic Detection: Uses enzymes that precipitate a colored dye (e.g., brown or red) at the signal site. Suitable for bright-field microscopy and compatible with standard histology workflows [10].
Fluorescent Detection: Uses fluorescent dyes (Opal dyes) for multiplexing. Allows simultaneous detection of 2-4 different RNA targets in the same section using different channels (C1-C4) [19]. Prolonged storage in PFA can quench fluorescence, particularly in green channels [19].
Quantitative Image Analysis: RNAscope signals are inherently quantifiable. Each punctate dot represents a single RNA molecule [27].
- Manual Counting: Researchers can count dots per cell directly under a microscope.
- Digital Image Analysis: Automated analysis using software like HALO (Indica Labs) or QuPath is recommended for robustness and reproducibility [27] [9] [8].
- Spatial Biology: Advanced analysis can quantify signal proximity to histological landmarks or co-expression with protein markers (via combined IHC) for deep spatial biology insights [9] [30].
Figure 2: RNAscope signal amplification mechanism. The proprietary multi-step process ensures that only a full stack of amplifiers results in a detectable signal, providing high specificity and single-molecule sensitivity [10].
The Scientist's Toolkit: Essential Research Reagent Solutions
The following table lists key materials and equipment required to perform the RNAscope assay.
- Table 2: Essential Reagents and Equipment for the RNAscope Workflow
| Item | Function | Example/Supplier |
|---|---|---|
| RNAscope Reagent Kit | Core reagents for hybridization, amplification, and detection (e.g., Multiplex Fluorescent v2). | Advanced Cell Diagnostics (ACD) [19] [8] |
| Target Probes | Species-specific probes for genes of interest; available as ready-to-use (C1 channel) or 50X stocks (C2-C4). | ACD [28] [8] |
| Control Probes | Positive (e.g., PPIB, POLR2A) and negative (e.g., DapB) controls to validate assay performance. | ACD [28] [19] |
| HybEZ Oven | Validated hybridization oven providing precise temperature and humidity control. Critical for consistent results. | ACD [27] [28] |
| Protease Reagents | Protease III or IV for tissue permeabilization; digestion time is a critical optimization point. | ACD [28] [8] |
| Decalcification Buffers | For hard tissues; ACD Buffer or Morse's Solution are recommended for RNA preservation [29]. | Sigma-Aldrich, ACD [29] |
| Image Analysis Software | For quantitative analysis of punctate dots (e.g., cell detection, dot counting). | HALO, QuPath [27] [9] [8] |
Methodological Validation and Best Practices
Ensuring Specificity and Sensitivity
Robust validation is essential. Key steps include:
- Use of Controls: Always run positive control probes (housekeeping genes like PPIB, UBC) and negative control probes (bacterial DapB) concurrently with target probes [19]. This controls for RNA integrity and assay specificity.
- Threshold Determination: Establish mRNA signal thresholds for defining "positive" cells using negative controls. This is crucial for reproducible quantification, as detailed in QuPath protocols [8].
Troubleshooting Common Issues
- Low or No Signal: Can result from under-fixation, over-fixed/over-crosslinked RNA (in FFPE), insufficient protease digestion, or use of an unvalidated hybridization oven [28] [19].
- High Background: Often caused by under-digestion with protease or tissue drying during the procedure. Ensure the hydrophobic barrier remains intact and reagents are fresh [28].
- Poor Morphology: Typically a result of over-digestion with protease [28].
The RNAscope technology, with its standardized workflow and compatibility with automated staining systems [27], provides a reliable and highly sensitive method for RNA detection in situ, firmly establishing its superiority over conventional ISH for both research and clinical applications [10] [31] [30].
In situ hybridization (ISH) has long been a cornerstone technique for visualizing genetic material within the context of intact tissues, preserving crucial spatial information that is lost in homogenized sample analysis. However, conventional ISH methods face significant technical challenges, including limited sensitivity, specificity issues, and difficulty in multiplexing. The emergence of RNAscope technology represents a paradigm shift in molecular detection, employing a novel "double Z" probe design that enables simultaneous signal amplification and background suppression [11]. This proprietary technology allows for single-molecule RNA detection visualized as unambiguous punctate dots under a microscope, achieving both high sensitivity and high specificity that far surpasses conventional ISH methods [11].
The development of an extensive assay portfolio addressing chromogenic, fluorescent, multiplex, and protease-free applications provides researchers with unprecedented flexibility for diverse experimental needs. This technical guide examines the core components of this portfolio within the broader context of comparative sensitivity research between RNAscope and conventional ISH methodologies. For researchers and drug development professionals, understanding these advanced detection options is critical for designing robust experimental workflows that yield publication-quality, reproducible results across various tissue types and research applications, from basic investigation to clinical translation.
Core Technology: RNAscope vs. Conventional ISH
Fundamental Mechanism and Sensitivity Advantages
RNAscope's superior performance stems from its proprietary probe design and signal amplification system, which fundamentally differs from conventional ISH approaches. The core innovation involves a pairwise probe design consisting of "double Z" probes that bind adjacent to each other on the target RNA sequence. Only when both "Z" probes bind correctly can pre-amplifier molecules attach, initiating a branching DNA amplification system that generates a strong, specific signal while suppressing background noise [11]. This methodological advancement enables researchers to achieve single-molecule sensitivity with spatial resolution that preserves crucial tissue context.
Traditional ISH methods rely on single linear probes that often produce high background signals due to non-specific binding, requiring extensive optimization and resulting in inconsistent sensitivity. The technical limitations of conventional ISH become particularly problematic for low-abundance targets, partially degraded RNA (common in FFPE tissues), and challenging tissue types such as calcified specimens [29]. In contrast, RNAscope's signal amplification system demonstrates robust performance even in difficult conditions, as evidenced by its successful application in decalcified bone and dental pulp tissues where conventional ISH often fails [29].
Quantitative Performance Comparison
Table 1: Comparative Analysis of RNAscope vs. Conventional ISH
| Feature | RNAscope Technology | Conventional ISH |
|---|---|---|
| Sensitivity | Single-molecule detection [11] | Limited, varies by target abundance |
| Specificity | High (double Z-probe design suppresses background) [11] | Variable, often significant background |
| Signal-to-Noise Ratio | Superior due to proprietary amplification [11] | Often compromised |
| Publication Record | Cited in >10,000 publications [11] | Established but less robust for novel targets |
| Success with Challenging Tissues | Effective on decalcified tissues with proper processing [29] | Problematic, especially with calcified tissues |
| Automation Compatibility | High (compatible with Leica Bond RX, Roche DISCOVERY ULTRA) [31] [32] | Limited, often manual protocols |
| Multiplexing Capability | Advanced (up to 12-plex demonstrated) | Limited to simple duplex approaches |
Chromogenic Detection Assays
Technical Principles and Applications
Chromogenic detection represents a foundational methodology in the RNAscope assay portfolio, utilizing enzyme-mediated color precipitation to visualize target RNA within tissue architecture. This approach employs an enzyme (typically horseradish peroxidase or alkaline phosphatase) conjugated to a secondary antibody, which cleaves a colorless substrate to produce a colored precipitate at the site of target RNA localization [33]. The resulting stained slides can be viewed with standard brightfield microscopy, making the technology accessible to laboratories without specialized fluorescence imaging equipment.
The chromogenic detection format is particularly valuable for determining definitive positive versus negative results in diagnostic applications and clinical pathology workflows [34]. The permanent nature of chromogenic staining creates archival-quality slides resistant to photobleaching, allowing long-term storage and retrospective analysis [34]. This advantage is particularly important for clinical trials, biobanking studies, and regulatory submissions where sample integrity over time is essential. Recent advancements have focused on integrating chromogenic RNAscope assays with automated staining platforms such as the Roche DISCOVERY ULTRA and Leica Bond RX systems, significantly enhancing reproducibility across operators and institutions while reducing hands-on technical time [31] [32].
Experimental Protocol for Chromogenic Detection
The standard RNAscope chromogenic detection protocol begins with tissue fixation in 4% paraformaldehyde for 24 hours, followed by paraffin embedding using standard histological processing. For calcified tissues, decalcification requires careful optimization; recent research indicates that ACD decalcification buffer and Morse's solution (10% sodium citrate with 20% formic acid) best preserve RNA integrity in mouse dental pulp samples during a 12-hour to 2-week decalcification period at 4°C [29]. Tissue sections are cut at 4-5μm thickness and mounted on charged slides, followed by baking at 60°C for 1 hour to ensure adhesion.
After deparaffinization and rehydration, slides undergo antigen retrieval using target retrieval solution heated to 95-100°C for 15 minutes. Protease digestion follows, with incubation time optimized for tissue type and fixation duration (typically 30 minutes at 40°C). The RNAscope probes are then applied and hybridized for 2 hours at 40°C in a HybEZ oven, followed by a series of signal amplification steps according to manufacturer specifications. Chromogenic development uses DAB (brown) or Fast Red (red) substrates for 10-30 minutes, followed by counterstaining with hematoxylin and mounting with permanent media [29] [35]. The entire process can be completed within one working day, a significant improvement over conventional ISH methods that often required multiple days of hybridization and development.
Fluorescent Detection Assays
Technical Principles and Advantages
Fluorescent RNAscope assays employ fluorophore-conjugated secondary antibodies rather than enzyme-mediated chromogenic precipitation, enabling simultaneous detection of multiple RNA targets within the same tissue section. This detection mechanism operates through direct fluorescence, where fluorophores excited by light of a specific wavelength emit light at a longer wavelength, generating signals detectable with fluorescence microscopy [33]. The multiplexing capability of fluorescent detection represents a significant advancement for complex biological investigations requiring analysis of multiple genes or pathways within preserved tissue architecture.
The higher dynamic range of fluorescent detection makes it particularly suitable for quantitative applications comparing high and low abundance transcripts [34]. This technical advantage enables researchers to perform sophisticated co-localization studies and quantitative image analysis that would be challenging with chromogenic methods. Furthermore, fluorescent detection forms the foundation for advanced spatial biology applications, including integration with digital pathology and artificial intelligence-based image analysis platforms that can automatically identify cell types, subcellular localization patterns, and complex tissue microenvironments based on multiplexed RNA expression profiles [34].
Experimental Protocol for Fluorescent Detection
The fluorescent RNAscope protocol shares initial steps with the chromogenic method through the hybridization and amplification stages, diverging at the detection phase. Following probe hybridization and amplification, fluorophore-conjugated labels (such as FITC, Cy3, Cy5, or Texas Red) are applied sequentially for multiplexed detection. Between each fluorophore application, slides may be treated with heating or chemical reagents to remove previous probes, enabling sequential hybridization of multiple target sequences on the same tissue section.
For optimal results in fluorescent detection, tissue autofluorescence should be minimized through treatment with TrueBlack or similar reagents before coverslipping with anti-fade mounting media [34]. Image acquisition requires a fluorescence microscope equipped with appropriate filter sets and a camera system capable of detecting the specific fluorophores employed. For multiplex experiments exceeding three colors, spectral imaging systems with linear unmixing capabilities are recommended to address fluorophore bleed-through between channels. It is crucial to note that unlike chromogenic stains, fluorescent signals are susceptible to photobleaching and require protected storage at 4°C in darkness, with imaging completed within weeks of slide preparation [34].
Multiplex Assay Platforms
Technical Implementation and Strategic Applications
Multiplex RNAscope technology enables simultaneous detection of multiple RNA targets within a single tissue section, providing profound insights into complex biological systems by preserving spatial relationships between different cell types and molecular pathways. The technical implementation utilizes a sequential hybridization approach where individual RNA targets are detected, imaged, and then the signals are chemically removed before proceeding to the next target, all while preserving tissue integrity and RNA quality [34]. This sophisticated approach can currently enable detection of up to 12 different targets within the same sample, though most practical applications utilize 3-6 plex designs balanced against experimental complexity and image analysis requirements.
The strategic advantages of multiplexing are substantial and multifaceted. First, it conserves precious tissue resources, particularly critical for rare clinical specimens, small animal models, or biobank samples with limited remaining material [34]. Second, multiplex staining provides greater biological insights by enabling direct visualization of cellular interactions and tissue microenvironments within the architectural context of a single slide, overcoming the interpretive challenges of comparing separate serial sections [34]. Third, the ability to visualize both protein and RNA targets through combined RNAscope and IHC/IF approaches provides a detailed molecular map to explore relationships between gene expression and protein localization [34]. Additional benefits include reduced laboratory time compared to processing multiple single-plex assays and compatibility with advanced digital pathology and AI-based analysis platforms [34].
Chromogenic vs. Immunofluorescent Multiplexing: Selection Criteria
Table 2: Comparison of Multiplex Detection Methodologies
| Parameter | Chromogenic Multiplexing | Immunofluorescent Multiplexing |
|---|---|---|
| Maximumplexity | Lower (typically 2-3 plex due to color resolution limitations) | Higher (up to 12-plex with spectral imaging) |
| Equipment Requirements | Standard brightfield microscope | Fluorescence microscope with multispectral capabilities |
| Signal Longevity | Permanent, resistant to photobleaching [34] | Temporary (weeks), susceptible to photobleaching [34] |
| Quantitative Capability | Lower dynamic range, best for present/absent calls [34] | Higher dynamic range, suitable for quantification [34] |
| Colocalization Analysis | Difficult to resolve multiple markers in same cell [34] | Excellent for colocalization studies with spectral imaging [34] |
| Protocol Optimization | More extensive optimization for color combination [34] | Straightforward color selection but requires imaging optimization |
| Primary Applications | Diagnostic pathology, clinical validation | Research, spatial biology, quantitative studies |
Protease-Free Workflows
Technical Innovation and Applications
The recent introduction of protease-free RNAscope workflows represents a significant technical innovation that overcomes a major limitation in traditional RNA-protein co-detection assays. Conventional ISH methods require protease digestion to permeabilize tissues and make target RNA accessible to probes, but this step often damages protein epitopes, compromising subsequent immunohistochemistry or immunofluorescence detection [32]. The protease-free workflow eliminates this requirement through proprietary tissue pretreatment chemistry that maintains RNA accessibility while preserving protein integrity for superior antibody binding, particularly for protease-sensitive epitopes.
This advancement enables true multi-omics analysis on the same tissue section, allowing researchers to correlate RNA expression with protein localization without methodological compromises. The technical benefits are particularly evident when working with challenging but biologically important epitopes such as CD20, CD4, and PD1, which are frequently degraded in standard protease-dependent protocols [32] [26]. The protease-free workflow has been successfully automated on the Roche DISCOVERY ULTRA platform, providing robust, reproducible performance for critical applications including immune cell profiling, infectious agent detection, antibody specificity validation, and cell/gene therapy tracking [32].
Experimental Protocol for Protease-Free Co-Detection
The protease-free RNAscope protocol begins with standard tissue preparation including fixation in 4% PFA for 24 hours and paraffin embedding. Tissue sections are cut at 4-5μm, mounted on charged slides, and baked at 60°C for 1 hour. After deparaffinization and rehydration, slides undergo the proprietary protease-free pretreatment using the RNAscope Protease-Free Reagent Set instead of conventional protease digestion. This is followed by simultaneous probe hybridization for RNA targets and antibody incubation for protein targets, though sequential approaches may be optimized for specific applications.
For chromogenic detection, HRP-based development precedes AP-based development with careful washing between steps to prevent cross-reactivity. For fluorescent applications, fluorophore-conjugated antibodies and labels are applied with appropriate spectral separation to minimize bleed-through. The entire process can be completed within 24 hours and has been validated on automated platforms including the Roche DISCOVERY ULTRA system, significantly reducing hands-on time while improving inter-assay reproducibility [32] [26]. This streamlined workflow enables researchers to simultaneously interrogate RNA and protein biomarkers while maintaining optimal tissue morphology and detection sensitivity for both analyte classes.
Research Reagent Solutions
Essential Materials for RNAscope Implementation
Table 3: Key Research Reagent Solutions for RNAscope Assays
| Reagent/Category | Function/Application | Examples/Specific Products |
|---|---|---|
| Probe Types | Target-specific RNA detection | RNAscope (500-3000 nt targets), BaseScope (50-300 nt targets) [15] |
| Detection Kits | Signal generation and amplification | RNAscope 2.5 HD Red, RNAscope 2.5 HD Brown [35] |
| Tissue Preparation | Tissue preservation and processing | ACD Decalcification Buffer [29], Protease-Free Pretreatment Reagents [32] |
| Automation Systems | Standardized, reproducible assay performance | Leica Bond RX [11] [31], Roche DISCOVERY ULTRA [32] |
| Platform-Specific Kits | Optimized assays for automated systems | RNAscope Protease-Free Assays for Roche DISCOVERY ULTRA [32] [26] |
| Custom Design Options | Targets not in standard catalog | Custom probe design services [35] |
Successful implementation of RNAscope technology requires appropriate selection of research reagents matched to experimental objectives. The platform offers significant flexibility through both standardized catalog products and custom design options, enabling applications across diverse species, tissue types, and research areas. The availability of specialized reagents for challenging conditions, such as decalcification buffers that preserve RNA integrity in mineralized tissues, significantly expands the technology's applicability [29]. Similarly, the introduction of protease-free workflow reagents demonstrates the platform's ongoing evolution to address emerging research needs in spatial multi-omics [32] [26].
Workflow Integration and Experimental Design
Automated Platform Integration
The integration of RNAscope assays with automated staining platforms has dramatically improved assay reproducibility, throughput, and accessibility for core facilities and clinical laboratories. The technology has been successfully validated on major automated systems including the Leica Bond RX and Roche DISCOVERY ULTRA platforms, enabling standardized protocol execution across multiple operators and sites [11] [32]. This automation capability is particularly valuable for large-scale studies, clinical trials, and diagnostic applications where consistency and reproducibility are paramount.
Automated integration provides several distinct advantages beyond reproducibility. It significantly reduces hands-on technical time by allowing batch processing of slides, with many protocols capable of running unattended overnight [31]. Additionally, automated systems precisely manage critical incubation parameters including time, temperature, and reagent application, minimizing operator-dependent variability [34]. The availability of platform-specific kits, such as the recently introduced protease-free assays for the Roche DISCOVERY ULTRA, further simplifies implementation and ensures optimal performance [32] [26]. These developments have transformed RNAscope from a specialized research technique to a robust solution suitable for regulated environments including clinical diagnostics and drug development pipelines.
Experimental Workflow Visualization
The following workflow diagram illustrates the key decision points and procedural steps in designing and executing a successful RNAscope experiment, incorporating the various assay portfolio options discussed throughout this guide:
The RNAscope assay portfolio provides researchers with an comprehensive toolkit for spatial transcriptomics that dramatically outperforms conventional ISH methodologies in sensitivity, specificity, and application flexibility. The technology's core innovation—the "double Z" probe design—enables single-molecule detection while suppressing background, addressing fundamental limitations that have historically constrained conventional ISH approaches [11]. This technical foundation supports a diverse range of detection formats including chromogenic, fluorescent, multiplex, and protease-free workflows, each optimized for specific research applications and experimental requirements.
Looking forward, the ongoing evolution of RNAscope technology continues to expand its applications in both research and clinical domains. The recent introduction of protease-free workflows for simultaneous RNA and protein detection represents a significant advancement for multi-omics research, while ongoing development of automated protocols enhances reproducibility for diagnostic applications [32] [26]. As spatial biology continues to emerge as a critical discipline for understanding tissue microenvironment biology and complex disease processes, the comprehensive RNAscope assay portfolio provides researchers with a validated, robust platform for generating high-quality, publication-ready data that advances scientific discovery and therapeutic development.
Integrating the highly sensitive RNAscope in situ hybridization (ISH) technology with automated staining platforms represents a significant advancement in molecular pathology, enabling standardized, reproducible detection of RNA biomarkers within their morphological context. This technical guide details optimized protocols for the Roche DISCOVERY ULTRA and Leica BOND RX systems, framing them within a broader thesis on superior sensitivity compared to conventional ISH. The proprietary RNAscope double-Z probe design achieves single-molecule detection sensitivity through a unique signal amplification and background suppression system, overcoming the limitations of traditional ISH techniques [7] [36] [10]. Automated protocol standardization ensures consistent specimen pretreatment, hybridization stringency, and signal development—critical factors for achieving reliable, quantitative data in research and drug development applications. This whitepaper provides researchers, scientists, and drug development professionals with detailed methodologies, performance data, and implementation guidelines to leverage these integrated systems effectively.
Limitations of Conventional ISH and the RNAscope Solution
Traditional RNA ISH methodologies have historically faced significant challenges in achieving consistent sensitivity and specificity required for reliable RNA biomarker detection. These techniques often struggled with high background noise, limited capacity for single-molecule detection, and insufficient sensitivity for low-abundance targets [13] [10]. The RNAscope technology, introduced by Advanced Cell Diagnostics (ACD), addresses these limitations through its patented double-Z probe design and branched DNA (bDNA) signal amplification system [10]. This design creates an amplification cascade only when two adjacent probe pairs bind to the target sequence, providing exceptional specificity that virtually eliminates background noise while enabling visualization of individual RNA molecules [36]. This technical breakthrough provides a 8,000-fold signal amplification capacity while maintaining morphological context, a critical advantage over "grind-and-bind" methods like qPCR and NGS that lose spatial information [7] [13].
Fundamental Principles of RNAscope Technology
The core innovation of RNAscope lies in its probe design and signal amplification mechanism. The double-Z probes consist of three elements: a target-specific lower region that hybridizes to the RNA of interest, a spacer sequence, and a tail that binds to pre-amplifier molecules [13]. This configuration requires two probes to bind adjacent to each other on the target RNA before signal amplification can initiate, creating a fundamental specificity checkpoint not present in conventional ISH [36]. Each successfully bound probe pair then initiates a multi-stage amplification process where pre-amplifiers attach to the Z-probe tails, followed by amplifier sequences, and finally labeled probes (chromogenic or fluorescent) [13]. The result is a highly specific signal where each dot represents an individual RNA molecule, enabling both qualitative localization and quantitative assessment at single-cell resolution [7] [10].
RNAscope vs. Conventional ISH: A Sensitivity Comparison
Systematic Performance Evaluation
A systematic review comparing RNAscope to established gold standard methods provides compelling evidence for its superior performance characteristics. The review, which analyzed 27 retrospective studies, confirmed that RNAscope demonstrates high sensitivity and specificity with excellent concordance rates between 81.8%-100% when compared to qPCR, qRT-PCR, and DNA ISH methodologies [13]. However, the concordance with immunohistochemistry (IHC) was notably lower (58.7%-95.3%), primarily reflecting the different biomolecules measured by each technique (RNA versus protein) rather than a deficiency in either method [13].
Table 1: Performance Comparison Between RNAscope and Conventional Techniques
| Technique | Sensitivity | Specificity | Morphological Context | Single-Cell Resolution | Quantification Capability |
|---|---|---|---|---|---|
| RNAscope ISH | High (single-molecule detection) | High (100% with specific probes) | Excellent | Yes | Semi-quantitative (dots/cell) |
| Traditional ISH | Moderate to Low | Variable (high background) | Good | Limited | Limited |
| qPCR/qRT-PCR | High | High | None | No | Quantitative (bulk analysis) |
| IHC | Moderate | Variable (antibody-dependent) | Excellent | Yes | Semi-quantitative |
Key Advantages in Research and Diagnostic Applications
The enhanced sensitivity of RNAscope provides several distinct advantages for research and potential diagnostic applications. First, it enables detection of low-abundance targets that may be undetectable by conventional ISH or IHC, including viral RNAs with low viral loads, weakly expressed genes, and splice variants [7] [36]. Second, the capacity for precise cellular localization allows researchers to identify specific cell types harboring viral reservoirs or expressing biomarkers of interest, providing insights into disease mechanisms and heterogeneity [7] [13]. Third, the multiplexing capability enables simultaneous detection of multiple RNA targets within the same tissue section, facilitating studies of co-infections, host-pathogen interactions, and complex gene regulatory networks [7] [37]. These advantages position RNAscope as a transformative technology particularly valuable for infectious disease research, oncology, neuroscience, and drug development programs where spatial context of gene expression is critical.
Automated Platform Integration: Protocols and Procedures
Roche DISCOVERY ULTRA Integration
The Roche DISCOVERY ULTRA system provides a fully automated platform for RNAscope assays, offering enhanced reproducibility and throughput for research applications. Proper integration requires attention to several critical instrument parameters and reagent specifications to ensure optimal performance [38].
Key Instrument Configuration Guidelines:
- Slide Cleaning Function: This option must be disabled in the software settings to prevent disruption of the assay [38].
- Bulk Solution Management: All bulk solution containers must be thoroughly rinsed and purged with recommended buffers before running RNAscope assays. Specifically, the DISCOVERY 1X SSC Buffer diluted 1:10 must be used—the Benchmark 10X SSC Buffer is not compatible [38].
- Temperature Control: The instrument's independent temperature control for each slide drawer maintains optimal hybridization conditions without cross-interference between simultaneous runs [38].
- Decontamination Protocol: Regular instrument decontamination every three months is essential to prevent microbial growth in fluidic lines that could compromise assay performance [38].
Tissue Pretreatment Optimization: Pretreatment conditions require optimization based on tissue fixation quality. For tissues fixed according to ACD recommendations (16-32 hours in fresh 10% NBF), standard pretreatment protocols typically suffice. However, deviation from these conditions necessitates adjustment of Pretreat 2 (boiling) and/or protease treatment times [38]. The software enables manual touchpoints at multiple protocol stages, providing flexibility for specialized applications while maintaining automation benefits [39].
Leica BOND RX Integration
The Leica BOND RX system offers another automated platform for RNAscope implementation, with specific configuration requirements distinct from the DISCOVERY ULTRA system.
Standardized Protocol Parameters:
- Standard Pretreatment: 15 minutes Epitope Retrieval 2 (ER2) at 95°C followed by 15 minutes Enzyme (Protease) at 40°C [38].
- Mild Pretreatment Alternative: 15 minutes ER2 at 88°C and 15 minutes Protease at 40°C for delicate tissues or optimally fixed specimens [38].
- Extended Pretreatment: For suboptimally fixed or challenging tissues, increase ER2 time in 5-minute increments and Protease time in 10-minute increments while maintaining standard temperatures (e.g., 20 minutes ER2 at 95°C + 25 minutes Protease at 40°C) [38].
Reagent Configuration: The system requires specific detection kits depending on the assay format: RNAscope 2.5 LS Brown assays must use Leica's Bond Polymer Refine Detection kits, while LS Red assays require Bond Polymer Refine Red Detection kits. Substitution with other chromogen kits is not recommended as it compromises assay performance [38]. The "Mock probe" and "Bond wash" containers must be user-filled with 1x Bond Wash Solution according to manufacturer specifications [38].
Universal Workflow and Quality Control
Both automated platforms share common workflow elements and quality control requirements essential for successful RNAscope implementation. The incorporation of appropriate control probes is critical for validating assay performance on every run.
Table 2: Essential Control Probes for RNAscope Assay Validation
| Control Type | Target | Expected Result | Purpose | Interpretation Guidelines |
|---|---|---|---|---|
| Positive Control (PPIB) | Human peptidylprolyl isomerase B | Score ≥2 with relatively uniform signal | Assess RNA integrity and assay performance | Successful staining indicates adequate RNA quality |
| Positive Control (POLR2A) | RNA polymerase II subunit A | Score appropriate for low-expression genes | Alternative for low-copy targets | Validates detection of low-abundance transcripts |
| Positive Control (UBC) | Ubiquitin C | Score ≥3 | For highly expressed genes | Confirms assay sensitivity for high-copy targets |
| Negative Control (dapB) | Bacterial dihydrodipicolinate reductase | Score <1 (no staining) | Assess background/non-specific binding | Indicates proper specificity; high signal suggests issues |
Automated Workflow Diagram:
Essential Research Reagent Solutions
Successful implementation of automated RNAscope protocols requires specific reagent systems and materials validated for compatibility with the technology. Deviations from recommended reagents can significantly impact assay performance and reliability.
Table 3: Essential Research Reagent Solutions for Automated RNAscope
| Reagent Category | Specific Product/Requirement | Function | Platform Compatibility |
|---|---|---|---|
| Slide Type | Superfrost Plus slides | Optimal tissue adhesion during stringent processing | DISCOVERY ULTRA & BOND RX |
| Barrier Pen | ImmEdge Hydrophobic Barrier Pen (Vector Labs) | Maintains reagent containment without dissolving | Manual pretreatment steps |
| Mounting Media | CytoSeal XYL (Brown assay)EcoMount or PERTEX (Red assay) | Preserves signal and tissue morphology | Platform independent |
| Detection Kits | Bond Polymer Refine Detection (Brown)Bond Polymer Refine Red Detection (Red) | Chromogen development for signal visualization | BOND RX specific |
| Bulk Buffers | DISCOVERY 1X SSC Buffer (diluted 1:10)RiboWash Buffer (diluted 1:10) | Stringency washes and reagent dilution | DISCOVERY ULTRA specific |
| Control Probes | PPIB, POLR2A, UBC (positive)dapB (negative) | Assay performance validation | DISCOVERY ULTRA & BOND RX |
Technical Troubleshooting and Optimization Guidelines
Common Implementation Challenges
Despite the standardized protocols, researchers may encounter specific challenges during automated RNAscope implementation. Understanding these issues and their solutions is crucial for maintaining assay reliability.
Insufficient Signal Intensity:
- Potential Causes: Inadequate permeabilization, RNA degradation, suboptimal fixation, or insufficient protease digestion [38].
- Solutions: Optimize protease concentration and incubation time; verify RNA integrity with positive controls; ensure proper tissue fixation (16-32 hours in fresh 10% NBF); extend antigen retrieval time incrementally [38].
High Background Noise:
- Potential Causes: Over-digestion with protease, excessive probe concentration, inadequate stringency washes, or tissue drying [38].
- Solutions: Reduce protease treatment duration; verify proper probe dilution; ensure sufficient wash buffer volumes and correct temperature during washes; maintain tissue hydration throughout the procedure [38].
Tissue Detachment:
- Potential Causes: Inappropriate slide type, excessive protease treatment, or mechanical disruption during processing [38].
- Solutions: Use only Superfrost Plus slides; optimize protease concentration and time; ensure proper drying of tissues before runs; handle slides carefully during manual steps [38].
Specimen Preparation Guidelines
Consistent specimen preparation is fundamental to successful RNAscope results. Tissues should be fixed in 10% neutral buffered formalin (NBF) within a 10:1 fixative-to-tissue ratio for 16-32 hours at room temperature [37] [38]. Prolonged fixation can compromise RNA accessibility and require extended retrieval times, while insufficient fixation may lead to RNA degradation and tissue morphology issues [37]. For tissues requiring alternative fixatives (e.g., Davidson's for eyes and testes), additional validation of pretreatment conditions is necessary [37]. Storage of paraffin blocks at lower temperatures (4°C or -20°C) preserves RNA integrity, particularly for blocks older than one year [37].
The integration of RNAscope technology with automated platforms like the Roche DISCOVERY ULTRA and Leica BOND RX systems represents a significant advancement in molecular pathology, providing researchers with a powerful tool for spatial gene expression analysis. The standardized protocols detailed in this whitepaper enable highly sensitive and specific RNA detection within morphological context, addressing critical limitations of conventional ISH methodologies. While current evidence demonstrates RNAscope's reliability as a complementary technique alongside established methods like IHC and PCR [13], ongoing validation studies and cost-benefit analyses will further define its role in clinical diagnostics. The technology's capacity for multiplex detection, compatibility with routine FFPE specimens, and single-molecule sensitivity positions it as an invaluable asset for drug discovery, biomarker validation, and translational research programs where cellular context of gene expression drives critical decisions.
The accurate detection and localization of RNA biomarkers within their histopathological context is crucial for advancing research in cancer biology, infectious diseases, and neuroscience. While in situ hybridization (ISH) techniques theoretically provide this capability, conventional RNA ISH methods have historically faced significant limitations in sensitivity and specificity, restricting their widespread research and clinical application [40]. These technical constraints have particularly hampered the detection of low-abundance transcripts, which are characteristic of many critical biomarkers, therapeutic targets, and viral pathogens.
The development of RNAscope technology represents a paradigm shift in RNA ISH, introducing a novel probe design and signal amplification system that fundamentally addresses the sensitivity challenges of conventional approaches [40]. This technical guide provides an in-depth comparison between RNAscope and conventional ISH methodologies, detailing the mechanistic basis for enhanced performance and presenting quantitative data validating its superior sensitivity. We further illustrate diverse research applications across biomarker development, infectious disease pathogenesis, and neuroscience, providing detailed experimental protocols to facilitate implementation within rigorous research settings. The comprehensive analysis presented herein establishes RNAscope as a transformative technology that enables researchers to visualize RNA expression with single-molecule sensitivity while fully preserving tissue morphology.
Technical Mechanisms: RNAscope vs. Conventional ISH
Fundamental Limitations of Conventional ISH
Conventional RNA in situ hybridization techniques suffer from two primary technical shortcomings that limit their reliability and sensitivity. First, they often lack sufficient signal amplification to detect low-copy RNA molecules against background noise. Second, and more critically, they exhibit significant non-specific background hybridization from probes binding to off-target sequences, which obscures genuine signal detection [40]. This signal-to-noise problem is particularly pronounced in formalin-fixed, paraffin-embedded (FFPE) tissues, where nucleic acid cross-linking and fragmentation further reduce hybridization efficiency. The resulting sensitivity limitations have historically restricted conventional ISH primarily to highly expressed targets, such as Epstein-Barr virus (EBV)-derived transcripts EBER1/2, while failing to reliably detect the majority of cellular mRNAs that express at moderate to low levels [40].
RNAscope's Novel Probe Design and Amplification System
RNAscope technology addresses these limitations through a proprietary double-Z probe design strategy that enables simultaneous signal amplification and background suppression [40]. This system employs paired "Z" probes that must bind contiguously to the target RNA molecule—a statistically unlikely event for nonspecific hybridization. Only when both probes bind correctly do they form a complete binding site for the preamplifier molecule, initiating a hybridization chain reaction that ultimately generates amplifiable signals [41].
Table 1: Comparison of RNAscope and Conventional ISH Technologies
| Feature | Conventional ISH | RNAscope Technology |
|---|---|---|
| Probe Design | Single linear probes | Paired "double-Z" probes |
| Signal Amplification | Limited or none | Hybridization-based amplification |
| Background Suppression | Minimal; relies on stringency washes | Built into probe design |
| Single-Molecule Sensitivity | Rarely achieved | Routinely demonstrated [40] |
| Compatible Sample Types | Limited by fixation | FFPE, frozen, cell preparations [42] |
| Multiplexing Capacity | Limited | Up to 4-plex with standard kits [40] |
The RNAscope workflow incorporates several innovations that collectively enable its superior performance. First, custom software automatically selects target probe sequences with compatible melting temperatures and minimal cross-hybridization potential to off-target sequences [40]. Second, the system typically utilizes 10-20 probe pairs targeting approximately 1 kb of the RNA molecule, providing added robustness against variable target accessibility and partial RNA degradation [40]. Finally, the detection system offers flexibility with either chromogenic dyes for bright-field microscopy or fluorescent dyes for multiplex analysis and higher resolution imaging [40].
Figure 1: RNAscope Signal Amplification Mechanism. The double-Z probe design requires two probes to bind contiguously to the target RNA before preamplifier binding can occur, initiating a hybridization cascade that ultimately generates detectable signal [40].
Quantitative Performance Comparison
Sensitivity and Specificity Metrics
Direct comparisons between RNAscope and conventional ISH demonstrate remarkable differences in detection capabilities. RNAscope achieves single-molecule visualization sensitivity while preserving tissue morphology, a feat rarely accomplished with conventional methods [40]. This sensitivity enables detection of individual viral particles in infected cells, even during early infection stages with low viral loads [7]. The proprietary probe design strategy provides exceptional specificity, allowing accurate discrimination among highly related viral species and strains, which is particularly valuable for pathogen research [7].
The signal-to-noise ratio advantage of RNAscope stems from its fundamental design principle: it is highly unlikely that nonspecific hybridization events will juxtapose a pair of target probes along an off-target mRNA molecule to form the complete 28-base hybridization site required for preamplifier binding [40]. This mechanistic advantage translates to significantly reduced background noise compared to conventional ISH, where nonspecific probe binding frequently generates false-positive signals that complicate interpretation.
Performance in Challenging Samples
RNAscope demonstrates particular utility with FFPE tissues, the most common archival format in pathology. While formalin fixation causes RNA fragmentation and cross-linking that challenges conventional ISH, RNAscope probes are specifically designed to detect these fragmented RNA molecules [19]. However, recent research indicates that RNA degradation in FFPET occurs in an archival duration-dependent fashion, with high-expression genes like PPIB showing more pronounced degradation than low-to-moderate expressors like POLR2A and HPRT1 [19]. This finding underscores the importance of implementing appropriate housekeeping gene controls when designing RNAscope experiments, especially with older archival samples.
Table 2: RNAscope Signal Integrity in Archival Tissues [19]
| Gene Target | Expression Level | Degradation in FFPET | Recommended Application |
|---|---|---|---|
| PPIB | High | Most pronounced | Sample quality assessment |
| UBC | High | Significant | Sample quality assessment |
| POLR2A | Moderate | Moderate | Reliable for quantification |
| HPRT1 | Low | Least pronounced | Most stable for archival tissue |
A 2025 systematic assessment of RNA-FISH signals in breast cancer samples demonstrated that while RNAscope signals in FFPET are lower than in fresh frozen tissue (FFT) in an archival duration-dependent fashion, the technology remains capable of generating quantifiable data even in suboptimal samples [19]. This study further validated that performing sample quality checks using housekeeping genes is essential for ensuring accurate RNAscope results, particularly when working with extensive archival collections [19].
Diverse Research Applications
Cancer Biomarker Development
In cancer research, RNAscope has proven invaluable for detecting and validating RNA biomarkers within their histopathological context. The technology enables direct visualization of gene expression patterns in tumor cells while preserving the surrounding tissue architecture, providing critical insights into tumor heterogeneity and microenvironment interactions [40]. This capability is particularly valuable for assessing biomarkers in complex tumor samples containing mixed cell populations, where grind-and-bind methods like RT-PCR may yield misleading results due to averaging effects across different cell types [40].
For HER2 amplification assessment in breast cancer, RNAscope offers a robust platform compatible with bright-field microscopy, providing a cost-effective and scalable solution for routine pathology [43]. Automated ISH analysis in combination with machine learning approaches shows particular promise for improving accuracy and reducing manual labor in HER2 status evaluation [43]. The technology's compatibility with standard bright-field microscopy makes it accessible for pathology laboratories without specialized fluorescence imaging equipment, facilitating broader implementation in both research and clinical settings.
Infectious Disease Pathogenesis
RNAscope provides exceptional utility in viral pathogenesis research by enabling direct detection of viral RNA within infected tissues and cells. Its single RNA molecule detection capability allows identification of individual viral particles despite low or undetectable viral loads, making it particularly valuable for studying early infection events and reservoir identification [7]. The platform has been successfully applied to diverse viral pathogens including HIV/SIV, Zika, hepatitis C virus (HCV), and Epstein-Barr virus (EBV) [7] [35].
Researchers at UCSF School of Medicine have utilized RNAscope to "visualize and quantify HIV RNA with single-copy resolution" in HIV-infected humanized mice, combining ISH with immunohistochemistry to characterize tissue and cellular compartments where the virus resides [35]. This application demonstrates how RNAscope enables researchers to establish the etiology and pathogenesis of viral diseases by coupling molecular detection with morphological context, elucidating viral transmission pathways, pathogen-host interactions, and localization of hidden reservoirs [7].
Neuroscience Targets
In neuroscience research, RNAscope has emerged as a powerful tool for mapping gene expression within complex neural circuits. The technology enables precise localization of mRNA markers implicated in neurological function and disease at single-cell resolution [41]. This capability is particularly valuable for understanding cell-type-specific gene expression patterns in heterogeneous brain tissues, where traditional bulk analysis methods obscure critical cellular-level information.
Researchers at Vanderbilt University have employed RNAscope to study "microglia during postnatal development in the hypothalamus to understand how they're interacting with AgRP neurons and facilitating establishment of neural circuits that control food intake" [35]. The proprietary "double Z" probe design in combination with advanced signal amplification enables highly specific and sensitive detection of target RNA, with each dot visualizing a single RNA transcript, providing clear answers while seamlessly fitting into existing workflows [41]. This high resolution and low background have made RNAscope particularly valuable for multiplex fluorescent assays combined with confocal microscopy in neural tissues [35].
Experimental Protocols
RNAscope Assay Procedure for FFPE Tissues
The standard RNAscope protocol for FFPE tissues involves sequential hybridization steps that build upon the double-Z probe design [40]. The following detailed methodology has been optimized for archival clinical specimens:
Sample Preparation: Cut 5-μm thick tissue sections and mount onto slides. Deparaffinize in xylene followed by dehydration through an ethanol series [40].
Pretreatment: Boil slides in citrate buffer (10 mmol/L, pH 6) for 15 minutes using a hot plate maintained at 100°C to 103°C. Rinse in deionized water and immediately treat with protease (10 μg/mL) at 40°C for 30 minutes in a HybEZ hybridization oven [40]. Protease concentration and incubation time may require optimization for different tissue types.
Probe Hybridization: Incubate slides with target probes in hybridization buffer A (6× SSC, 25% formamide, 0.2% lithium dodecyl sulfate, blocking reagents) at 40°C for 3 hours [40]. Typically, 20 probe pairs targeting approximately 1 kb of the RNA molecule are used for optimal signal generation.
Signal Amplification: Perform sequential hybridization with the following components, washing with buffer (0.1× SSC, 0.03% lithium dodecyl sulfate) three times between each step [40]:
- Preamplifier (2 nmol/L) in hybridization buffer B (20% formamide, 5× SSC, 0.3% lithium dodecyl sulfate, 10% dextran sulfate, blocking reagents) at 40°C for 30 minutes
- Amplifier (2 nmol/L) in hybridization buffer B at 40°C for 15 minutes
- Label probe (2 nmol/L) in hybridization buffer C (5× SSC, 0.3% lithium dodecyl sulfate, blocking reagents) for 15 minutes
Signal Detection: For chromogenic detection using DAB, incubate with HRP-conjugated label probe followed by DAB reaction. Counterstain with hematoxylin [40]. For fluorescent detection, use fluorophore-conjugated label probes compatible with your imaging system.
Figure 2: RNAscope Workflow for FFPE Tissues. The standardized procedure includes critical pretreatment steps to expose target RNA, followed by sequential hybridization and amplification steps that generate detectable signals [40].
Multiplex Fluorescent RNAscope Protocol
For detecting multiple RNA targets simultaneously, the RNAscope Multiplex Fluorescent v2 Assay enables visualization of up to four different targets in a single sample [19]. The protocol builds upon the standard method with the following key considerations:
Probe Design: Utilize probes with different tail sequences recognized by distinct signal amplification systems for each target [40].
Hybridization Strategy: Incubate with equimolar amounts of target probes for multiple genes simultaneously. The same preamplifier, amplifier, and label probe concentrations are used as in single-plex assays [40].
Signal Development: Use fluorophores with non-overlapping emission spectra (e.g., Opal 520, 570, 620, and 690) to label different targets [19]. Apply fluorophores sequentially with appropriate washing between applications.
Image Acquisition: Acquire images using a multispectral imaging system such as the Vectra Polaris Automated Quantitative Pathology Imaging System within two weeks after completing RNAscope assays [19]. This timeframe preserves fluorescence intensity for accurate quantification.
Controls and Quality Assessment
Incorporating appropriate controls is essential for validating RNAscope results. The following controls should be included in every experiment [40] [19]:
Positive Control: Endogenous housekeeping genes like UBC, PPIB, POLR2A, or HPRT1 assess both tissue RNA integrity and assay procedure. Positive staining with signals easily visible under a 10× objective indicates adequate sample quality [40] [19].
Negative Control: The bacterial gene dapB serves as a negative control to assess non-specific background signal [40].
Sample Quality Assessment: Given the archival duration-dependent RNA degradation observed in FFPET, performing sample quality checks using housekeeping genes is strongly recommended to ensure accurate results interpretation [19].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagent Solutions for RNAscope Applications
| Reagent/Equipment | Function | Application Notes |
|---|---|---|
| RNAscope Probe Sets | Target-specific detection | Custom probes available in 2 weeks; catalog probes for >100 viruses and numerous human genes [7] [42] |
| HybEZ Hybridization Oven | Temperature-controlled hybridization | Provides consistent 40°C environment critical for specific hybridization [40] |
| Multiplex Fluorescent v2 Kit | Simultaneous detection of multiple targets | Enables 4-plex detection using different fluorophores [19] |
| Housekeeping Gene Probes | Sample quality control | UBC, PPIB, POLR2A, HPRT1 assess RNA integrity [19] |
| dapB Negative Control Probe | Background assessment | Bacterial gene probe confirms specificity [40] |
| Automated Platforms | High-throughput processing | Compatible with Leica BOND RX, Roche DISCOVERY ULTRA [14] |
| Protease-Free Assays | Sensitive protein epitope preservation | Enables RNA-protein co-detection; available on Roche Discovery ULTRA [26] |
RNAscope technology represents a significant advancement over conventional ISH methods, offering researchers unprecedented capability to visualize RNA expression within morphological context. Its proprietary double-Z probe design provides the sensitivity and specificity necessary to detect low-abundance transcripts that were previously challenging to visualize in situ. The technology's compatibility with FFPE tissues, multiplexing capabilities, and adaptability to automated platforms make it particularly valuable for both basic research and translational applications.
As spatial biology continues to evolve, RNAscope stands as a cornerstone technology that enables precise mapping of gene expression patterns in diverse research contexts. Its demonstrated applications in cancer biomarker validation, infectious disease pathogenesis, and neuroscience underscore its versatility and robust performance. By following the detailed experimental protocols outlined in this guide and implementing appropriate quality controls, researchers can reliably harness the power of this technology to advance our understanding of gene expression in health and disease.
Optimizing Performance: RNAscope Troubleshooting, Controls, and Quality Assessment
Within the context of a broader thesis comparing the sensitivity of RNAscope to conventional in situ hybridization (ISH), it is paramount to recognize that superior technical sensitivity can be entirely negated by suboptimal sample handling. The revolutionary RNAscope technology, with its proprietary signal amplification and background suppression system, represents a major advance over traditional RNA ISH, enabling single-molecule detection in intact cells [44] [45]. However, this advanced detection capability is critically dependent on the integrity of the target RNA within the tissue specimen. Pre-analytical variables—encompassing all steps from tissue collection to slide preparation—directly govern RNA integrity and accessibility, thereby forming the fundamental determinants of assay success and the validity of any comparative sensitivity analysis. This guide details the core pre-analytical factors that researchers must optimize to ensure data reliability in studies leveraging RNAscope's enhanced sensitivity for research and diagnostic applications.
Tissue Fixation and Processing: Preserving the Analytical Target
The initial steps of tissue preservation are arguably the most critical in the pre-analytical chain. The primary goal of fixation is to immobilize RNA in situ while preserving tissue morphology, a balance that is easily compromised.
Fixation Parameters
Optimal Fixative: Fresh 10% Neutral Buffered Formalin (NBF) is the universally recommended fixative for RNAscope assays [44] [46]. Its consistency and buffering capacity prevent acid-induced RNA degradation. Fixation Duration: Tissues should be fixed for 16–32 hours at room temperature [44] [46]. Under-fixation leads to poor morphological preservation and significant RNA loss during storage, while over-fixation causes excessive nucleic acid cross-linking, impeding probe access [46]. A 2024 study demonstrated that RNAscope signals progressively decrease with extremely prolonged formalin fixation, with signal intensity and percent area of signal dropping after 180 days and becoming undetectable at 270 days [47]. Tissue Specifications: For effective penetration of fixative and reagents, tissues should be trimmed to a thickness of 3–4 mm before immersion in a sufficient volume of fixative [46].
Tissue Processing and Archival
Following fixation, tissue processing and storage conditions introduce additional variables that impact RNA quality.
- Processing: Dehydration through a graded series of ethanol and xylene, followed by infiltration with paraffin, should be performed with paraffin held at no more than 60°C to prevent heat-induced RNA degradation [46].
- Archival Duration: Formalin-fixed paraffin-embedded (FFPE) tissue blocks can be stored for extended periods, but RNA degradation occurs in an archival duration-dependent fashion [19]. Despite this degradation, RNAscope has proven capable of detecting RNA in FFPE tissues stored at room temperature for up to 15 years, underscoring its robustness for retrospective studies [47]. In contrast, fresh frozen tissue (FFT) biobanking better preserves nucleic acids but requires costly low-temperature storage [19].
Table 1: Impact of Archival Duration on RNAscope Signal in FFPE vs. FFT
| Tissue Type | Storage Condition | Impact on RNAscope Signal | Key Research Findings |
|---|---|---|---|
| Formalin-Fixed Paraffin-Embedded (FFPE) | Room Temperature | Signal decreases over time [19]. | Signal detectable after 15 years of block storage [47]; significant degradation observed in high-expressor genes in breast cancer samples archived for 7+ years [19]. |
| Fresh Frozen Tissue (FFT) | -80°C or Liquid Nitrogen | Superior signal preservation compared to FFPE [19]. | Requires more cumbersome handling and expensive storage infrastructure [19]. |
Tissue Pretreatment Optimization: Unlocking Target Accessibility
Even perfectly fixed tissues require carefully optimized pretreatment to make the target RNA accessible to the probes without destroying the tissue or the nucleic acids. This step is where most assay failures occur and thus requires systematic optimization.
Standard Pretreatment Workflow
The standard RNAscope pretreatment protocol involves three key steps after sectioning:
- Baking: FFPE slides are baked to melt paraffin and ensure tissue adhesion.
- Dehydration: Slides are passed through xylene and ethanol to remove residual paraffin.
- Antigen Retrieval: Slides are heated in a specific retrieval reagent (e.g., RNAscope 1X Target Retrieval Reagent) to break formalin-induced cross-links. This step is performed at 98–102°C and is critical for signal intensity [19] [48].
- Protease Digestion: A protease (e.g., Protease III) is applied to permeabilize the tissue by digesting proteins, thereby allowing probe access. This step must be performed at 40°C [44]. The duration of protease treatment is one of the most sensitive variables requiring optimization.
Optimization Strategies for Challenging Samples
When sample preparation history is unknown or suboptimal, or when working with novel tissue types, a qualification experiment using positive and negative control probes is strongly recommended [44] [49]. The table below outlines common issues and their tailored optimization strategies, derived from experimental data across various tissues.
Table 2: Tissue Pretreatment Optimization Guide
| Tissue Scenario | Observed Problem | Recommended Optimization | Experimental Evidence |
|---|---|---|---|
| Over-fixed or Hard Tissues (e.g., bone, tooth) | Weak or no signal | Extended Protease digestion: Increase time in 5–10 minute increments [44].Extended Antigen Retrieval: For automated assays, increase boiling time in 5-minute increments [44]. | In decalcified rodent incisors, only specific decalcification buffers (ACD buffer, Morse's solution) preserved RNA integrity for successful RNAscope, while traditional methods (EDTA, formic acid) failed [50]. |
| Under-fixed or Delicate Tissues (e.g., brain, plant) | Tissue loss, degradation, high background | Reduced Protease digestion: Decrease digestion time [51].Milder Antigen Retrieval: For automated systems, reduce temperature to 88°C [44].Longer Fixation: Plant tissues required 1 hour in 10% NBF vs. standard 15 minutes [51]. | In barley leaf sections, reducing protease time minimized tissue disintegration. A backing step with semi-frozen OCT compound prevented section detachment [51]. |
| FFPE Blocks with Long Archival Duration | Weak signal | Extended Protease digestion: Primary adjustment to recover signal from aged, cross-linked samples [44]. | A study on archived breast cancer FFPETs (2013–2020) showed archival duration-dependent signal loss, most pronounced in high-expressor genes [19]. |
Essential Quality Control and Experimental Design
Robust experimental design for RNAscope mandates the inclusion of proper controls to distinguish technical failure from true biological negativity.
Control Probes and Their Interpretation
- Positive Control Probes: Housekeeping genes (HKGs) validate sample RNA quality and assay technique. The choice of HKG should mirror the expected expression level of the target gene [49].
- PPIB (Cyclophilin B): Medium expression (10-30 copies/cell). The recommended flexible option for most tissues [49].
- POLR2A: Low expression (3-15 copies/cell). Ideal for low-expression targets or proliferating tissues like tumors [49].
- UBC (Ubiquitin C): High expression (>20 copies/cell). Should only be used for high-expression targets, as its signal may persist even in suboptimal conditions, giving false confidence [49].
- Negative Control Probe: The bacterial dapB gene should yield no staining (score <1) in properly fixed tissue, confirming low background [44] [49].
A successful quality control check requires the positive control probe (e.g., PPIB) to generate a score of ≥2 with relatively uniform signal, and the negative control (dapB) a score of <1 [44].
The Scientist's Toolkit: Key Research Reagent Solutions
The following table catalogues essential materials and their functions for a successful RNAscope experiment, as evidenced by published protocols.
Table 3: Essential Reagents and Materials for RNAscope
| Item | Function / Purpose | Technical Notes |
|---|---|---|
| Superfrost Plus Slides | Microscope slides with enhanced tissue adhesion. | Critical to prevent tissue detachment during the rigorous protocol; other slide types are not recommended [44] [48]. |
| ImmEdge Hydrophobic Barrier Pen | Creates a hydrophobic barrier around the tissue section. | Maintains reagent volume over the tissue; the only pen validated for use throughout the RNAscope procedure [44] [48]. |
| HybEZ Oven / Humidity Control Tray | Maintains optimum humidity and temperature (40°C) during hybridization and incubation steps. | Required to prevent slides from drying out, which can cause high background and assay failure [44]. |
| Positive & Negative Control Probes | Qualify sample RNA integrity and assay performance. | PPIB/dapB is a standard combination. Essential for troubleshooting and validating results [44] [49]. |
| RNAscope Target Probes | Gene-specific probes for the RNA target of interest. | Designed as "Z" probes binding to ~1000 bases of the target, enabling single-molecule detection [51]. |
| Protease III | Enzyme for tissue permeabilization. | Digests proteins to allow probe access to RNA; time and temperature (40°C) are critical [48]. |
| Specific Mounting Media (e.g., EcoMount, PERTEX, ProLong Gold) | Preserves fluorescence and mounts coverslip. | Varies by assay type (chromogenic vs. fluorescent); using the incorrect media can quench signal [44] [19] [48]. |
The enhanced sensitivity of RNAscope over conventional ISH is not a guarantee but a potential that must be unlocked through meticulous attention to pre-analytical factors. The journey from tissue collection to the final stained slide is a chain of interdependent steps, each capable of introducing variability that compromises RNA integrity and, consequently, experimental conclusions. As demonstrated across diverse tissues—from archived human breast cancer and decalcified teeth to plant leaves—the universal application of standardized fixation, coupled with tailored pretreatment optimization and rigorous quality control, is non-negotiable. By adopting the systematic, evidence-based approach outlined in this guide, researchers can fully leverage the power of RNAscope, ensuring that their data reflects true biological expression with the sensitivity and spatial resolution that this advanced technology promises.
In the evolving landscape of RNA in situ hybridization (ISH), the superior sensitivity and specificity of the RNAscope technology represent a significant advancement over conventional ISH methods. This technical guide delineates a comprehensive quality assurance framework utilizing the control probes PPIB, POLR2A, UBC, and dapB. Within the context of formalin-fixed, paraffin-embedded (FFPE) tissues, we provide detailed methodologies for integrating these controls into experimental workflows, complete with quantitative scoring guidelines and troubleshooting protocols. This strategy ensures the reliability of RNA detection, which is paramount for high-stakes research and drug development.
Traditional RNA ISH methods have long been hampered by limitations in sensitivity and specificity, often failing to reliably detect low-abundance RNA transcripts without significant background noise [40]. The grind-and-bind approach of methods like RT-PCR, while sensitive, sacrifices crucial spatial context, rendering it impossible to localize gene expression to specific individual cells within a complex tissue architecture [7] [40].
The RNAscope platform addresses these limitations through its patented double Z probe design [36] [40]. This technology employs a pair of "Z" probes that must bind contiguously to the target RNA to initiate a hybridization-based signal amplification cascade. This design inherently suppresses background by ensuring that signal generation is contingent upon two independent probe binding events, making non-specific amplification highly improbable [36] [40]. This innovation enables single-molecule visualization in situ, preserving morphological context while achieving a level of sensitivity required for modern molecular pathology and research [6] [40].
The implementation of a rigorous control strategy is not merely a supplementary step but a foundational component for validating this sensitive technology. Proper use of the control probes PPIB, POLR2A, UBC, and dapB is critical for qualifying sample integrity, verifying technical assay performance, and ensuring the accurate interpretation of experimental results.
Control Probe Characteristics and Selection Criteria
The selection of appropriate control probes is the first critical step in assay design. The recommended controls serve distinct purposes and are characterized by their different expression levels and specific applications.
Table 1: RNAscope Positive Control Probe Profiles
| Control Probe | Expression Level (Copies/Cell) | Primary Application | Interpretation of Results |
|---|---|---|---|
| UBC (Ubiquitin C) | High (>20) | Technical workflow control; best for high-expression targets. | Score ≥3 indicates successful assay run. May signal success even with suboptimal RNA quality. |
| PPIB (Cyclophilin B) | Medium (10-30) | Sample and RNA quality control; most flexible and recommended option. | Score ≥2 indicates good sample quality and technical performance. |
| POLR2A (RNA Polymerase II) | Low (3-15) | Rigorous sample quality control; for low-expression targets or challenging tissues (e.g., tumors, retina). | Score ≥2 indicates high-quality sample suitable for detecting low-abundance targets. |
The universal negative control probe, dapB, targets a bacterial gene from Bacillus subtilis and should not generate signal in properly prepared human or animal tissue samples. A successful assay yields a dapB score of <1, confirming minimal background staining [52] [49] [25].
Table 2: RNAscope Control Probe Selection Guide
| Experimental Scenario | Recommended Positive Control | Rationale |
|---|---|---|
| Initial assay validation and workflow check | UBC and PPIB | UBC confirms technical execution; PPIB provides a more rigorous check on sample quality. |
| Routine analysis of most tissue types | PPIB | Offers a balanced, reliable assessment of both sample quality and assay performance. |
| Targeting a low-abundance RNA | POLR2A | Matching control sensitivity to target sensitivity ensures the sample is qualified for low-copy detection. |
| Working with proliferative tissues (e.g., tumors) | POLR2A | Functions well as a low-copy control in these contexts. |
| Analyzing highly degraded samples | UBC | High copy number may still generate a signal, helping to troubleshoot assay failure. |
Experimental Protocol for Control Implementation
Sample Preparation Essentials
The foundation of a successful RNAscope assay is proper sample preparation. For FFPE tissues, ACD recommends fixing tissue blocks of 3-4 mm thickness in fresh 10% Neutral-Buffered Formalin (NBF) for 16-32 hours at room temperature [25]. Tissues are then dehydrated, cleared, and infiltrated with paraffin. Sections should be cut at 5 ±1 μm and mounted on Fisher Scientific SuperFrost Plus slides to prevent tissue loss during the assay [25]. Sectioned slides must be stored with desiccant and analyzed within three months for optimal results.
RNAscope Assay Workflow
The RNAscope procedure can be performed manually or on automated staining systems (e.g., Leica BOND RX, Roche DISCOVERY) and is typically completed in a single day [52] [44]. The key stages are consistent across platforms:
Key considerations for the protocol:
- Protease Digestion: Temperature must be maintained at 40°C for optimal permeabilization without damaging tissue morphology [44].
- Hybridization System: The HybEZ Hybridization System is required for manual assays to maintain optimum humidity and temperature during probe hybridization [44].
- Reagent Freshness: Always use fresh ethanol, xylene, and formalin. Older reagents can compromise staining quality [44].
- Slide Drying: Avoid letting slides dry out between steps, as this increases non-specific background [44].
Control Experiment Methodology
For a comprehensive quality control check, researchers should run several control slides in parallel with experimental samples:
- Technical Workflow Control: Use a reference cell pellet slide (e.g., Human HeLa Cell Pellet, Cat. No. 310045) with PPIB (positive) and dapB (negative) probes to verify the assay is performed correctly [25].
- Sample Quality Control: For each new sample or batch, run consecutive sections with:
- The positive control probe (PPIB recommended) to assess RNA integrity.
- The negative control probe (dapB) to determine background levels.
- The experimental target probe(s).
This control strategy allows for direct comparison and accurate interpretation of the experimental results.
Data Interpretation and Scoring Guidelines
RNAscope assay results are interpreted using a semi-quantitative scoring system that focuses on the number of punctate dots per cell rather than signal intensity. Each dot represents a single RNA molecule [52] [25].
Table 3: RNAscope Scoring Guidelines for Control Probes
| Score | Dot Count Criteria | Interpretation |
|---|---|---|
| 0 | No staining or <1 dot per 10 cells | Negative/Negligible expression |
| 1 | 1-3 dots per cell | Low expression |
| 2 | 4-9 dots per cell; very few dot clusters | Moderate expression |
| 3 | 10-15 dots per cell; <10% dots in clusters | High expression |
| 4 | >15 dots per cell; >10% dots in clusters | Very high expression |
For an assay to be considered successful and the sample qualified, the following outcomes are required:
- Positive Control (PPIB): Score of ≥2 with relatively uniform signal throughout the sample.
- Positive Control (UBC): Score of ≥3.
- Negative Control (dapB): Score of <1, indicating minimal to no background staining [25].
Samples that do not meet these criteria require pretreatment optimization before proceeding with experimental probes.
Troubleshooting and Optimization Strategies
A standardized workflow for qualifying samples, particularly those with unknown or suboptimal fixation history, is crucial for assay success.
Common issues and solutions:
- Low or No Signal with PPIB/POLR2A: This indicates suboptimal RNA exposure. Extend the epitope retrieval (ER2) time in 5-minute increments and/or the protease treatment time in 10-minute increments while keeping temperatures constant (e.g., 20 min ER2 at 95°C and 25 min Protease at 40°C) [44].
- High Background with dapB: This suggests over-permeabilization or non-specific binding. Reduce protease treatment time and ensure all reagents, especially ethanol and xylene, are fresh [44].
- Inconsistent Staining Across Tissue: This can result from uneven tissue sectioning or fixation. Ensure consistent fixation times and section thickness, and always use recommended SuperFrost Plus slides [25].
Essential Research Reagent Solutions
The following table catalogues the critical reagents and materials required for implementing this control strategy successfully.
Table 4: Essential Research Reagent Solutions for RNAscope QA
| Reagent / Material | Function / Purpose | Examples / Catalog References |
|---|---|---|
| Control Probes | Verify assay performance and sample quality. | PPIB, POLR2A, UBC (positive); dapB (negative) [49]. |
| Control Slides | Technical control for assay workflow. | Human Hela Cell Pellet (#310045), Mouse 3T3 Cell Pellet (#310023) [25]. |
| RNAscope Assay Kits | Core reagents for detection. | RNAscope 2.5 HD/LS Reagent Kits (BROWN/RED) [52]. |
| Specialized Slides | Prevent tissue loss during stringent protocol. | Fisher Scientific SuperFrost Plus Slides [25]. |
| Hybridization System | Maintain optimal temperature and humidity for probe binding. | HybEZ Hybridization System [44]. |
| Hydrophobic Barrier Pen | Create a well around tissue section to retain reagents. | ImmEdge Hydrophobic Barrier Pen [44]. |
| Automated Platforms | Provide standardized, high-throughput staining. | Leica BOND RX, Roche DISCOVERY XT/ULTRA [52] [44]. |
The implementation of a systematic control strategy using PPIB, POLR2A, UBC, and dapB is a critical enabler for leveraging the full potential of the RNAscope platform. This framework provides researchers and drug development professionals with a validated, practical path to ensure data integrity, reproduce experimental findings, and confidently translate RNA biomarkers from discovery to clinical application. By adhering to these quality assurance protocols, the scientific community can fully capitalize on the superior sensitivity of RNAscope over conventional ISH, driving forward our understanding of gene expression within its native morphological context.
In the evolving landscape of molecular pathology and genetic research, RNAscope in situ hybridization (ISH) represents a significant advancement over conventional ISH methodologies, offering unrivaled sensitivity and specificity for RNA visualization within intact tissues [7] [10]. This proprietary technology employs a unique double-Z probe design and signal amplification system that enables single-molecule RNA detection while preserving crucial morphological context [36] [53]. Despite its technological sophistication, researchers often encounter technical challenges including signal abnormalities, excessive background noise, and tissue detachment that can compromise experimental outcomes. This technical guide examines these common obstacles within the broader thesis of RNAscope's enhanced sensitivity over conventional ISH, providing evidence-based troubleshooting protocols and solutions tailored for research scientists and drug development professionals engaged in precision molecular detection.
Understanding RNAscope Technology and Its Sensitivity Advantages
The foundational innovation of RNAscope technology lies in its proprietary probe design strategy that fundamentally differs from conventional ISH approaches. The double-Z probe architecture requires two adjacent probe pairs to bind specifically to the target RNA before signal amplification can occur, creating a built-in mechanism for enhanced specificity [36]. This design strategy significantly reduces non-specific binding and background noise that frequently plague traditional ISH methods [10] [36].
Compared to conventional ISH, RNAscope offers:
- Single-molecule sensitivity enabling detection of low-abundance transcripts [53]
- High specificity with minimal background signals [53]
- Preservation of tissue morphology for spatial context [7]
- Multiplexing capability for simultaneous detection of multiple RNA targets [7] [53]
The technology's superior performance is particularly evident in formalin-fixed, paraffin-embedded (FFPE) tissues, where RNA degradation and crosslinking typically challenge conventional ISH methods [10] [19]. RNAscope probes are specifically engineered to detect fragmented RNA, making them ideally suited for archived clinical samples [19].
Establishing a Foundation for Success: Pre-Assay Considerations
Sample Quality Assessment and Validation
Before initiating RNAscope experiments, rigorous sample qualification is essential. Research demonstrates that RNA degradation in FFPE tissues occurs in an archival duration-dependent fashion, with high-expressor housekeeping genes like UBC and PPIB showing the most pronounced degradation [19]. Implement a standardized validation protocol using control probes:
- Positive controls: PPIB, POLR2A, or UBC to assess RNA integrity [44]
- Negative control: Bacterial dapB to evaluate background noise [44]
- Acceptance criteria: Successful PPIB staining should generate a score ≥2, while dapB should yield a score <1 [44]
Table 1: RNAscope Semi-Quantitative Scoring Guidelines
| Score | Criteria | Interpretation |
|---|---|---|
| 0 | No staining or <1 dot/10 cells | Negative |
| 1 | 1-3 dots/cell | Low expression |
| 2 | 4-9 dots/cell, none or very few dot clusters | Moderate expression |
| 3 | 10-15 dots/cell, <10% dots in clusters | High expression |
| 4 | >15 dots/cell, >10% dots in clusters | Very high expression |
Critical Sample Preparation Requirements
Proper sample preparation establishes the foundation for successful RNAscope experiments. Adherence to these specific requirements prevents common problems including tissue detachment:
- Slide selection: Use only Superfrost Plus slides; other slide types may result in tissue detachment [44]
- Fixation: Employ fresh 10% neutral buffered formalin (NBF) with fixation times of 16-32 hours [44]
- Barrier pens: Use exclusively ImmEdge Hydrophobic Barrier Pen; other pens may fail during the procedure [44]
- Mounting media: Select appropriate media - xylene-based for Brown assays, EcoMount or PERTEX for Red and 2-plex assays [44]
Troubleshooting Common Technical Challenges
Addressing Signal Issues: No Signal, Weak Signal, or Irregular Signal Patterns
Signal abnormalities represent the most frequent challenge in RNAscope experiments. The solutions vary based on the specific signal problem encountered:
No Signal Detection
- Verify probe quality: Ensure probes are warmed to 40°C before use to dissolve precipitates that form during storage [44]
- Confirm enzymatic amplification: Apply all amplification steps in the correct sequence; omitting any step will result in no signal [44]
- Validate detection chemistry: For automated systems, ensure proper reagents are used - Leica Bond Polymer Refine Detection for Brown assays and Bond Polymer Refine Red Detection for Red assays [44]
- Check equipment calibration: On Ventana systems, disable the "Slide Cleaning" option and verify hybridization temperatures [44]
Weak or Faint Signal
- Optimize pretreatment: For over-fixed tissues, increase Protease treatment time in 10-minute increments while maintaining 40°C [44]
- Adjust antigen retrieval: Extend boiling time (Pretreat 2) in 5-minute increments for over-fixed tissues [44]
- Verify reagent freshness: Use fresh ethanol and xylene solutions; degraded reagents diminish signal strength [44]
- Assess RNA integrity: Run housekeeping gene controls; weak control signals indicate sample degradation [19]
Table 2: Troubleshooting Guide for Signal Issues
| Problem | Potential Causes | Recommended Solutions |
|---|---|---|
| No signal | Probe precipitation, skipped steps, incorrect temperatures | Warm probes to 40°C, verify protocol adherence, calibrate equipment |
| Weak signal | Over-fixation, suboptimal pretreatment, degraded reagents | Increase protease time (10-min increments), extend retrieval time, use fresh reagents |
| Irregular signal pattern | Incomplete hybridization, drying artifacts, inadequate humidity | Maintain 40°C during protease step, ensure hydrophobic barrier integrity, keep humidifying paper wet |
Mitigating Background Noise: Nonspecific Staining and High Background
Excessive background noise compromises signal interpretation and quantification. The unique double-Z probe design of RNAscope inherently reduces nonspecific binding, but background issues can persist:
Persistent Background Staining
- Validate probe specificity: Always include dapB negative control; background should score <1 [44]
- Optimize protease treatment: Under-treatment increases background, while over-treatment damages tissue morphology [44]
- Adjust wash stringency: For Ventana systems, use DISCOVERY 1X SSC Buffer only (diluted 1:10); do not use Benchmark 10X SSC Buffer [44]
- Control assay humidity: Maintain adequate humidity by keeping humidifying paper wet in the Humidity Control Tray [44]
Specific Background Patterns
- Nuclear background: Often indicates excessive protease digestion; reduce protease time [44]
- Diffuse cytoplasmic staining: Suggests inadequate washing; increase wash duration or temperature [44]
- Edge artifacts: Caused by drying; ensure hydrophobic barrier remains intact throughout procedure [44]
Preventing Tissue Detachment: Structural Integrity Challenges
Tissue detachment during the rigorous RNAscope procedure represents a significant problem, potentially resulting in complete sample loss:
Primary Prevention Strategies
- Slide selection: Use only Superfrost Plus slides; other slide types cannot withstand the stringent conditions [44]
- Fixation optimization: Under-fixed tissues detach more readily; ensure proper fixation in fresh 10% NBF for 16-32 hours [44]
- Temperature management: During antigen retrieval, avoid thermal shock by directly placing slides in room temperature water to stop the reaction [44]
- Proper mounting: For Brown assays, use only xylene-based mounting media; other media may not provide adequate adhesion [44]
Secondary Reinforcement Approaches
- Barrier pen integrity: Use only ImmEdge Hydrophobic Barrier Pen and confirm the barrier remains intact throughout the procedure [44]
- Gentle handling: Flick or tap slides to remove residual reagent, but never allow slides to dry completely [44]
- Equipment verification: For automated systems, ensure proper maintenance and calibration to prevent mechanical tissue damage [44]
RNAscope Experimental Workflow
The following diagram illustrates the critical steps in the RNAscope workflow, highlighting key decision points for troubleshooting common challenges:
Systematic Approaches to Signal Optimization
Automated Platform-Specific Optimization
Ventana DISCOVERY XT/ULTRA Systems
- Instrument maintenance: Perform decontamination protocols every three months to prevent microbial growth in fluid lines [44]
- Bulk solution management: Replace all bulk solutions with recommended buffers before running RNAscope assays [44]
- Software configuration: Uncheck the "Slide Cleaning" option and maintain recommended hybridization temperatures without adjustment [44]
Leica BOND RX Systems
- Standard pretreatment: 15 minutes Epitope Retrieval 2 (ER2) at 95°C and 15 minutes Enzyme (Protease) at 40°C [44]
- Mild pretreatment alternative: 15 minutes ER2 at 88°C and 15 minutes Protease at 40°C for sensitive samples [44]
- Extended pretreatment: For over-fixed tissues, increase ER2 time in 5-minute increments and Protease time in 10-minute increments [44]
Sample Quality-Dependent Protocol Modifications
Recent research demonstrates that RNA degradation in FFPE tissues follows archival duration-dependent patterns, with high-expressor genes (UBC, PPIB) degrading more significantly than moderate expressors (POLR2A, HPRT1) [19]. This evidence informs specific protocol adjustments:
- Short archival duration (<2 years): Apply standard pretreatment protocols [19]
- Medium archival duration (2-5 years): Increase protease treatment by 10-15 minutes [19]
- Long archival duration (>5 years): Combine extended protease treatment with reduced antigen retrieval temperature (88°C instead of 95°C) [44] [19]
Essential Research Reagent Solutions
Table 3: Critical Research Reagents for RNAscope Experiments
| Reagent/Category | Specific Product | Function and Importance |
|---|---|---|
| Microscope Slides | Superfrost Plus slides | Prevents tissue detachment; essential for protocol success [44] |
| Control Probes | PPIB, POLR2A, UBC (positive); dapB (negative) | Validates RNA integrity, assay performance, and specificity [44] |
| Barrier Pen | ImmEdge Hydrophobic Barrier Pen | Maintains reagent containment; prevents drying artifacts [44] |
| Mounting Media | CytoSeal XYL (Brown), EcoMount/PERTEX (Red) | Preserves signal and tissue integrity; media-specific requirements [44] |
| Detection Kits | mRNA DAB Detection Kit (HRP), mRNA RED Detection Kit (AP) | Enzyme-substrate system for signal generation [54] |
| Pretreatment Reagents | mRNA Sample Prep Kit | Standardizes antigen retrieval and protease digestion [54] |
The sophisticated design of RNAscope technology provides substantial advantages over conventional ISH methods, particularly in sensitivity and specificity, but requires meticulous attention to technical details to overcome common challenges. Successful implementation hinges on systematic approaches to signal optimization, background reduction, and tissue preservation throughout the experimental workflow. By adhering to the specific protocols and troubleshooting strategies outlined in this guide—including proper sample qualification, reagent selection, and platform-specific optimization—researchers can consistently generate reliable, publication-quality data that leverages the full potential of this transformative technology. The integration of these evidence-based solutions ensures that RNAscope continues to advance scientific discovery across research domains from basic science to drug development, particularly as the methodology evolves toward broader clinical applications.
In situ hybridization (ISH) techniques, pivotal for gene expression analysis, have been transformed by the advent of RNAscope, a technology offering single-molecule sensitivity and high specificity through its proprietary signal amplification. This technical guide examines the evolution from manual scoring to digital quantification of RNAscope assays, comparing the open-source QuPath platform and the commercial HALO software. Framed within a broader thesis on RNAscope versus conventional ISH sensitivity, we present experimental protocols and quantitative data demonstrating that both platforms achieve high concordance (correlation coefficients >0.89) for critical analytical modules while exhibiting distinct operational characteristics. Manual methods, though accessible, suffer from subjectivity and limited throughput, whereas digital pathology platforms provide reproducible, cell-by-cell quantification essential for rigorous biomarker research and drug development. Our analysis confirms that digital quantification with either platform reliably surpasses manual scoring in precision, throughput, and analytical depth, enabling more sensitive detection of gene expression changes in response to therapeutic interventions.
The quantification of gene expression via in situ hybridization has historically presented significant challenges. Conventional ISH techniques, relying on digoxigenin (DIG) or radioactive probes, are limited by non-specific binding, background noise, and an inability to detect low-abundance transcripts [13]. The introduction of RNAscope in 2012 represented a paradigm shift, utilizing a unique double-Z probe design that enables specific hybridization and powerful signal amplification—achieving up to 8,000-fold amplification through a branched DNA (bDNA) cascade [13]. This technological advancement fundamentally altered the sensitivity standards for RNA detection in tissue contexts.
As the sensitivity of detection methodologies advanced, so too did the requirements for quantification. Manual scoring of chromogenic or fluorescent signals under a microscope, while straightforward, introduces substantial inter-observer variability and becomes prohibitively time-consuming for large-scale studies. The advent of digital pathology platforms like HALO and QuPath addresses these limitations by providing automated, quantitative analysis that preserves the spatial context of gene expression while offering unprecedented reproducibility [55] [56].
Within drug development, particularly in immuno-oncology and neurodegenerative disease research, precise quantification of biomarker expression at single-cell resolution has become indispensable for patient stratification, target engagement assessment, and understanding therapeutic mechanisms [57] [58]. This whitepaper provides researchers with a technical framework for selecting and implementing appropriate quantification methodologies for RNAscope assays, with specific attention to their application in sensitivity comparisons between RNAscope and conventional ISH techniques.
RNAscope Technology: Foundations for Quantification
Technical Principles and Advantages
RNAscope's superior performance characteristics stem from its core design principles. The technology employs paired "Z" probes, each comprising three elements: a target-hybridizing region, a spacer sequence, and a tail for pre-amplifier binding [13]. This architecture requires two independent probes to bind adjacent regions of the target RNA before signal amplification can commence, ensuring exceptional specificity [13].
The amplification cascade begins when pre-amplifiers bind to the Z-probe tails, followed by the sequential binding of multiple amplifiers and subsequently labeled probes (chromogenic or fluorescent) [13]. Each RNA molecule can accommodate 20 Z-probe dimers, ultimately resulting in the attachment of up to 400 labeled probes per dimer and enabling single-molecule detection [13]. This engineered system achieves both 100% sensitivity and 100% specificity under optimal conditions, dramatically outperforming conventional ISH, particularly for low-abundance targets [13].
Signal Characteristics and Detection Requirements
RNAscope signals manifest as punctate dots, each representing an individual RNA molecule [13]. In samples with high gene expression, these dots may form clusters that challenge discrimination by manual scoring but can be resolved through digital algorithms [56]. Proper imaging is critical for accurate quantification:
- Chromogenic assays: Standard brightfield microscopy or digital slide scanners at 20× or 40× magnification are recommended [59].
- Fluorescent assays: High-numerical-aperture objectives (40× air or oil immersion) are preferred, with specialized camera systems (e.g., cooled CCD with >65% quantum efficiency) for low-expression targets or autofluorescent tissues [59].
The fundamental unit of quantification across manual and digital methods remains the discrete dot, with expression levels reported as dots per cell or dots per unit area, enabling direct comparison between approaches [13] [56].
Manual Scoring: Traditional Methodology
Established Scoring Protocols
Manual scoring of RNAscope assays typically follows manufacturer-recommended approaches that involve visual assessment of dot density and distribution across multiple representative regions [13]. A semi-quantitative scoring system is commonly employed:
- Score 0: No staining or <1 dot per 10 cells
- Score 1: 1-3 dots per cell (focal distribution)
- Score 2: 4-9 dots per cell (moderate, clustered distribution)
- Score 3: ≥10 dots per cell (abundant, frequently clustered)
- Score 4: Too numerous to count, with clusters encompassing >10% of cellular area
This approach requires pathologists to mentally average staining patterns across multiple high-power fields (typically 3-5 regions) while accounting for tissue heterogeneity [13].
Manual scoring introduces multiple potential sources of error and variability:
- Observer fatigue: Prolonged microscopy sessions diminish scoring consistency, particularly in large studies.
- Inter-observer variability: Subjective threshold differences between technicians affect reproducibility.
- Sampling bias: Inadequate sampling of heterogeneous tissues yields non-representative expression estimates.
- Limited dynamic range: The semi-quantitative 0-4 scale cannot resolve subtle expression differences potentially significant in therapeutic contexts.
- Inability to multiplex: Manual discrimination of multiple targets in multiplex assays is exceptionally challenging.
Despite these limitations, manual scoring remains a valuable validation tool and may suffice for binary (positive/negative) classifications in assays with strong signal-to-noise ratios.
Digital Analysis Platforms
HALO: Commercial Solution
HALO (Indica Labs) is a commercial digital pathology platform offering specialized modules for RNAscope analysis, including ISH, FISH, and multiplex IF modules [55] [59]. The platform employs both traditional image analysis and AI-based segmentation for nuclear, membrane, and cytoplasmic identification [55].
Key Analysis Capabilities:
- Automated dot counting with user-definable size and intensity thresholds
- Cell segmentation and subcellular dot localization (nuclear, cytoplasmic, membrane)
- Multiplex analysis for up to 12 targets in fluorescent assays [59]
- H-score calculation and expression histogram generation
- Spatial analysis modules for proximity and infiltration assessment [55] [59]
The platform features a user-friendly interface with pre-trained AI networks for segmentation that require minimal parameter adjustment, making it accessible to users without programming expertise [55].
QuPath: Open-Source Alternative
QuPath is an open-source software designed for quantitative pathology analysis, offering comprehensive tools for RNAscope quantification without licensing costs [56]. Its functionality includes complete workflow support from color deconvolution to final cell-by-cell RNA quantification [56].
Key Analysis Capabilities:
- Detection of individual and clustered RNA dots [56]
- Subcellular dot-count-based and optical-density-based quantification [56]
- Scriptable workflow for batch processing [56]
- Integration with external tools like CytoMap for advanced spatial analysis [60] [57]
- Support for brightfield and fluorescence assays, including multiplex experiments [56]
While QuPath offers exceptional flexibility, it typically requires scripting knowledge and manual pipeline setup to maximize its capabilities, presenting a steeper learning curve than HALO [57].
Platform Interoperability and Data Management
Both platforms support whole-slide image formats from major scanner manufacturers, including SVS, NDPI, MRXS, and CZI formats [55] [61]. Each maintains interactive links between cell-level data and spatial context, enabling researchers to visually verify automated findings by clicking on data points to review corresponding cell images [55] [62].
Table 1: Technical Comparison of Digital Analysis Platforms
| Feature | HALO | QuPath |
|---|---|---|
| Licensing | Commercial | Open-source |
| User Interface | Graphical, module-based | Script-based with GUI elements |
| Learning Curve | Moderate | Steeper (requires scripting for advanced features) |
| Analysis Speed | High-throughput with batch processing | Varies with workflow complexity |
| Segmentation Approach | Pre-trained AI networks & traditional | Traditional & machine learning |
| Multiplex Capacity | Up to 12-plex with specialized modules | Virtually unlimited with scripting |
| Spatial Analysis | Built-in modules for proximity and infiltration | Requires external tool integration (e.g., CytoMap) |
| Cost | Significant licensing fees | Free |
| Support | Commercial technical support | Community-based and documentation |
Experimental Protocols for Digital Quantification
Sample Preparation and Image Acquisition
Tissue Processing Protocol:
- Use formalin-fixed paraffin-embedded (FFPE) or fresh frozen tissues sectioned at 4-5μm thickness [13].
- Perform RNAscope according to manufacturer protocols using appropriate positive (PPIB, Polr2A, UBC) and negative (dapB) control probes [13].
- For brightfield detection, use chromogenic substrates (DAB, Fast Red); for fluorescence, select fluorophores with minimal tissue autofluorescence (e.g., Opal dyes) [59].
- Counterstain appropriately: hematoxylin for chromogenic, DAPI for fluorescent assays [59].
Image Acquisition Parameters:
- Scan slides at 20× magnification for high-expression targets, 40× for low-expression targets [59].
- Ensure consistent exposure times across compared samples within the same experiment.
- For multiplex fluorescence, use spectral unmixing or sequential exposure to minimize bleed-through [59].
- Save images in standardized formats (e.g., SVS, TIFF) with embedded pixel size calibration [61].
HALO Analysis Workflow for RNAscope
The following workflow utilizes the HALO ISH or FISH modules:
- Module Selection: Choose the appropriate ISH (brightfield) or FISH (fluorescence) module based on assay type [55].
- Tissue Classification: Apply a tissue classifier to exclude non-relevant regions (e.g., stroma, artifacts) or to define specific compartments (e.g., tumor vs. non-tumor) [55].
- Cell Segmentation: Optimize nuclear segmentation parameters using the real-time tuning feature, selecting from pre-trained AI networks or traditional algorithms [55].
- Dot Detection: Define dot detection parameters (size, intensity thresholds) using positive and negative controls for calibration [59].
- Phenotype Assignment (for multiplex assays): Assign cell phenotypes based on marker co-expression patterns using the intuitive grid interface [55].
- Analysis Execution: Run batch processing for multiple images using saved analysis settings [55].
- Data Export: Export cell-by-cell data, including dot counts per cell, cellular locations, and H-scores, in spreadsheet format for statistical analysis [55].
QuPath Analysis Workflow for RNAscope
The following protocol adapts QuPath for optimal RNAscope quantification:
- Image Import and Calibration: Import whole-slide images and verify pixel size calibration under the Image tab [61].
- Annotation: Manually annotate regions of interest or use automated detection tools [56].
- Cell Detection: Perform cell detection using the "Cell Detection" command, selecting the appropriate nuclear stain channel and optimizing parameters for your tissue type [56].
- Dot Detection: Implement a dot detection script using the "Subcellular Detection" extension, adjusting intensity and size thresholds based on control samples [56].
- Classification (optional): Apply machine learning classifiers to distinguish cell types or regions if needed [56].
- Data Extraction: Export measurements including cells per annotation, dots per cell, and dot locations for further analysis [56].
- Batch Processing: Apply the optimized script to multiple images using the "Batch Processing" feature [56].
Digital Analysis Workflow Selection
Comparative Performance Data
Quantitative Platform Comparisons
Recent studies have directly compared quantification outputs between HALO and QuPath to validate their performance. In a multiplex immunofluorescence study of prostate cancer utilizing a tissue microarray with 192 cores, both platforms demonstrated excellent concordance across three key analytical modules [60] [57].
Table 2: Performance Metrics from Comparative Studies
| Analysis Module | Correlation Coefficient | Study Context | Sample Size |
|---|---|---|---|
| Immune Cell Density | R > 0.89 | Prostate Cancer TMA | 192 cores [57] |
| Spatial Organization | R > 0.89 | Prostate Cancer TMA | 192 cores [57] |
| Nearest Neighbor Analysis | R > 0.89 | Prostate Cancer TMA | 192 cores [57] |
| Tau Quantification (Object Density) | ρ > 0.70 | Neurodegenerative Tissue | 70 slides [58] |
| Tau Quantification (Percent Positivity) | Strong correlation with Braak/CERAD | Neurodegenerative Tissue | 70 slides [58] |
In neuropathology applications, QuPath demonstrated superior correlation with Braak staging, while HALO showed excellent alignment with CERAD scoring [58]. Percent positivity emerged as the most reliable metric across both platforms, showing strong correlations with established neuropathological standards while object and optical densities showed more variability between platforms [58].
Sensitivity Comparisons: RNAscope vs. Conventional ISH
The systematic review by PMC8710359 provides critical data on RNAscope performance compared to conventional ISH and other gold standard techniques [13]. When compared to PCR-based methods and DNA ISH, RNAscope demonstrated high concordance rates ranging from 81.8% to 100% [13]. However, concordance with immunohistochemistry was lower (58.7%-95.3%), reflecting the different biomolecules (RNA vs. protein) measured by these techniques [13].
These findings underscore RNAscope's superior sensitivity compared to conventional ISH, particularly for low-abundance targets, while highlighting the importance of platform selection for specific application requirements.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents for RNAscope and Digital Quantification
| Reagent/Category | Specific Examples | Function/Purpose |
|---|---|---|
| RNAscope Assay Kits | RNAscope 2.5 HD Reagent Kit (BROWN/RED), RNAscope Fluorescent Multiplex Kit, RNAscope HiPlex12 Reagents | Signal amplification and detection for specific assay formats |
| Control Probes | PPIB (moderate expression), Polr2A (low expression), UBC (high expression), dapB (negative) | Assay validation, RNA integrity assessment, background determination [13] |
| Tissue Preparation | Formalin, paraffin, OCT compound (frozen tissue) | Tissue preservation and sectioning support |
| Detection Reagents | DAB chromogen, Fast Red, Opal fluorophores (520, 570, 620, 650, 690) | Signal generation for brightfield or fluorescence detection |
| Counterstains | Hematoxylin (brightfield), DAPI (fluorescence) | Nuclear visualization and cell segmentation reference |
| Mounting Media | Aqueous mounting medium (fluorescence), Permanent mounting medium (brightfield) | Slide preservation and signal stability |
| Analysis Software | HALO (with ISH/FISH modules), QuPath | Digital quantification and data extraction |
| Image Management | HALO Link, Aperio ImageScope, Proprietary scanner software | Slide digitization, storage, and management |
Digital analysis platforms have revolutionized RNAscope quantification, providing researchers with tools that surpass manual scoring in accuracy, throughput, and reproducibility. Both HALO and QuPath offer robust solutions for gene expression quantification, with the choice between them depending on specific research constraints and requirements.
For drug development professionals requiring standardized, high-throughput analysis with dedicated technical support, HALO provides a commercial solution with optimized workflows and validation frameworks. For academic researchers and those requiring maximum flexibility, custom workflows, and integration with specialized spatial analysis tools, QuPath offers unparalleled capabilities without licensing costs.
Critical considerations for implementation include:
- Validation: Establish quantification parameters using appropriate controls and correlate with complementary methods when possible.
- Personnel expertise: Assess programming capabilities for QuPath implementation versus need for turnkey solutions with HALO.
- Budget constraints: Evaluate initial and recurring costs against project scope and duration.
- Regulatory requirements: Consider platform validation needs for regulated environments.
The demonstrated superiority of RNAscope over conventional ISH techniques, coupled with rigorous digital quantification, empowers researchers to detect subtle gene expression changes with unprecedented sensitivity and spatial context. This technical advancement continues to expand possibilities in biomarker discovery, therapeutic development, and diagnostic applications across disease states.
This technical guide has presented a comprehensive framework for scoring and quantification method selection, supporting the broader research objective of demonstrating RNAscope's enhanced sensitivity compared to conventional ISH through rigorous, quantifiable methodologies.
Evidence-Based Validation: Systematic Reviews and Comparative Performance Data
The accurate detection of gene expression is fundamental to cancer diagnostics, treatment selection, and prognostic assessment. For years, immunohistochemistry (IHC), quantitative polymerase chain reaction (qPCR), and DNA in situ hybridization (DNA ISH) have served as cornerstone techniques in clinical diagnostics and research. However, each method has inherent limitations, including semiquantitative output, loss of spatial context, and inter-laboratory variability, driving the need for more precise and reliable alternatives [13] [63].
RNAscope in situ hybridization (ISH) represents a significant technological advancement, enabling the visualization and quantification of RNA transcripts within intact tissue morphology with single-molecule sensitivity [13] [36]. This whitepaper synthesizes systematic review findings and recent research to evaluate the concordance of RNAscope with established gold standard methods, framing this analysis within a broader thesis on its enhanced sensitivity over conventional ISH techniques.
RNAscope Technology: A Primer on Superior Sensitivity
Core Principle and Design
RNAscope is a novel, branched DNA (bDNA)-based ISH technology specifically engineered to overcome the key limitations of traditional ISH: poor sensitivity and high background noise [13]. Its exceptional performance stems from a proprietary double-Z probe design and signal amplification system:
- ZZ Probe Specificity: Each target is detected by a probe pair ("Z" probes) that must bind adjacent sites on the target RNA simultaneously. This dual-binding requirement is the foundation of the technique's high specificity, as it minimizes non-specific background—a single probe binding off-target will not initiate amplification [36].
- Signal Amplification: Upon successful dual probe binding, a pre-amplifier molecule attaches, followed by multiple amplifier molecules. Each amplifier can then bind numerous enzyme-linked labels (chromogenic or fluorescent). This cascade results in up to 8,000-fold signal amplification, enabling the detection of individual RNA molecules as distinct dots [13] [36].
Workflow and Quality Control
The standardized RNAscope workflow ensures robust and reproducible results. Key stages include:
- Sample Preparation: Optimized for formalin-fixed, paraffin-embedded (FFPE) tissues, fresh frozen tissues (FFT), and cell blocks [13] [19].
- Target Hybridization: Z probes hybridize to the target RNA sequence.
- Signal Amplification: A series of sequential amplification steps enhance the signal.
- Signal Detection and Visualization: Detection via chromogenic or fluorescent methods, compatible with bright-field and fluorescence microscopy [13].
Crucially, the assay incorporates positive and negative control probes (e.g., bacterial DapB) to validate RNA integrity, assay performance, and specificity for every run [13].
Concordance Analysis: RNAscope vs. Gold Standard Methods
A systematic review comparing RNAscope to established gold standards confirmed it is a highly sensitive and specific method with a high concordance rate across different platforms [13].
Table 1: Summary of Concordance Rates Between RNAscope and Gold Standard Techniques
| Gold Standard Method | Concordance Rate (CR) with RNAscope | Key Context and Findings |
|---|---|---|
| qPCR / qRT-PCR | 81.8% - 100% [13] | High concordance; RNAscope provides superior spatial resolution. |
| DNA ISH | 81.8% - 100% [13] | High concordance for gene detection. |
| Immunohistochemistry (IHC) | 58.7% - 95.3% [13] | Lower CR attributed to measuring different molecules (RNA vs. protein) and post-transcriptional regulation. |
| Fluorescence ISH (FISH) | 97.3% [64] | Demonstrated in resolving equivocal HER2 status in breast carcinoma. |
Concordance with IHC
The concordance between RNAscope and IHC is variable and generally lower than with molecular techniques. This is primarily because IHC detects proteins, while RNAscope detects RNA transcripts—distinct molecules separated by post-transcriptional and post-translational regulatory mechanisms [13]. Discrepancies can reveal biologically meaningful information about translational control. In practice, RNAscope serves as an excellent reflex test to resolve ambiguous IHC results [64].
Concordance with PCR-Based Methods (qPCR/ddPCR)
RNAscope shows very high concordance with quantitative PCR methods. A 2023 study on breast cancer biomarkers (ER, PR, HER2, Ki67) found RT-qPCR had high concordance with IHC (88.0%-94.4% for ER, PR, HER2), demonstrating the reliability of mRNA-based quantification [65]. Similarly, a 2024 study using droplet digital PCR (ddPCR) for ERBB2 copy number quantification reported an accuracy of 93.7%-94.1% compared to clinical ISH status [63].
The critical advantage of RNAscope over PCR is the preservation of spatial context. While PCR requires tissue homogenization, losing cellular and subcellular localization data, RNAscope allows quantification within specific cell types and reveals tumor heterogeneity [13] [64].
Concordance with DNA ISH (FISH, CISH, SISH)
RNAscope demonstrates excellent concordance with DNA ISH techniques for gene fusion or amplification detection. A large-scale study on ALK fusions in NSCLC found that next-generation sequencing (NGS) assays and RNAscope offered higher specificity and positive predictive value compared to standard FISH/IHC [66]. In breast cancer, RNAscope quantification of HER2 mRNA was 97.3% concordant with FISH and superior to qPCR in cases with intratumoral heterogeneity or equivocal FISH results [64].
Table 2: Comparative Analysis of Gene Detection Techniques in Clinical Studies
| Study Focus | Techniques Compared | Key Concordance Finding | Clinical/Research Utility |
|---|---|---|---|
| HER2 in Breast Cancer [64] | RNAscope vs. FISH, qPCR, IHC | 97.3% concordance with FISH; superior to qPCR in heterogeneous cases. | Resolves equivocal HER2 status; quantifies single-cell heterogeneity. |
| ALK in NSCLC [66] | NGS (Oncomine) vs. IHC/FISH (Standard of Care) | High sensitivity; significantly higher specificity and PPV than standard tests. | More specific determination of ALK rearrangements for targeted therapy. |
| ERBB2 in Breast Cancer [63] | ddPCR vs. ISH (FISH) | 93.7% - 94.1% accuracy for determining HER2 status. | Identified "ultrahigh" CN group with worse survival post-trastuzumab. |
| Multicenter HER2 Testing [67] | SISH/CISH/qPCR vs. FISH | 97%, 98%, and 95% concordance for SISH, CISH, and qPCR (vs. FISH ratio). | Alternative techniques show excellent performance on core biopsies. |
Experimental Protocols for Concordance Assessment
Protocol: RNAscope for HER2 Status Resolution in Breast Carcinoma
This protocol is adapted from a key study demonstrating RNAscope's utility in resolving equivocal HER2 status [64].
- Sample Preparation: Use 4-5 μm sections from FFPE breast carcinoma tissue blocks. Bake slides at 60°C for 1 hour, followed by deparaffinization in xylene and ethanol gradients.
- Pretreatment and Proteolysis: Perform heat-induced epitope retrieval and protease treatment to permeabilize tissues and expose target RNA, per manufacturer's instructions (Advanced Cell Diagnostics).
- Probe Hybridization: Apply HER2 RNAscope target probe. Include positive control (PPIB) and negative control (DapB) probes. Hybridize in a HybEZ oven at 40°C for 2 hours.
- Signal Amplification: Perform the RNAscope amplification steps according to the kit protocol (e.g., RNAscope 2.5 HD Reagent Kit).
- Detection and Counterstaining: For bright-field detection, use DAB chromogen and hematoxylin counterstain. For fluorescent detection, use fluorophore-conjugated labels and a compatible counterstain (e.g., DAPI).
- Quantitative Analysis: Scan slides and use image analysis software (e.g., Halo, QuPath) to quantify single-cell RNA signals. Determine HER2 status based on established mRNA copy number thresholds.
Protocol: Systematic Comparison with IHC, qPCR, and DNA ISH
This protocol outlines a general framework for comparative studies, as seen in the systematic review [13].
- Sample Cohort Selection: Obtain a well-characterized set of human tissue samples (e.g., FFPE blocks) representing the disease of interest. Ensure ethical approval and informed consent.
- Parallel Testing:
- RNAscope: Perform as described in Section 4.1.
- IHC: Perform on consecutive tissue sections using validated antibodies and scoring systems (e.g., ASCO/CAP guidelines for HER2).
- qPCR/ddPCR: Extract RNA/DNA from mirror tissue sections or macrodissected samples. Perform reverse transcription and qPCR/ddPCR using TaqMan assays for the target genes and housekeeping genes for normalization [65] [63].
- DNA ISH (FISH/CISH): Perform according to standard clinical protocols using FDA-approved kits.
- Blinded Analysis: Have pathologists and scientists score each assay independently and in a blinded manner.
- Concordance Calculation: Calculate concordance rates, sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) using the gold standard (e.g., FISH or clinical consensus) as the reference.
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Reagent Solutions for RNAscope and Comparative Assays
| Item | Function/Description | Example Products/Genes |
|---|---|---|
| RNAscope Probe Sets | Target-specific ZZ probes designed for the gene of interest. | HER2, ERBB2, PPIB (control), UBC (control), POLR2A (control) [64] [19]. |
| RNAscope Kit Reagents | Contains all necessary reagents for the assay workflow: amplifiers, labels, and detection reagents. | RNAscope Multiplex Fluorescent v2 Kit, RNAscope 2.5 HD Reagent Kit [19]. |
| Positive Control Probes | Probes for ubiquitously expressed "housekeeping" genes to verify sample RNA integrity. | PPIB, POLR2A, UBC [13] [19]. |
| Negative Control Probe | A probe for a bacterial gene not present in human tissues to assess background noise. | DapB (dihydrodipicolinate reductase) [13] [19]. |
| IHC Antibodies | Validated primary antibodies for protein detection on consecutive sections. | Anti-HER2/ErbB2, Anti-ER (Estrogen Receptor), Anti-PR (Progesterone Receptor) [65]. |
| qPCR/ddPCR Assays | Primer and probe sets for quantitative DNA/RNA analysis of extracted nucleic acids. | TaqMan assays for ERBB2, CEP17, and reference genes (e.g., on chr. 2p13.1) [63]. |
| Image Analysis Software | Software for quantitative analysis of RNAscope signals (dot counting) and IHC staining. | Halo, QuPath, Aperio [13] [19]. |
Visualizing the RNAscope Workflow and Technology
The following diagram illustrates the core principle and procedural workflow of the RNAscope technology, highlighting the steps that contribute to its high sensitivity and specificity.
Evidence from systematic reviews and primary research solidifies RNAscope as a robust and highly concordant method when compared to traditional gold standards like IHC, qPCR, and DNA ISH. Its unique capability to provide single-cell, quantitative data within morphological context addresses critical limitations of conventional techniques, particularly in characterizing tumor heterogeneity and resolving equivocal diagnostic cases.
While not yet positioned to replace gold standards outright in all clinical scenarios, RNAscope serves as a powerful complementary and reflex diagnostic tool. Its high technical performance and growing adoption in research and clinical validation studies underscore its significant value for researchers, pathologists, and drug development professionals seeking to enhance the precision of gene expression analysis.
RNAscope in situ hybridization (ISH) technology represents a significant advancement in molecular pathology, offering a robust, automated platform for clinical diagnostics. With its proprietary probe design enabling single-molecule sensitivity and single-cell resolution within morphological context, RNAscope has transitioned from research to clinical applications through partnerships with diagnostic manufacturers and availability of Analyte Specific Reagents (ASRs). This technical assessment evaluates RNAscope's readiness for integration into pathology workflows, focusing on its technical validation, automated platforms, and growing menu of clinically relevant targets that support its adoption for diagnostic use.
Table 1: RNAscope Clinical Diagnostic System Overview
| Feature | Clinical Implementation | Significance for Pathology Workflows |
|---|---|---|
| Automation Platform | Optimized for Leica BOND III Advanced Staining Clinical Platform [68] | Full automation enables standardized, reproducible results compatible with existing clinical laboratory equipment [27] [68] |
| Probe Design | Proprietary "double Z" probe design for high specificity [6] [36] | Minimizes false positives/negatives; enables detection of highly homologous sequences [7] |
| Sensitivity | Single-molecule detection capability [6] [23] | Identifies low-abundance targets and individual viral particles despite low viral loads [7] |
| Assay Menu | 16 ASR probes available (including viral targets and controls) [68] | Growing menu for clinical disease states with 2-week custom probe design capability [7] [68] |
| Visualization | Chromogenic detection with bright-field microscopy [68] | Familiar review and analysis method for pathologists accustomed to IHC [68] |
| Throughput | Single-day workflow [27] | Supports timely diagnostic reporting compatible with clinical turnaround requirements |
Technology Foundation: The RNAscope Assay Principle
Double-Z Probe Design Mechanism
The foundational innovation of RNAscope technology lies in its proprietary double-Z probe design, which enables unprecedented specificity in RNA detection. This system utilizes paired Z-probes that must bind adjacent to each other on the target RNA sequence to generate a signal [36]. Each probe pair consists of two distinct "Z" shaped probes containing complementary sequences to the target RNA—if only one probe binds non-specifically, no amplification occurs, dramatically reducing background signal [36]. This mechanism provides the equivalent of a dual-key security system where both probes must correctly bind to initiate signal amplification [36].
The RNAscope system typically utilizes approximately 20 different probe pairs targeting the same RNA molecule, ensuring robust detection even if some probe pairs fail to hybridize [36]. This multi-probe strategy combined with the requirement for adjacent binding creates a system with both high sensitivity and exceptional specificity, capable of distinguishing between highly similar sequences such as viral strains and splice variants [7].
Signal Amplification and Detection
Following successful probe hybridization, the RNAscope assay employs a hierarchical signal amplification system that generates discrete punctate dots visible under standard microscopy. Each dot represents an individual RNA molecule, enabling both qualitative localization and quantitative assessment [23]. The amplification process occurs through these stages:
- Pre-amplifier Hybridization: The top sequences of correctly paired Z-probes form a binding site for a pre-amplifier molecule [36].
- Amplifier Assembly: Multiple amplifier molecules bind to each pre-amplifier, creating a branching structure [36].
- Label Probe Binding: Finally, label probes conjugated to enzymes or fluorophores attach to the amplifiers [36].
- Signal Development: For chromogenic detection, enzymatic reactions produce permanent precipitates; for fluorescent applications, fluorophores are directly visualized [68] [23].
This multi-stage amplification creates substantial signal from minimal starting material while maintaining specificity through the initial dual-probe requirement.
Figure 1: RNAscope Double-Z Probe Mechanism. The diagram illustrates the sequential process of specific RNA target detection requiring adjacent binding of two Z-probes for signal generation.
Automated Diagnostic Workflow Implementation
Integrated Staining Systems
RNAscope assays have been optimized for fully automated staining systems to ensure standardization and reproducibility essential for clinical diagnostics. The technology is compatible with major automated staining platforms including:
- Leica BOND RX Research Advanced Staining System [27]
- Roche Discovery Ultra and Discovery XT Automated IHC/ISH slide staining systems [27]
- Leica BOND III Advanced Staining Clinical Platform (for diagnostic use) [68]
The partnership between ACD and Leica Biosystems has been particularly significant for clinical implementation, resulting in a "fully-automated, plug and play RNA ISH that fits seamlessly with existing clinical laboratory equipment and workflows" [68]. This integration allows clinical laboratories to implement RNAscope technology without extensive workflow modifications.
Single-Day Procedural Timeline
The RNAscope procedure is designed for efficiency with a complete workflow achievable within a single day [27]. The streamlined process includes:
- Sample Preparation: FFPE tissue sections are mounted and deparaffinized
- Pretreatment: Target retrieval and protease digestion to expose RNA targets
- Probe Hybridization: Probe sets are applied and hybridized to target RNAs
- Signal Amplification: Hierarchical amplification creates detectable signal
- Detection: Chromogenic or fluorescent detection of RNA targets
- Counterstaining and Mounting: Preparation for pathological assessment
This efficient workflow supports timely diagnostic reporting compatible with clinical turnaround requirements.
Figure 2: RNAscope Clinical Workflow. The diagram outlines the standardized single-day procedure from sample preparation to pathologist review.
Clinical Validation and Quality Assurance
Control Systems for Diagnostic Accuracy
Robust control systems are essential for clinical implementation, and RNAscope protocols mandate running three slides per sample to ensure result validity [23]:
- Target Marker Panel: The actual diagnostic probe(s) of interest
- Positive Control Probe: Typically PPIB or UBC to verify RNA integrity and assay performance [68]
- Negative Control Probe: Bacterial dapB to confirm absence of non-specific signal and appropriate tissue preparation [68] [23]
This tripartite control system provides confidence in both positive and negative results, which is critical for diagnostic decision-making.
Quantitative Analysis Methods
RNAscope signals are visualized as discrete punctate dots, with each dot representing an individual RNA molecule [23]. This characteristic enables both semi-quantitative and quantitative analysis approaches:
- Semi-quantitative Scoring: Pathologist-based scoring systems evaluating signal intensity and distribution
- Digital Image Analysis: Automated quantification using platforms such as HALO software (Indica Labs) or Aperio RNA ISH Algorithm (Leica Biosystems) [27] [9]
- Spatial Biology Analysis: Advanced computational methods to evaluate expression patterns within tissue architecture [9]
The Professional Assay Services team at ACD provides comprehensive image analysis support, offering custom data outputs based on specific diagnostic needs [9].
Table 2: Diagnostic Validation Framework for RNAscope Assays
| Validation Component | Implementation in RNAscope | Clinical Utility |
|---|---|---|
| Analytical Sensitivity | Single RNA molecule detection capability [6] [23] | Detection of low-abundance targets; identification of individual infected cells [7] |
| Analytical Specificity | Double-Z probe design distinguishing highly homologous sequences [36] [7] | Accurate viral strain differentiation; splice variant discrimination [7] |
| Control Systems | Tripartite system (target, positive, negative) [23] | Ensures result validity; identifies preparation failures |
| Reproducibility | Automated staining protocols [27] [68] | Standardized inter-laboratory and inter-instrument results |
| Tissue Scope | Validated on FFPE tissues [68] | Compatibility with standard pathology specimen types |
Diagnostic Applications and Menu
Infectious Disease Pathogen Detection
RNAscope has demonstrated particular utility in viral detection, with ACD offering RNAscope probes for over 100 viruses and capability for new probe design and manufacture within two weeks [7]. This rapid response capability is especially valuable for emerging pathogens. Key advantages for infectious disease diagnostics include:
- Strain Differentiation: High specificity enables discrimination between highly related viral species/strains [7]
- Latency Detection: Flexible probe design targeting sense or anti-sense strands enables differentiation between latent and active infection stages [7]
- Co-infection Identification: Duplex and multiplex assays allow simultaneous detection of multiple pathogens [7]
- Reservoir Localization: Morphological context helps identify cellular reservoirs of persistent infection [7]
Current ASR probes include Cytomegalovirus (CMV), Epstein-Barr Virus (EBV), HPV genotypes (6, 11, 16, 18, 31, 33), and SARS-CoV-2 [68].
Research Applications with Diagnostic Potential
Beyond currently approved ASRs, RNAscope technology supports numerous research applications with significant diagnostic translation potential:
- Cell and Gene Therapy: Monitoring therapeutic oligonucleotide delivery and distribution [69]
- Oncology: Biomarker validation and tumor subtyping [6] [9]
- Neuroscience: Localization of low-abundance neuronal transcripts [6] [9]
- Therapeutic Development: Spatial biodistribution assessment of RNA therapeutics [69]
The technology's ability to detect both endogenous and synthetic small RNAs through variants like miRNAscope and BaseScope assays extends its utility across multiple diagnostic domains [69].
Comparative Advantages in Clinical Diagnostics
RNAscope Versus Traditional Detection Methods
When evaluated against established diagnostic techniques, RNAscope demonstrates several compelling advantages:
- Superior to PCR: While real-time PCR is sensitive and rapid, it lacks morphological context and provides expression levels averaged across cell populations [7]. RNAscope complements PCR data with single-cell resolution and spatial localization [7].
- Advantage Over IHC: Compared to immunohistochemistry, RNAscope offers greater sensitivity and specificity while eliminating dependence on antibody availability and quality [7]. The technology detects RNA directly without requiring protein expression or being limited by epitope preservation.
- Beyond NGS: Next-generation sequencing provides comprehensive profiling but lacks spatial context and requires complex infrastructure [7]. RNAscope offers targeted detection within morphological framework.
These comparative advantages position RNAscope as a valuable addition to the diagnostic toolkit, particularly in cases where spatial context, sensitivity, and specificity are simultaneously required.
Essential Research Reagent Solutions
Table 3: Key Components for RNAscope Implementation
| Component | Function | Examples/Formats |
|---|---|---|
| Detection Kits | Chromogenic or fluorescent signal generation | BOND RNAscope Detection Reagents [68] |
| Protease Reagents | Tissue pretreatment for target accessibility | BOND RNAscope Protease [68] |
| Target Probes | Specific detection of RNA targets | 16 ASR probes available [68]; >30,000 research probes [68] |
| Control Probes | Assay validation and quality control | PPIB (positive), dapB (negative) [68] [23] |
| Image Analysis Software | Signal quantification and interpretation | HALO, Aperio RNA ISH Algorithm [27] [9] |
| Automation Systems | Standardized assay performance | Leica BOND III, BOND RX [27] [68] |
RNAscope technology has achieved significant milestones in its transition to clinical diagnostics, with automated platforms, standardized controls, and a growing menu of ASR probes. The double-Z probe design foundation provides the sensitivity and specificity required for demanding diagnostic applications, particularly in infectious disease and oncology. While the current ASR menu continues to expand, the technology's fundamental advantages—single-molecule sensitivity, spatial context preservation, and rapid probe development—position it as a valuable complementary technology to existing diagnostic methods. As clinical validation studies accumulate and the probe menu expands, RNAscope is poised for increased adoption in pathology workflows where RNA biomarker detection with morphological correlation is clinically imperative.
Thesis Context: The detection of Epstein-Barr virus (EBV) through in situ hybridization (ISH) represents a critical diagnostic and research tool, particularly in oncology and virology. This technical guide frames a direct comparison within a broader thesis investigating the superior sensitivity and specificity of novel RNAscope technology against conventional Universal HRP/AP and traditional EBER ISH methodologies. For researchers and drug development professionals, selecting the optimal detection platform has profound implications for diagnostic accuracy, assay validation, and therapeutic development. The gold standard for diagnosing EBV infection has historically been histological analysis using EBV-encoded small RNAs in situ hybridization (EBER-ISH) [70]. However, traditional ISH methods face significant challenges in sensitivity, specificity, and technical complexity, driving the development of enhanced platforms like RNAscope that offer single-molecule visualization while preserving tissue morphology [10].
Technical Comparison of ISH Methodologies
Underlying Principles and Probe Design
Traditional EBER ISH: Conventional DNA ISH methods for EBV detection typically utilize biotinylated cDNA probes or fluorescein isothiocyanate-labeled oligonucleotides targeting the abundant non-polyadenylated EBER RNAs (EBER1 and EBER2) expressed during latent infection [71]. These methods rely on standard hybridization techniques with limited signal amplification, often resulting in insufficient sensitivity for low-copy targets and challenges with background noise.
Universal HRP/AP ISH: This approach employs enzyme-based signal detection systems using either horseradish peroxidase (HRP) or alkaline phosphatase (AP) enzymes conjugated to reporters. While offering improved visualization over basic ISH through chromogenic or fluorescent development, these methods still face limitations in background suppression and single-molecule detection capability.
RNAscope ISH: The RNAscope platform represents a fundamental advancement in ISH technology through its unique dual "Z" probe design strategy [10]. This proprietary system employs paired probes that must bind adjacent to each other on the target RNA sequence before signal amplification can proceed. This requirement provides exceptional specificity and enables dramatic signal amplification through a pre-amplifier and amplifier cascade, ultimately allowing visualization of individual RNA molecules with up to 8,000-fold signal amplification [13]. The technology's background suppression features effectively minimize non-specific binding, addressing a critical limitation of traditional ISH methods.
Performance Metrics and Comparative Data
Table 1: Quantitative Comparison of ISH Method Performance Characteristics
| Method | Sensitivity | Specificity | Target Localization | Single-Molecule Detection | Multiplexing Capability |
|---|---|---|---|---|---|
| Traditional EBER ISH | Moderate; requires highly expressed targets [71] | Variable; background noise concerns [13] | Nuclear (for EBER) | Not reliably achievable | Limited |
| Universal HRP/AP ISH | Improved over basic ISH | Moderate; enzyme-related background possible | Dependent on target | Limited | Moderate (with different enzymes) |
| RNAscope | High (single-molecule detection) [10] | Exceptional (100% reported) [13] | Precise subcellular | Yes [10] | Advanced (up to 12-plex with HiPlex) [72] |
Table 2: Experimental Validation Studies Comparing ISH Methods
| Study Context | Traditional Methods Performance | RNAscope Performance | Significance |
|---|---|---|---|
| EBV in Nasopharyngeal Carcinoma [71] | EBER oligonucleotides showed stronger signals than cDNA probes on routine sections | N/A in this study | Established EBER detection as more reliable than LMP1 immunohistochemistry |
| Clinical Diagnostic Applications [13] | Lower concordance with IHC (58.7-95.3%) due to protein vs. RNA measurement | High concordance with qPCR, qRT-PCR, and DNA ISH (81.8-100%) | Superior correlation with molecular detection methods |
| Hodgkin Lymphoma EBV Detection [73] | Variable performance across 5 commercial probes (30-67.8% positivity) | N/A in this study | Highlighted significant variability in traditional probe performance |
| HIV RNA Detection [35] | Limited sensitivity for single-copy detection | Enabled visualization and quantification of HIV RNA with single-copy resolution | Critical advancement for viral reservoir characterization |
Experimental Protocols for Direct Comparison
Sample Preparation and Tissue Processing
For a valid comparative analysis, consistent sample preparation across all methodologies is essential:
Tissue Collection and Fixation: Collect tissues and fix immediately in 10% neutral buffered formalin. The fixation time should be standardized (typically 18-24 hours) to minimize RNA degradation and ensure comparable results across platforms [13].
Embedding and Sectioning: Process fixed tissues through graded ethanol and xylene steps followed by paraffin embedding (FFPE). Section at 4-5μm thickness using RNAse-free techniques and mount on positively charged slides.
Slide Storage and Preservation: Store slides at 4°C or -20°C in a desiccated environment until use to preserve RNA integrity. Avoid prolonged storage at room temperature.
Traditional EBER ISH Protocol
The following protocol adapts established methodologies from comparative studies [71] [73]:
Deparaffinization and Hydration: Bake slides at 60°C for 30 minutes, followed by deparaffinization in xylene and rehydration through graded ethanol series to distilled water.
Permeabilization and Protein Digestion: Treat slides with proteinase K (10-20 μg/mL) at 37°C for 15-30 minutes to expose target nucleic acids. Optimal concentration and time require titration for each tissue type.
Hybridization: Apply biotinylated or digoxigenin-labeled EBER probes in hybridization buffer. Cover with parafilm and incubate overnight at 37-55°C in a humidified chamber. Probe selection significantly impacts results, with studies showing detection rates varying from 30% to 67.8% across different commercial probes [73].
Stringency Washes: Perform post-hybridization washes with SSC buffers at appropriate stringency temperatures to remove non-specifically bound probes.
Signal Detection: For chromogenic detection, incubate with streptavidin-HRP or anti-digoxigenin-AP conjugates followed by DAB (brown) or NBT/BCIP (red/blue) substrate development. Counterstain with hematoxylin, dehydrate, clear, and mount.
RNAscope ISH Protocol
The RNAscope workflow incorporates proprietary reagents and optimized conditions [10] [74]:
Pretreatment: Bake FFPE slides at 60°C for 1 hour, followed by deparaffinization and ethanol rehydration. Perform target retrieval using a specified retrieval solution (e.g., RNAscope Target Retrieval Reagents) at 98-102°C for 15 minutes, followed by protease digestion (RNAscope Protease Plus) for 30 minutes at 40°C.
Hybridization with Z Probes: Apply target-specific "Z" probe pairs (designed against EBER or other EBV targets) and incubate at 40°C for 2 hours in the HybEZ Hybridization System. The dual "Z" probe design requires binding of both segments adjacent to each other on the target RNA for signal amplification to initiate, ensuring high specificity.
Amplification Steps: The RNAscope HD 2.5 Brown assay utilizes a sequential amplification process:
- Amplifier 1: Incubate for 30 minutes at 40°C
- Amplifier 2: Incubate for 30 minutes at 40°C
- Amplifier 3: Incubate for 15 minutes at 40°C Each step builds upon the previous, creating substantial signal amplification (up to 8,000-fold) while maintaining low background through the requirement for probe pairing [13].
Signal Detection and Visualization: Develop signal using DAB chromogen for 10 minutes at room temperature, producing brown punctate dots corresponding to individual RNA molecules. Counterstain with hematoxylin, dehydrate, clear, and mount.
Quality Control: Include positive control probes (e.g., PPIB for moderate expression, POLR2A for low expression) and negative control probes (bacterial dapB gene) with each run to validate assay performance and tissue RNA integrity [13].
Workflow Comparison Diagram
Diagram 1: Comparative ISH Experimental Workflows. The RNAscope method incorporates a proprietary amplification system that enables superior signal detection while reducing hands-on time compared to traditional EBER ISH protocols.
Essential Research Reagent Solutions
Table 3: Key Research Reagents and Their Functions in ISH Detection
| Reagent Category | Specific Examples | Function & Importance | Technology Platform |
|---|---|---|---|
| Probe Systems | EBER cDNA probes [71], EBER oligonucleotides [71], "Z" probe pairs [10] | Target recognition; critical for sensitivity and specificity | All platforms |
| Detection Enzymes | Horseradish Peroxidase (HRP), Alkaline Phosphatase (AP) | Signal generation through chromogenic/fluorescent substrates | Traditional ISH, Universal HRP/AP |
| Amplification Systems | Pre-amplifier, Amplifier sequences [13] | Signal magnification enabling single-molecule detection | RNAscope |
| Chromogenic Substrates | Diaminobenzidine (DAB) [74], NBT/BCIP | Visual signal development for bright-field microscopy | All platforms (particularly chromogenic) |
| Control Probes | PPIB, POLR2A (positive) [13], dapB (negative) [13] | Assay validation, RNA quality assessment, background monitoring | RNAscope |
| Hybridization Systems | HybEZ Oven [35] [74] | Temperature control for optimal hybridization conditions | RNAscope |
Technical Implementation Guide
Signal Amplification Mechanism
The fundamental advantage of RNAscope technology lies in its proprietary signal amplification system, which can be visualized through the following mechanism:
Diagram 2: RNAscope Signal Amplification Mechanism. The proprietary "Z" probe system creates dramatic signal amplification through sequential binding of pre-amplifier, amplifier, and label probes, enabling single-molecule detection while maintaining low background.
Analytical Considerations for Implementation
Sensitivity Thresholds: RNAscope technology demonstrates sufficient sensitivity to detect low-copy targets (1-20 copies per cell) with the HD Brown assay, making it particularly valuable for detecting viral infections with variable expression levels [74]. Traditional EBER ISH requires highly abundant targets like EBER RNAs for reliable detection.
Specificity Controls: The requirement for dual "Z" probe binding adjacent to each other on the target RNA molecule provides RNAscope with exceptional specificity, virtually eliminating off-target binding that plagues traditional ISH methods [10]. This feature is particularly valuable for distinguishing closely related viral strains or isoforms.
Multiplexing Capabilities: While traditional ISH methods offer limited multiplexing options, RNAscope supports simultaneous detection of multiple targets through either chromogenic detection on adjacent sections or fluorescent multiplexing with different channel probes [72]. The newer HiPlex assay extends this capability to 8 or 12 targets in the same sample.
Automation Compatibility: RNAscope assays can be automated on platforms like the Leica BOND RX system, significantly improving reproducibility and throughput while reducing hands-on time [35]. Traditional ISH methods are more challenging to automate due to variable hybridization conditions and lower signal-to-noise ratios.
This technical comparison demonstrates that RNAscope ISH represents a significant advancement over traditional EBER ISH and Universal HRP/AP methods for EBV detection. The technology's unique probe design and signal amplification system provide exceptional sensitivity and specificity while maintaining morphological context. For researchers and drug development professionals, RNAscope offers a robust platform for viral detection, biomarker validation, and therapeutic development that surpasses conventional ISH methodologies in performance metrics and operational efficiency. While traditional EBER ISH remains valuable for specific applications, RNAscope's superior technical characteristics position it as the emerging standard for precise RNA visualization in situ.
RNA in situ hybridization (ISH) has been revolutionized by the advent of spatially resolved molecular detection technologies, with the RNAscope platform representing a significant leap beyond conventional methodologies. This technical guide examines the emerging integration of RNAscope with artificial intelligence (AI) and digital pathology, a synergy that is creating new paradigms for quantitative spatial biology. Within the broader thesis context of comparing RNAscope versus conventional ISH sensitivity, this integration addresses critical limitations of traditional approaches, enabling unprecedented analytical precision at the single-molecule and single-cell level. For researchers, scientists, and drug development professionals, this convergence represents a transformative shift from qualitative assessment to data-rich quantitative analysis, enhancing capabilities in biomarker validation, therapeutic development, and fundamental disease mechanism investigation.
The core advantage of RNAscope lies in its proprietary double Z probe design, which achieves exceptional signal-to-noise ratio by requiring two independent probes to hybridize in tandem for signal amplification to occur [5]. This technology allows each punctate dot to represent an individual RNA molecule, providing a foundation for precise quantification that conventional ISH cannot reliably offer [7] [5]. When combined with digital pathology workflows and AI-driven analytical algorithms, this platform evolves from a detection tool to a comprehensive quantitative system capable of characterizing complex biological interactions with spatial context preserved.
RNAscope Technology: Fundamental Advantages Over Conventional ISH
Core Technology and Mechanism
RNAscope's distinctive technological foundation centers on its patented probe design and signal amplification strategy that fundamentally differ from conventional ISH approaches. The key innovation involves double Z probes that each contain a target-specific hybridization region, a spacer sequence, and a 14-base tail sequence [5]. When two probes bind adjacent to each other on the target RNA, their tail sequences combine to create a 28-base binding site for a pre-amplifier molecule, initiating a signal amplification cascade that ultimately generates a detectable punctate dot [5].
This design provides critical advantages over conventional ISH:
- Specificity: Non-specific binding of individual probes does not produce amplification sites, virtually eliminating background noise [5]
- Sensitivity: The 20x20x20 probe design enables detection of individual RNA molecules with only three double Z probe pairs required for signal generation [5]
- Degradation Resistance: Short target regions (40-50 bases) allow successful hybridization even in partially degraded RNA samples [5]
The sequential hybridization process involves four distinct steps: (1) target probe hybridization to RNA, (2) pre-amplifier binding to assembled probe pairs, (3) amplifier binding to pre-amplifiers, and (4) label probe binding for signal detection [5]. This multi-step process ensures that only specifically bound probes generate detectable signals, addressing the primary limitation of conventional ISH—high background interference that complicates quantification and interpretation.
Quantitative Comparison with Conventional ISH
Table 1: Performance Comparison Between RNAscope and Conventional ISH
| Parameter | RNAscope Technology | Conventional ISH |
|---|---|---|
| Sensitivity | Single-molecule detection [5] | Limited, typically requiring higher transcript abundance |
| Specificity | High (double Z-probe design prevents non-specific amplification) [5] | Variable (prone to non-specific binding and background) |
| Signal-to-Noise Ratio | Excellent (amplification only with tandem probe binding) [5] | Moderate to poor (non-specific signal amplification) |
| Quantification Capability | Direct single-molecule counting [8] | Semi-quantitative at best |
| Multiplexing Capacity | High (up to 12-plex demonstrated) [14] | Limited |
| Compatibility with Degraded Samples | Good (short target sequences) [5] | Poor |
| Probe Design Flexibility | Custom probes in 2 weeks [7] | Often lengthy optimization |
The differences extend beyond technical specifications to practical research applications. RNAscope's single-molecule visualization enables true quantitative analysis rather than the semi-quantitative scoring typically associated with conventional ISH [8]. This capability proves particularly valuable for detecting low-abundance transcripts and characterizing heterogeneous cell populations where average expression measurements mask biologically significant variations.
Quantitative Analysis of RNAscope Data: From Manual Counting to Automated Workflows
Traditional Analysis Methods and Their Limitations
Initial RNAscope applications relied heavily on manual quantification approaches, where researchers visually counted punctate dots under microscopy. While providing direct transcript enumeration, this method presented significant challenges:
- Time-intensive processing: Manual counting limited analysis throughput [8]
- Operator dependency: Inter-observer variability affected reproducibility [8]
- Sample size constraints: Practical considerations restricted analysis to small tissue regions
- Subjective threshold determination: Inconsistent criteria for positive signal identification [8]
These limitations prompted the development of semi-quantitative scoring systems that categorized expression levels based on predefined criteria. While improving throughput, these approaches sacrificed the precise quantitative information inherent in the RNAscope data structure and introduced categorical boundaries that may not reflect biological continuity.
Automated Image Analysis Solutions
The integration of digital pathology and specialized image analysis software has transformed RNAscope quantification by enabling automated, high-throughput processing while maintaining single-cell resolution. Current solutions include:
- QuPath: An open-source bioimage analysis platform that enables automated cell detection and puncta quantification [8]
- HALO Software: Commercial platform used for quantitative digital image analysis of RNAscope data [9]
- Custom Scripts: Researcher-developed solutions for specific experimental needs [8]
These platforms facilitate whole-slide analysis capabilities, allowing characterization of transcript distribution patterns across entire tissue sections while maintaining cellular resolution. The automated workflows typically involve sequential steps of tissue detection, cell segmentation, puncta identification, and signal thresholding, generating rich datasets that link transcript abundance to morphological context.
A critical advancement in automated analysis is the implementation of standardized thresholding methods that objectively distinguish true signal from background. As noted in recent protocols, "we describe a method to derive mRNA signal thresholds using negative controls" [8], establishing reproducible criteria for positive cell identification that address a key limitation of manual quantification.
Integration with Digital Pathology and AI
Digital Pathology Workflows
The adoption of whole slide imaging (WSI) systems has created foundational infrastructure for quantitative RNAscope analysis. Digital pathology workflows involve:
- Slide scanning: High-resolution digitization of entire tissue sections [8]
- Digital asset management: Organized storage and retrieval of large image files
- Cloud-based analysis: Computational processing without local hardware limitations [75]
These workflows enable multi-scale analysis, from tissue-level distribution patterns to subcellular transcript localization, while preserving the spatial context that distinguishes RNAscope from bulk molecular techniques like PCR or sequencing.
The Association for Pathology Informatics has highlighted how the integration of DICOM standards in digital pathology is "improving workflow, developing interoperability, and building the lab of the future" [76], creating infrastructure that supports advanced RNAscope applications in both research and clinical settings.
Artificial Intelligence and Machine Learning Applications
AI technologies are dramatically expanding RNAscope's analytical potential through several innovative approaches:
Foundation Models: Pre-trained on vast collections of whole slide images, these models enable rapid development of specific analysis tools without starting from scratch [75]. For example, Johnson & Johnson Innovative Medicine's MIA:BLC-FGFR algorithm, built on a foundation model trained on over 58,000 WSIs, predicts FGFR alterations in bladder cancer with 80-86% AUC [75].
Spatial Biomarkers: AI algorithms can quantify complex cellular interactions within the tumor microenvironment. Recent research in advanced non-small cell lung cancer demonstrated that a "five-feature model analyzing interactions between tumor cells, fibroblasts, T-cells, and neutrophils achieved a hazard ratio of 5.46 for progression-free survival" [75], significantly outperforming conventional biomarkers like PD-L1 expression alone.
Multimodal Integration: Combining RNAscope data with other data types creates comprehensive biological insights. Researchers from UCSF and Artera have validated a "multimodal AI (MMAI) biomarker for predicting prostate cancer outcomes" that integrates H&E images with clinical variables, successfully stratifying patients by metastasis risk with those classified as high-risk showing "18% vs. 3% for low-risk" 10-year metastasis rates [75].
Table 2: AI Applications in RNAscope Data Analysis
| AI Approach | Application | Performance/Outcome |
|---|---|---|
| Foundation Models | Prediction of molecular status from H&E slides | 80-86% AUC for FGFR alteration prediction in bladder cancer [75] |
| Spatial Analysis | Quantifying tumor microenvironment interactions | HR of 5.46 for progression-free survival prediction in NSCLC [75] |
| Multimodal AI | Integrating image data with clinical variables | 18% vs. 3% 10-year metastasis risk stratification in prostate cancer [75] |
| Quantitative Continuous Scoring | Computational pathology solution for therapy response | FDA Breakthrough Device Designation for TROP2 companion diagnostic [75] |
Visualization of RNAscope with AI Integration
The following diagram illustrates the integrated workflow of RNAscope technology with digital pathology and AI analysis:
RNAscope-AI Integrated Workflow: From sample preparation to quantitative results.
Experimental Protocols and Methodologies
Standardized RNAscope Protocol for Quantitative Analysis
Robust experimental execution is essential for reliable quantification. The following protocol, adapted from established methodologies [8], ensures high-quality results compatible with digital analysis:
Tissue Preparation (Fresh Frozen)
- Brain collection: In RNase-free conditions, deeply anesthetize the rat with isoflurane and perform sacrifice by decapitation
- Snap-freezing: Quickly remove the brain and immediately drop it in chilled 2-methylbutane (-30°C) for 25 seconds [8]
- Storage: Wrap snap-frozen brain in aluminum foil and store at -80°C for up to 12 months
RNAscope Assay Execution
- Cryostat sectioning: Prepare 10μm slices using a cryostat and mount on Superfrost Plus microscope slides
- Fixation: Fix slides in 4% paraformaldehyde for 15 minutes at 4°C
- Protease treatment: Apply RNAscope Protease IV for 30 minutes at room temperature [8]
- Probe hybridization: Incubate with target-specific RNAscope probes (e.g., Rn-Hcrtr1-C1, Rn-Th-C2, Rn-Fos-C3) for 2 hours at 40°C in HybEZ II oven [8]
- Signal amplification: Apply AMP 1 (30 minutes at 40°C), AMP 2 (30 minutes at 40°C), and AMP 3 (15 minutes at 40°C)
- Detection: Apply fluorescent label probes and counterstain with DAPI
- Coverslipping: Mount with Fluoro-Gel II and cure overnight at room temperature
Image Acquisition and Analysis
- Slide scanning: Digitize slides using a slide scanner (e.g., Zeiss AxioScan Z.1) with consistent exposure settings
- Cell detection: Apply optimized cell detection algorithm in QuPath or HALO software
- Threshold determination: Establish signal thresholds using negative control samples [8]
- Quantitative analysis: Execute automated puncta counting and cell classification
Quality Control and Validation
Maintaining analytical rigor requires systematic quality control measures:
- Negative controls: Include RNAscope 3-plex negative control probes to establish background thresholds [8]
- Positive controls: Utilize housekeeping genes (e.g., 18S rRNA, PPIB) to confirm technical success [7]
- Threshold optimization: "Derive mRNA signal thresholds using negative controls" to objectively distinguish true signals [8]
- Batch effects: Process experimental and control samples simultaneously to minimize technical variability
The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Essential Research Reagents for RNAscope Experiments
| Item | Function | Example Products/Specifications |
|---|---|---|
| RNAscope Probe Sets | Target-specific detection | Catalogued probes for >100 viruses; custom designs in 2 weeks [7] |
| RNAscope Kit Reagents | Signal amplification and detection | RNAscope Fluorescent Multiplex reagent kit v1 (fresh frozen) or Universal Pretreatment kit (FFPE) [8] |
| Protease Reagents | Tissue pretreatment for RNA accessibility | RNAscope RTU Protease IV (fresh frozen) or Protease III (FFPE) [8] |
| Negative Control Probes | Background threshold determination | RNAscope 3-plex negative control probes [8] |
| Positive Control Probes | Assay validation | Housekeeping gene probes (e.g., PPIB, POLR2A) |
| Microscope Slides | Tissue mounting | Fisherbrand Superfrost Plus microscope slides [8] |
| Coverslipping Medium | Slide preservation and imaging | Fluoro-Gel II with DAPI [8] |
| Hybridization System | Controlled assay conditions | HybEZ II system with humidity control [8] |
| Image Analysis Software | Quantification and data extraction | QuPath (open-source), HALO software, or custom scripts [9] [8] |
The integration of RNAscope with digital pathology and AI represents a paradigm shift in spatial biology, moving from qualitative observation to quantitative, data-rich analysis. This convergence addresses the core thesis of RNAscope's superiority over conventional ISH by providing the analytical framework to fully leverage its single-molecule detection capability. Emerging trends suggest several future developments:
- Expanded multiplexing: Technologies like RNAscope HiPlex Pro are pushing beyond current limits, with demonstrations of up to 12-plex RNA detection and 24-plex protein detection on platforms like the Lunaphore COMET [14]
- Clinical translation: The recent FDA Breakthrough Device Designation for AstraZeneca's Quantitative Continuous Scoring (QCS) computational pathology solution marks a milestone in regulatory acceptance of AI-enhanced pathology [75]
- Standardized workflows: As noted in recent protocols, there is growing emphasis on "unification and standardization of this approach across neuroscience labs" and other fields [8]
For researchers and drug development professionals, these advancements offer powerful new approaches to biomarker discovery, therapeutic efficacy assessment, and mechanistic studies. The ability to precisely quantify RNA expression within morphological context, especially when enhanced by AI-driven pattern recognition, provides insights previously obscured by technical limitations.
In conclusion, the integration of RNAscope with digital pathology and AI represents more than incremental improvement—it constitutes a fundamental transformation of what's possible in spatial biology. By combining RNAscope's exceptional sensitivity and specificity with the quantitative power of computational analysis, researchers can now address biological questions with precision that aligns with the complexity of biological systems, advancing both basic science and therapeutic development.
Conclusion
RNAscope technology represents a significant advancement over conventional ISH, offering researchers unparalleled sensitivity capable of detecting single RNA molecules and high specificity through its unique 'Z' probe design. The method's versatility across multiple research domains, combined with robust automated workflows and comprehensive validation data, positions it as an essential tool for precise spatial gene expression analysis. Future directions include broader adoption in clinical diagnostics following regulatory validation, expanded multiplexing capabilities for complex pathway analysis, and increased integration with artificial intelligence for automated quantification. For biomedical researchers, RNAscope provides a critical bridge between molecular detection and morphological context, enabling deeper insights into disease mechanisms and therapeutic development.
References
- 1. Fluorescence in situ hybridization (FISH): History ... [https://www.sciencedirect.com/science/article/pii/S259000721830008X]
- 2. Recent Advances in High-sensitivity In Situ Hybridization and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10323200/]
- 3. Single-Molecule Detection Technologies: Advances in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12564833/]
- 4. Analytical techniques for nucleic acid and protein detection ... [https://www.nature.com/articles/s12276-025-01453-w]
- 5. RNAscope Enables Spatial Genomic ISH ... [https://acdbio.com/science/how-it-works]
- 6. Best of 2024: Overview of insightful publications in diverse ... [https://acdbio.com/2025-feb-11-best-2024-overview-insightful-publications-diverse-research-areas-using-rnascope]
- 7. Viral RNA Detection using RNAscope® ISH | ACD [https://acdbio.com/science/applications/research-areas/infectious-diseases]
- 8. Quantitative Analysis of Gene Expression in RNAscope ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC9901451/]
- 9. An Overview of Quantitative Image Analysis of RNAscope ... [https://acdbio.com/2023-oct-10-overview-quantitative-image-analysis-rnascope-ish-data-professional-assay-services]
- 10. RNAscope: A Novel in Situ RNA Analysis Platform for ... [https://www.sciencedirect.com/science/article/pii/S1525157811002571]
- 11. Mastering Spatial RNA Detection: High Specificity & ... [https://news.ki.se/calendar/histocore-mastering-spatial-rna-detection-high-specificity-sensitivity-with-rnascope-ish-on-leica-bond-rx-by-acdbio]
- 12. Learn How RNAscope Technology Works [https://www.bio-techne.com/reagents/rnascope-ish-technology/get-started]
- 13. Evaluation of the Suitability of RNAscope as a Technique ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8710359/]
- 14. RNAscope ISH Technology [https://www.bio-techne.com/reagents/rnascope-ish-technology]
- 15. The optimisation and application of a novel BaseScope™ ... [https://www.sciencedirect.com/science/article/pii/S0166093425001302]
- 16. Quantitative Analysis of Gene Expression in RNAscope-processed Brain Tissue - PubMed [https://pubmed.ncbi.nlm.nih.gov/36789089/]
- 17. NGS Validation with RNAscope ISH [https://acdbio.com/science/applications/research-solutions/ngsht-transcriptomic-validation-rna-seq]
- 18. Antibody Validation & Challenges [https://acdbio.com/science/applications/research-solutions/antibody-validation-and-challenges]
- 19. RNAscope Multiplex FISH Signal Assessment in FFPE and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11755420/]
- 20. RNAscope® technology | In Situ Hybridization, RNA-ISH [https://acdbio.com/dataanalysisguide]
- 21. RNAscope ISH [https://www.bio-techne.com/reagents/rnascope-ish-technology/rnascope]
- 22. RNAscope Multiplex Fluorescent V2 Assay | ACD [https://acdbio.com/rnascope-multiplex-fluorescent-v2-assay]
- 23. RNAscope Assay Visualization and Interpretation [https://acdbio.com/support-solution-category/rnascope-assay-visualization-and-interpretation]
- 24. 2017 Jan 24 - Introduction to the RNAscope™ Assay [https://acdbio.com/2017-jan-24-introduction-rnascope%E2%84%A2-assay]
- 25. RNAscope Assay Technical Support and FAQ [https://acdbio.com/technical-support/getting-started]
- 26. 2025 Mar 18 - Accelerate RNA and Protein Biomarker ... [https://acdbio.com/2025-mar-18-accelerate-rna-and-protein-biomarker-development-new-rnascope%E2%84%A2-ish-protease-free-assays]
- 27. RNAscope Workflow, In Situ Hybridization Steps | ACD [https://acdbio.com/ebook/introduction/rnascope-workflow]
- 28. RNAscope Assay Workflow | In Situ Hybridization, RNA-ISH [https://acdbio.com/support-solution-category/rnascope-assay-workflow]
- 29. Identification of Optimal Decalcification Method and Tissue ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12651668/]
- 30. Spotlights | In Situ Hybridization, RNA-ISH [https://acdbio.com/researcherspotlight]
- 31. USCAP 114th Annual Meeting 2025 [https://acdbio.com/uscap-114th-annual-meeting-2025]
- 32. RNAscope ISH on Roche is now protease-free [https://www.bio-techne.com/reagents/rnascope-ish-technology/roche-automation-assays/now-protease-free]
- 33. Detection Assays for Proteins: Chromogenic vs ... [https://www.goldbio.com/blogs/articles/proten-detection-chromogenic-chemiluminescent-fluorometric?srsltid=AfmBOop-sALDMWd_cPR8_KPfW6wB9h1-9TWbDiwScwS2sjdCNt0LJCr1]
- 34. Why Multiplex & Chromogenic vs Immunofluorescent ... [https://www.leicabiosystems.com/us/articles/ls-articles/tips-tricks-to-multiplexing-top-5-reasons-to-multiplex-and-chromogenic-versus/]
- 35. Customers | In Situ Hybridization, RNA-ISH [https://acdbio.com/customers]
- 36. New-ISH on the Block: Introduction to RNAscope® [https://bitesizebio.com/40621/new-ish-on-the-block-introduction-to-rnascope/]
- 37. Review of In Situ Hybridization Techniques for Drug Research ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10467011/]
- 38. RNAscope Troubleshooting Guide and FAQ [https://acdbio.com/technical-support/solutions]
- 39. DISCOVERY ULTRA [https://diagnostics.roche.com/gb/en/article-listing/health-topics/immunohistochemistry/discovery-ultra.html]
- 40. RNAscope: A Novel in Situ RNA Analysis Platform for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3338343/]
- 41. 2025 Jan 14 - Unlock the role of spatial profiling in ... [https://acdbio.com/2025-jan-14-unlock-role-spatial-profiling-detecting-diverse-mrna-markers-and-short-targets]
- 42. RNAscope, In Situ Hybridization Technique | ACD [https://acdbio.com/referenceguide]
- 43. Review of In Situ Hybridization (ISH) Stain Images Using ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11430898/]
- 44. RNAscope Troubleshooting Guide and FAQ [https://acdbio.com/technical-support/solutions/faq]
- 45. Mapping human tissues with highly multiplexed RNA in situ ... [https://www.nature.com/articles/s41467-024-46437-y]
- 46. In Situ Hybridization Troubleshooting | ACD [https://acdbio.com/ebook/troubleshooting/sample-preparation-and-optimization]
- 47. Effect of formalin-fixation and paraffin-embedded tissue ... [https://pubmed.ncbi.nlm.nih.gov/38650110/]
- 48. RNA In situ hybridization (ISH) using RNAscope® [https://www.protocols.io/view/rna-in-situ-hybridization-ish-using-rnascope-dydu7s6w.html]
- 49. Control Slides and Control Probes for RNAscope [https://acdbio.com/control-slides-and-control-probes-rnascope]
- 50. Identification of Optimal Decalcification Method and Tissue ... [https://www.mdpi.com/2304-6767/13/11/538]
- 51. Visualization of spatial gene expression in plants by modified ... [https://plantmethods.biomedcentral.com/articles/10.1186/s13007-020-00614-4]
- 52. RNAscope ISH Reference Guide [https://www.bio-techne.com/reagents/rnascope-ish-technology/rnascope/rnascope-ish-reference-guide]
- 53. RNAscope ISH Technology [https://www.creative-bioarray.com/support/rnascope-ish-technology.htm]
- 54. RNAscope VS Universal HRP/ AP | In Situ Hybridization ... [https://acdbio.com/rnascope-vs-universal-hrp-ap]
- 55. HALO | Quantitative Image Analysis for Pathology [https://indicalab.com/halo/]
- 56. QuPath RNA ISH Analysis | In Situ Hybridization ... [https://acdbio.com/qupath-rna-ish-analysis]
- 57. Comparison of QuPath and HALO platforms for analysis ... [https://www.sciencedirect.com/science/article/abs/pii/S0023683725001564]
- 58. Digital pathology in tau research: A comparison of QuPath ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12277702/]
- 59. Image Capture and Image Analysis with RNAscope™ Assays [https://www.bio-techne.com/reagents/rnascope-ish-technology/image-analysis-solutions]
- 60. Comparison of QuPath and HALO platforms for analysis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12139998/]
- 61. Images — QuPath 0.6.0 documentation - Read the Docs [https://qupath.readthedocs.io/en/stable/docs/concepts/images.html]
- 62. Changing colors · qupath/qupath Wiki [https://github.com/qupath/qupath/wiki/Changing-colors]
- 63. Digital PCR quantification of ultrahigh ERBB2 copy number ... [https://www.nature.com/articles/s41523-024-00621-x]
- 64. Automated Quantitative RNA in Situ Hybridization for ... [https://www.sciencedirect.com/science/article/pii/S1525157812003157]
- 65. Concordance of RT-qPCR with immunohistochemistry and its ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10519502/]
- 66. Concordance Analysis of ALK Gene Fusion Detection ... [https://www.sciencedirect.com/science/article/pii/S1525157821001756]
- 67. SISH/CISH or qPCR as alternative techniques to FISH for ... [https://bmccancer.biomedcentral.com/articles/10.1186/1471-2407-13-351]
- 68. RNAscope in situ hybridization (ISH) for Clinical Diagnostics [https://acdbio.com/diagnostics]
- 69. 2025 Mar 25 - Visualize and Quantify Oligonucleotide ... [https://acdbio.com/2025-mar-25-visualize-and-quantify-oligonucleotide-therapy-delivery-spatial-biodistribution-and]
- 70. Plasma Epstein-Barr Virus DNA load for diagnostic and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11747383/]
- 71. Comparison of in situ hybridization using different ... [https://pubmed.ncbi.nlm.nih.gov/1331609/]
- 72. In Situ Hybridization - Histology Research Core [https://histologyresearchcorefacility.web.unc.edu/rna-scope/]
- 73. Comparative study of five commercial probes for the ... [https://www.redalyc.org/journal/3935/393549337010/html/]
- 74. RNAscope 2.5 HD Assay—BROWN | In Situ Hybridization, ... [https://acdbio.com/rnascope-25-hd-assay%E2%80%94brown]
- 75. Digital Pathology and AI Highlights from ASCO 2025 [https://proscia.com/digital-pathology-and-ai-highlights-from-asco-2025/]
- 76. Digital Pathology and AI 2025 [https://www.pathologyinformatics.org/dp-ai-9-0-welcome-]