Xenopus Fate Mapping: A Definitive Guide to Blastomere Identification for Developmental Biology and Biomedical Research
This comprehensive guide details the principles and practice of creating and interpreting fate maps in Xenopus laevis and tropicalis embryos.
Xenopus Fate Mapping: A Definitive Guide to Blastomere Identification for Developmental Biology and Biomedical Research
Abstract
This comprehensive guide details the principles and practice of creating and interpreting fate maps in Xenopus laevis and tropicalis embryos. It covers the foundational history and significance of blastomere lineage tracing, modern methodological approaches including microinjection and live imaging, and best practices for troubleshooting common experimental challenges. Furthermore, it provides a critical validation framework comparing fate mapping techniques and their applications in modeling human disease and screening teratogens. Designed for researchers and drug development professionals, this article serves as an essential resource for leveraging Xenopus embryos in developmental studies and translational research.
Understanding Xenopus Fate Maps: History, Principles, and Core Concepts in Lineage Tracing
What is a Fate Map? Defining Blastomere Identity and Developmental Potential
This technical guide explores the concept of a fate map, a foundational tool in developmental biology that delineates the prospective fate and developmental potential of embryonic cells. Framed within a broader thesis on Xenopus laevis embryo research, we detail the methodologies for blastomere identification, the transition from fate to specification and commitment, and the experimental paradigms used to construct quantitative fate maps. This whitepaper serves as a resource for researchers and drug development professionals investigating cell lineage, morphogenesis, and teratogenicity.
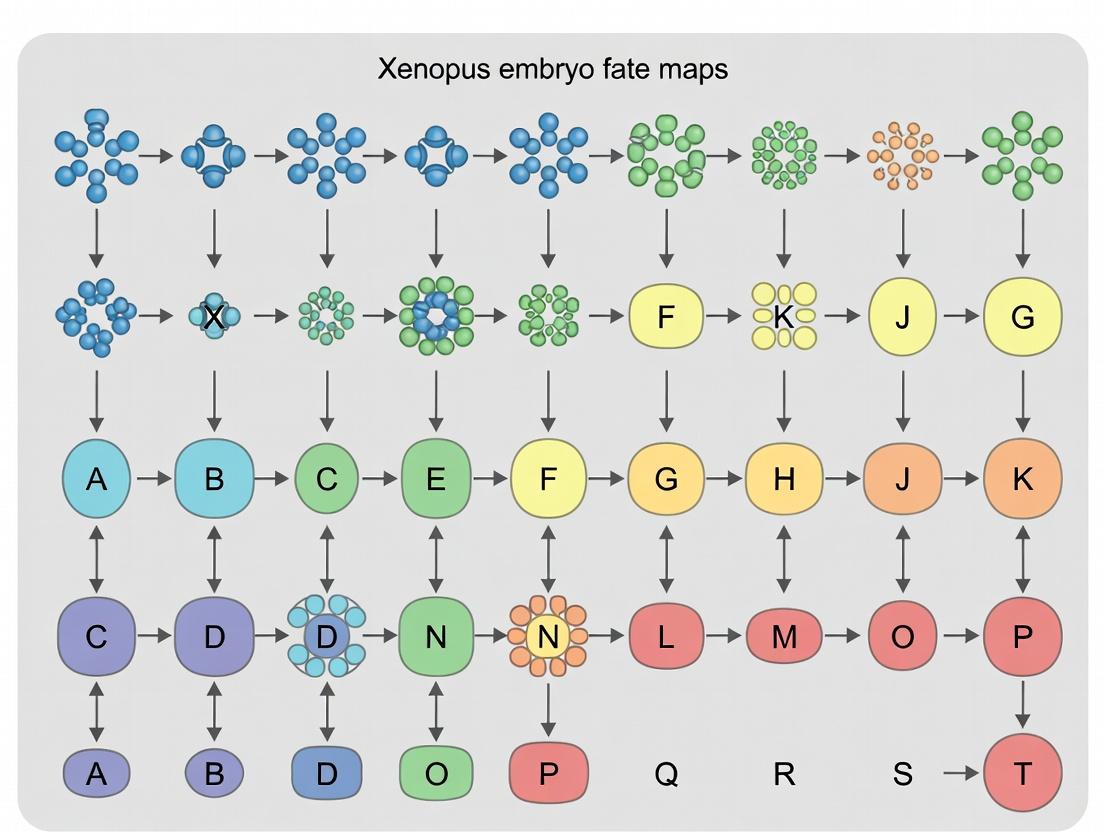
A fate map is a diagrammatic representation of an embryo at a specific developmental stage, indicating the future tissue or organ contributions (prospective fate) of defined regions or individual cells. In the context of Xenopus research, fate maps are instrumental for understanding the origin of germ layers and the spatial organization of the embryonic body plan. The core inquiry extends beyond descriptive fate to assess developmental potential (the range of possible fates a cell can adopt if experimentally challenged) versus restrictive specification (the progressive limitation of that potential). Constructing a fate map is thus the first critical step in blastomere identification, linking spatial coordinates to eventual functional identity.
Historical and Modern Techniques for Fate Mapping inXenopus
Classic Lineage Tracing Methods
- Vital Dye Marking: Injection of non-diffusible, non-toxic dyes (e.g., DiI, Fluorescein-dextran) into individual blastomeres.
- Radioactive/Enzymatic Tracing: Injection of tritiated thymidine or horseradish peroxidase.
- Function: Short-term lineage tracking through gastrulation and neurulation.
Contemporary Genetic and Imaging Approaches
- mRNA Injection: Synthetic mRNA encoding fluorescent proteins (e.g., GFP, mCherry) or photoactivatable proteins.
- Genome Editing Integration: Using CRISPR/Cas9 to insert reporter constructs (e.g., H2B-GFP) into specific loci for stable, heritable labeling.
- Intravital Time-Lapse Microscopy: Enables high-resolution, four-dimensional tracking of labeled blastomeres in living embryos.
Core Experimental Protocols for Blastomere Analysis
Protocol 3.1: Fluorescent Dextran Lineage Tracing in 32-Cell StageXenopusEmbryos
- Embryo Preparation: Obtain Xenopus embryos, dejelly using cysteine solution, and culture in 0.1x MMR to the desired stage (e.g., stage 6, 32-cell).
- Microinjection Setup: Prepare a 2-5 nL injection solution of lysine-fixable Fluorescein- or Rhodamine-dextran (10-20 mg/mL in nuclease-free water).
- Blastomere Injection: Using a calibrated microinjector and fine glass needle, target the specific blastomere of interest (identified by established naming conventions, e.g., A1, B1, C1, D1 tiers). Inject into the animal cytoplasm.
- Fixation and Imaging: Allow embryos to develop to the stage of interest. Fix in 4% PFA for 2 hours at room temperature. Image using a fluorescence stereomicroscope or confocal microscope.
- Fate Analysis: Document the localization of fluorescent signal in germ layers and tissues. Pool data from multiple embryos to generate a probabilistic fate map.
Protocol 3.2: Single-Cell Transplantation for Testing Developmental Potential
- Donor and Host Preparation: Label donor embryo blastomeres vitally with a fluorescent lineage tracer (as in Protocol 3.1). Prepare age-matched host embryos (unlabeled).
- Microsurgery: Using a sharp glass needle and hair loop, carefully excise a single labeled blastomere from the donor.
- Transplantation: Transplant the isolated blastomere into a novel location in the host embryo (e.g., transplant a vegetal cell into the animal region).
- Culture and Analysis: Culture the host embryo and analyze the contribution of the transplanted cell at later stages. Differentiation in the new location reveals potency; maintenance of original fate indicates specification.
Quantitative Fate Map Data forXenopus laevis(32-Cell Stage)
The tables below summarize canonical fate contributions, based on seminal and modern studies.
Table 1: Prospective Fate of Dorsal Tier Blastomeres (Animal View)
| Blastomere | Primary Germ Layer Contribution | Major Tissue/Organ Derivatives (Probability >70%) |
|---|---|---|
| D1 (Dorsal-Animal) | Ectoderm | Anterior CNS (Forebrain, Retina), Epidermis |
| D2 (Dorsal-Marginal) | Mesoderm & Endoderm | Anterior Dorsal Mesoderm (Prechordal Plate), Pharyngeal Endoderm |
| C1 (Dorsal-Vegetal) | Endoderm & Mesoderm | Dorsal Endoderm (Gut), Axial Mesoderm (Notochord) |
Table 2: Developmental Potential Assay via Blastomere Transplantation
| Blastomere Origin (32-cell) | Host Transplant Site | Resultant Fate (Common Outcome) | Interpretation of Potential |
|---|---|---|---|
| V1 (Ventral-Vegetal) | Dorsal Marginal Zone | Ventral Blood Island Mesoderm | Specified: Fate restricted to ventral mesendoderm. |
| A1 (Animal) | Vegetal Pole | Epidermis only | Restricted: Cannot form endoderm or mesoderm. |
| C1 (Dorsal-Vegetal) | Animal Pole | Diverse tissues (Neural, Mesodermal) | Retains Broad Potential: Inductive signals can redirect fate. |
The Molecular Basis of Fate: Key Signaling Pathways
Fate specification is governed by the asymmetric distribution of maternal determinants and subsequent inductive signaling. Key pathways are diagrammed below.
Diagram Title: Wnt/β-Catenin Signaling in Dorsal Axis Specification
Diagram Title: TGF-β/Nodal Signaling for Mesendoderm Induction
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Xenopus Fate Mapping Research
| Reagent/Material | Function & Application in Fate Mapping |
|---|---|
| Lysine-Fixable Fluorescein-Dextran (10,000 MW) | High molecular weight, membrane-impermeant tracer for long-term lineage labeling. Remains in injected cell and its progeny. |
| Capped, Polyadenylated Synthetic mRNA (e.g., H2B-GFP) | For ubiquitous nuclear-localized labeling. Enables precise cell tracking and quantification in living embryos. |
| Morpholino Oligonucleotides or CRISPR/Cas9 Components | Gene knockdown or knockout tools to test the function of specific genes in blastomere specification. |
| Recombinant Growth Factors (e.g., Activin A, BMP4) | Used in animal cap assays to test the inductive capacity and response potential of blastomeres. |
| Paraformaldehyde (PFA, 4%) | Standard fixative for preserving embryo morphology and fluorescence post-lineage tracing. |
| Manually Pulled Glass Capillary Needles | For precise microinjection into single blastomeres without causing lethal damage. |
Integration with Drug Development and Toxicology
Fate maps provide a predictive framework for developmental toxicology. By understanding the normal fate and signaling environment of a blastomere, researchers can design assays to test how pharmaceutical compounds or environmental toxins disrupt cell specification, migration, and tissue formation. Xenopus offers a medium-throughput vertebrate model for such teratogenicity screens, where perturbed fate maps can serve as a quantifiable endpoint for developmental toxicity.
Within the broader thesis on Xenopus embryo fate maps and blastomere identification research, the historical legacy of lineage analysis forms the essential foundation. This in-depth technical guide examines the seminal studies that established Xenopus laevis as a premier model for understanding vertebrate embryonic development, cell fate determination, and the molecular logic of pattern formation.
Seminal Lineage Tracing Studies: Data and Impact
The following table summarizes key quantitative findings from historic lineage analysis experiments that mapped blastomere fates.
Table 1: Foundational Xenopus laevis Lineage Tracing Studies
| Study (Authors, Year) | Key Technique | Stage Analyzed | Major Finding (Quantitative Summary) | Contribution to Fate Map |
|---|---|---|---|---|
| Nakamura & Kishiyama, 1971 | Vital dye injection (Niagara blue) | 1- to 32-cell | Mapped approximate contributions of early blastomeres to larval structures. | First comprehensive fate map for early cleavage stages. |
| Moody, 1987 | Intracellular injection of horseradish peroxidase (HRP) | 32- and 64-cell | Identified exact lineage of ~90% of cells in the neural plate; defined precursor domains for specific neuronal types. | Established a detailed fate map for the nervous system with clonal resolution. |
| Dale & Slack, 1987 | Fluorescent dextran lineage tracers | 8- to 64-cell | Defined fate of identified blastomeres up to tadpole stage; mapped territories for epidermis, neural tissue, mesoderm, and endoderm. | Created the standard "fate map" for the 32-cell stage embryo, widely used for decades. |
| Kimmel & Law, 1985 | Microinjection of single blastomeres with HRP | Early cleavage | Traced division patterns and cell movements; provided precise timing and geometry of early development. | Added dynamic dimension to static fate maps, emphasizing cell lineage and morphogenesis. |
Detailed Experimental Protocols
Protocol 1: Intracellular Horseradish Peroxidase (HRP) Injection (Moody, 1987)
This protocol allows for permanent, high-resolution tracing of a blastomere's progeny.
- Embryo Preparation: Fertilize Xenopus eggs in vitro. Dejelly embryos chemically (e.g., 2% cysteine hydrochloride, pH 8.0) and culture in 0.1x Modified Barth's Saline (MBS) until desired cleavage stage (e.g., 32-cell).
- Micropipette Preparation: Pull glass capillary tubes to a fine tip. Backfill with a 4% solution of HRP (Type VI) in 0.2 M KCl.
- Microinjection: Mount embryo in a depression slide with fine nylon mesh in 1x MBS. Using a micromanipulator and pressure injector, penetrate a single, identified blastomere. Inject approximately 4-10 nl of HRP solution, visualized by a slight expansion of the cell.
- Fixation and Development: Allow embryo to develop to desired stage (e.g., neural plate). Fix in 2.5% glutaraldehyde in 0.1 M phosphate buffer (pH 7.4) for 1 hour. Rinse thoroughly.
- Histochemical Reaction: Incubate embryos in 0.5 mg/ml diaminobenzidine (DAB) with 0.01% hydrogen peroxide in Tris buffer. Reaction proceeds for 10-30 minutes until brown precipitate forms in HRP-containing cells.
- Analysis: Clear embryo in glycerol or section. HRP-labeled cells are permanently stained brown and can be mapped under a microscope.
Protocol 2: Fluorescent Dextran Lineage Tracing (Dale & Slack, 1987)
This protocol enables live and fixed visualization of clones.
- Tracer Solution: Prepare a 10-20 mg/ml solution of lysine-fixable fluorescent dextran (e.g., Fluorescein or Rhodamine conjugated, 10,000 MW) in distilled water.
- Injection: Follow steps 1-3 from Protocol 1, injecting ~5 nl of fluorescent dextran solution into the target blastomere of an 8- to 64-cell embryo.
- Live Imaging: Culture injected embryos and observe under a fluorescence dissection or compound microscope at successive stages to trace clone location and morphology.
- Fixation and Counterstaining: Fix embryos at the terminal stage in 4% paraformaldehyde. For whole-mount imaging, clear in Murray's clear (benzyl benzoate:benzyl alcohol, 2:1). Optionally, counterstain nuclei with DAPI.
- Imaging: Analyze using epifluorescence or confocal microscopy.
Signaling Pathways in Cell Fate Specification
The fate of blastomeres mapped in lineage studies is determined by conserved signaling pathways.
Lineage Analysis Experimental Workflow
The general workflow for a classical lineage tracing experiment.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Xenopus Lineage Analysis
| Reagent/Material | Function in Experiment |
|---|---|
| Lineage Tracers (Fluorescent Dextrans, e.g., FITC-/Rhodamine-dextran, 10kDa lysine-fixable) | Injected into blastomeres; they are metabolically inert, do not pass through gap junctions, and allow visualization of all progeny. Fluorescent version enables live imaging. |
| Horseradish Peroxidase (HRP, Type VI) | A classic enzymatic tracer. Injected and later developed with DAB to produce a permanent, high-resolution stain visible by light microscopy. |
| Synthetic mRNAs (e.g., for GFP, lineage-specific markers) | Co-injected with tracer to link lineage to molecular manipulation (e.g., overexpression, dominant-negative). GFP provides a live tracer. |
| Morpholino Oligonucleotides | Antisense molecules for knocking down specific gene functions in the injected lineage. Allows fate mapping under loss-of-function conditions. |
| Modified Barth's Saline (MBS) | Standard physiological saline for culturing Xenopus embryos post-injection, maintaining osmotic balance and health. |
| Agarose Injection Molds | Used to create wells or grooves to immobilize embryos during microinjection without damage. |
| Diethylpyrocarbonate (DEPC)-treated Water | RNase-free water for preparing solutions for RNA/DNA injection to prevent degradation of sensitive nucleic acids. |
| Diaminobenzidine (DAB) | Chromogenic substrate for HRP. Forms an insoluble brown precipitate upon enzymatic reaction, marking the lineage. |
1. Introduction
This technical guide details the anatomical and experimental framework for blastomere identification in early vertebrate embryos, specifically contextualized within a broader thesis on fate mapping and lineage tracing in Xenopus laevis. The period from the 2-cell to the 32-cell stage is critical, as it encompasses the establishment of the basic embryonic axes and the segregation of major lineage progenitors. Precise identification of individual blastomeres at these stages is foundational for experimental embryology, enabling targeted microinjections, ablation studies, and the construction of high-resolution fate maps that inform models of cell specification and morphogenesis.
2. Key Developmental Stages and Blastomere Nomenclature
The early cleavages in Xenopus follow a stereotypic pattern. The first cleavage furrow bisects the grey crescent, establishing the future dorsal-ventral axis. Subsequent cleavages are meridional and equatorial, dividing the embryo into identifiable blastomeres with predictable fates.
Table 1: Key Cleavage Stages and Blastomere Characteristics in Xenopus laevis
| Stage (Cell #) | Approximate Time Post-Fertilization (NF Stage*) | Cleavage Planes | Key Anatomical Landmarks for Orientation | Major Lineage Segregation |
|---|---|---|---|---|
| 2-cell | ~1.5 hours (NF 2) | 1st: Meridional, through animal-ventral pole. | Grey crescent (future dorsal side) located perpendicular to the first cleavage furrow. | Establishes left-right halves. |
| 4-cell | ~2 hours (NF 3) | 2nd: Meridional, perpendicular to the 1st. | Four equal-sized blastomeres in animal view. Dorsal blastomeres contain most of the grey crescent. | Distinguishes dorsal from ventral halves. |
| 8-cell | ~2.5 hours (NF 4) | 3rd: Latitudinal (equatorial), nearer the animal pole. | Two tiers: 4 animal (smaller) and 4 vegetal (larger) blastomeres. | Separates animal (ectodermal) from vegetal (endodermal) fates. |
| 16-cell | ~3 hours (NF 5) | 4th: Two meridional, then one latitudinal. | Four tiers: Animal (A), Vegetal (V), and intermediate tiers. Distinction of dorsal-vegetal (DV) signaling center. | Further subdivision of ectoderm and mesendoderm progenitors. |
| 32-cell | ~4 hours (NF 6) | 5th: Mixed meridional and latitudinal. | Complex, tiered arrangement. Blastomeres are named by a standard code (e.g., A1, B1, C1, D1 for animal tier). | Refined progenitor zones for specific tissues (e.g., neural, notochord, somite). |
NF: Nieuwkoop and Faber (1967) normal table of *Xenopus development.
3. Experimental Protocols for Blastomere Identification and Fate Mapping
Protocol 1: Vital Dye Labeling for Lineage Tracing
- Objective: To mark individual blastomeres and trace their descendants through later development.
- Materials: Fine glass capillary needles, pneumatic microinjector, injection plate with agarose wells, vital dyes (e.g., Lysinated Rhodamine Dextran, Fluorescein Dextran), 3-5% Ficoll in 1x MMR.
- Procedure:
- Dejelly embryos chemically or manually in 2% cysteine (pH 8.0).
- Align and immobilize the embryo in an agarose well on an injection plate submerged in 3-5% Ficoll/1x MMR.
- Using a micromanipulator, insert a capillary needle loaded with 10-20 nL of fluorescent dextran (e.g., 10 mg/mL) into the target blastomere.
- Apply a brief pressure pulse to deliver the dye. The dye should fill the blastomere without leaking.
- After injection, gently transfer embryos to 0.1x MMR for recovery and culture until the desired stage.
- Analyze using fluorescence microscopy at successive stages to map descendant cell locations.
Protocol 2: Targeted Microinjection of Morpholino Oligonucleotides or mRNA
- Objective: To perturb gene function in a lineage-specific manner.
- Materials: As in Protocol 1, plus Morpholino oligonucleotides or in vitro transcribed mRNA.
- Procedure:
- Prepare embryos and immobilize as in Protocol 1.
- Identify the target blastomere based on known fate maps (e.g., inject dorsal-vegetal blastomeres to affect organizer formation).
- Inject a calibrated volume (typically 5-10 nL) of the reagent (e.g., 1-5 ng Morpholino, 50-200 pg mRNA) into the desired blastomere.
- Culture embryos and score phenotypes, comparing injected to uninjected control siblings.
4. Blastomere Fate Maps and Signaling Pathways
Fate mapping experiments have established the general contribution of early blastomeres. The dorsal-vegetal (DV) blastomeres at the 32-cell stage are a crucial source of inductive signals, including Nodal and Wnt ligands, which pattern the mesoderm and endoderm. The specification of these lineages relies on conserved signaling pathways.
Table 2: Representative Blastomere Fates at the 32-Cell Stage (Adapted from Moody, 1987)
| Blastomere Tier & Code | Primary Germ Layer Contribution | Major Tissue Derivatives |
|---|---|---|
| Animal (A-tier) | Ectoderm | Epidermis, anterior central nervous system. |
| Animal (B-tier) | Ectoderm/Mesoderm | Posterior CNS, neural crest, head mesoderm. |
| Vegetal (C-tier) | Mesendoderm | Heart, kidney, lateral/ventral mesoderm, gut endoderm. |
| Vegetal (D-tier: Dorsal) | Organizer (Mesendoderm) | Notochord, prechordal plate, pharyngeal endoderm. |
Short Title: Signaling from Dorsal Blastomere to Organizer
Short Title: Blastomere Identification and Targeting Workflow
5. The Scientist's Toolkit: Essential Research Reagents and Materials
Table 3: Key Research Reagent Solutions for Xenopus Blastomere Studies
| Item | Function/Application |
|---|---|
| 1x MMR (Marc's Modified Ringer's) | Standard saline solution for embryo culture and microinjection setups. |
| Ficoll PM 400 (3-5% in 1x MMR) | Injection medium; increases viscosity to reduce leakage and buffer embryos during microinjection. |
| Fluorescent Lineage Tracers (e.g., Lysinated Rhodamine Dextran, FDA) | Vital dyes for labeling individual blastomeres to trace cell lineage and fate. |
| Morpholino Antisense Oligonucleotides | Gene-specific knockdown tools to inhibit mRNA translation or splicing in a lineage-restricted manner. |
| Capped, Polyadenylated Synthetic mRNA | For targeted overexpression of wild-type or mutant proteins in specific blastomeres. |
| Agarose Plates with Micro-wells | For immobilizing and orienting embryos during microinjection procedures. |
| Glass Capillary Microneedles | For precise delivery of reagents into single blastomeres with minimal damage. |
| Horseradish Peroxidase (HRP) | A non-fluorescent tracer used for high-resolution lineage analysis via histochemical staining. |
| Whole-Mount In Situ Hybridization (WMISH) Reagents | To analyze gene expression patterns resulting from lineage-specific manipulations. |
The Classic Nieuwkoop and Faber Normal Table as a Foundational Reference
Within the field of Xenopus developmental biology, the construction of accurate embryo fate maps and the precise identification of blastomeres are fundamental to dissecting the mechanisms of cell specification, morphogen signaling, and tissue patterning. The broader research thesis posits that quantitative, high-resolution fate mapping, integrated with modern molecular techniques, is essential for validating and extending classical embryological concepts. At the core of this pursuit lies the Normal Table of Xenopus Development (Nieuwkoop and Faber, 1967). This work provides the definitive morphological staging series against which all experimental manipulations, observations, and molecular data must be standardized. This whitepaper details its ongoing critical role as a foundational reference for contemporary research in embryology and drug development toxicology.
Core Reference: The Normal Table
Nieuwkoop and Faber's table meticulously describes 66 normative stages (0-66) of Xenopus laevis development, from fertilized egg to feeding tadpole. It is defined by invariant, easily observable external morphological features (e.g., cleavage patterns, neural plate closure, tailbud elongation). Its quantitative rigor provides the temporal framework for developmental research.
Table 1: Key Developmental Milestones in the Nieuwkoop and Faber Staging Series
| Stage Range | Descriptive Name | Key Morphological Criteria | Critical Periods for Research |
|---|---|---|---|
| 1-5 | Cleavage | Division into blastomeres (2, 4, 8, 16, 32 cells). | Blastomere fate mapping, early asymmetry studies. |
| 6-9 | Blastula | Formation of blastocoel cavity. | Mesoderm induction signaling studies. |
| 10-12.5 | Gastrula | Dorsal lip formation, blastopore closure. | Axis formation, organizer experiments. |
| 13-21 | Neurula | Neural plate, folds, and tube formation. | Neural patterning, placode specification. |
| 22-33 | Tailbud | Progressive elongation of tailbud. | Organogenesis, cell migration assays. |
| 34-45 | Tadpole | Heartbeat, circulation, gill circulation. | Functional assays, high-throughput screening. |
| 46-66 | Larval | Feeding, limb bud development. | Long-term developmental toxicity studies. |
Application in Experimental Protocols
Protocol A: Blastomere Lineage Tracing and Fate Mapping (Classical Method)
- Embryo Collection & Staging: Obtain embryos via natural mating or in vitro fertilization. Culture in 0.1x MMR or Modified Barth's Saline (MBS). Stage live embryos precisely under a dissecting microscope using the Nieuwkoop and Faber criteria.
- Blastomere Labeling: At the desired cleavage stage (e.g., 8-cell, Stage 4), remove the vitelline membrane manually with forceps. Prepare a solution of a vital dye (e.g., lysinated fluorescein-dextran, FLDx) at 10-20 mg/mL in distilled water.
- Microinjection: Back-fill a sharp glass capillary needle with the dye solution. Using a micromanipulator and picospritzer or pressure injector, inject approximately 5-10 nL of dye solution into the target blastomere(s). The identification of blastomeres (e.g., A1, B3, C1) is defined relative to the pigmentation and cleavage patterns standardized in the Normal Table.
- Tracking & Analysis: Return embryos to culture medium. Allow development to proceed. At subsequent stages (fixed according to the Normal Table), analyze the distribution of the fluorescent label in fixed or live embryos using fluorescence microscopy. Document the contribution of the injected blastomere to specific tissues, building a fate map.
Protocol B: Chemical Teratogen Screening Using a Normal Table Framework
- Synchronized Embryo Exposure: Raise embryos to the precise developmental stage required for the target process (e.g., Stage 10 for gastrulation screens, Stage 22 for early organogenesis). The Normal Table ensures cohort synchronization.
- Compound Administration: At the target stage, expose groups of embryos (in triplicate) to a range of compound concentrations diluted in culture medium. Include vehicle controls.
- Morphological Scoring: At defined post-exposure intervals (e.g., 24, 48, 72 hours), score all surviving embryos against the expected normative morphology for their chronological stage as per the Normal Table.
- Data Quantification: Record specific malformations (e.g., delayed blastopore closure, shortened axis, edema, microcephaly). Calculate LC50 (lethal concentration 50) and EC50 (effective concentration for malformation 50) values. The precise staging allows correlation of insult timing with specific phenotypic outcomes.
Visualization of Key Concepts
(Diagram Title: Mesoderm Patterning Cascade in Xenopus)
(Diagram Title: Embryo Research Workflow with Normal Table)
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Xenopus Embryo Fate Mapping & Perturbation Studies
| Reagent/Material | Function/Application | Key Detail |
|---|---|---|
| Modified Barth's Saline (MBS) | Standard culture medium for post-cleavage embryos. | Provides ionic balance and pH stability; used for compound exposures. |
| Holtfreter's Solution | Classical culture medium, often used for early embryos. | Simulates natural ionic environment; used in micromanipulation. |
| Lysinated Fluorescein Dextran (FLDx) | Vital lineage tracer for blastomere injection. | High molecular weight prevents diffusion; marks daughter cells. |
| mRNA for Morphogens (e.g., BMP4, Noggin, β-catenin) | Gain-of-function analysis. | Synthetic mRNA injected to perturb signaling pathways. |
| Antisense Morpholino Oligonucleotides (MOs) | Transient knockdown of specific gene products. | Blocks translation or splicing; used for loss-of-function. |
| Para-Nitro-Phenyl-Phenylalanine (pNP-Phe) | Chemical inhibitor of Nodal signaling. | Used for temporally controlled pathway inhibition. |
| Dispase | Protease for gentle removal of vitelline membranes. | Essential for microsurgery and transplantation experiments. |
| Paraformaldehyde (PFA) | Fixative for embryo preservation. | Used prior to in situ hybridization or immunostaining. |
| Anti-Histone H3 Phospho-Ser10 Antibody | Mitotic cell marker. | Labels dividing cells in whole-mount immunofluorescence. |
| Digoxigenin-labeled RNA probes | In situ hybridization for gene expression patterning. | Spatial mapping of transcripts in fixed embryos. |
This whitepaper establishes Xenopus laevis and Xenopus tropicalis as premier models for fate mapping and blastomere identification research, a core pillar of comparative embryology. The utility of Xenopus stems from its unique combination of experimental accessibility, conserved vertebrate developmental pathways, and the foundational knowledge provided by its precisely mapped fate maps. This document provides a technical guide within the context of ongoing thesis research aimed at refining blastomere lineage contributions and their regulatory logic.
Core Advantages for Fate Mapping
Xenopus embryos offer distinct technical advantages that are quantified in the following table.
Table 1: Quantitative Advantages of Xenopus for Embryological Studies
| Parameter | Xenopus laevis | Xenopus tropicalis | Significance for Fate Mapping |
|---|---|---|---|
| Egg Diameter | ~1.2 - 1.3 mm | ~0.7 - 0.8 mm | Large size enables microsurgery & targeted blastomere injection. |
| Cleavage Cycle (Early) | ~30 minutes at 23°C | ~20 minutes at 23°C | Rapid, synchronous divisions allow high-resolution temporal tracking. |
| Genome Size | ~3.1 Gb (pseudotetraploid) | ~1.7 Gb (diploid) | tropicalis: simpler genetics; laevis: gene redundancy for functional studies. |
| Time to Gastrulation | ~9 hours post-fertilization (hpf) | ~6 hpf | Fast development enables rapid assay of cell fate decisions. |
| Clutch Size | 1000-4000 eggs/female | 1000-3000 eggs/female | High yield provides statistical power for lineage tracing experiments. |
Foundational Fate Map and Key Methodologies
The classic Xenopus fate map, established via vital dye tracing and single blastomere injection, delineates the contributions of specific blastomeres at the 32- to 64-cell stage to primary germ layers and tissues.
Experimental Protocol 1: Iontophoretic Lineage Tracing with Fluorescent Dextrans
- Purpose: To label and track the progeny of a single blastomere through development.
- Materials: Sharp glass microelectrode, Iontophoresis unit, Fluorescent lysinated dextran (e.g., FITC, Rhodamine, 10,000 MW), Injection/micromanipulation rig.
- Procedure:
- Prepare a 5% solution of fluorescent dextran in distilled water.
- Backfill a sharp glass microelectrode with the dye solution.
- Mount the embryo in a depression slide with a solution like 3% Ficoll in 1x MMR.
- At the desired cleavage stage (e.g., 32-cell), identify the target blastomere.
- Impale the blastomere with the electrode and apply a pulsed, negative current (e.g., -1 µA, 500 ms pulses for 10-20 seconds) to eject the negatively charged dextran.
- Culture the embryo and image at subsequent stages using fluorescence microscopy to trace labeled descendants.
Experimental Protocol 2: Targeted Blastomere Injection of mRNA/Morpholinos
- Purpose: To perturb gene function in a specific lineage for functional fate mapping.
- Materials: Picospritzer or pressure injector, Fine glass capillary needles, Synthetic mRNA or Gene-specific Morpholino oligonucleotides.
- Procedure:
- Generate synthetic capped mRNA in vitro or design a translation-blocking Morpholino against the target gene.
- Load the reagent into a glass capillary needle.
- De-jelly embryos chemically (e.g., with cysteine) and align in grooves on an agarose plate.
- Under a dissecting microscope, orient the embryo to visualize blastomere boundaries.
- Using a micromanipulator, inject a calibrated nanoliter volume (e.g., 4-10 nL) directly into the cytoplasm of the target blastomere at the appropriate stage.
- Score phenotypic consequences in the expected descendant tissues during gastrula/neurula stages.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Xenopus Fate Mapping Research
| Reagent/Material | Function | Example/Notes |
|---|---|---|
| Fluorescent Lysinated Dextrans | Long-term, non-diffusible lineage tracer. | FDA-approved (Fluorescein), RLD (Rhodamine); 10kDa MW prevents gap-junctional transfer. |
| Capped Synthetic mRNA | Gain-of-function analysis in specific lineages. | In vitro transcribed from linearized plasmid with cap analog (e.g., mMessage mMachine kit). |
| Morpholino Oligonucleotides | Loss-of-function via translational or splicing blockade. | Designed to target 5'UTR or splice junctions of gene of interest; requires careful controls. |
| CRISPR-Cas9 Components | Targeted gene knockout in specific blastomeres. | Cas9 protein + sgRNA injected into 1- or 2-cell embryo or later into a targeted blastomere. |
| Horseradish Peroxidase (HRP) | Historic short-term lineage tracer. | Injected iontophoretically; visualized via enzyme reaction (DAB) in fixed specimens. |
| Agarose Injection Plates | Immobilize embryos for microinjection. | Molded plates with rows of conical wells to hold and orient embryos. |
| MMR or NAM Solution | Embryo culture medium. | Provides appropriate ionic strength and pH for normal development. |
Visualizing Key Signaling Pathways in Fate Specification
Fate maps are not static but are regulated by conserved signaling pathways. The following diagrams, generated with Graphviz DOT language, illustrate core pathways manipulated in fate mapping studies.
Diagram 1: Nodal Signaling in Mesendoderm Specification
Diagram 2: Experimental Workflow for Blastomere Fate Mapping
Xenopus remains indispensable for high-resolution fate mapping due to its optimal embryological features, established methodologies, and the deep foundational knowledge of its blastomere lineages. Integration of classic lineage tracing with modern functional perturbations (CRISPR, morpholinos) allows researchers to not only map fate but also dissect the gene regulatory networks that instruct it. This positions Xenopus as a critical model for advancing our understanding of vertebrate development and the ontogeny of organ systems, with direct relevance to biomedical research.
Practical Techniques for Xenopus Blastomere Labeling, Imaging, and Fate Analysis
The establishment of comprehensive fate maps in Xenopus embryos is foundational for developmental biology and teratogenicity screening in drug development. Precise blastomere identification and lineage tracing are critical for understanding the origins of tissues and organs. This technical guide compares three core tracing methodologies—fluorescent dextrans, mRNA reporters, and CRISPR-Cas9 lineage reporters—detailing their applications, experimental protocols, and integration into modern fate mapping research.
Comparative Analysis of Tracer Technologies
The table below summarizes the key quantitative and qualitative parameters for each lineage tracing method.
Table 1: Quantitative Comparison of Lineage Tracers
| Parameter | Fluorescent Dextrans | mRNA Reporters (e.g., GFP) | CRISPR-Cas9 Lineage Reporters |
|---|---|---|---|
| Temporal Resolution | Immediate (passive diffusion) | Delayed (requires translation, ~2-4 hrs) | Permanent activation post-cleavage |
| Persistence | Up to ~48 hrs (dilutes with division) | Up to tadpole stages | Heritable, permanent |
| Detection Threshold | High signal-to-noise | Moderate to High | Dependent on editing efficiency |
| Typical Injection Volume | 5-20 nL per blastomere | 5-20 nL per blastomere | 5-20 nL per blastomere |
| Inheritance Pattern | Asymmetric upon division | Symmetric upon division | Clonal (marks all progeny) |
| Best Use Case | Short-term lineage tracing, cell grafting | Fate mapping, overexpression studies | Long-term lineage analysis, clonal fate maps |
Table 2: Qualitative Comparison of Lineage Tracers
| Aspect | Fluorescent Dextrans | mRNA Reporters | CRISPR-Cas9 Lineage Reporters |
|---|---|---|---|
| Mechanism | Passive labeling | Transcriptional/Translational | Genomic editing |
| Ease of Use | Simple | Moderate | Technically complex |
| Perturbation Risk | Low (inert) | Moderate (may overload translation) | High (potential off-target edits) |
| Multiplexing Potential | High (multiple colors) | Moderate | Very High (multiple target sites) |
| Cost | Low | Low to Moderate | High |
Detailed Experimental Protocols
Protocol 1: Microinjection of Fluorescent Dextrans for Blastomere Labeling
- Preparation: Pull borosilicate glass capillaries to create sharp injection needles. Backfill with mineral oil.
- Tracer Loading: Frontfill the needle tip with ~2 µL of 10-25 mg/mL lysine-fixable fluorescent dextran (e.g., Texas Red, FITC) dissolved in nuclease-free water.
- Embryo Preparation: Obtain Xenopus laevis or tropicalis embryos via standard hormone-induced mating. Dejelly embryos chemically (e.g., 2% cysteine, pH 8.0) and array in injection chambers filled with 1x Marc's Modified Ringer (MMR) or 3% Ficoll solution.
- Microinjection: Using a pneumatic picopump, calibrate injection volume (5-20 nL) against a micrometer scale. For fate mapping at the 32- to 64-cell stage, target specific blastomeres using well-established dorsal-ventral and animal-vegetal maps.
- Post-Injection Care: After injection, transfer embryos to 0.1x MMR and incubate at 14-18°C until the desired developmental stage.
- Imaging: Fix embryos in 4% PFA or image live using a fluorescence stereomicroscope or confocal microscope.
Protocol 2: mRNA-Based Lineage Reporting with Nuclear Localized GFP (nlGFP)
- mRNA Synthesis: Linearize a plasmid containing nlGFP cDNA downstream of an SP6 or T7 promoter. Perform in vitro transcription using an mMESSAGE mMACHINE kit. Purify mRNA via phenol/chloroform extraction or lithium chloride precipitation. Resuspend in nuclease-free water at a final concentration of 500-1000 ng/µL.
- Microinjection: Follow steps 1, 3-5 from Protocol 1, injecting 5-20 nL of mRNA solution per blastomere. Include a tracer dye (e.g., 0.05% Texas Red dextran) in the injection mix for immediate confirmation of delivery.
- Incubation & Analysis: Allow embryos to develop until the target stage. GFP fluorescence typically becomes detectable after the mid-blastula transition. Image live or after fixation (GFP signal is fixable with PFA).
Protocol 3: CRISPR-Cas9-Based Lineage Tracing Using a Barcode Editor
- gRNA and Cas9 Preparation: Design a single-guide RNA (sgRNA) targeting a benign, repetitive genomic locus (e.g., the tyrosinase gene in albino lines). Synthesize sgRNA via in vitro transcription. Purify Cas9 protein or use Cas9-encoding mRNA.
- Donor Template Design: Synthesize a short single-stranded DNA (ssDNA) donor oligonucleotide containing a fluorescent protein reporter (e.g., H2B-mCherry) flanked by homology arms (30-50 bp) matching the target locus. The reporter must be preceded by a strong, ubiquitous promoter (e.g., EF1α).
- Injection Mix: Co-inject Cas9 protein (100-200 pg), sgRNA (50-100 pg), and donor ssDNA (10-20 pg) in a total volume of 5-20 nL into the target blastomere at the 1- or 2-cell stage for whole-embryo labeling, or at later stages for restricted clones.
- Screening & Validation: Raise injected embryos. Screen for fluorescence at the tailbud stage. Validate precise genomic integration via PCR and sequencing from fin-clip or embryo tail samples.
Research Reagent Solutions
Table 3: Essential Research Toolkit for Lineage Tracing in Xenopus
| Reagent/Material | Function/Application |
|---|---|
| Lysine-Fixable Dextrans | Inert, high MW fluorescent conjugates for short-term, non-diffusible lineage tracing. |
| mMESSAGE mMACHINE Kit | High-yield, capped mRNA synthesis for in vitro transcription of reporter mRNAs. |
| Recombinant Cas9 Protein | For precise CRISPR-Cas9 editing; offers rapid activity and reduces mRNA translation delay. |
| CRISPRclean SgRNA Kit | Production of high-purity, stable sgRNAs for genome editing. |
| Homology-Directed Repair (HDR) Donor Oligos | Single-stranded DNA templates for precise, reporter knock-in at target loci. |
| Ficoll PM400 | Injection medium to reduce leakage and improve embryo survival post-injection. |
| Phenol Red Tracer Dye | Visual aid for confirming successful microinjection delivery. |
Visualizing Tracer Methodologies and Workflows
Decision Workflow for Tracer Selection
Fluorescent Dextran Injection Workflow
CRISPR-Cas9 HDR Reporter Knock-In Mechanism
This technical guide details precise microinjection protocols for targeting specific blastomeres in early Xenopus laevis embryos. The methodology is foundational for a broader thesis research program aimed at refining the Xenopus fate map and investigating the mechanisms of blastomere fate specification. By enabling the targeted delivery of lineage tracers, morpholino oligonucleotides (MOs), CRISPR-Cas components, or mRNA into identified blastomeres, these techniques allow for the functional testing of hypotheses regarding the roles of specific cells and signaling pathways in early development. This precision is critical for advancing research in developmental biology and for early-stage drug discovery that utilizes Xenopus as a model for teratogenicity and pathway modulation.
Blastomere Identification and Fate Map Reference
Successful targeting requires precise identification of blastomeres based on their cleavage pattern and known fate. The following table summarizes the canonical 32-cell stage fate map for Xenopus laevis.
Table 1: Xenopus laevis 32-Cell Stage Blastomere Fate Summary
| Blastomere Tier | Major Blastomere Designations | Primary Tissue Fate | Key Identifying Features |
|---|---|---|---|
| Animal | A1, A2, etc. | Epidermis, Neural Plate, Sensory Placodes | Pigmented, small cells at the animal pole. |
| Equatorial | B1, B2, etc. | Paraxial Mesoderm, Heart, Kidney, Blood. | Boundary between pigmented animal and vegetal hemispheres. |
| Vegetal | C1, C2, etc. | Endoderm (Gut lining). | Large, unpigmented/yolky cells at the vegetal pole. |
| Dorsal Vegetal | D1, D2, etc. | Endoderm, "Organizer" precursors (e.g., Spemann's Organizer). | Dorsal side, identified by earlier first cleavage furrow and lighter pigmentation gradient. |
Note: The "D" tier blastomeres (particularly D1) are critical for dorsal-axial structures. The first cleavage furrow normally bisects the future dorsal-ventral axis.
Core Experimental Protocol: Targeted Microinjection
Materials and Reagent Preparation
- Embryos: De-jellied Xenopus laevis embryos, staged according to Nieuwkoop and Faber.
- Injection Needles: Borosilicate glass capillaries (e.g., 1.0 mm OD, 0.58 mm ID) pulled to a fine, bevelled point.
- Microinjector: A pressure-based microinjector system with a foot pedal for precise control.
- Injection Solution: Your reagent of interest (e.g., fluorescent dextran lineage tracer, MO, mRNA) in nuclease-free water or a mild buffer. Critical: Include a tracer dye (e.g., 0.05% Texas Red dextran for non-fluorescent reagents) to confirm delivery.
- Injection Dish: A dish with a silicone elastomer (Sylgard) base or an agarose mold with depressions to hold embryos in position.
Step-by-Step Procedure
Step 1: Embryo Orientation and Immobilization.
- Transfer a de-jellied embryo at the desired cleavage stage (2-cell to 32-cell) to an injection dish filled with 1x Modified Barth's Saline (MBS) or 3% Ficoll in 0.5x MBS.
- Using fine forceps or a hair loop, gently rotate the embryo so the target blastomere is facing upward and is accessible. For vegetal injections, a shallow agarose well can hold the embryo at an angle.
Step 2: Needle Loading and Calibration.
- Back-fill 2-3 µL of injection solution into the needle using a microloader tip.
- Mount the needle on the injector. Using the microscope's eyepiece reticule, calibrate the injection volume by measuring the diameter of the droplet expelled into oil. A typical volume for a single blastomere at the 8-32 cell stage is 2-10 nL.
Step 3: Targeting and Microinjection.
- Lower the needle and position it adjacent to the target blastomere.
- Using a swift, controlled motion, pierce the cell membrane. The vegetal hemisphere membrane is tougher and may require a quicker jab.
- Immediately depress the foot pedal to deliver the calibrated volume. A slight swelling of the blastomere confirms delivery.
- Withdraw the needle cleanly.
Step 4: Post-Injection Care.
- Allow injected embryos to recover in 3% Ficoll/0.5x MBS for 1-2 hours to facilitate wound healing.
- Wash and transfer embryos to 0.1x MBS for normal development until the desired analysis stage.
Validation and Analysis Workflow
A standard experimental workflow for fate mapping or functional analysis is depicted below.
Diagram 1: Targeted Blastomere Injection & Analysis Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Targeted Blastomere Experiments
| Item | Function & Purpose | Example/Note |
|---|---|---|
| Fluorescent Dextran Conjugates | Lineage Tracing. Inert, cell-impermeable dyes to label progeny of the injected blastomere. | Texas Red (TxR), Fluorescein (FITC), or Cascade Blue dextran (10,000 MW). |
| Morpholino Oligonucleotides (MOs) | Gene Knockdown. Block translation or splicing of target mRNA with high specificity. | Requires careful dose optimization and mismatch control MOs. |
| CRISPR-Cas9 Components | Gene Knockout. Induce targeted mutations in specific cell lineages. | Cas9 protein + sgRNA co-injection for immediate activity. |
| Synthetic mRNA | Gene Overexpression/Misexpression. Express wild-type or mutant proteins in a lineage-restricted manner. | Must be capped (m7G) and polyadenylated for stability. |
| Dominant-Negative/Active Constructs | Pathway Modulation. Constitutively inhibit or activate signaling pathways in targeted cells. | e.g., Constitutively Active BMP Receptor (caBMPR). |
| Fluorescent Tracer Dye | Injection Control. Visualize successful delivery, even when primary reagent is not fluorescent. | 0.05% TxR dextran in all injection solutions. |
| Agarose/Sylgard Dishes | Embryo Immobilization. Secure embryos in precise orientation for reliable targeting. | Custom-made with molds or poured layers. |
Key Signaling Pathways Studied via Blastomere Targeting
Targeted injection is powerful for probing early embryonic signaling. A major pathway in dorsal-ventral patterning is the Wnt/β-catenin pathway, active in dorsal vegetal (D tier) blastomeres.
Diagram 2: Wnt/β-Catenin Pathway in Dorsal Blastomeres
Live Imaging and Time-Lapse Microscopy for Dynamic Fate Mapping
Dynamic fate mapping represents a paradigm shift from static histological snapshots to a four-dimensional understanding of embryogenesis. Within Xenopus laevis and tropicalis research, this approach is revolutionizing our ability to trace blastomere lineages with high temporal resolution, directly testing and refining classic fate maps. The core thesis is that live imaging integrated with computational analysis provides an unambiguous, quantitative record of cell behaviors—divisions, migrations, and fate restrictions—that static methods can only infer. This technical guide details the methodologies enabling this transformation for researchers and drug development professionals investigating developmental mechanisms and teratogenicity.
Core Imaging Methodologies and Quantitative Comparison
The choice of imaging modality is dictated by the trade-off between spatial resolution, temporal resolution, phototoxicity, and depth penetration. The following table summarizes key quantitative parameters for the most relevant techniques in Xenopus studies.
Table 1: Quantitative Comparison of Live Imaging Modalities for Xenopus Embryos
| Modality | Spatial Resolution (XY/Z) | Typical Frame Interval (Temporal Resolution) | Max Imaging Depth | Key Advantage for Fate Mapping | Primary Limitation |
|---|---|---|---|---|---|
| Widefield Epifluorescence | ~300 nm / ~500 nm | 30 sec - 5 min | ~100 µm (with clearing) | Low phototoxicity, high speed | Out-of-focus blur, limited depth |
| Confocal (Point-Scanning) | ~180 nm / ~500 nm | 2 - 10 min | ~150 µm | Optical sectioning, excellent contrast | Slower speed, higher photobleaching |
| Spinning Disk Confocal | ~180 nm / ~500 nm | 30 sec - 2 min | ~150 µm | High-speed optical sectioning | Lower light throughput vs. point scanning |
| Light-Sheet Fluorescence (LSFM) | ~200 nm / ~400 nm | 10 - 60 sec | Whole embryo (mm scale) | Extreme low phototoxicity, high speed | Complex sample mounting, lower XY resolution |
| Two-Photon Microscopy | ~300 nm / ~800 nm | 2 - 10 min | >500 µm | Deep tissue penetration, reduced scattering | Expensive, slower, potential heat deposition |
Detailed Experimental Protocols
Protocol: Generating Fluorescently Labeled Blastomeres for Time-Lapse
This protocol details lineage tracing via targeted mRNA injection, a cornerstone for Xonopus dynamic fate mapping.
- Template and Probe Preparation: Clone the coding sequence for a photostable fluorescent protein (e.g., mNeonGreen, mScarlet, H2B-mCherry) into a vector containing the Xenopus β-globin 3' UTR for stability. Generate mRNA using an mMESSAGE mMACHINE SP6/T7 kit. Purify via LiCl precipitation and resuspend in nuclease-free water. Store at -80°C.
- Embryo Preparation: Obtain Xenopus embryos via standard hormone-induced mating or in vitro fertilization. Dejelly embryos in 2% cysteine (pH 7.9-8.0) and rinse thoroughly in 0.1x MMR or Ficoll solution.
- Microinjection: At the desired cleavage stage (e.g., 4-, 8-, or 16-cell), calibrate injection volume to 4-10 nL per blastomere using a micrometer. Back-load the injection needle with mRNA (typically 50-200 pg for nuclear labels, 200-500 pg for cytoplasmic). Inject into the desired lineage (animal pole for ectoderm, vegetal for endoderm, equatorial for mesoderm).
- Mounting for Imaging: At the appropriate stage (post-gastrulation for deep imaging), anesthetize embryos in 0.02% MS-222. For long-term imaging (>6 hours), embed in 0.8-1.2% low-melting-point agarose within a fluorinated ethylene propylene (FEP) tube or glass-bottom dish. Use 0.1x MMR + 6% Ficoll as imaging medium to maintain osmolarity.
Protocol: Long-Term Time-Lapse Acquisition on a Spinning Disk System
This workflow is optimized for balancing resolution and viability over 12-24 hours.
- Microscope Setup: Use a spinning disk confocal system equipped with a 20x air (NA 0.8) or 25x water-immersion (NA 1.05) objective. Environmental chamber set to 14-18°C (for X. laevis) or 22-26°C (for X. tropicalis).
- Acquisition Parameters:
- Laser Power: Use the minimum power required for a clear signal (typically 1-10% of laser output).
- Exposure Time: 50-200 ms per channel.
- Z-stacks: Acquire stacks with 3-5 µm steps to cover the entire depth of the labeled population.
- Time Interval: 3-5 minutes for early cleavage stages; 5-10 minutes during gastrulation and neurulation.
- XY Resolution: Set to 1024 x 1024 or 2048 x 2048 pixels.
- Focus Stabilization: Activate hardware-based autofocus systems (e.g., laser-based or software-defined) to compensate for focal drift.
- Multi-Position Imaging: If imaging multiple embryos, use a motorized stage to cycle through predefined positions.
Visualization of Workflows and Pathways
Diagram 1: Dynamic Fate Mapping Experimental Workflow
Diagram 2: Key Signaling Pathways in Xenopus Blastomere Patterning
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Materials for Xenopus Dynamic Fate Mapping
| Item | Function & Rationale | Example/Format |
|---|---|---|
| Photostable FP mRNAs | Lineage tracing with minimal photobleaching over long timescales. | H2B-mCherry/mNeonGreen: Nuclear labeling for precise tracking. Membrane-targeted GFP: For analyzing cell shape and contact. |
| Morpholino Oligos / CRISPR-Cas9 | Perturb gene function to test fate specification mechanisms in a traced lineage. | Gene-specific Morpholino: Knockdown. Cas9 Protein + gRNA: Knockout in injected blastomeres. |
| Low-Melt Agarose | Physically immobilize embryo for imaging while permitting gas exchange. | 1.0% in 0.1x MMR/3% Ficoll: Standard embedding for post-gastrula stages. |
| MS-222 (Tricaine) | Reversible anesthetic to prevent embryo movement during mounting. | 0.01-0.02% Solution: In imaging medium. |
| Phenol Red-Free Medium | Imaging medium without autofluorescence. | 0.1x MMR Modified: Or commercial amphibian saline. |
| Ficoll PM-400 | Increases medium viscosity; reduces osmotic stress and supports development. | 3-6% Solution: Added to imaging medium. |
| Fluorescent Dextran | Short-term lineage tracer or fluid space marker. | 70 kDa Tetramethylrhodamine Dextran: Injected into blastocoel or cells. |
| Nuclei Tracking Software | Automated segmentation and tracking of cell nuclei across 4D datasets. | TrackMate (Fiji), Imaris, arivis Vision4D: For lineage tree reconstruction. |
Whole-Mount In Situ Hybridization and Immunohistochemistry for Fixed Tissue Analysis
Within the context of Xenopus laevis and Xenopus tropicalis embryo fate mapping and blastomere identification research, whole-mount in situ hybridization (WMISH) and whole-mount immunohistochemistry (IHC) are indispensable techniques. They allow for the spatial localization of mRNA transcripts and proteins, respectively, in three-dimensional fixed embryos. This guide provides an integrated technical framework for applying these methods to answer critical questions about blastomere specification and lineage commitment.
Core Principles and Applications in Blastomere Research
WMISH utilizes labeled antisense RNA probes complementary to a target mRNA sequence, enabling the visualization of gene expression domains. Whole-mount IHC employs antibodies to detect specific protein antigens. In fate map studies, these techniques are used to:
- Validate lineage-specific gene expression predicted by microinjection or lineage tracing.
- Correlate protein localization with embryonic patterning events.
- Analyze the effects of morpholino oligonucleotides, CRISPR/Cas9 mutations, or drug treatments on molecular markers.
Experimental Protocols
A. Fixed Tissue Preparation forXenopusEmbryos
- Fixation: Collect embryos at desired Nieuwkoop and Faber (NF) stage. Fix in MEMFA (0.1 M MOPS pH 7.4, 2 mM EGTA, 1 mM MgSO₄, 3.7% formaldehyde) for 1-2 hours at room temperature or overnight at 4°C.
- Dehydration: Wash in PBS, then progressively dehydrate through a methanol series (25%, 50%, 75% in PBS, then 100% methanol). Embryos can be stored at -20°C in 100% methanol for months.
- Rehydration & Permeabilization: Rehydrate through a descending methanol series to PBS. For WMISH, treat with 10 μg/mL Proteinase K in PBS (duration is stage-dependent: e.g., NF10-12: 5-10 min; NF20-25: 15-20 min). Immediately refix in 4% paraformaldehyde for 20 minutes and wash. For IHC, permeabilize with 0.1% Triton X-100 in PBS (PBST).
B. Whole-Mount In Situ Hybridization (Colorimetric)
- Pre-hybridization: Equilibrate embryos in hybridization buffer (HB: 50% formamide, 5x SSC, 1 mg/mL Torula RNA, 100 μg/mL heparin, 1x Denhardt's, 0.1% Tween-20, 0.1% CHAPS) at 60-65°C for ≥4 hours.
- Hybridization: Incubate with digoxigenin (DIG)- or fluorescein-labeled antisense RNA probe (1-5 ng/μL) in HB at 60-65°C for 12-48 hours.
- Post-Hybridization Washes: Stringent washes to remove unbound probe: 2x SSC, 50% formamide at 60°C; then MABT (100 mM Maleic acid, 150 mM NaCl, 0.1% Tween-20, pH 7.5).
- Immunological Detection: Block in MABT + 2% Boehringer Blocking Reagent. Incubate with anti-DIG/anti-fluorescein Alkaline Phosphatase (AP)-conjugated Fab fragments (1:2000-1:5000) overnight at 4°C.
- Color Reaction: Wash extensively in MABT, then AP buffer (100 mM Tris pH 9.5, 100 mM NaCl, 50 mM MgCl₂, 0.1% Tween-20). Develop in AP buffer with NBT/BCIP (or BM Purple/Magenta-Phos) substrate in the dark. Monitor reaction and stop with PBST + 1 mM EDTA.
- Post-staining: Refix in MEMFA, clear in Murray's clear (2:1 Benzyl Benzoate:Benzyl Alcohol), and mount.
C. Whole-Mount Immunohistochemistry (Fluorescent)
- Blocking: After permeabilization, block embryos in PBST + 10% normal goat serum (or species-appropriate serum) + 1% BSA for 1-2 hours at room temperature.
- Primary Antibody Incubation: Incubate with primary antibody (e.g., anti-GFP, anti-phospho-Histone H3, anti-β-catenin) diluted in blocking solution, typically for 24-48 hours at 4°C with gentle agitation.
- Washing: Wash extensively (6-8 changes over 24 hours) with PBST.
- Secondary Antibody Incubation: Incubate with fluorophore-conjugated secondary antibody (e.g., Alexa Fluor 488, 568, 647) diluted in blocking solution, protected from light, for 24 hours at 4°C.
- Washing & Counterstaining: Wash as in step 3. Counterstain nuclei with DAPI (1 μg/mL in PBS) for 30 minutes if required. Wash again.
- Mounting: Clear and mount in 80% glycerol/PBS or specialized clearing media (e.g., CUBIC, ScaleS2) for confocal microscopy.
Data Presentation: Key Quantitative Parameters
Table 1: Critical Variables in Xenopus Whole-Mount Protocols
| Parameter | WMISH Typical Range | IHC Typical Range | Impact on Outcome |
|---|---|---|---|
| Fixation Time | 1-2 hrs (RT) to O/N (4°C) | 1-2 hrs (RT) to O/N (4°C) | Under-fixation: tissue degradation. Over-fixation: reduced probe/antibody penetration & antigenicity. |
| Proteinase K Time | Stage-dependent: 5-25 min | Not typically used | Critical for probe penetration. Must be optimized per embryo stage. |
| Hybridization Temp | 60-70°C | N/A | Stringency: higher temp reduces non-specific binding. |
| Antibody Incubation | O/N at 4°C | 24-48 hrs at 4°C | Longer incubations often needed for deep penetration into whole embryos. |
| Color/Detection Time | 30 min - 24 hrs | N/A | Must be monitored closely to avoid high background. |
Table 2: Common Detection Systems & Reagents
| System | Target | Conjugate | Common Substrate/Channel | Application |
|---|---|---|---|---|
| Colorimetric AP | DIG-labelled probe | Anti-DIG-AP | NBT/BCIP (purple) | Single mRNA localization |
| Fluorescent | Primary Antibody | Alexa Fluor 488, 568, 647 | 488nm, 561nm, 640nm lasers | Protein co-localization, 3D reconstruction |
| Fluorescent | Fluorescein-labelled probe | Anti-Fluorescein-AP | Fast Red TR/Vector Red (red fluor) | Multiple mRNA detection |
Visualization: Pathways and Workflows
Diagram 1: Integrated Workflow for WMISH and IHC in Xenopus
Diagram 2: Core Mechanism of Colorimetric WMISH Detection
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Xenopus Whole-Mount Analysis
| Item | Function | Key Considerations for Fate Mapping |
|---|---|---|
| MEMFA Fixative | Crosslinks and preserves embryonic morphology and nucleic acid/protein integrity. | Preferred over PFA alone for Xenopus; provides better morphology for later stages. |
| Proteinase K | Digests proteins to permeabilize the vitelline membrane and allow probe penetration. | Critical optimization point. Time must be carefully titrated by embryo stage to avoid damage. |
| Hybridization Buffer (with Formamide) | Creates stringent conditions for specific binding of RNA probe to target mRNA. | High-quality formamide and RNA blockers (Torula RNA) reduce non-specific background. |
| DIG/FLU RNA Labeling Mix | For in vitro transcription of labeled antisense RNA probes. | Freshly synthesized probes yield highest sensitivity. Aliquots should be ethanol-precipitated for long-term storage. |
| Anti-DIG-AP Fab fragments | Highly specific antibody fragments for detecting digoxigenin-labeled probes. | Fab fragments penetrate embryonic tissue better than whole IgG molecules. |
| NBT/BCIP Stock Solution | Chromogenic substrate for Alkaline Phosphatase (AP). | Precipitate forms at sites of mRNA localization. BM Purple is a newer, more sensitive alternative. |
| Normal Goat Serum | Protein block to prevent non-specific binding of antibodies in IHC. | Must match the host species of the secondary antibody. |
| Fluorophore-conjugated Secondary Antibodies (e.g., Alexa Fluor) | Highly sensitive and photostable detection of primary antibodies. | Choose antibodies pre-adsorbed against Xenopus proteins to reduce background. Use different species for multiplexing. |
| Mounting Media with DAPI | Preserves fluorescence and counterstains nuclei for orientation. | For 3D imaging, use clearing-appropriate media (e.g., 80% glycerol, ScaleS2). |
Congenital defects arise from disruptions in the complex choreography of early embryonic development. A fundamental resource for understanding this process is the embryonic fate map, which describes the developmental potential of specific blastomeres. Research using the Xenopus embryo has been instrumental in creating high-resolution fate maps due to its large, experimentally tractable blastomeres and external development. This whitepaper posits that the integration of classical Xenopus blastomere fate maps with modern molecular, genomic, and computational techniques provides a powerful, cross-species framework for modeling human congenital disease pathways and identifying novel therapeutic targets.
XenopusFate Maps: The Foundational Blueprint
The classic Xenopus fate map, established through lineage tracing and blastomere isolation experiments, assigns specific tissue fates to each cell of the 32- to 64-cell stage embryo.
Table 1: Developmental Fate of Key Xenopus Blastomeres at the 32-Cell Stage
| Blastomere (Nomenclature) | Primary Germ Layer Contribution | Major Tissue/Organ Fates | Associated Human Congenital Defects if Disrupted |
|---|---|---|---|
| D1 (Dorsal Vegetal) | Endoderm, Mesoderm | Foregut, heart, prechordal plate | Holoprosencephaly, laterality defects |
| V1 (Ventral Vegetal) | Endoderm | Hindgut, blood islands | Gut malformations, anemia |
| D2 (Dorsal Animal) | Ectoderm, Mesoderm | Brain, neural tube, somites | Neural tube defects (spina bifida, anencephaly) |
| V2 (Ventral Animal) | Ectoderm | Epidermis, cement gland | Ectodermal dysplasias |
| C1 (Equatorial) | Mesoderm | Kidney, muscle, notochord | Renal agenesis, musculoskeletal defects |
Experimental Protocol: Lineage Tracing with Fluorescent Dextrans
- Microinjection Setup: Pull capillary needles and calibrate a microinjector. Position a dejellied Xenopus embryo (stage 3-4, 32-cell) on a agarose-coated dish.
- Tracer Injection: Backfill a needle with 2-5 nL of 10-20 mg/mL lysine-fixable fluorescent dextran (e.g., Texas Red). Using a micromanipulator, penetrate the target blastomere membrane and deliver the tracer.
- Embryo Culture: Allow injected embryos to develop in 0.1x MMR solution at 14-18°C until the desired stage (e.g., tadpole, stage 45).
- Fixation & Imaging: Fix embryos in 4% paraformaldehyde, wash, and clear using glycerol or Murray's clear. Image using a fluorescence stereomicroscope or confocal microscope to visualize descendant cell populations.
From Fate Map to Disease Pathway: Key Signaling Nodes
Developmental pathways that govern cell fate decisions in mapped blastomeres are frequently mutated or dysregulated in congenital diseases. The following pathways are critical.
Diagram 1: From signaling to congenital defect.
Table 2: Key Developmental Pathways & Associated Congenital Defects
| Signaling Pathway | Primary Blastomere/Role in Xenopus | Key Genes | Associated Human Congenital Defects |
|---|---|---|---|
| Nodal | Dorsal Vegetal (D1); mesendoderm induction | NODAL, LEFTY1, FOXH1 | Heterotaxy, congenital heart disease |
| BMP/TGF-β | Ventral blastomeres; dorsoventral patterning | BMP4, SMADs, CHRD | Cleft palate, brachydactyly |
| Wnt/β-catenin | Dorsal blastomeres; axis formation | WNT3A, β-CATENIN, APC | Caudal regression syndrome |
| Sonic Hedgehog | Derived from dorsal mesoderm; neural patterning | SHH, PTCH1, GLI2 | Holoprosencephaly, polydactyly |
Experimental Protocol: Targeting Pathways in Specific Blastomeres
- Morpholino/CRISPR Design: Design a morpholino oligonucleotide (MO) or sgRNA against the translational start site of the target gene (e.g., shha).
- Targeted Delivery: Co-inject the MO/sgRNA with a lineage tracer (fluorescent dextran) into a specific blastomere (e.g., D1 for SHH-related defects) at the 2- or 4-cell stage, targeting its descendant field.
- Phenotypic Analysis: Score resulting tadpoles for specific malformations (e.g., cyclopia, loss of ventral neural cell types). Quantify penetrance (% affected) and expressivity (severity score 0-5).
- Molecular Validation: Perform in situ hybridization or qPCR on sibling embryos for downstream target genes (e.g., nkx2.2 for SHH) to confirm pathway disruption.
The Modern Toolkit: Integrating Fate Maps with Omics
Current research integrates fate mapping with single-cell transcriptomics and CRISPR screening to model polygenic diseases.
Diagram 2: Integrative omics and perturbation workflow.
Table 3: Quantitative Omics Data from Xenopus Disease Models
| Experiment Type | Target Lineage | Number of Cells Profiled | Key Dysregulated Genes Identified | Disease Model |
|---|---|---|---|---|
| scRNA-seq | D1-derived prechordal plate | 5,120 | SHH, FGF8, SIX3 downregulation | Holoprosencephaly |
| ATAC-seq | C1-derived kidney precursor | N/A (bulk assay) | 1,243 chromatin regions opened | Renal dysplasia |
| Proteomics | Whole embryo (BMP4 morphant) | ~4,000 proteins quantified | 12% of proteome significantly altered | Cleft lip/palate |
The Scientist's Toolkit: Essential Research Reagents
Table 4: Key Research Reagent Solutions for Fate Map-Based Disease Modeling
| Reagent / Material | Function & Application in Fate Mapping | Example Product / Specification |
|---|---|---|
| Fluorescent Lineage Tracers | Labeling specific blastomeres for fate tracking; often co-injected with perturbations. | Lysine-fixable Dextran, Tetramethylrhodamine (10,000 MW). |
| Morpholino Oligonucleotides | Transient, sequence-specific knockdown of gene expression in early embryos. | Gene-specific, Vivo-Morpholinos for enhanced stability. |
| CRISPR-Cas9 Components | Permanent gene knockout or editing in specific lineages. | Cas9 protein, synthetic sgRNA, homology-directed repair templates. |
| Xenopus Embryo Manipulation Tools | Microinjection and microsurgery for precise experimental intervention. | Borosilicate glass capillaries, microinjector, agarose-coated dishes. |
| Single-Cell Isolation Kits | Dissociating and preparing labeled lineages for downstream omics. | 10x Genomics Chromium Chip B, validated dissociation enzymes. |
| Whole-Mount In Situ Hybridization Kits | Spatial visualization of gene expression patterns in mutant embryos. | DIG RNA labeling mix, anti-DIG-AP Fab fragments, NBT/BCIP substrate. |
| High-Content Imaging Systems | Automated, quantitative phenotypic screening of embryos. | Confocal microscope with environmental chamber, automated image analysis software (e.g., CellProfiler). |
Application in Drug Discovery & Therapeutic Target Identification
The Xenopus fate map model enables medium-throughput screening for compounds that can rescue congenital defect phenotypes.
Experimental Protocol: Small Molecule Rescue Screen
- Generate Disease Model: Create a cohort of embryos with a targeted defect (e.g., >70% penetrance of neural tube closure defect via vangl2 MO injection into D2 lineage).
- Compound Library Addition: At the onset of gastrulation (stage 10), array embryos into 96-well plates containing small molecules from a library (e.g., FDA-approved drugs).
- Phenotypic Scoring: At tadpole stage (45), score each well for rescue of the primary defect using automated bright-field and fluorescence imaging (to confirm lineage targeting).
- Hit Validation & Mechanistic Study: Confirm hits in independent experiments. Use in situ hybridization or Western blot to assess restoration of the native molecular pathway.
This approach directly links a mapped blastomere, its associated disease pathway, and potential therapeutic modulators, offering a rapid, cost-effective bridge between basic developmental biology and clinical application for congenital diseases.
Solving Common Fate Mapping Challenges: Precision, Consistency, and Data Interpretation
This guide is framed within the ongoing research to refine the Xenopus laevis and Xenopus tropicalis embryonic fate maps. Precise blastomere identification and targeting are foundational to this work, as mis-injection leads to erroneous lineage tracing, flawed gene function analysis, and compromised data on cell fate determination. Accurate targeting is thus critical for generating reliable fate maps and for applications in developmental biology and drug discovery screening.
Blastomere Identification: The Foundation of Accuracy
Successful injection begins with correct blastomere identification at the desired cleavage stage (e.g., 2-, 4-, 8-, 16-, 32-cell).
Key Identification Landmarks:
- Animal-Vegetal Axis: The pigmented animal hemisphere vs. the less-pigmented vegetal hemisphere.
- Cleavage Planes: The first cleavage plane is vertical and divides the gray crescent. The second is vertical and perpendicular to the first. The third is horizontal and equatorial.
- Blastomere Size: Vegetal blastomeres are larger than animal blastomeres.
- Pigmentation Gradients: Useful for distinguishing dorsal (lighter due to gray crescent) from ventral (more pigmented) blastomeres.
Visual Aids: Use standardized diagrams (e.g., Nieuwkoop and Faber plates) side-by-side with the embryo. Fluorescent lineage tracers (e.g., FITD, RLD) from previous injections can confirm identity.
| Source of Error | Consequence | Corrective Action |
|---|---|---|
| Misidentification | Injecting the wrong lineage; corrupting fate map data. | Use multiple landmarks (pigmentation, size, cleavage orientation). Confirm with time-lapse records of early cleavages. |
| Needle Placement Error | Leakage into adjacent blastomeres or extracellular space. | Calibrate needle opening (10-15 µm). Use a sharp, beveled tip. Perform test injections in a practice embryo/oocyte. |
| Embryo Orientation | Inaccessible target blastomere. | Use agarose wells or hair loops to gently rotate and stabilize the embryo in the optimal position. |
| Developmental Staging | Variability in injection timing leads to different developmental contexts. | Stage precisely according to Nieuwkoop and Faber. Account for temperature (e.g., stage 4 at 18°C vs. 23°C). |
| Needle Clogging | No delivery or sudden explosive delivery. | Use filtered solutions and minerals. Apply positive pressure before penetrating. Clear tip by gently touching the holding pipette. |
Quantitative Data on Injection Parameters
Optimal physical parameters vary by embryo stage and target. Below is a summary table based on current literature and practice.
Table 1: Standardized Injection Parameters for Xenopus Blastomeres
| Target Stage | Needle Tip Diameter (µm) | Injection Volume (nL) | Tracer/Dye Amount | Pressure (psi) / Duration (ms) |
|---|---|---|---|---|
| 1-cell | 10-15 | 5-10 nL | 50-100 pg mRNA | 10-15 psi / 50-100 ms |
| 2-8 cell | 5-10 | 1-2 nL per blastomere | 10-25 pg mRNA | 8-12 psi / 20-50 ms |
| 16-32 cell | 3-7 | 0.5-1 nL per blastomere | 5-15 pg mRNA | 5-10 psi / 10-30 ms |
| Morula | <5 | <0.5 nL | 1-5 pg morpholino | 3-7 psi / 5-15 ms |
Note: Volume is the most critical parameter. Calibrate the needle by measuring the diameter of a droplet injected into oil.
Core Experimental Protocol for Fate Mapping via Blastomere Injection
Aim: To label a specific blastomere lineage for fate mapping.
Materials: See "The Scientist's Toolkit" below. Protocol:
- Prepare Injection Needles: Pull capillary glass to a fine point. Break or bevel to desired diameter (Table 1). Back-fill with ~2 µL of injection solution (tracer + experimental reagent).
- Prepare Embryos: Obtain dejellied embryos staged to the desired cleavage period. Transfer to an injection dish with 3% Ficoll in 0.1x MMR to prevent dehydration.
- Stage & Orient: Under a stereomicroscope, precisely stage the embryo. Use an agarose mold or hair loop to orient the embryo so the target blastomere is accessible and the cleavage planes are visible.
- Calibrate: Mount the needle on the injector. Set pressure parameters (Pulse Pressure, Balance Pressure, Time). Inject into mineral oil to create a droplet and measure its diameter to calculate volume.
- Inject: Lower the needle, pierce the vitelline membrane and blastomere wall at a shallow angle. Apply the injection pulse. A slight swelling of the blastomere confirms delivery. Withdraw smoothly.
- Post-Injection Care: After all injections, transfer embryos to 0.1x MMR with antibiotics. Allow to develop to the stage of observation.
- Validation: Image at successive stages using fluorescence microscopy to trace lineage. For fate maps, co-inject a lineage tracer (e.g., lacZ mRNA, fluorescent dextran) with your experimental molecule.
Diagram Title: Blastomere Injection Workflow for Fate Mapping
Signaling Pathways in EarlyXenopusPatterning
Accurate targeting requires understanding the key signaling centers. Mis-injection can aberrantly activate or inhibit these pathways.
Diagram Title: Core Patterning Signals in Early Xenopus Embryo
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Blastomere Injection & Fate Mapping
| Reagent/Material | Function & Application | Key Consideration |
|---|---|---|
| Fluorescent Dextran (e.g., FDA, RDA) | Lineage Tracer. Inert, non-diffusible marker to label injected blastomere and all its progeny. | Choose fixable (amine-reactive) for later immunohistochemistry. |
| Synthetic mRNA (e.g., GFP, lacZ) | Lineage Tracer & Protein Overexpression. Can be co-injected with other molecules to label cells and manipulate gene function. | Must be capped and polyadenylated for stability. Purify to remove contaminants. |
| Morpholino Oligonucleotides | Gene Knockdown. Used to inhibit translation or splicing of specific maternal/early zygotic transcripts. | Requires careful controls (standard control, 5-mispair, rescue). Dose-dependent. |
| CRISPR-Cas9 Components (gRNA, Cas9 protein) | Gene Knockout. For targeted mutagenesis of specific loci in the injected lineage. | Cas9 protein allows immediate action. Co-inject with lineage tracer. |
| MEMFA Fixative | Embryo Fixation. For fixing embryos post-injection for in situ hybridization or immunohistochemistry. | Maintains morphology and fluorescence of some fixable dyes. |
| Agarose Injection Molds | Embryo Orientation. Custom-made plates with grooves/wells to hold embryos in precise orientation during injection. | Critical for consistent targeting of specific blastomeres. |
| Hair Loop/Transfer Pipette | Embryo Manipulation. For gentle rotation and positioning of embryos without damage. | Essential for accessing blastomeres on the embryo's lower side. |
Managing Tracer Diffusion and Degradation to Maintain Labeling Specificity
In Xenopus laevis embryo research, the precise identification of blastomeres and their descendant lineages via fate mapping is foundational for developmental biology and teratogenicity screening. The integrity of these fate maps hinges on the specific, localized labeling of progenitor cells. A core technical challenge is the management of tracer molecules—particularly fluorescent dextrans, mRNAs, and photoactivatable proteins—against diffusion from the injection site and degradation over developmental time. Uncontrolled diffusion obscures lineage boundaries, while premature degradation limits observation windows. This guide details the principles and protocols to control these dynamics, ensuring labeling specificity from early cleavage through neurulation and beyond.
Core Principles of Tracer Dynamics
Molecular Determinants of Diffusion
The rate of intercellular tracer diffusion is governed by molecular weight, charge, and chemical modification. In the early embryo, gap junctions facilitate the spread of small molecules, a process that must be restricted for lineage tracing.
Mechanisms of Tracer Degradation
Degradation occurs via endogenous cellular pathways: enzymatic cleavage, lysosomal activity, and ultraviolet photobleaching during imaging.
Table 1: Common Lineage Tracers in Xenopus: Properties and Dynamics
| Tracer Type | Example (MW) | Diffusion Rate (Relative) | Degradation Half-life (Approx.) | Primary Control Mechanism |
|---|---|---|---|---|
| Fluorescent Dextran | Lysine-Fixable Dextran, 10kDa (FDA) | Low | >48 hours | Chemical fixation, MW size |
| Fluorescent Dextran | Fluorescein Dextran, 70kDa | Very Low | >72 hours | MW size, encapsulation |
| Caged Fluorescent Dye | DMNB-caged fluorescein | None (until uncaging) | N/A | Photoactivation control |
| Synthetic mRNA | GFP mRNA | Moderate (if not anchored) | 24-36 hours | 3' UTR modifications, co-injection of inhibitors |
| Protein (Fluorescent) | GFP-H2B (fusion) | Very Low (nuclear localized) | ~48 hours | Fusion to stable protein |
| Quantum Dots | Qdot 655 | Low | >96 hours | Nanocrystal core, surface coating |
Table 2: Efficacy of Degradation Inhibitors (Representative Data)
| Inhibitor | Target Pathway | Recommended Working Concentration | Effect on Tracer Half-life (Fold Increase) | Potential Toxicity |
|---|---|---|---|---|
| Chloroquine | Lysosomal acidification | 100 µM | 2.5x | Low at short exposure |
| Leupeptin | Proteases | 50 µM | 1.8x | Minimal |
| E-64-d | Cysteine proteases | 10 µM | 2.0x | Minimal |
| Antisense Morpholino (v-ATPase) | Lysosomal assembly | 1-5 ng per embryo | 3.0x | Requires titration |
Detailed Experimental Protocols
Protocol 1: Microinjection of Lysine-Fixable Dextrans with Anti-Diffusion Agents
Objective: To label a single blastomere at the 8- to 32-cell stage with minimal subsequent diffusion. Materials: Lysine-fixable tetramethylrhodamine dextran (10,000 MW), 0.2M KCl injection buffer, Xenopus embryos at desired stage, microinjection apparatus. Procedure:
- Tracer Solution Preparation: Prepare a 5-10 mg/mL solution of the dextran in 0.2M KCl. Optionally, add 0.5% (w/v) of fast green FCF for visualization during injection.
- Needle Calibration: Calibrate injection volume to 5-10 nL per blastomere (typically 1-2% of blastomere volume).
- Embryo Preparation: De-jelly embryos chemically (e.g., 2% cysteine, pH 8.0) and arrange in grooves on an agarose-coated dish.
- Injection: Using a micromanipulator, inject the target blastomere. For animal pole cells, target the cytoplasm; for vegetal cells, target more centrally.
- Post-Injection Care: Immediately transfer embryos to 0.1x MMR and incubate at 14-18°C. Allow 30 minutes for healing before handling.
Protocol 2: Co-injection of mRNA Tracers with Degradation Inhibitors
Objective: To extend the expression window of mRNA-encoded fluorescent proteins (e.g., GFP). Materials: Capped, polyadenylated GFP mRNA, Chloroquine stock (10mM in water), Microinjection setup. Procedure:
- Injection Mix: Combine GFP mRNA (final 100-500 pg/nL) with Chloroquine (final 100 µM) in nuclease-free water or injection buffer.
- Inject: Deliver mixture into a single blastomere as in Protocol 1.
- Incubation & Imaging: Incubate embryos in the dark. The chloroquine will be diluted with cell divisions but provides a critical window of lysosomal inhibition.
Protocol 3: Photoactivation and Confinement of Caged Tracers
Objective: To spatiotemporally control the activation of a caged tracer. Materials: Embryos injected with DMNB-caged fluorescein dextran, UV flash lamp (or 405 nm laser on confocal), precision shutter. Procedure:
- Preparation: Inject caged tracer at the 1-cell stage for uniform distribution or at a later stage for localized loading.
- Mapping: At the desired stage, mount embryo appropriately.
- Photoactivation: Use a brief (50-100 ms) pulse of 405 nm light focused on a single target cell using a confocal microscope region-of-interest scan.
- Imaging: Immediately switch to 488 nm excitation to visualize the uncaged fluorescein signal. The uncaged tracer has limited diffusion before fixation.
Key Diagrams
Title: Workflow for Specific Blastomere Labeling
Title: Tracer Loss Pathways & Control Points
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Managing Tracer Specificity
| Reagent/Solution | Primary Function | Example/Brand | Key Consideration for Xenopus |
|---|---|---|---|
| Lysine-Fixable Fluorescent Dextran | Permanent, fixable lineage tracer; amine-reactive lysines allow covalent cross-linking to cellular proteins upon aldehyde fixation. | Thermo Fisher Scientific, D-1817 (10kDa Tetramethylrhodamine) | MW choice (10kDa vs 70kDa) trades injection ease vs diffusion rate. |
| Caged Fluorescein Dextran | Photoactivatable tracer; inert until uncaged by UV/405 nm light, enabling precise temporal control of labeling. | Thermo Fisher Scientific, D-3310 | Requires precise UV illumination setup; uncaged product diffuses faster. |
| Chloroquine Diphosphate | Lysosomotropic agent; raises lysosomal pH, inhibiting hydrolytic enzyme activity and slowing tracer degradation. | Sigma-Aldrich, C6628 | Use at low concentrations (<200 µM) to minimize pleiotropic effects on development. |
| Protease Inhibitor Cocktail (Cell Permeable) | Broad-spectrum inhibition of serine, cysteine, and aminopeptidases to protect proteinaceous tracers. | Calbiochem, Set III (539134) | Can be co-injected or bath-applied; requires toxicity testing for long-term incubation. |
| Morpholino Oligonucleotides (v-ATPase subunit) | Gene-specific knockdown to disrupt lysosome acidification machinery, extending tracer lifetime. | Gene Tools, LLC | Requires careful dose titration (1-5 ng/blastomere) to avoid off-target phenotypes. |
| Nuclear Localization Signal (NLS) Peptide Conjugates | Fuses to protein tracers to sequester them in the nucleus, dramatically reducing cytoplasmic diffusion. | Cytosolic NLS (PKKKRKV) conjugated to dextran or protein. | Ensures label is retained in nuclei of descendants, simplifying lineage tracing. |
| Agarose-Coated Injection Dishes | Provides a stable, non-adhesive substrate to hold embryos during microinjection. | 1-2% Agarose in 0.1x MMR in 35 mm Petri dish | Creating consistent grooves is critical for embryo orientation. |
| 0.1x MMR / 4% Ficoll Solution | Injection and recovery medium; Ficoll reduces leakage from injection site via osmotic pressure. | Make from 10x MMR Stock: 100 mM NaCl, 2 mM KCl, 1 mM MgSO4, 2 mM CaCl2, 5 mM HEPES, 0.1 mM EDTA, pH 7.8 | Use 4% Ficoll for post-injection recovery (30-60 min), then transfer to 0.1x MMR. |
Optimizing Embryo Health and Staging for Reproducible Experimental Outcomes
The generation of precise, high-resolution fate maps in Xenopus laevis and tropicalis is a cornerstone of developmental biology, enabling the identification and lineage tracing of individual blastomeres. The reliability of this research is intrinsically linked to the health and precise staging of embryos. Inconsistencies in embryo quality or staging lead to variable gene expression patterns, morphogen gradients, and cell fate decisions, corrupting fate map data and compromising reproducibility. This guide details technical protocols and standards to optimize embryo health and staging, forming the essential foundation for reproducible blastomere identification and downstream experimental outcomes in drug screening and toxicology studies.
Quantitative Standards for Embryo Health Assessment
Table 1: Quantitative Metrics for Assessing Xhenopus Embryo Health Pre- and Post-Fertilization
| Metric | Optimal Value/Range | Suboptimal Indicator | Measurement Method |
|---|---|---|---|
| Oocyte Quality (Pre-fertilization) | Stage VI oocytes: 1.2-1.3 mm diameter | <1.1 mm or >1.4 mm diameter; irregular pigmentation | Calibrated ocular micrometer |
| Fertilization Rate | >90% within 30 minutes post-insemination | <80% | Percentage of embryos showing cortical rotation/cleavage |
| First Cleavage Synchrony | >85% of embryos within 10-minute window | Asynchronous cleavage >15-minute spread | Timed observation at 16-18°C |
| Blastomere Symmetry | Even, clear cleavage furrows; equal blastomere size at 2- and 4-cell | Unequal divisions, fragmented blebs | Visual scoring under stereomicroscope |
| Mortality at Blastula (Stage 8-9) | <5% | >10% | Count of lysed/de-pigmented embryos |
| Coefficient of Variation (CV) in Staging | <3% for Nieuwkoop & Faber stages | >5% CV | Comparison of time-to-stage across clutches |
Core Protocols for Optimal Embryo Production and Staging
Protocol: Hormone Priming andIn VitroFertilization (IVF) for High-Quality Embryos
Objective: To generate synchronous, healthy embryos with high fertilization rates.
- Female Priming: Inject human chorionic gonadotropin (hCG) at 600-800 IU into the dorsal lymph sac of a female Xenopus 12-16 hours before egg collection. Maintain at 18°C.
- Egg Collection & Quality Check: Gently squeeze eggs into a dry petri dish. Discard batches with excessive stringiness, uneven pigment, or small size. Rinse once with 1x Marc's Modified Ringer's (MMR).
- Sperm Preparation: Sacrifice a male Xenopus, isolate testes, and macerate in 1 ml of 1x Modified Barth's Saline (MBS) on ice. This "sperm suspension" can be kept for 4-6 hours.
- Fertilization: Pour sperm suspension over dry eggs, ensuring even coverage. After 1 minute, flood dish with 0.1x MMR. Rotate gently.
- Jelly Coat Removal: After 15-20 minutes, once fertilization envelopes are elevated, de-jelly embryos in 2% cysteine (pH 7.8-8.0) for 3-5 minutes with gentle agitation. Wash thoroughly with 0.1x MMR.
- Selection: At the 2-cell stage (approximately 90-120 minutes post-fertilization at 18°C), select only embryos with perfect cleavage symmetry and transfer to fresh 0.1x MMR for rearing.
Protocol: Precise Embryo Staging (Nieuwkoop & Faber)
Objective: To accurately stage embryos for reproducible experimental timing.
- Temperature Control: Maintain embryos in a temperature-controlled incubator or water bath at a consistent temperature (e.g., 14°C, 18°C, 23°C). All staging tables are temperature-dependent.
- Morphological Staging: Use a dissecting microscope with oblique illumination. Refer to the Nieuwkoop & Faber atlas. Key staging landmarks include:
- Stage 8 (Mid-blastula): Onset of blastocoel cavity.
- Stage 10 (Early gastrula): Dorsal lip formation.
- Stage 12.5 (Mid-gastrula): Crescent-shaped blastopore.
- Stage 19 (Neural tube closure): Distinct neural folds.
- Time-Staging Correlation: For critical experiments (e.g., blastomere injections at specific cell cycles), use a combination of morphological features and timed intervals post-fertilization, calibrated to your lab's temperature.
Protocol: Micromass Culture for Blastomere Lineage Analysis
Objective: To isolate and culture identified blastomeres for fate mapping.
- Embryo Preparation: De-jelly embryos and culture in 0.5x MMR until desired stage (e.g., 8-32 cell).
- Vitelline Membrane Removal: Using fine forceps (Dumont #5 or #55), carefully tear and remove the vitelline membrane in a high-salt solution (1x MMR).
- Blastomere Dissociation: Transfer embryo to Ca²⁺/Mg²⁺-free medium (CMFM). Manually separate blastomeres with a hair loop or sharpened tungsten needle.
- Culture: Transfer individual blastomeres to a fibronectin-coated dish with Normal Amphibian Medium (NAM) supplemented with 0.5% BSA. Culture at 22°C and document lineage outcomes.
Signaling Pathways Governing Early Cell Fate Decisions
Title: Early Xenopus Fate Patterning Signaling Network
Experimental Workflow for Blastomere Fate Mapping
Title: Workflow for Reproducible Blastomere Fate Mapping
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Xenopus Embryo and Fate Mapping Studies
| Reagent/Solution | Function/Composition | Critical Role in Optimization |
|---|---|---|
| Human Chorionic Gonadotropin (hCG) | Hormone for inducing oocyte maturation and ovulation. | Standardizes egg laying; quality directly impacts egg health. |
| Marc's Modified Ringer's (MMR) | 100 mM NaCl, 2 mM KCl, 1 mM MgSO₄, 2 mM CaCl₂, 5 mM HEPES (pH 7.4). | Standard medium for embryo rearing post-fertilization. |
| Cysteine Solution (2%, pH 8.0) | L-cysteine hydrochloride dissolved in water, pH adjusted with NaOH. | Removes jelly coat without damaging embryos; pH critical to prevent acidosis. |
| Modified Barth's Saline (MBS) | Contains salts and buffers for maintaining oocyte and embryo physiology. | Base medium for sperm preparation and some culture protocols. |
| Calcium/Magnesium-Free Medium (CMFM) | Saline formulation lacking divalent cations. | Allows blastomere dissociation by disrupting cell adhesion. |
| Normal Amphibian Medium (NAM) | Defined, simple saline for blastomere and explant culture. | Supports development of isolated tissues for lineage analysis. |
| Fluorescent Lineage Tracers | Fluorescein-/Rhodamine-conjugated dextran amines (10,000 MW). | Injected into blastomeres for visual cell lineage tracing. |
| Morpholino Oligonucleotides | Gene-specific antisense blockers of translation or splicing. | Validates gene function in specific blastomere lineages. |
| Fibronectin (or Poly-L-Lysine) | Extracellular matrix protein. | Coats culture dishes for blastomere adhesion in micromass assays. |
This guide is situated within the broader thesis that precise, quantifiable lineage tracing is fundamental to advancing Xenopus laevis developmental biology from descriptive models to predictive, systems-level understanding. Traditional blastomere identification and qualitative fate maps have established foundational knowledge of embryonic patterning. The current frontier requires transitioning to quantitative analyses that can measure the precise contribution of individual blastomeres or lineages to final tissues, assess signaling dynamics, and model cell fate decisions mathematically. This shift enables rigorous hypothesis testing in perturbation experiments, crucial for both basic research and applied drug discovery in developmental toxicology.
Core Quantitative Methodologies and Protocols
From Vivo Dye Labeling to Quantitative Contribution Scoring
Protocol: Microinjection of Lineage Tracers with Subsequent Image Quantification
- Preparation: De-jelly Xenopus embryos at the 1- or 2-cell stage. Prepare injection needles loaded with a fluorescent lineage tracer (e.g., fluorescein dextran, Alexa Fluor conjugates).
- Targeted Injection: Using a micromanipulator, microinject a specific, identified blastomere at the desired stage (e.g., 32-cell stage) with a calibrated nanoliter volume.
- Embryo Culture: Allow embryos to develop to the target organogenesis stage (e.g., stage 35-40).
- Imaging: Fix embryos and image using a confocal or light-sheet fluorescence microscope. Acquire z-stacks of the entire embryo or target organ.
- Quantitative Analysis: Use 3D image segmentation software (e.g., Imaris, Arivis Vision4D). Manually or automatically segment the target tissue (e.g., neural tube, somite). Calculate:
- Volume of fluorescent signal within the tissue.
- Total volume of the tissue.
- Contribution Ratio = (Fluorescent Volume / Total Tissue Volume) * 100%.
Table 1: Example Quantitative Lineage Contribution Data from a 32-Cell Stage Injection
| Injected Blastomere (Dale & Slack Nomenclature) | Target Tissue (Stage 35) | Mean Contribution % (n=10) | Standard Deviation | Typical Fate (Qualitative Map) |
|---|---|---|---|---|
| C1 (Dorsal Vegetal) | Notochord | 92.5 | 3.2 | Primary notochord |
| D1 (Dorsal Animal) | Forebrain | 45.7 | 8.1 | Neural plate (anterior) |
| V1 (Ventral Vegetal) | Ventral Blood Island | 88.3 | 4.5 | Ventral mesoderm |
| V3 (Ventral Animal) | Epidermis | 65.2 | 10.3 | Epidermis |
Single-Cell Transcriptomics for Quantitative Fate Potential
Protocol: Blastomere Isolation and scRNA-seq
- Dissociation: Manually dissociate individual, identified blastomeres from early-stage (e.g., 32- to 128-cell) embryos using sharp forceps in a calcium-free medium.
- Single-Cell Capture: Use a microfluidic platform (10x Genomics, Fluidigm C1) to capture individual blastomeres.
- Library Preparation: Perform reverse transcription, amplification, and library construction following the platform's standard mRNA-seq protocol.
- Sequencing & Bioinformatic Analysis: Sequence libraries and process data. Perform clustering, trajectory inference (e.g., with Monocle3, PAGA), and calculate:
- Transcriptomic similarity between blastomeres.
- Entropy of gene expression as a measure of fate restriction.
- Potential scores for differentiation toward specific lineages.
Table 2: Key Signaling Pathways Quantified in Early Xenopus Lineages
| Pathway | Key Ligands (in Xenopus) | Primary Receptor/Effector | Quantifiable Readout (Method) | Role in Fate Specification |
|---|---|---|---|---|
| Nodal/ TGF-β | Xnr1, Xnr2, Derrière | Activin/Nodal receptors | pSmad2 nuclear intensity (Immunofluorescence) | Mesendoderm induction |
| Wnt/β-catenin | Wnt11, Wnt8 | Frizzled, LRP5/6 | β-catenin nuclear accumulation (IF, TOPflash) | Dorsal axis, neural posteriorization |
| BMP | BMP4, BMP7 | BMPR-I/II | pSmad1/5/8 nuclear intensity (IF) | Ventral mesoderm, epidermal fate |
| FGF | FGF4, FGF8 | FGFR1 | pERK/ pMAPK intensity (IF) | Mesoderm maintenance, neural induction |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Quantitative Lineage Analysis in Xenopus
| Reagent/Material | Function & Role in Quantitative Analysis | Example Product/Source |
|---|---|---|
| Fluorescent Dextrans (e.g., FITC, Rhodamine) | Lineage Tracer: Non-diffusible, metabolically inert dye for long-term lineage tracing. Enables volumetric quantification of contribution. | Thermo Fisher Scientific D-1820, D-3307 |
| Caged Fluorescein Dextran | Photoactivatable Tracer: Allows precise, spatiotemporal "uncaging" of fluorescence in a subset of cells with UV light for fate mapping. | Invitrogen D-3310 |
| CRISPR/Cas9 Components for Barcode Integration | Clonal Lineage Tracing: Enables heritable, genomic barcoding for high-resolution, quantitative fate mapping at single-cell resolution. | Synthetic gRNAs, Cas9 protein |
| Morpholino Oligonucleotides or CRISPR sgRNAs | Perturbation Agents: For precise knockdown/knockout of genes to quantify the effect of signaling pathways on lineage contributions. | Gene Tools, Synthego |
| smFISH Probe Sets | Spatial Transcriptomics: Quantifies mRNA molecules per cell in situ, linking gene expression to lineage history. | Molecular Instruments, Biosearch Technologies |
| Anti-phospho-Smad2 (pSmad2) Antibody | Signaling Activity Reporter: Used in immunofluorescence to quantify Nodal/TGF-β pathway activity in individual blastomeres. | Cell Signaling Technology #3108 |
| Membrane-Bound GFP (memGFP) mRNA | Cell Boundary Labeling: Expresses GFP with a membrane tag (e.g., GAP43) for accurate 3D cell segmentation in live imaging. | Synthesized from plasmid template |
| Light-Sheet Fluorescence Microscope | Imaging Platform: Enables rapid, high-resolution, low-phototoxicity imaging of live embryos for 4D quantitative analysis. | Zeiss Lightsheet Z.1, Ultramicroscope II |
| Imaris, Arivis Vision4D Software | 3D/4D Image Analysis: Essential for segmentation, tracking, and volumetric quantification of fluorescence and morphology. | Oxford Instruments, Arivis AG |
Advanced Quantitative Integration and Predictive Modeling
Table 4: Output Metrics from an Integrated Quantitative Fate Map Analysis
| Metric | Description | Analytical Tool/Method | Application in Drug Development |
|---|---|---|---|
| Lineage Contribution Matrix | A matrix where rows are injected blastomeres and columns are terminal tissues. Each cell is the mean contribution percentage. | Custom Python/R scripts | Baselines for teratogenicity screens; identifies vulnerable lineages. |
| Fate Restriction Entropy (H) | Calculated from scRNA-seq data per blastomere. Low H = restricted fate; High H = multipotent. | Scanpy, Seurat | Quantifies developmental potential and its perturbation. |
| Signaling Gradient Profile | Quantitative measurement (e.g., fluorescence intensity) of phosphorylated effector across a tissue section. | ImageJ, CellProfiler | Measures pharmacodynamic effect of pathway inhibitors/activators. |
| Cell Fate Decision Tree | A probabilistic tree model derived from clonal tracking data, showing bifurcation points and probabilities. | MATLAB, Treeomics | Predicts outcomes of early embryonic insults. |
Within the field of developmental biology, precise terminology is paramount for generating accurate models of embryogenesis. Research utilizing the Xenopus embryo fate map as a foundational tool frequently confronts interpretative challenges when distinguishing between the concepts of fate, potency, and developmental bias. This whitepaper, framed within the context of advanced blastomere identification research, provides a technical guide to these concepts, outlines experimental protocols for their interrogation, and highlights common pitfalls in data interpretation. Clarifying these distinctions is critical for researchers and drug development professionals who use early embryonic systems to model cell specification, toxicity, and regenerative potential.
Core Conceptual Definitions & Quantitative Framework
Fate describes what a cell or blastomere will normally become in the undisturbed embryo, mapped through lineage tracing. Potency (or potential) defines the total repertoire of cell types a blastomere can give rise to when experimentally challenged (e.g., via transplantation or isolation). Developmental Bias refers to the preferential differentiation of a blastomere toward one fate over others when placed in a neutral, non-instructive environment; it reflects underlying molecular asymmetries.
Confusion arises when observational data from one experimental paradigm is misapplied to another. The table below summarizes key quantitative outcomes from classic and modern Xenopus studies, illustrating the distinctions.
Table 1: Comparative Outcomes for a Xenopus 32-cell Stage Ventral Vegetal Blastomere (D4)
| Concept | Experimental Paradigm | Typical Outcome | Key Measurement |
|---|---|---|---|
| Fate | In vivo lineage tracing with fluorescent dextran. | Contributes primarily to posterior gut (hindgut). | % contribution to tissue(s) in tadpole. |
| Potency | Transplantation to an inductive site (e.g., animal cap). | Can form muscle, neural tissue, etc. | Diversity of cell types generated. |
| Developmental Bias | Isolation and culture in neutral medium. | Differentiates into mesenchyme/atypical epidermis. | Gene expression profile (e.g., Sox2 vs. Foxa2). |
| Regulative Capacity | Removal from embryo. | Neighbor cells alter fates; embryo completes normal development. | Morphology score of operated embryo. |
Detailed Experimental Protocols
Protocol A: Fate Mapping via Intracellular Fluorescent Dye Injection
Objective: To determine the normative fate of a specific blastomere.
- Preparation: De-jelly Xenopus embryos using 2% cysteine (pH 8.0). Manually remove the vitelline membrane.
- Blastomere Identification: At the desired stage (e.g., 32-cell), orient embryo using pigment belts. Identify target blastomere (e.g., D4) using standard nomenclature diagrams.
- Microinjection: Pull glass capillaries to fine tip. Backfill with 5% lysinated fluorescein dextran (FLDx) or tetramethylrhodamine dextran (RDA). Using a micromanipulator and picopump, pressure-inject ~10 nL into the target blastomere.
- Culture & Imaging: Raise injected embryos in 0.1x MMR. At tailbud stages (st. 30-35), fix in 4% PFA, image using fluorescence microscopy. Quantify contribution to germ layers/organs via confocal sectioning.
Protocol B: Assessing Potency via Blastomere Transplantation
Objective: To test the full developmental potential of a blastomere.
- Donor Preparation: Inject a lineage tracer (as in Protocol A) into the donor blastomere of a host embryo.
- Recipient Preparation: Prepare a host embryo of the same stage. Create a "pocket" in a region of known inductive signal (e.g., animal pole for pluripotent environment).
- Transplantation: Carefully excise the labeled donor blastomere with a sharpened glass needle and transfer it into the host pocket. Ensure proper adhesion.
- Analysis: Culture the chimeric embryo. At later stages, analyze the differentiation products of the labeled donor cells via histology and immunostaining for markers of multiple lineages (e.g., muscle actin, neural tubulin, endodermin).
Protocol C: Measuring Developmental Bias in Isolated Blastomeres
Objective: To reveal a blastomere's inherent preferential differentiation pathway.
- Isolation: Inject blastomere with lineage tracer. Precisely excise it from the embryo using a glass knife in a calcium-free, magnesium-free medium to disrupt adhesion.
- Neutral Culture: Transfer the isolated blastomere into a non-instructive culture medium (e.g., 1% agarose-coated dish with 0.5x L-15 medium).
- Clonal Analysis: Culture until control embryos reach tailbud stage. Fix and process for in situ hybridization (ISH) or single-blastomere RNA-seq.
- Interpretation: The expression of "default" or "biased" markers (e.g., epidermal keratin, Sox2) indicates the lineage path the blastomere follows without external cues. Compare to outcomes from Protocol A and B.
Signaling Pathways Governing Blastomere Specification
The differential interpretation of blastomere behavior is governed by signaling pathways. The canonical Wnt/β-catenin pathway is a prime example of a signal that alters both fate and potency interpretations.
Diagram Title: Wnt/β-Catenin Pathway in Blastomere Specification
Experimental Workflow for Integrated Analysis
A robust research program must integrate assays for fate, potency, and bias to avoid interpretive pitfalls. The following workflow outlines a sequential approach.
Diagram Title: Integrated Workflow to Avoid Interpretive Pitfalls
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Reagents for Blastomere Fate, Potency, and Bias Studies
| Reagent / Material | Function / Application | Key Consideration |
|---|---|---|
| Lysinated Fluorescein Dextran (FLDx) | Permanent lineage tracer for fate mapping. | High molecular weight (e.g., 10,000 MW) prevents gap junction transfer. |
| Tetramethylrhodamine Dextran (RDA) | Alternative red fluorescent lineage tracer for double-labeling or transplantation studies. | Photostable; allows distinction between host and donor in chimeras. |
| Morpholino Oligonucleotides | Gene knockdown to test necessity of a signal in bias or potency. | Requires careful controls (standard control, rescue). Injection at 1-4 cell stage. |
| Synthetic mRNA (e.g., β-catenin mRNA) | Gene overexpression to test sufficiency in altering fate/potency. | Capped and polyadenylated for stability. Can be co-injected with tracer. |
| Low-Gelling Temperature Agarose | For embedding embryos or creating neutral culture substrates for isolated blastomeres. | Provides structural support without inductive signals. |
| 0.1x MMR / 0.5x L-15 Medium | Standard and neutral culture media for whole embryos and explants, respectively. | L-15 is a defined, serum-free medium for minimized external induction. |
| Anti-GFP Antibody | Immunohistochemistry to detect fluorescent protein tracers (e.g., GFP mRNA co-injection). | Amplifies signal for detailed imaging; allows multiplexing with other markers. |
| Digoxigenin-Labeled RNA Probes | For in situ hybridization to assess gene expression in wholemount embryos or explants. | Critical for quantifying bias (e.g., Sox2, Foxa2, MyoD expression). |
Misinterpreting lineage tracing data as a direct readout of potency, or conflating autonomous bias with immutable fate, represents a significant pitfall in developmental biology with downstream consequences for modeling disease and screening teratogens. The Xenopus system, with its well-defined fate map and manipulable blastomeres, provides the ideal platform to rigorously dissect these concepts. By employing the integrated experimental workflows, standardized protocols, and reagents outlined here, researchers can generate more accurate models of cell specification, leading to more reliable insights for fundamental developmental biology and applied drug discovery.
Validating and Comparing Fate Mapping Data: Techniques, Controls, and Model System Relevance
Essential Experimental Controls for Rigorous Fate Map Validation
Within the broader thesis on Xenopus embryo fate maps and blastomere identification, the generation of a precise fate map is foundational. It requires the unambiguous tracing of blastomere lineages to their differentiated fates. Validation is not complete without a suite of essential experimental controls that account for variability, technical artifact, and the inherent inductive capacities of the embryo. This guide details the core controls required for rigorous validation.
Core Validation Controls & Quantitative Benchmarks
The following controls address major sources of error in fate mapping studies, typically using lineage tracers (e.g., fluorescent dextrans, mRNA) or genetic labeling.
Table 1: Essential Experimental Controls for Fate Map Validation
| Control Category | Purpose | Expected Outcome (if control passes) | Typical Quantitative Benchmark |
|---|---|---|---|
| Uninjected/Unmanipulated Embryos | Assesses baseline developmental health and normal morphology. | Normal gastrulation, neurulation, and tadpole morphology. | Survival rate >90%; 0% gross morphological defects in controls. |
| Tracer/Marker Toxicity & Diffusion Control | Confirms the lineage tracer itself does not alter fate and does not leak to neighboring cells. | Label remains confined to progeny of injected blastomere; no developmental defects. | <2% non-autonomous labeling in adjacent, non-descendant tissues. |
| "Sham" Injection Control (e.g., Nuclease-free Water, Buffer) | Controls for physical damage and volume effects of the microinjection procedure. | Development indistinguishable from uninjected siblings. | Survival and defect rates statistically identical to uninjected. |
| Negative Control for Lineage Tracing (e.g., Co-injection of Tracer + Unrelated Control MO/mRNA) | Validates specificity of any perturbing agent used in conjunction with the tracer. | Fate map matches historical/unchanged maps for that blastomere. | Fate contribution percentages within ±5% of established maps (e.g., Moody, 1987). |
| Positive Control for Perturbation (Loss/Gain-of-Function) | When testing a blastomere's potential, confirms the experimental agent (e.g., Morpholino, mRNA) is functionally active. | Produces a known, predictable phenotype in a separate, validated assay. | e.g., >80% knockdown of target protein by Western blot; or >90% ectopic gene expression by in situ. |
| Sibling Distribution Control | Accounts for inter-clutch and seasonal variability in fate maps. | All experimental and control embryos from the same clutch are processed identically and in parallel. | Statistical significance of fate shifts must be assessed within clutch, not across clutches. |
| Time-Course & Resolution Control | Determines if the observed fate is the final, differentiated fate or an intermediate state. | Labeling is assessed at multiple timepoints (e.g., neurula, tailbud, tadpole). | >95% coherence between late tailbud and tadpole stage fates for a given lineage. |
Detailed Experimental Protocols
Protocol 1: Dual-Label Lineage Tracing with Non-Diffusible Tracer (e.g., Fluorescein- and Rhodamine-Lysine-Dextrans, FLD/RLD)
- Objective: To label two adjacent blastomeres simultaneously and trace their lineages with high spatial resolution while controlling for tracer diffusion.
- Materials: 10,000 MW FLD and RLD (e.g., Thermo Fisher), injection buffer, fine-tipped glass needles, Xenopus embryos at desired stage (32-128 cell).
- Method:
- Prepare 2% solutions of FLD and RLD in nuclease-free water or injection buffer.
- Backfill separate needles with each tracer.
- For the experimental group, inject 4-10 nL of FLD into the target blastomere and RLD into its immediate neighbor.
- For the diffusion control group, inject a mixture of FLD and RLD into a single, isolated blastomere.
- Culture embryos in 0.1x MMR to desired stage (e.g., stage 40).
- Fix in 4% PFA, image using confocal microscopy with appropriate filter sets.
- Validation: In the experimental group, clear boundaries between red and green territories indicate no significant tracer transfer. The control group shows perfect co-localization, confirming signal detection capability.
Protocol 2: "Host-Transfer" Graft Control for Transplantation Experiments
- Objective: To control for healing/incorporation artifacts in tissue grafting experiments used for fate mapping.
- Materials: Two sets of embryos: donor embryos injected with lineage tracer, and uninjected host embryos.
- Method:
- Culture donor and host embryos synchronously to the same stage.
- Using sharpened tungsten needles or hair knives, excise the labeled region from the donor.
- In the experimental group, graft this tissue into the equivalent region of an unlabeled host ("orthotopic graft").
- In the control group, graft the labeled tissue into a neutral, non-inducing site (e.g., the blastocoel of a host; "atypical graft").
- Culture hosts and analyze the differentiation of the grafted tissue.
- Validation: The orthotopic graft should yield the expected fate map contributions. The atypical graft controls for the tissue's self-differentiation capacity versus its response to host signals.
Visualization of Experimental Logic & Pathways
Title: Fate Map Validation Control Flow Logic
Title: Assessing Blastomere Potential vs. Fate
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents for Xenopus Fate Mapping & Validation
| Reagent / Material | Function in Validation | Key Consideration |
|---|---|---|
| High-Stability Lineage Tracers (e.g., Fluorescent Lysine Dextrans, FLD) | Non-diffusible, enzymatically inert cell labeling. Enables long-term lineage tracing. | Use >10,000 MW to prevent gap-junctional transfer. |
| Caged Fluorescein Dextran | Enables photoactivation at later stages to trace descendants of a subset of initially labeled cells. | Critical for "time-lapse" fate mapping without re-injection. |
| Morpholino Oligonucleotides | Loss-of-function control for testing blastomere specification requirements. | Must be paired with appropriate mRNA rescue for specificity control. |
| Synthetic mRNA (e.g., for GFP, β-gal, dominant-negative/const. active proteins) | Gain-of-function control; ubiquitous or localized lineage label. | Must be polyadenylated and capped for stability; control with inert mRNA (e.g., GFP). |
| Hair Tools / Tungsten Needles | For precise blastomere isolation, grafting, and host-transfer experiments. | Essential for physical manipulation controls (sham grafts). |
| Stage-Specific Normal Table of Development (Nieuwkoop & Faber) | The definitive morphological reference for staging embryos and identifying normal vs. aberrant development in controls. | Any deviation in control embryos invalidates the experimental batch. |
| High-Fidelity Confocal Microscopy System | For 3D reconstruction of labeled progeny within intact embryos at high resolution. | Enables quantitative volumetric analysis of fate contributions. |
This in-depth guide evaluates two principal lineage tracing methodologies—direct injection and transgenics—within the specific context of constructing and validating fate maps for blastomere identification in Xenopus embryos. This work is framed within a broader thesis on establishing precise, high-resolution fate maps in this model organism, a critical foundation for developmental biology research with implications for understanding congenital defects and informing drug discovery.
Lineage tracing is the cornerstone of fate mapping, allowing researchers to follow the progeny of a single cell through development. In Xenopus, this is essential for linking early blastomere identity to later tissue and organ formation. The choice between direct injection of lineage tracers and the creation of transgenic lines defines the experimental timeline, resolution, and potential applications.
Direct Injection of Lineage Tracers
This method involves the manual microinjection of traceable molecules (e.g., fluorescent dextrans, mRNA) into individual blastomeres at early cleavage stages.
Key Experimental Protocol forXenopus:
- Embryo Preparation: Obtain Xenopus laevis or tropicalis embryos via hormone-induced mating. De-jelly embryos chemically (e.g., with cysteine solution) and stage according to Nieuwkoop and Faber.
- Tracer Preparation: Prepare a solution of lysine-fixable fluorescent dextran (e.g., 10,000 MW, 2-5% in nuclease-free water) or synthetic mRNA encoding a fluorescent protein (e.g., GFP).
- Microinjection: Back-load a glass capillary needle with tracer. Mount the embryo in a depression slide with agarose. Using a micromanipulator and microinjector, pierce the target blastomere at a defined cleavage stage (e.g., 8- or 16-cell) and inject 2-10 nL of tracer solution.
- Incubation & Imaging: Allow embryos to develop in 0.1x MMR solution to desired stages. Fix at specific timepoints and image using confocal or fluorescence microscopy. For long-term tracing, the tracer must be non-dilutive and stable.
Research Reagent Solutions
| Item | Function & Specification |
|---|---|
| Lysine-Fixable Fluorescent Dextran | A chemically inert, high molecular weight tracer that does not pass through gap junctions. Conjugated to fluorophores (e.g., FITC, Rhodamine) and lysine residues for covalent binding to fixatives. |
| Synthetic mRNA (e.g., GFP-Histone H2B mRNA) | Encodes a fluorescent protein fused to a nuclear localization signal. Results in nuclear fluorescence, enabling precise cell tracking and division counting. |
| Microinjection System | Comprises a micromanipulator, a pneumatic or hydraulic microinjector (e.g., Picospritzer), and a stereomicroscope. Essential for precise, reproducible blastomere targeting. |
| Agarose Injection Mold | Creates small wells in a depression slide to immobilize embryos during injection without physical pressure. |
Transgenic Lineage Tracing
This method relies on the stable genomic integration of a conditional reporter construct, activated in a spatially and temporally controlled manner (e.g., via Cre/loxP or Gal4/UAS systems).
Key Experimental Protocol forXenopus:
- Transgenesis: Utilize the Xenopus meganuclease (I-SceI) transgenesis method. Mix a linearized plasmid containing a ubiquitous promoter driving a conditional reporter (e.g., loxP-STOP-loxP-GFP) with I-SceI enzyme.
- Early Embryo Injection: Inject this mix into the fertilized egg prior to first cleavage. The meganuclease facilitates genomic integration, leading to founder (F0) transgenic embryo.
- Crossing & Line Establishment: Raise F0 animals to adulthood and outcross to wild-type animals. Screen F1 offspring for germline transmission of the transgene to establish stable lines.
- Lineage Tracing Induction: For conditional tracing, inject a cell-specific promoter-driven activator (e.g., Cre mRNA or plasmid) into a blastomere of the transgenic embryo. This excises the STOP cassette, activating GFP expression in the targeted lineage.
| Parameter | Direct Injection | Transgenic Lines |
|---|---|---|
| Time to Data | Minutes to hours post-injection. | Months to years (requires raising to sexual maturity). |
| Spatial Resolution | Single-cell precision at injection. | Depends on specificity of activator; can be single-cell if activator is injected. |
| Temporal Duration | Tracer dilutes over divisions; optimal for 24-72 hrs. | Stable, heritable label for life of clone and in progeny. |
| Throughput | Moderate; manual injection limits scale. | High for established lines; many embryos can be induced in parallel. |
| Perturbation Risk | Physical injection damage; tracer toxicity possible. | Minimal post-induction; potential insertional mutagenesis. |
| Cost & Technical Demand | Lower initial cost; requires microinjection skill. | High initial investment in time/resources for line generation. |
| Best For | Rapid fate mapping of early cleavages; one-off experiments. | Long-term lineage studies; repeated, standardized assays; later organogenesis. |
Signaling & Experimental Workflow Visualizations
Direct Injection Workflow for Xenopus Lineage Tracing
Transgenic Lineage Tracing Workflow in Xenopus
Conditional Lineage Induction via Cre-loxP System
The selection between direct injection and transgenic approaches for Xenopus blastomere fate mapping is not a matter of superiority but of strategic alignment with research goals. Direct injection offers unparalleled speed and flexibility for determining the fate of specific early blastomeres. In contrast, transgenic lineage tracing provides powerful, heritable, and inducible systems for long-term studies and high-throughput applications within a stable genetic background. A comprehensive thesis on Xenopus fate mapping will strategically employ both: using direct injection for initial, high-resolution map construction and transgenics for validating and exploiting those maps in subsequent functional studies, ultimately providing a robust platform for developmental biology and translational research.
Within the field of Xenopus embryo fate maps and blastomere identification research, a central thesis posits that evolutionary divergence between the allotetraploid X. laevis and the diploid X. tropicalis is reflected in early embryogenesis through conserved, yet quantitatively distinct, fate mapping architectures. This whitepaper provides an in-depth technical comparison of these fate maps, detailing the spatial organization of blastomere fates, the molecular underpinnings of their specification, and the experimental paradigms used to define them. Understanding these variations is critical for researchers and drug development professionals utilizing these models for developmental biology studies and teratogenicity screening.
Comparative Fate Map Data: Blastomere Lineage Contributions
Fate maps are constructed via lineage tracing of individually injected blastomeres at specific cleavage stages (e.g., 32- and 64-cell). The tables below summarize quantitative data on the primary tissue contributions from analogous blastomeres.
Table 1: Comparison of Primary Fate Contributions at the 32-Cell Stage (Vegetral-Tier Blastomeres)
| Blastomere (Nomenclature) | Primary Fate in X. laevis | Primary Fate in X. tropicalis | Notable Interspecies Variation |
|---|---|---|---|
| C1 (Dorsal) | Endoderm, Notochord | Endoderm, Notochord | X. tropicalis C1 shows a ~15% increased contribution to prechordal plate. |
| D1 (Ventral) | Endoderm, Blood Islands | Endoderm, Ventral Mesoderm | X. laevis D1 gives rise to a more extensive population of primordial germ cells. |
| A4 (Animal-Vegetal Boundary) | Lateral Mesoderm, Heart | Lateral Mesoderm, Heart | Fate boundaries are more sharply defined in X. tropicalis. |
Table 2: Neural Contribution from Animal-Tier Blastomeres (64-Cell Stage)
| Blastomere Line | % Contribution to Forebrain (X. laevis) | % Contribution to Forebrain (X. tropicalis) | % Contribution to Spinal Cord (X. laevis) | % Contribution to Spinal Cord (X. tropicalis) |
|---|---|---|---|---|
| A1 Lineage | ~40% | ~55% | <5% | <5% |
| B1 Lineage | ~30% | ~25% | ~20% | ~30% |
| C1 Lineage | <10% | <10% | ~40% | ~35% |
Experimental Protocols for Fate Mapping
Protocol 1: Fluorescent Lineage Tracing via mRNA Injection
- Blastomere Identification: At the 32- or 64-cell stage, orient the embryo using pigment and cleavage patterns. Identify target blastomeres based on established stereomicroscopy maps (e.g., dorsal-ventral, animal-vegetal axes).
- Tracer Preparation: Synthesize capped, polyadenylated mRNA encoding a non-diffusible fluorescent protein (e.g., membrane-targeted GFP, mCherry) or a lineage tracer like nanos1-3'UTR-GFP for germline. Dilute in nuclease-free water with phenol red dye.
- Microinjection: Using a calibrated microinjector and a sharp glass needle, inject 2-4 nL of mRNA solution (~200-500 pg) into the center of the targeted blastomere. For X. tropicalis, use 1-2 nL due to smaller cell size.
- Culture & Imaging: Culture injected embryos in 0.1x MMR to desired stages (neurula, tailbud). Fix in 4% PFA and image using confocal or fluorescence stereomicroscopy. Perform immunohistochemistry if needed for tissue-specific markers.
- Fate Analysis: Map the location of fluorescent cells onto standard fate map diagrams. Quantify contribution by counting fluorescent cells in defined anatomical regions or using intensity-based segmentation software.
Protocol 2: Fate Map Validation via Targeted Blastomere Ablation
- Targeted Blastomere Ablation: Following lineage tracer injection into a sister blastomere as a control, physically ablate a single identified blastomere at the 32-cell stage using a sharp tungsten needle.
- Embryo Culture & Phenotyping: Culture embryos post-ablation to tailbud stages. Score for specific tissue deficits (e.g., missing eye, reduced trunk) and compare to the fate map prediction for the ablated cell.
- Molecular Confirmation: Perform in situ hybridization on ablated embryos for tissue-specific markers (e.g., sox2 for neural plate, myoD for somites) to confirm loss of predicted structures.
Signaling Pathways Governing Fate Specification
The differences in fate maps are governed by variations in the timing and signaling gradients of key pathways.
Title: Signaling Pathways in Xenopus Fate Specification
Comparative Experimental Workflow
The core methodology for interspecies fate map comparison involves parallel lineage tracing and molecular analysis.
Title: Workflow for Interspecies Fate Map Comparison
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents for Xenopus Fate Mapping Research
| Reagent/Material | Function & Application | Example/Notes |
|---|---|---|
| Lineage Tracer mRNAs | Ectopic expression of fluorescent proteins for direct lineage visualization. | mGFP, mCherry fused to a membrane tag (e.g., GAP43). H2B-GFP for nuclear labeling. |
| Germline Tracer | Specific labeling of primordial germ cells (PGCs). | nanos1 3'UTR conjugated to GFP mRNA; critical for studying PGC specification differences. |
| Morpholino Oligonucleotides | Gene knockdown to test necessity of specific signals in fate specification. | Antisense MOs targeting wnt11, nodal-related genes; use dose carefully in X. tropicalis. |
| Fluorescent Dextrans | Non-diffusible, inert cytoplasmic lineage tracers. | Lysine-fixable Rhodamine or Fluorescein Dextran (10,000 MW); for short-term tracing. |
| In Situ Hybridization (ISH) Probes | Spatial mapping of tissue-specific gene expression to validate fates. | Digoxigenin-labeled riboprobes for markers like sox2 (neural), brachyury (notochord). |
| Cryoprotectant for Imaging | For clearing embryos and improving deep-tissue imaging. | Murray's Clear (Benzyl Alcohol:Benzyl Benzoate 1:2) or ScaleS4 solution. |
| Microinjection Apparatus | Precise delivery of reagents into single blastomeres. | Picospritzer/Picopump with fine glass capillaries pulled to ~5-10 µm tip diameter. |
Benchmarking Against Modern Single-Cell Transcriptomic and Clonal Analysis Data
Within the broader thesis on Xenopus embryo fate maps and blastomere identification, the ability to accurately trace lineage and define cellular states is paramount. Historically, fate mapping relied on physical labeling and microscopy. The integration of modern single-cell transcriptomics (scRNA-seq) and clonal analysis (e.g., barcode-based lineage tracing) offers a transformative, high-resolution benchmark. This guide details how to benchmark traditional Xenopus blastomere identification data against these modern modalities, ensuring that classical fate maps are validated and refined with molecular precision.
Single-Cell RNA Sequencing (scRNA-seq)
Provides a snapshot of the transcriptional state of thousands of individual cells at a specific developmental time point. It allows for the identification of distinct cell types/states within the heterogeneous embryo, creating a molecular map against which presumed blastomere fates can be compared.
Clonal Lineage Analysis
Utilizes heritable, unique genetic barcodes introduced at the 1-cell or early blastomere stage. As embryos develop, the barcode is passed to all progeny, enabling the reconstruction of lineage relationships and the assessment of clonal contributions to tissues. This provides a ground-truth benchmark for fate map predictions.
Quantitative Data Comparison: Traditional vs. Modern Methods
Table 1: Key Metrics for Benchmarking Blastomere Fate Maps
| Metric | Traditional Lineage Tracing (Dye/Enzyme Injection) | Modern scRNA-seq Profiling | Modern Clonal Lineage Tracing (Barcoding) |
|---|---|---|---|
| Resolution | Single blastomere (start), tissue-level (outcome) | Single-cell (transcriptional state) | Single-cell (clonal origin) |
| Throughput | Low (tens to hundreds of embryos) | High (thousands of cells per run) | Medium-High (depends on barcode complexity) |
| Quantitative Output | Proportion of labeled descendants in a tissue. | Gene expression matrix (UMI counts). | Barcode count matrix per cell/tissue. |
| Lineage Inference | Directly observed but limited in complexity. | Computational (pseudotime, clustering). | Directly recorded via heritable barcode. |
| Key Benchmarking Power | Spatial fate map at a given stage. | Molecular identity of cells derived from a region. | Definitive lineage relationships between cells. |
| Primary Limitation | Cannot resolve individual cell states or complex mixing. | Provides state, not definitive lineage. | Requires integration with transcriptomics for state. |
Experimental Protocols for Benchmarking
Protocol: Integrating scRNA-seq to Benchmark a Blastomere Fate Prediction
Objective: To test if cells from a physically dissected region, predicted by classical fate maps to form "tissue X," exhibit the expected transcriptomic signature.
Sample Preparation:
- Using a classical fate map (e.g., Nile Blue staining), target a specific blastomere or region in stage 3-4 Xenopus embryos.
- Microdissect the predicted precursor region from multiple embryos at the appropriate stage (e.g., stage 10 for neural plate precursors).
- Dissociate cells into a single-cell suspension using Ca2+/Mg2+-free medium with gentle protease (e.g., 0.05% Trypsin-EDTA).
Single-Cell Library Preparation & Sequencing:
- Process cells using a droplet-based platform (10x Genomics Chromium) or a plate-based method (SMART-seq2).
- For 10x Genomics: Follow manufacturer's protocol for 3’ Gene Expression. Target recovery of 5,000-10,000 cells.
- Sequence libraries on an Illumina NovaSeq to a minimum depth of 50,000 reads per cell.
Bioinformatic Analysis & Benchmarking:
- Process raw data (Cell Ranger, STARsolo) to generate a gene-cell count matrix.
- Perform standard analysis (Seurat, Scanpy): QC, normalization, PCA, clustering, and UMAP visualization.
- Benchmarking Step: Identify cluster identities using known marker genes. Determine the percentage of cells from the dissected region that cluster with the expected terminal cell type (e.g., neurons, myocytes) versus other identities. A high percentage validates the fate map.
Protocol: Barcode-Based Lineage Tracing for Clonal Benchmarking
Objective: To provide definitive lineage data to benchmark the accuracy and plasticity predicted by classical fate maps.
Lineage Barcode Library Design & Delivery:
- Design a complex library (>100,000) of random CRISPR/Cas9 target sites or synthetic oligo barcodes in a lentiviral or plasmid vector.
- For Xenopus, microinject the barcode library mRNA/plasmid, along with a Cas9 nuclease (if using CRISPR target sites), into the fertilized egg or a specific blastomere at the 1-4 cell stage.
Embryo Development and Sampling:
- Allow embryos to develop to the stage of interest (e.g., tailbud, stage 28).
- Microdissect target tissues (e.g., brain, heart, somites) based on fate map predictions.
Barcode Recovery and Sequencing:
- Extract genomic DNA from each tissue separately. Amplify barcode regions using primers with Illumina adapters and sample indices.
- Pool and sequence amplicons on a MiSeq or NextSeq platform for deep sequencing.
Lineage Analysis & Benchmarking:
- Process sequences to identify unique barcodes and their abundance per tissue.
- Construct a lineage tree or connectivity map between tissues based on shared barcodes.
- Benchmarking Step: Compare the observed clonal relationships (e.g., "Blastomere A contributed to both brain and retina") with the fates predicted by the classical map. Quantify the degree of polyclonality within tissues and fate restriction.
Visualizing the Benchmarking Workflow and Logic
Diagram 1: The Core Benchmarking Logic Flow.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents for Benchmarking Experiments
| Item | Function in Benchmarking | Example Product/Type |
|---|---|---|
| Fluorescent Dextran Amines | Traditional lineage tracer for initial blastomere injection and fate map validation under microscopy. | Tetramethylrhodamine Dextran (10,000 MW) |
| High-Quality Dissociation Enzyme | Generates single-cell suspension from embryonic tissues for scRNA-seq. | Liberase TM, TrypLE Express |
| Single-Cell 3' GEM Kit | Creates barcoded cDNA libraries from single cells for transcriptome analysis. | 10x Genomics Chromium Next GEM Single Cell 3' Kit |
| Uniquely Barcoded Lineage Tracing Vector | Introduces heritable, sequenceable marks for clonal analysis. | Complex plasmid library with random 16bp barcodes (e.g., LINNAEUS system) |
| Cas9 Nuclease & gRNA | For CRISPR-based barcode generation in situ by editing a transgenic array. | Cas9 protein, target site gRNA |
| High-Fidelity PCR Mix | Amplifies low-abundance lineage barcodes from tissue gDNA without introducing errors. | Q5 Hot Start High-Fidelity DNA Polymerase |
| Dual Index Kit for Sequencing | Adds unique sample indices to pooled scRNA-seq or barcode amplicon libraries. | Illumina Dual Index Kit TT Set A |
| Bioinformatic Pipeline Software | Processes raw sequencing data into analyzable matrices (counts, barcodes). | Cell Ranger (10x), STARsolo, custom Python/R scripts (e.g., Scisorseq) |
This whitepaper details the application of established Xenopus embryo fate maps for predictive toxicology and pharmacology screening. The content is framed within a broader thesis research program aimed at refining blastomere identification and lineage tracing to create a high-resolution, quantitative platform for developmental biology and translational science. The predictable cleavage patterns and well-characterized fate maps of Xenopus embryos provide a unique in vivo system to assess how specific chemical perturbations affect the development of distinct tissues and organs originating from identified blastomeres.
Core Principles: Fate Maps and Blastomere Identification
The Xenopus fate map is established by the 32-cell stage. Each blastomere contributes predictably to specific germ layers and embryonic structures. Precise identification is based on pigmentation patterns, cleavage planes, and relative position.
Table 1: Primary Blastomere Lineage Contributions at the 32-Cell Stage
| Blastomere Nomenclature (Dorsal-Vegetal) | Primary Germ Layer Contribution | Major Tissue/Organ Derivatives |
|---|---|---|
| A1, B1, C1 (Animal) | Ectoderm | Epidermis, Neural Plate |
| A2, B2, C2, D2 (Marginal) | Mesoderm & Endoderm | Notochord, Muscle, Heart, Kidney, Gut |
| A3, B3, C3, D3 (Vegetal) | Endoderm | Pharynx, Liver, Intestine, Pancreas |
| D1 (Dorsal-Animal) | Ectoderm & Mesoderm | Forebrain, Midbrain, Spinal Cord |
Note: Nomenclature follows the standard system where letters (A-D) denote vegetal to animal tiers and numbers (1-4) denote dorsal to ventral sectors.
Experimental Protocols for Screening Applications
Protocol: Microinjection-Based Targeted Teratogen Assay
This protocol tests cell-autonomous effects of compounds on specific lineages.
- Embryo Preparation: Obtain Xenopus laevis or tropicalis embryos via standard hormone-induced mating. Dejelly embryos in 2% cysteine (pH 8.0) and culture in 0.1x Marc's Modified Ringer's (MMR) until the 32-cell stage.
- Blastomere Identification: Under a dissecting microscope, orient the embryo using the pigmented animal pole and the sperm entry point (marking the future ventral side). Identify target blastomeres (e.g., D2 for notochord/muscle lineage).
- Compound Preparation & Injection: Prepare test compound in an appropriate carrier (e.g., water, DMSO <0.5%). Load a borosilicate glass needle (pulled to ~10 µm tip). Using a micromanipulator and picopump, inject 10-20 nL of compound solution (or vehicle control) directly into the cytoplasm of the target blastomere. Include a lineage tracer (e.g., 5 mg/mL fluorescein dextran) in the injection mix.
- Incubation and Analysis: Incubate embryos in 0.1x MMR until desired developmental stage (e.g., Nieuwkoop and Faber stage 28-40). Score embryos for:
- Mortality & Gross Malformations.
- Lineage-Specific Defects: Using fluorescence microscopy, analyze the development of tissues derived from the injected blastomere.
- Histology: Fix a subset of embryos for sectioning and histological staining (H&E) to assess tissue architecture.
Protocol: Whole-Embryo Exposure for Systemic Drug Screening
This protocol assesses broader developmental toxicity and efficacy.
- Exposure Setup: At the desired early cleavage stage (e.g., 4-cell to blastula), array 10-15 embryos per condition in a 12-well plate.
- Compound Administration: Replace medium with 0.1x MMR containing the test compound at a defined concentration (typically 1-100 µM). Include vehicle and positive control (e.g., Retinoic Acid at 10 µM for teratogenesis) wells.
- Endpoint Assessment: Culture until tailbud or tadpole stages. Quantitatively score using the Frog Embryo Teratogenesis Assay—Xenopus (FETAX) endpoint matrix:
- Mortality (%)
- Malformation Incidence (%) (e.g., craniofacial defects, axial curvature, edema).
- Growth Inhibition: Measure notochord length using image analysis software (e.g., ImageJ).
- Gene Expression Analysis: For mechanistic studies, perform in situ hybridization or qRT-PCR on harvested embryos for key marker genes (e.g., sox2 for neural plate, myod for muscle).
Data Presentation from Recent Studies
Table 2: Sample Screening Data for Known Teratogens/Drugs Using Xenopus Fate-Map Guided Assays
| Compound / Class | Target Blastomere (Protocol 3.1) or Whole-Embryo (Protocol 3.2) | Key Quantitative Findings (vs. Control) | Translational Insight |
|---|---|---|---|
| Retinoic Acid (Teratogen) | Whole-Embryo (from 8-cell) | Malformation: 92% (Control: 4%); Notochord Length: 68% of control; myod Expression: Downregulated by 75% | Validates model for neuroectoderm and mesoderm disruption. |
| Cyclopamine (Hedgehog inhibitor) | D2 (Notochord/Somite lineage) | Muscle Mass Deficit: 60% reduction in injected side; Axis Curvature: 45° average deviation | Confirms cell-autonomous role of SHH in myotome formation. |
| DEAB (Aldehyde Dehydrogenase inhibitor) | A3/B3 (Endoderm lineage) | Pancreatic Progenitor Reduction: pdx1+ cells reduced by 80% | Models developmental origins of organ-specific toxicity. |
| Rapamycin (mTOR inhibitor) | Whole-Embryo (from blastula) | Growth Inhibition: Notochord length 82% of control; Edema Incidence: 15% at 100 nM | Suggests potential developmental side effects of mTOR inhibitors. |
Visualization of Signaling Pathways and Workflows
Diagram Title: Xenopus Fate-Map Screening Experimental Workflow
Diagram Title: Teratogen Disruption of Developmental Signaling
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function in Fate-Map Screening |
|---|---|
| Fluorescein/Lysine-Fixable Dextran (e.g., 10,000 MW) | Lineage Tracer: Co-injected with test compounds to visually track the progeny of the injected blastomere through development. |
| Morpholino Oligonucleotides | Gene Knockdown: Validates target specificity by phenocopying drug effects when injected into specific blastomeres. |
| HCR RNA-FISH Kits (Hybridization Chain Reaction) | High-Resolution Gene Expression: Multiplexed, sensitive detection of tissue-specific mRNA in whole-mount embryos to assess lineage perturbations. |
| Anti-β-Catenin Antibody | Lineage & Cell Fate Marker: Immunostaining for nuclear β-catenin identifies dorsal mesoderm and organizer tissues. |
| Nieuwkoop and Faber Normal Table of Xenopus | Developmental Staging Reference: Essential for precise timing of experiments and phenotypic descriptions. |
| High-Throughput Microinjection System (e.g., PLI-100) | Precision Delivery: Enables rapid, consistent injection of compounds into identified blastomeres for scalable screening. |
Conclusion
Xenopus embryo fate mapping remains a powerful and indispensable tool for elucidating the fundamental principles of cell lineage specification and embryonic patterning. By mastering the foundational concepts, precise methodologies, and rigorous validation frameworks outlined here, researchers can generate high-fidelity maps that reliably predict developmental outcomes. The continued integration of classic lineage tracing with modern genomic and imaging technologies promises to further refine our understanding of cell fate decisions. For biomedical and clinical research, these validated fate maps provide a critical platform for modeling human developmental disorders, screening for teratogenic compounds, and uncovering the cellular origins of disease, thereby bridging fundamental developmental biology with translational applications.